Abstract
Objectives
The objectives were to evaluate the pharmacokinetics (PK) of subcutaneous (SC) and intravenous (IV) dolasetron and the pharmacodynamics (PD) of SC dolasetron in healthy cats.
Methods
Five cats with unremarkable complete blood count, serum biochemistry and urinalyses were utilized. In the PK study, cats received 0.8 mg/kg SC and IV dolasetron in a crossover format. Serum samples were obtained via a jugular catheter at 0, 0.25, 0.5, 1, 2, 4, 8, 12, 24, 36 and 48 h after the administration of dolasetron. Dolasetron and the active metabolite hydrodolasetron were measured using liquid chromatography/tandem mass spectrometry. Non-compartmental PK analysis was performed. In the PD study, SC dolasetron (0.8 mg/kg and 1.0 mg/kg) and saline were administered 30 mins prior to administration of 0.44 mg/kg intramuscular xylazine in a randomized three-way crossover. Number of emetic events, lip licks, time to onset of emesis and visual nausea score were scored by a blinded observer.
Results
In the PK study, dolasetron was quickly metabolized to the active metabolite hydrodolasetron, limiting assessment of dolasetron PK parameters. Median (range) PK parameters for IV hydrodolasetron were as follows: maximum serum concentration (Cmax) 116 ng/ml (69–316 ng/ml), time to maximum concentration (Tmax) 0.5 h (0.3–0.5 h), half-life 3.3 h (2.9–7.2 h) and area under the curve until the last measurable concentration (AUClast) 323 h/ng/ml (138–454 h/ng/ml). Median (range) PK parameters for SC hydrodolasetron were as follows: Cmax 67.9 ng/ml (60.4–117 ng/ml), Tmax 0.5 h (0.5–1.0 h), half-life 3.8 h (2.9–5.3 h) and AUClast 437 h/ng/ml (221.5–621.8 h/ng/ml). There was no significant difference in exposure to hydrodolasetron between the routes of administration. With regard to PD, when dolasetron was administered prior to xylazine, there was no significant difference in the mean number of emetic events, lip licks, time to onset of emesis or visual nausea score when compared with saline.
Conclusions and relevance
Administration of 0.8 mg/kg dolasetron does not maintain serum concentrations of active metabolite for 24 h. Administration of dolasetron at 0.8 mg/kg and 1 mg/kg did not prevent xylazine-induced vomiting. Additional feline dose studies are needed to determine if a higher dose is efficacious.
Introduction
Dolasetron is a 5-hydroxytryptamine type 3 (5-HT3) receptor antagonist that blocks serotonin-induced nausea and emesis in both the gastrointestinal tract and central nervous system. 1 Dolasetron was developed to treat chemotherapy-induced emesis in humans and is also recommended as an antiemetic in cats. 2 Empirical dosing in cats has been previously described as 0.6–1.0 mg/kg intravenously (IV) once daily but pharmacokinetic (PK) or pharmacodynamic (PD) studies have not been performed to confirm the recommended dosage.2,3 The mechanism of action and indications for use of dolasetron are similar to ondansetron. In feline patients dolasetron may hold the additional benefit of a longer half-life and decreased dosing intervals over ondansetron, which has an average half-life of 1.8 h IV, 1.2 h orally and 3.2 h subcutaneously (SC), and requires dosing every 6–8 h. 4
Hydrodolasetron, the predominant active metabolite of dolasetron, has an affinity for the 5-HT3 receptor that is 50-fold greater than that of dolasetron, ensuring tight binding to antagonize this nausea and emesis-triggering receptor. 5 Renal excretion is the primary route of elimination for dolasetron and hydrodolasetron. However, in human patients with renal impairment dose reduction is not recommended.6,7 Furthermore, the safety profile of this 5-HT3 antagonist is considered excellent in humans with adverse effects (headache, constipation or diarrhea in humans) being mild and transient.1,3 Anecdotally, no significant side effects have been reported in feline patients. 3 The goal of this study was to gain additional information about the PK and PD of IV and SC administration of dolasetron to determine which route and dose would be the most effective in feline patients.
Materials and methods
PK study
Five healthy research cats with unremarkable physical examination, complete blood count, chemistry and urinalysis were used. All portions of the PK and PD project were approved by the Institutional Care and Use Committee at Colorado State University. For all PK studies a jugular catheter was placed under ketamine/butorphanol sedation (20 mg ketamine/cat IV, 0.1 mg/kg butorphanol IV) 16 h prior to study initiation for ease of sample collection. Cats received 0.8 mg/kg IV and 0.8 mg/kg SC dolasetron in a crossover manner with 6 weeks between crossover. Serum samples (from 1.5 ml whole blood) were obtained via jugular catheter using a three-syringe sample technique at 0, 0.25, 0.5, 1, 2, 4, 8, 12, 24, 36 and 48 h after administration of dolasetron. Serum was collected by centrifugation and stored at −80ºC until analysis of dolasetron and its active metabolite, hydrodolasetron, serum concentrations using liquid chromatography/tandem mass spectrometry (LC/MS/MS).
PD study
This was a randomized, double-blinded, saline-controlled crossover study for which the same five healthy research cats were used. Each cat received a total of three treatments in a pre-randomized order: SC saline (used as placebo), SC dolasetron at 0.8 mg/kg and SC dolasetron at 1.0 mg/kg with at least 1 week between crossover. Treatments were administered 30 mins prior to administration of 0.44 mg/kg intramuscular (IM) xylazine.
The number of separate productive emetic events, number of retches (unproductive), lip licks, drooling, time to onset of emesis and visual nausea score were scored by a single, blinded observer from the time of xylazine administration to the time of recumbency. Visual nausea score was a subjective, non-validated assessment based on body posture, ear position and facial expression, lethargy/stillness, restlessness, exaggerated swallowing, salivation and lip licking (0 = no nausea, 1 = mild nausea, 2 = moderate nausea and 3 = most severe nausea possible).
After sedation to the point of recumbency occurred, cats were reversed with 200–300 μg/cat atipamezole IM. Maropitant was planned as a rescue drug for any cats in which intractable emesis or further signs of overt nausea were noted, but this was not needed.
Statistical analysis of nausea score, emetic events, number of lip licks and time to onset of emesis were measured using a one-way ANOVA non-parametric test (Friedman) with Dunn’s post-hoc analysis in GraphPad Prism version 7 software.
PK analysis materials
Dolasetron mesylate hydrate and an internal standard, ondansetron HCl, were obtained from Sigma. Hydrodolasetron was obtained from Toronto Research Chemicals. All other chemicals and solvents used in the analysis were of reagent or higher quality and obtained from Fisher Scientific. Initial stocks (1 mg/ml) were prepared in dimethyl sulfoxide and subsequent dilutions were prepared in 50:50 acetonitrile (ACN):Milli-Q water.
Sample extraction
Blank serum was collected from cats of similar age and used to make a standard curve ranging from 1–1000 ng/ml for both dolasetron and hydrodolasetron (5 µl of each appropriate standard). Serum samples or blank serum (50 µl) were spiked with 5 µl internal standard (ondansetron at 250 ng/ml). Samples were prepared for analysis by protein precipitation by adding 50 µl of ACN containing 0.1% formic acid followed by vortexing for 10 mins, then centrifuged at 14,000 × g for 10 mins. Supernatants were transferred to high-performance liquid chromatography (HPLC) vials with inserts and analyzed by LC/MS/MS.
Mass spectrometry
Positive ion electrospray ionization mass spectra were obtained with a MDS Sciex 3200 Q-TRAP triple quadrupole mass spectrometer (Applied Biosystems) with a turbo ionspray source interfaced to an Agilent 1200 Series Binary Pump SL HPLC system. Samples were chromatographed with a Sunfire C8, 5 µm (4.6 × 50 mm column; Waters) protected by a C18 guard cartridge (4.0 × 2.0 mm; Phenomenex). An LC gradient was employed with mobile phase A consisting of 0.1% formic acid in Milli-Q water and mobile phase B consisting of 50:50 ACN:methanol with 0.1% formic acid. Chromatographic resolution was achieved by increasing mobile phase B linearly from 2% to 85% from 1 to 1.5 mins, maintaining at 85% from 1.5 to 2.75 mins, decreasing linearly from 85% to 2% from 2.75 to 4 mins, followed by re-equilibration of the column at 2% mobile phase B from 4 to 5 mins. The LC flow rate was 1 ml/min, the sample injection volume was 20 µl and the analysis run time was 5 mins.
The mass spectrometer settings for dolasetron, hydrodolasetron and ondansetron, respectively, were optimized as follows: turbo ion spray temperature, 550°C; ion spray voltage, 5500 V; declustering potential, 55, 60 and 39 V; entrance potential, 4.9, 5.1 and 8.3 V; collision energy, 37, 41 and 36 V; collision cell entrance potential, 13, 13 and 22 V; collision cell exit potential, 1.5, 2.4 and 2.2 V; curtain gas (N2), 10 units; collision gas (N2), 2; nebulizer gas (N2), 40 units; and auxiliary gas (N2), 60 units. The predominant product ion for dolasetron was m/z 164.3, for hydrodolasetron it was m/z 166.4 and for ondansetron it was m/z 170.2. Samples were quantified in the MRM mode monitoring ion transitions m/z 325.2 → 164.3 for dolasetron, m/z 327.2 → 166.4 for hydrodolasetron and m/z 294.3 → 170.2 for the internal standard, ondansetron.4,8 The dwell times for each ion transition were 500 ms. Q1 and Q3 were both operated in unit resolution mode.
Data analysis
Quantitation of dolasetron and hydrodolasetron was based on linear standard curves in spiked blank serum and 1/x2 weighting of linear regression. Parameters for the assessment of assay performance were calculated as follows:
The average accuracy and precision, respectively, between analysis batches (n = 3) were 95.9% and 4% for dolasetron, and 98.2% and 6% for hydrodolasetron. Accuracy and precision measurements were within the minimum accuracy (85%) and precision (± 20%) ranges for acceptable analytical method validation. 9 For the current analyses the lower limit of quantitation based on analytical method validation guidelines was 0.75 ng/ml. Non-compartmental and compartmental modeling was applied for the calculation of PK parameters using Phoenix WinNonlin software, version 6.3 (Certara). PK parameters half-life and area under the curve until the last measurable concentration (AUClast) for the active metabolite hydrodolasetron were compared between IV and SC routes of administration using Wilcoxon sign rank test in GraphPad Prism version 7.
Results
PK
Dolasetron was administered at 0.8 mg/kg IV in five cats and 0.8 mg/kg SC in four cats as one cat’s jugular catheter malfunctioned before blood sampling could be performed. PK parameters for dolasetron and active metabolite hydrodolasetron after administration of 0.8 mg/kg dolasetron IV and SC are summarized in Table 1. Dolasetron was quickly metabolized to hydrodolasetron with only two cats for each administration route having measurable concentrations beyond 2 h, limiting the calculation of dolasetron PK parameters (Figure 1). There was no significant difference in half-life or exposure (AUClast) to the active metabolite between the two routes of administration (Figure 2). The median bioavailability of SC dolasetron was 37% (range 19–44%). The median bioavailability of SC hydrodolasetron was 149% (range 56–315%) and demonstrated significant variability.
Pharmacokinetic parameters for dolasetron and active metabolite hydrodolasetron after administration of 0.8 mg/kg dolasetron intravenously (IV) and subcutaneously (SC)

There was no significant difference in parameters between the two routes of administration when half-life and area under the curve until the last measurable concentration (AUClast) for the active metabolite hydrodolasetron were compared between the IV and SC routes of administration using Wilcoxon signed rank test in GraphPad Prism software
NP = not performed; Cmax = maximum plasma concentration; Tmax = time to maximum concentration; Tlast = time to last measurable serum concentration
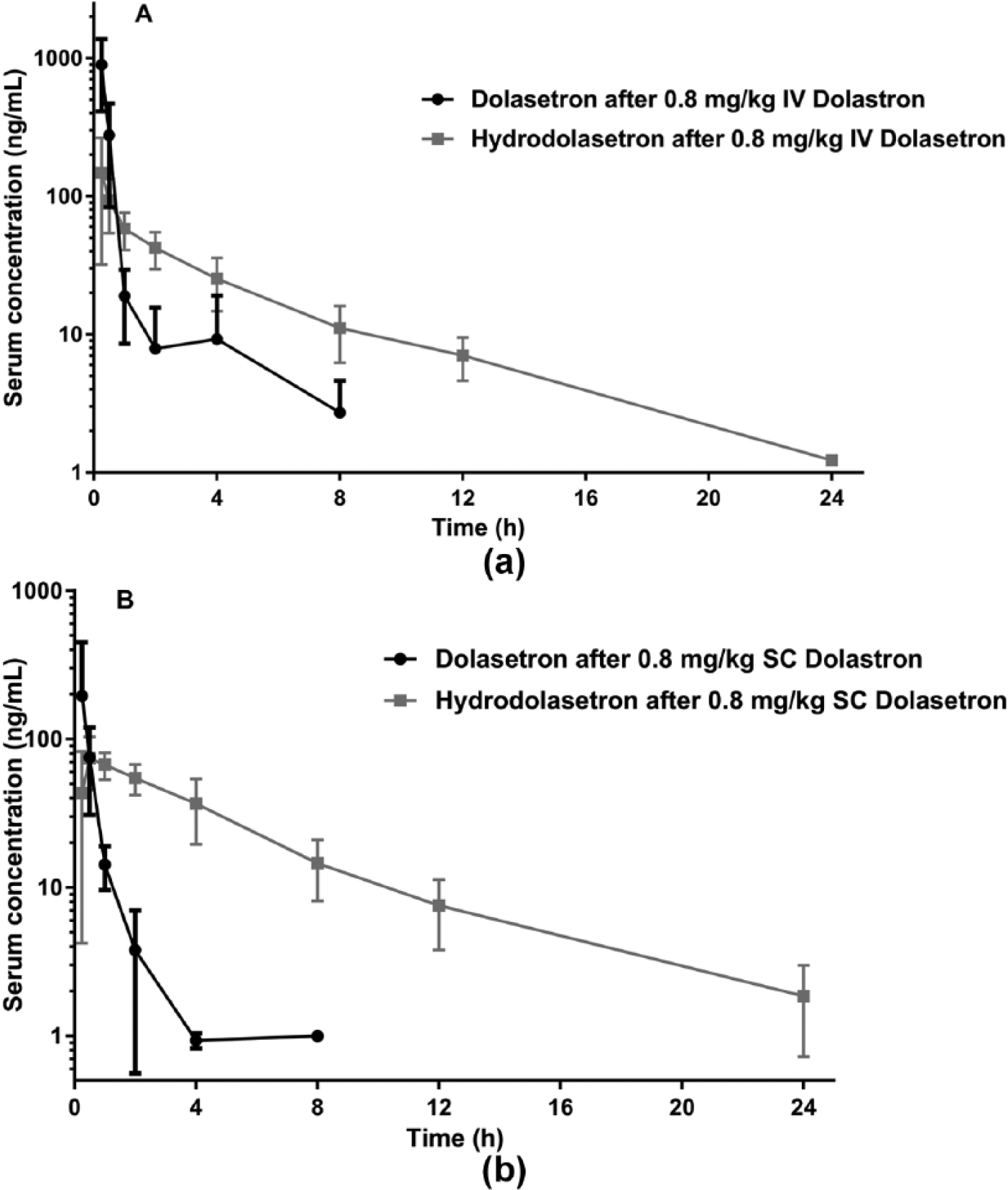
Drug serum concentration curves for dolasetron and active metabolite hydrodolasetron after administration of 0.8 mg/kg dolasetron (a) intravenously (IV) and (b) subcutaneously (SC). These figures are mean and SD

Serum hydrodolasetron concentrations following 0.8 mg/kg intravenous (IV) and subcutaneous (SC) dolasetron administration. There was no significant difference in metabolite exposure between the IV and SC route of administration. These figures are mean and SD
PD
When 0.8 mg/kg dolasetron, 1 mg/kg dolasetron and saline were administered SC 30 mins prior to 0.44 mg/kg xylazine IM in a randomized three-way crossover manner, there was no statistically significant difference in number of emetic events, time to onset of emesis or visual nausea score compared with saline (Figure 3). Median number of emetic events recorded for saline were 2 (range 1–2) emetic events vs 1 (range 0–2) event for 0.8 mg/kg SC and 1 (range 0–2) emetic event for 1.0 mg/kg SC. In one cat emesis was prevented with both doses of dolasetron vs two emetic events with saline; however, this cat still continued to display lip licking and moderate nausea on the visual nausea score. Only one cat experienced retching (during the saline administation); therefore, retching could not be compared between treatments. No drooling was observed at any time.

Pharmacodynamics of dolasetron after administration of saline, and 0.8 and 1 mg/kg subcutaneous dolasetron 30 mins prior to 0.44 mg/kg xylazine intramuscularly in a randomized three-way crossover. There was no statistically significant difference in the number of: (a) emetic events; (b) time to onset of emesis; (c) number of lip licks; or (d) visual nausea score compared with saline. Administration of dolasetron did prevent emesis in one cat. These figures are mean and SD
Discussion
The purpose of this study was to investigate the PK and PD of IV and SC administration of dolasetron to inform effective use in feline patients. After administration of 0.8 mg/kg dolasetron, the drug was quickly metabolized to the predominant active metabolite hydrodolasetron with only two cats in each administration route having measurable dolasetron serum concentrations beyond 2 h. This limited the assessment of dolasetron PK parameters, and is similar to results of studies in other species where dolasetron functions as a prodrug.10,11 In the present study no significant difference was found in hydrodolasetron drug exposure between IV and SC routes of administration; however, bioavailability of SC hydrodolasetron was extremely variable ranging from 56–315%. The suitability of once-daily dosing by either route is questionable considering only one cat had measurable serum hydrodolasetron concentrations 24 h after IV administration, and only 2/4 cats had measureable 24 h after SC administration. In all three cats these concentrations were near the lower limit of detection and likely clinically insignificant (1–2 ng/ml), assuming that serum concentration is correlated with clinical effect. This also assumes that hydrodolasetron is the active metabolite as it is in other species. In addition, the half-life of dolasetron in cats is shorter than it is in humans (4–8 h; Table 2), for whom daily dosing is recommended. 5
Comparison of pharmacokinetic parameters for hydrodolasetron across species (mean ± SD)

Cats appear to have a shorter half-life than humans, and relatively lower drug exposure (area under the curve) than dogs and humans
AUClast = area under the curve until the last measurable concentration
IV = intravenously; AUCinf = area under the curve extrapolated to infinity; Cmax = maximum serum concentration; Tmax = time to maximum concentration
Contrary to expectations, SC administration of dolasetron at 0.8 mg/kg and 1 mg/kg did not significantly reduce the mean number of xylazine-induced emetic events, number of lip licks, time to onset of emesis or nausea score when compared with saline. Although the PD study involved a small number of cats and statistical comparisons were likely underpowered, a more effective result on a per cat basis was anticipated. Possible explanations for why dolasetron failed to palliate xylazine-induced emesis include ineffective time of dose administration, inapplicable model of emesis and inadequate dose for cats. However, the first two are less likely given current information for cats. Ineffective time of dose administration (ie, 30 mins before xylazine administration) seems unlikely as the time of drug administration for the PD study was chosen based on the results of the PK study where the median Tmax was 30 mins. This is similar to Tmax in dogs and humans (Table 2) for whom dolasetron pretreatment is recommended 30 mins prior to chemotherapy or anesthesia administration.10,12 Previous work in cats demonstrated that the 5-HT3 drug ondansetron palliates xylazine-induced emesis (0.66 mg/kg IM) when given 1 h ahead of time at 0.2 0.4 and 0.8 mg/kg IM. 13
The xylazine-induced emesis model was chosen for this study in order to compare dolasetron to maropitant, which has been demonstrated to be effective in blocking emesis in this model. 14 In the maropitant study cats were administered maropitant at a dose of 1 mg/kg SC, IV and orally 2 h before xylazine, and a reduction in emetic events of 76.4%, 100% and 90%, respectively, was seen. 14 In comparison, in the previously discussed study on the effects of ondansetron on xylazine-induced emesis, it should be noted that emesis was not entirely prevented by administration of ondansetron and, even at the highest dose, emesis was reduced to a mean ± SEM of 2.67 ± 0.3 vs 5.3 ± 1.1 for saline. This represents a roughly 50% reduction in emetic events. 13 In the current study the administration of dolasetron resulted in approximately 30% reduction in emesis. If dolasetron is a drug that does not, in fact, act upon, or acts weakly upon, central emetic stimuli, then this is also important information as this would then not be a good drug choice for diseases mediated in the chemoreceptor trigger zone (ie, uremic vomiting). In a study in which ondansetron was compared with maropitant in dogs, ondansetron’s mechanism of action was described as more peripheral than central. 15 Similarly, maropitant and dolasetron would need to be compared in the same emesis model in cats once an appropriate dose of dolasetron is identified. Altogether, maropitant is possibly a better choice owing to its ability to act both peripherally and centrally, as it may be difficult in the clinical setting to distinguish whether emesis is centrally or peripherally mediated. 14
As administration of dolasetron prevented emesis in one cat in this study, the likelihood that a higher dose may be more effective should be explored. In humans 1.8 mg/kg is the most commonly reported effective dose. 11 When comparing PK data across species (Table 2) it could be argued that cats seem to achieve a relatively lower AUC than dogs or humans when dose comparison calculations are performed. Given the relatively strong safety profile for the drug in humans, and no anecdotal adverse event reports in cats, dose-escalation studies would be helpful to determine if better efficacy could be achieved.
Conclusions
The suitability of once-daily dosing of dolasetron by either the IV or SC route is questionable. Furthermore, SC administration of dolasetron at 0.8 mg/kg and 1 mg/kg did not adequately prevent xylazine-induced emesis. In addition, previous literature has suggested that a higher dose is needed for control of active emesis vs prophylaxis.3,11 As the majority of clinical use would typically be for control of active vomiting, overall it appears dolasetron is not an adequate antiemetic drug for use in feline patients at the dose used in this study. Additional dose studies in cats are needed to determine if a higher dose is efficacious.
Footnotes
Author note
Previously presented in abstract form at ACVIM Annual Forum 2016, Denver Colorado and AAFP Conference 2016 Washington, DC.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funded, in part, by the Young Investigator Grant program in the Center for Companion Animal Studies, Frankie’s Fund and the Angelo Fund for Feline Therapeutics. Drug analysis was carried out by the Pharmacology Shared Resource, supported by the University of Colorado Cancer Center Support Grant (P30 CA046934).


