Abstract
Objectives
The objective of the study was to measure the preoperative and postoperative renal pelvic size and describe the ultrasound findings following successful decompression of a ureteral obstruction using the subcutaneous ureteral bypass (SUB) device in cats.
Methods
This retrospective study assessed the measurement of the renal pelvis of 27 cats with unilateral (n = 21) or bilateral (n = 6) ureteral obstruction before (pre-t0) and after placement of a SUB during short- (before t0 + 3 months) and long-term (after t0 + 3 months) follow-up. Several qualitative ultrasound parameters were recorded for each cat. At both intervals, the last ultrasound examination was used for qualitative criteria and the mean pelvic size was recorded. The complications observed during follow-up were divided into obstructive and non-obstructive.
Results
No qualitative ultrasound parameter was statistically significant. The presence of retroperitoneal or peritoneal effusion was rarely seen (n = 4/25 during the short term and n = 1/14 during the long term). Hyperechogenicity of the perirenal adipose tissue decreased in the long term. A statistically significant decrease in the width of the renal pelvis was noted in the short- (2.4 mm, range 0–7.0 mm) and long-term (1.7 mm, range 0–3.5 mm) follow-ups compared with the preoperative value (11.7 mm, range 0.9–41 mm). Three months following SUB placement, each cat without an obstructive complication had a pelvic width ⩽3.5 mm.
Conclusions and relevance
Renal pelvic distension is at least partially reversible when ureteral obstruction is treated by placement of a SUB. Ultrasound monitoring is a useful tool to detect obstructive complications.
Introduction
Ureteral obstruction is a potentially life-threatening condition in cats. It leads to restriction of urine flow, resulting in hydronephrosis and hydroureter. If bilaterally obstructed, it may cause severe azotemia. In case of unilateral ureteral obstruction and azotemia, decreased function of the contralateral kidney should be suspected. Ureteral obstruction most commonly occurs secondarily to stones or strictures, but may also result from surgical trauma, mucus and mucopurulent plugs or neoplasia.1,2 Recently, an association between upper urinary tract stones and feline chronic kidney disease has been reported. 3
The goal of treating ureteral obstruction is to relieve the obstruction and improve glomerular filtration in a timely manner to avoid permanent nephron loss. Treatment of ureteral obstruction can be divided into medical or surgical approaches. Surgical options are commonly indicated since initial medical management is often unsuccessful. 4 Given the high complication rate associated with traditional surgical techniques (ureterotomy, ureteral resection and anastomosis, ureteronephrectomy), other surgical options, such as ureteral stenting or a subcutaneous ureteral bypass (SUB), have been explored and resulted in improved outcomes.5–8 In our practice, SUB placement has become an important tool in the treatment of feline ureteral obstruction. Complications related to SUB placement include urine leakage from the nephrostomy or cystostomy tubes, leakage around the SUB port, occlusion of the SUB device catheters or the port with a blood clot, stones, mineral debris or a kink, recurrent urinary tract infections and sterile cystitis. 9
To our knowledge, the renal pelvic size following SUB device placement and the ultrasound appearance over time of the SUB device and urinary tract have yet to be reported. In a recent paper, 5 SUB placement was considered successful if short- and long-term renal ultrasound showed decompression and/or improvement in the renal pelvic diameter compared with preoperative measurements. In this study, preoperative pelvic dilation was not a predictor of overall survival. The authors proposed that renal pelvic measurements of 2–7 mm should be expected postoperatively. Renomegaly is not pathognomonic for a ureteral obstruction, and abdominal palpation is an unreliable method for detection of renomegaly. 10 Ultrasonographically, ureteral obstructions are mainly characterized by hydronephrosis and hydroureter proximal to the location of the obstruction.1,11 A retrospective study reported a mean maximal pelvic width of 10.9 mm in cats (n = 17) and 24.9 mm in dogs (n = 6) with urinary outflow obstruction. 12 Given the lack of available data, a persistent distended renal pelvis following correction of the obstruction may be interpreted as normal and related to the SUB device, especially if the obstruction was severe and long-standing prior to placement. In our experience, however, renal pelvic width returns to normal values within 24 h of SUB placement. Therefore, persistent pyelectasia postoperatively may be a sign of continued obstruction.
The aim of this study was to measure the preoperative and postoperative renal pelvic size and describe the ultrasound findings following successful decompression of a ureteral obstruction using the SUB device in cats.
Materials and methods
Case selection
Medical records of all cats treated with a SUB for either a partial or complete ureteral obstruction at the University of Montreal from 2010–2015 were reviewed using a computerized medical record search. The terms bypass, SUB, ureteral obstruction and port were used for patient screening. Cats were included if they had a diagnosis of benign ureteral obstruction and underwent placement of a SUB by one of the authors (MD) at the Centre Hospitalier Universitaire Vétérinaire of the University of Montreal or at the Centre Vétérinaire Rive-Sud (n = 1). The diagnosis of ureteral obstruction was established by a clinician (MD), based on the concomitant presence of clinical signs, azotemia and ultrasound evidence of hydronephrosis, and ureteral distension proximal to the location of the obstruction. The surgical procedure was referred as t0. Relevant information including signalment, history, imaging findings and clinical biochemistry (including creatinine values) data were recorded preoperatively, short term (within the first 3 months following t0) and long term (after the first 3 months following t0) for each cat. To meet the inclusion criteria, one ultrasound examination performed by a radiologist with images available for review had to be recorded for each cat before surgery (pre-t0) and at least once during the short- (between t0 and t0 + 3 months) and/or long-term (after t0 + 3 months) follow-up periods. This time threshold of 3 months between short and long term was arbitrarily decided by the authors based on the usual clinical follow-up recommendations given at the institution. Typically, cats are followed closely in the immediate postoperative period and re-checked at 1 and 3 months following the procedure. Long-term follow-up frequency is based on clinical signs and usually performed every 3–6 months.At each ultrasound follow-up, urine was taken from the SUB for culture and the device was flushed under ultrasound guidance to ensure patency. The port sites were surgically prepared and a Huber needle, connected to an extension, three-way valve and two syringes (one empty and one containing agitated sterile 0.9% saline), was inserted into the subcutaneous port and 1–2 ml of urine was collected for culture. One milliliter of agitated saline was then injected into the port with the ultrasound probe over the kidney. Patency was established by seeing saline microbubbles fill the pelvis. The 1 ml of urine was then aspirated and the procedure repeated this time with the ultrasound probe over the bladder to ensure patency of the cystotomy tube. Perioperative complications were recorded and classified as obstructive or non-obstructive. This was established depending on the patency of the SUB device when flushed with saline during ultrasound follow-up. If a second surgery had to be performed owing to obstruction, this was recorded as t0, and the same classification for postoperative ultrasound examinations was respected. In the case of bilateral obstruction and bilateral SUB placement, kidneys were considered independent to each other and parameters were analysed separately. Pelvic dilation and ultrasound parameters were recorded separately.
SUB placement
A standard commercial SUB kit (SUB 100IK kit; Norfolk Vet Products) was placed in all cats of this study. This kit is specifically designed for cats and consists of a 6.5 Fr locking loop catheter inserted into the caudal pole of the renal pelvis and a 7 Fr bladder catheter both connected to a subcutaneous shunting port.
The surgical placement of the SUB was performed through a ventral midline laparotomy, as previously described. 13 An 18 G intravenous catheter was advanced through the caudal pole of the renal pelvis until urine was obtained; a sample was collected for culture. Diluted 50/50 iodinated contrast was injected through the catheter into the renal pelvis under intraoperative fluoroscopy. A hydrophilic angled guidewire (Infiniti Medical) was placed through the catheter and looped in the renal pelvis or directed into the ureter. The catheter was removed and the nephrostomy tube was advanced over the wire into the renal pelvis under fluoroscopic guidance. Once in place, the guidewire was removed, the loop of the nephrostomy tube was made by securing the string, and the tube and disk were glued to the caudal pole of the kidney with sterile cyanoacrylate glue. A purse-string suture was placed at the bladder apex, a stab incision using an #11 blade was made at the center of the purse string and the 7 Fr cystotomy catheter with a hollow trocar was advanced into the bladder. Four additional simple interrupted sutures were placed full thickness through the bladder wall and the Dacron disk. Sterile cyanoacrylate glue was also used to secure the catheter. Stab incisions through the abdominal wall on the side of the obstruction allowed passage of the nephrostomy tube caudally and cystotomy tube cranially. The tubes were connected to the port placed in a small subcutaneous pocket and the port was sutured to the abdominal wall. The entire system was flushed with iodinated contrast diluted 50/50 under fluoroscopic guidance to ensure patency of the system and identify any leaks.
Images analysis
Ultrasound examination was performed by board-certified radiologists. Images were acquired with two different devices (ATL HDI 5000 [Phillips Medical] and Aplio 400 [Toshiba Medical]) used at the University of Montreal between 2010 and 2015. All cats were positioned in dorsal recumbency, the hair was clipped from the ventral aspect of the abdomen, and scanning was performed with 5–8 MHz curvilinear or 5–11 MHz linear transducers with the aid of acoustic gel. Images were retrospectively reviewed by two board-certified radiologists (PP and SS) and analyzed concurrently with the original report.
Pelvic distension was measured according to the method described by D’Anjou et al (Figure 1a). 12 Multiple qualitative parameters were also recorded. The presence and appearance (echogenicity) of free fluid in the retroperitoneal and peritoneal spaces were recorded. The echogenicity of the adipose tissue surrounding the kidney (Figure 1b), the ureter and the urinary bladder were noted. The echogenicity of the adipose tissue surrounding the kidney, especially at the caudal pole, surrounding the ureter and the urinary bladder were noted. The appearance, orientation and position of the access port were recorded. The appearance of the Dacron nephrostomy tube disk at the level of the caudal pole of the kidney, as well as the site where the tube passed through the abdominal wall, were evaluated (presence of free fluid, thickening of the wall, hyperechoic adipose tissue). The appearance and path of the bladder and kidney catheters (visualization of two continuous echogenic double parallel lines) were recorded. Ureteral wall thickness, appearance (smooth or irregular), its degree of distension (entire ureteral diameter measured in a transverse plan) and content (anechoic, echogenic, mineralized) were recorded. The subcutaneous tissues surrounding the access port were evaluated for their echogenicity (Figure 1c).

(a) Ultrasonographic image of a feline kidney in a transverse section with a preoperative pelvic distension of 8 mm. (b) Ultrasonographic image of a feline kidney in a longitudinal section 2 months after subcutaneous ureteral bypass placement with a pelvic distension of 1.7 mm. Note the nephrostomy catheter (white arrowhead) and the perirenal steatitis (black arrowhead) at the caudal pole of the kidney. (c) Ultrasonographic image of a subcutaneous port (P). Note the adjacent subcutaneous steatitis (black arrowheads)
For cats that underwent multiple abdominal ultrasound examinations prior to surgery, the images immediately prior to surgery were reviewed. When several ultrasound examinations were available in the same follow-up time period (short or long term), mean pelvic size was calculated, whereas the last ultrasound examination was used for qualitative criteria. A binary coding of qualitative parameters (0 or 1) was assigned for statistical purposes (Table 1).
Binary code was attributed to the qualitative ultrasound parameters (0 or 1) observed in the various time periods

RP = retroperitoneal space; P = peritoneal space
Statistical analysis
Outcomes of interest included age, body weight, sex, cause of the ureteral obstruction, pelvic size and the previously described qualitative ultrasound parameters. A repeated-measures linear model was used to assess pelvic dilation over time (pre-t0, short term and long term) followed by Tukey’s post-hoc tests to compare pairs of means. For the analysis of creatinine values, a repeated-measures linear model on the log10 transformed data was used with time as a within-subject factor and type of SUB device (unilateral or bilateral) as a between-subject factor. This was followed by a priori contrasts to compare pairs of means using the Benjamini–Hochberg sequential procedure to adjust the alpha level downward. A linear mixed model with the individual as a random effect was used to evaluate the linear relationship between log10-transformed renal pelvic size and creatinine values over all time points. McNemar’s test was used to compare the binary-coded quantitative parameters of each group with each other. The level of statistical significance was set at 0.05 throughout.
Results
Cats
Twenty-seven cats met the inclusion criteria. Thirty-three kidneys were recorded, as six cats had bilateral ureteral obstruction with bilateral SUB placement. In cats with unilateral obstruction, the left ureter was obstructed in 10 cats and the right in 11 cats. The population consisted of neutered males (n = 8) and spayed females (n = 19). Breeds represented included domestic shorthair (n = 19), Siamese (n = 3), Burmese (n = 2), Persian (n = 1), Tonkinese (n = 1) and domestic longhair (n = 1). The average age at the time of ureteral obstruction was 7.4 years (range 3–17 years) and the average body weight was 5.0 kg (range 2.3–10 kg). In total, 90 ultrasound examinations were reviewed.
Postoperative ultrasound follow-up
An average number of 2.2 ultrasound examinations were recorded for each cat postoperatively. Twenty-four cats (30 kidneys) had at least one ultrasound examination in the short-term period. Thirteen cats (15 kidneys) had at least one ultrasound examination in the long-term period. Ten cats (12 kidneys) had at least one ultrasound examination in the short- and long-term periods. Fourteen cats (18 kidneys) had at least one ultrasound examination in the short-term period with no long-term evaluation and three cats (three kidneys) had at least one ultrasound examination in the long-term period with no short-term evaluation. The mean postoperative time to the short-term examination was 1.6 months. The mean postoperative time to the long-term examination was 8.8 months.
Creatinine values
The mean ± SD creatinine value among the entire cat population was 438 ± 261 µmol/l (range 145–1035 µmol/l, n = 26 cats) at pre-t0. In the short- and long-term periods, the mean ± SD creatinine value was 236 ± 120 µmol/l (range 112–730 µmol/l, n = 26 cats) and 210 ± 42 µmol/l (range 112–304 µmol/l, n = 21 cats), respectively. Among cats with unilateral obstruction and unilateral SUB device, the mean ± SD creatinine value was 406 ± 270 µmol/l (range 145–1035 µmol/l, n = 20 cats) at pre-t0. In the short- and long-term periods, mean ± SD creatinine values were 209 ± 63 µmol/l (range 112–351 µmol/l, n = 20 cats) and 202 ± 39 µmol/l (range 112–267 µmol/l, n = 17 cats), respectively. Among cats with bilateral obstruction and a bilateral SUB device, the mean ± SD creatinine value was 545 ± 219 µmol/l (range 337–868 µmol/l, n= 6 cats) at pre-t0. In the short- and long-term periods, the mean ± SD creatinine values were 328 ± 209 µmol/l (range 176–730 µmol/l, n = 6 cats) and 243 ± 46 µmol/l (range 198–304 µmol/l, n = 4 cats), respectively. These results are summarized in Table 2. Appendix 1 (supplementary material) shows the creatinine value pre-t0 for each of the 27 cats, the average creatinine value over the short-term follow-up and the average creatinine value over the long-term follow-up.
Creatinine values (µmol/l)

Data are mean ± SD
SUB = subcutaneous ureteral bypass; Pre-t0 = before surgery
Among all cats treated with a SUB device (including unilateral and bilateral SUB devices) that had no obstructive complication over the follow-up period, the creatinine value at pre-t0 was significantly higher compared with the short- (P = 0.0009) and long-term (P = 0.0004) mean values. However, no significant difference was present between the short- and long-term mean values (P = 0.50). Among cats treated with a unilateral SUB device that had no obstructive complication, the creatinine value at pre-t0 was significantly higher compared with the short- (P <0.0001) and long-term (P <0.0001) mean values. However, no significant difference was present between the short- and long-term mean values (P = 0.92). No significant difference was present between cats treated with unilateral or bilateral SUB devices, for any time period.
Ultrasound appearance of the urinary tract and SUBs
The echogenicity of the urine was the only qualitative parameter with statistical significance between the two time periods. The urine in the bladder was anechoic in 48% cats preoperatively and in 20.8% during the short-term period (P = 0.025). Therefore, 79.2% of the cats had urine either echogenic or with mineralized content in the postoperative short-term period. No other qualitative ultrasound parameter showed statistical significance throughout the time periods. Throughout the three time periods, a retroperitoneal or peritoneal effusion was rarely seen. It was present in <16% of cats. Loss of corticomedullary distinction was noted in >70% of cases throughout all three time periods. Perirenal fat was noted as hyperechoic in 70.5% at pre-t0 and in 51.8% in the short-term and 42.8% in the long term. The fat surrounding the subcutaneous port was hyperechoic in 18.5% of cats in the short term but was not recorded in the long term. The bladder wall was considered normal in 87.5% of cats at pre-t0. Thickening or irregular aspect of the bladder wall was noted in 25% of cats in the short-term and in 22.2% of cats in the long term. The qualitative parameters are summarized in Table 3.
Summary of the qualitative parameters at the various time intervals in the scanned population

OC = obstructive complication; bc = binary code; RP = retroperitoneal space; P = peritoneal space; Pre-t0 = before surgery
Size of the renal pelvis
Mean ± SD pelvis size on the obstructed side was 11.7 ± 8.4 mm (range 0.9–41 mm, n = 33 kidneys) at pre-t0. In the short- and long-term periods, mean ± SD pelvis size was 2.4 ± 1.8 mm (range 0–7.0 mm, n = 30 kidneys) and 1.7 ± 0.9 mm (range 0–3.5 mm, n = 15 kidneys), respectively. When mean values and 95% confidence intervals (CIs) were evaluated for the scanned population, there was a distinct difference over various time intervals (Figure 2). A significant effect of time on this value was observed (P <0.0001). A significant difference was found between renal pelvis size at pre-t0 and the short term (P <0.0001), as well as between pre-t0 and the long term (P <0.0001). However, no significant difference was present between the short- and long-term mean values. Among the cats without obstructive complication, 93% of kidneys had a pelvic size ⩽5 mm in the short term and all of them had a pelvic size ⩽3.5 mm in the long term. Appendix 2 (supplementary material) shows the pelvic size pre-t0 for each of the 27 cats, the average pelvic size over the short-term follow-up and the average pelvic size over the long-term follow-up.
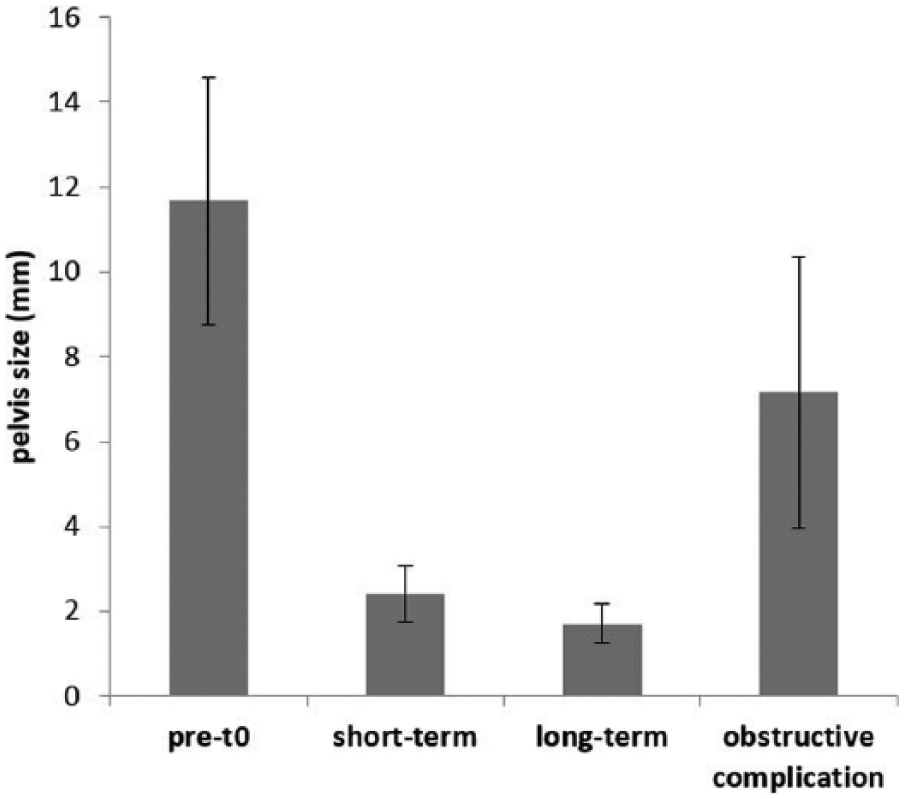
Mean pelvic size at the various time intervals in the scanned population. Error bars represent 95% confidence intervals of the mean. pre-t0 = before surgery
Relationship between the renal pelvis size and creatinine
When an individual was assessed as a random effect, a positive and statistically significant relationship (P = 0.002) was noted between the renal pelvic size and creatinine value.
Complications/concurrent disease
Eleven cats (40.7%) did not have any complications during the postoperative follow-up.
Nine cats (33.3%) had non-obstructive complications, including hematuria (n = 3), urinary tract infection (n = 3), uremic gastritis (n = 1), inflammatory reaction around the port (n = 1) and multicentric lymphoma (n = 1). Urinary tract infection was diagnosed based on a positive urine culture obtained from the SUB port during a routine flush procedure. Uremic gastritis was suspected in one cat based on the occurrence of vomiting, azotemia and an hyperechoic thickened gastric wall on ultrasound examination. The diagnosis of multicentric lymphoma was based on histopathological analysis following necropsy of the cat. The mean time to a non-obstructive complication was 6.4 months (range 0.5–21 months).
Eight cats (29.6%) had an obstructive complication. This was evidenced by non-patency (inability to see microbubbles and drain either the renal pelvis, bladder or both) of the SUB device during the ultrasound-guided flush procedure. The mean time between the detection of an obstruction and the previous normal ultrasound examination was 2.6 months. In cats without obstruction, the mean time between ultrasound examinations was 2.1 months. There was no statistical difference between these two means. Five of the cats with an obstructive complication had a kinked catheter, and successful surgical repositioning or replacement of the device was undertaken. The three remaining cats had, respectively, a partial SUB obstruction of unknown origin (suspected mineral debris, n = 2) and suspected renal carcinoma with secondary carcinomatosis (n = 1). One of the partial SUB obstructions spontaneously resolved 6 months after diagnosis; the other was euthanized because of progressive azotemia but had no ultrasound examination follow-up available. The suspicion of renal carcinoma with secondary carcinomatosis in the third cat was based on ultrasound evaluation 2 months after the placement of the SUB device. Humane euthanasia was elected and necropsy was declined by the owners.
The mean time to appearance of an obstructive complication was 3.7 months. At the time of obstruction, the mean ± SD creatinine value was 283 ± 97 µmol/l (range 176–407 µmol/l, n = four cats). The mean ± SD pelvic size was 7.2 ± 3.6 mm (range 4–15 mm, n = six kidneys). When mean value and 95% CIs were evaluated for the cats with obstructive complication, there was a distinct difference with short- and long-term intervals (Figure 2). Two of the five catheter replacements or repositionings were performed during the immediate or short-term postoperative period (2 and 15 days post-t0, respectively). Three of the five catheter replacements or repositionings were performed during the long-term period (mean time period of 8.7 months post-t0, 4–18 months). The renal pelvic measurements for the five obstructed cats are presented in Appendix 2 (supplementary material).
Discussion
When assessed over the long term, the renal pelvis measured ⩽3.5 mm in the absence of an obstructive complication in all of our cats. In the short term, 93% of kidneys from cats without an obstructive complication had a renal pelvis that measured ⩽5 mm. Whether in the short- or long-term periods, free fluid was rarely noticed and not associated with a complication, although a larger number of cases would be necessary to evaluate this further. The decrease over time in hyperechogenicity of the perirenal and periureteral adipose tissue was not statistically significant and likely reflected decreased perioperative inflammation over time.
The creatinine value of cats treated with a SUB device decreased postoperatively compared with preoperative values. This is consistent with previous reports.5,7 When separately assessed, there was no significant difference between the creatinine values of cats treated with bilateral SUB devices over time. No significant difference was noted between cats treated with unilateral or bilateral SUB devices. It is difficult to draw conclusions given the low number of cats treated with bilateral SUB devices in our population (n = 6). The wide range of individual values presented in Appendix 1 (supplementary material) is likely the result of dehydration, variable subcutaneous or intravenous fluid administration but, most importantly, varying severity of the underlying renal disease.
The mean preoperative renal pelvic measurement in cats with ureteral obstruction was 11.7 mm. This is similar to values reported by previous retrospective study with a mean value of 10.9 mm in obstructive cats. 12 Another retrospective study reported that complete resolution of obstructive pyelectasia should not be expected in cats, with renal pelvic measurements of 2–7 mm being expected postoperatively. 5 A similar observation was also published based on nine cats whose ureteral obstruction was treated with the placement of ureteral stents. 14 In a recent paper, 15 6/13 dogs stented with double pigtail ureteral stents for obstructive pyonephrosis were ultrasonographically followed at short- (14–30 days after the procedure) and long-term (>30 days) intervals. The authors noted a median postoperative pelvic transverse measurement of 6.5 mm and considered this to be minimal pelvic dilation and successful pelvic decompression. Such values have yet to be published in cats. As the duration of the ureteral obstruction could not be determined, no correlation can be made here regarding the length of obstruction and pelvic size pre- and postoperatively.
Renal pelvic size in cats with normal renal function has been reported. In a previous study, 12 a group of 10 cats with normal renal function had a mean pelvic size of 1.8 ± 0.8 mm. This study also reported a mean ± SD pelvic size of 2.3 ± 0.9 mm in cats with normal renal function and evidence of diuresis. In our study, pelvic sizes close to normal were observed in many cats within a short postoperative time period and in the majority of cats during the long-term period. The mean ± SD postoperative renal pelvic measurement in cats with ureteral obstruction at the short- and long-term follow-ups were 2.4 ± 1.8 and 1.7 ± 0.9 mm, respectively. These values were both significantly lower than preoperative values. Our results show that the distension of the renal pelvic is at least partially reversible when ureteral obstruction is relieved. A positive correlation between renal pelvic size and the creatinine values were observed. This may be explained by a greater degree of obstruction causing a greater renal pelvic distension and decreased function in that kidney. Successful treatment of feline ureteral obstruction with a SUB device is thus associated with decreased postoperative azotemia and pelvic renal size. Postoperative renal pelvic measurements in our study showed that a dilated pelvis in cats may decrease considerably following decompression.
In women, one pregnancy in 500 is complicated by urinary calculi. Clinical manifestations secondary to ureteral obstruction caused by stones mostly occur during the second trimester. Ultrasound is the imaging modality of choice and severe hydronephrosis (>2 cm) is often observed. 16 Conservative medical treatment is often sufficient, but a double pigtail stent may be placed. The renal pelvis returns to normal size within 6–12 weeks following medical or interventional management.
Although this study does not provide an absolute pelvic size confirming obstruction, a pelvic size >5 mm following SUB placement could suggest an obstructive complication. If an obstruction is suspected, ultrasound-guided saline flush or contrast flush under fluoroscopic guidance can be used to confirm and identify the site of the obstruction. Following pelvic size over time in an individual cat can also aid in the early identification of an obstruction. One of our cases illustrates this well as an obstruction was diagnosed with a renal pelvic measuring 4 mm. During each of the previous follow-up ultrasound examinations, the pelvis measured 1 mm. It was not possible from our data to determine whether SUB flush frequency had an effect on the incidence of obstruction.
Ureteral measurements may be important in the assessment of obstructive complications and may be as or more important than renal pelvic size. The authors have observed mildly dilated renal pelvises with markedly distended ureters in ureterally obstructed cats. However, this was a recent observation in our practice. It was still unknown at the time most of the ultrasound examinations were performed. Therefore, only a few ultrasound evaluations had ureteral images available for retrospective evaluation. Since then, we routinely evaluate ureters in cats treated with SUB devices. In our clinical experience, some cats with concurrent ureteral obstruction and chronic kidney disease do not show marked pelvic distension despite severe ureteral obstruction. 11 Capsular and parenchymal fibrosis may explain the lack of distension in these cats.
Among our cats with obstructive complications, one was suspected of having a renal carcinoma with secondary carcinomatosis 2 months following placement of the SUB device. At the time of the pre-t0 ultrasound evaluation, there was no suspicion of any underlying renal neoplastic disease, even on retrospective review of available images. A subcutaneous fluid port associated with a poorly differentiated soft tissue sarcoma has recently been reported in a cat. 17 In this case report, a 20-year-old male castrated domestic longhair was evaluated for assessment of chronic kidney disease diagnosed 10 years prior. A non-healing ulcerated mass was noted at the site of a GIF tube, which was previously placed over a 5-year period. Histopathological analysis following necropsy revealed a fibrosarcoma. To our knowledge, a renal neoplasia associated with the SUB device has not been reported. Although this possibility was not ruled out, it was considered less likely by the authors, given the short period of time between the two ultrasounds. We believe it is likely that renal changes were too minor to be detected on preoperative ultrasound evaluation and therefore missed.
This study outlines ultrasound as an effective tool for the evaluation of renal pelvic distension. In cats treated with a SUB device, pelvic size postoperatively is often close to previously reported pelvic sizes in cats with normal renal function. Therefore, the detection of an increased pelvic size should alert the clinician to the risk of obstruction and the need for an ultrasound-guided flush. Ultrasound allows assessment of patency of the SUB device by visualization of saline microbubbles filling the renal pelvis and the urinary bladder during a flush procedure.
This study has a number of limitations, many related to its retrospective nature. Retrospective reviewing of the still images by another radiologist may have led to differing descriptive interpretations. The ureter has not been consistently assessed in most cats. A relatively small number of cats met the inclusion criteria. Inter-operator variability likely affected ultrasound measurements of the renal pelvis. The number of examinations and examination frequency was not standardized. Available long-term follow-up evaluations were limited in our population. Fourteen cats (18 kidneys) had no long-term ultrasound follow-up. Although a small range of pelvic sizes (0–3.5 mm) was noted during long-term evaluation, these values were recorded in only half of our total population (13/27 cats, 15/33 kidneys). This may explain, in part, the loss of power and the lack of statistically significant results, especially for the qualitative criteria. Given its retrospective nature, duration of obstruction and renal function prior to presentation for a ureteral obstruction was not available in the majority of cats.
Conclusions
Ultrasonography appears to be an effective tool in the monitoring of cats following SUB placement. Ultrasound allows for both qualitative and quantitative assessment of the urinary tract, which may help detect complications. Our study revealed that pelvic distension secondary to ureteral obstruction is at least partially reversible following decompression with a SUB. Our study also showed that ultrasound is an important tool in the detection of obstructive complications, and a renal pelvic width of ⩽3.5 mm 3 months postoperatively is expected in cats following SUB placement.
Supplemental Material
Click here for Supplementary Appendix 1
Creatinine values (μmol/l)
Supplemental Material
Click here for Supplementary Appendix 2
Pelvis size (mm)
Footnotes
Acknowledgements
The authors thank Dr Guy Beauchamp for assistance with statistical analysis.
Author note
Presented in abstract form at the The Annual European Veterinary Diagnostic Imaging Meeting, Wroclaw, Poland, August–September 2016.
Supplementary material
Appendix 1. Creatinine values (µmol/l)
Appendix 2. Pelvis size (mm)
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
