Abstract
Objectives
The purpose of this study was to determine differences in normal feline pituitary dimensions in brachycephalic and mesocephalic cats as a basis for establishing cryohypophysectomy in cats.
Methods
Measurements were performed on sagittal T2-weighted and transverse post-gadolinium T1-weighted or T1 3D fast field echo-weighted MRI images. A total of 32 brachycephalic and 27 mesocephalic cats were examined. Inter-observer reproducibility was assessed by t-test and Bland–Altman analysis.
Results
The cats were 0.6–15.9 years of age with a body weight range of 1.84–6.60 kg. For brachycephalic cats, the mean pituitary gland sagittal height was 2.15 ± 0.15 mm, pituitary gland transverse height was 2.42 ± 0.21 mm, pituitary gland transverse width was 4.44 ± 0.27 mm and pituitary gland sagittal length was 3.14 ± 0.30 mm. In mesocephalic cats, the pituitary gland dimensions were 2.94 ± 0.16 mm, 3.09 ± 0.26 mm, 4.73 ± 0.31 mm and 4.88 ± 0.30 mm for pituitary gland sagittal height, transverse height, transverse width and sagittal length, respectively. There was a highly significant correlation between brachycephalic and mesocephalic cats and pituitary gland height and length (P <0.0001), respectively. Sex also had an effect on pituitary gland measurements. Neutering status had no significant effect on hypophyseal measurements. Age had a significant influence on pituitary gland height, width and length in the brachycephalic population. Inter-observer reproducibility was good to excellent.
Conclusions and relevance
The different pituitary measurements in brachycephalic and mesocephalic cats has to be considered if surgery comes into question. There are ranges in pituitary gland sizes, even among the mesocephalic cat population. Thus, exact measuring of the pituitary gland is crucial before any surgical intervention.
Introduction
Neoplastic changes of the pituitary gland produce various disorders in domestic cats, including acromegaly and Cushing’s syndrome or pituitary-dependent hyperadrenocorticism (PDH).1,2 Both hormonal diseases may lead to poorly regulated insulin-resistant diabetes mellitus.1,3,4 Medical treatment options exist for different pituitary gland-related diseases, but proper clinical trials are lacking.5–8 In cats with PDH, the most common treatment is adrenalectomy. 9 Even that is a symptomatic treatment modality, and does not consider the underlying problem of a primary pituitary gland tumour.
In human medicine, hypophysectomy is the treatment of choice for Cushing’s disease. 10 This surgical procedure has also been developed in veterinary medicine as a state-of-the-art therapy, 11 as medication and radiation therapy are not effective in all cases.1,5,7,12 This technique has a number of potential complications, 13 the most concerning of which is uncontrollable haemorrhage from damage to the cavernous sinus. 14 Therefore, the development of alternative methods may be warranted. Using a permanently cooled surgical cryoprobe placed in the centre of the tumour (cryohypophysectomy), 15 the neoplastic tissue could be destroyed while avoiding the risk of uncontrollable haemorrhage.16,17 Convection of cold around the cryoprobe tip continues until thermal equilibrium is reached. It is crucial for the surgeon to control the cryolesion, 15 to avoid damage to the hypothalamus. 18 Owing to the nature of the microsurgical approach, visualisation of the freezing front is not possible. Temperature gradient and impedance measuring devices require the invasive placement of thermocouples or electrodes,15,19 and ultrasonography can be used to visualise the spread of the ice front; 15 however, these methods are inappropriate for surgical interventions of the pituitary gland. Other options like CT- or MRI-controlled cryosurgical units can be used, but these are associated with high radiation exposure or exorbitant costs.19,20 Consequently, it seems more reasonable to determine the time required for the growing frozen mass to encompass the whole pituitary gland while sparing the hypothalamus. Therefore, it is important that the precise dimensions of the gland in cats be known with certainty.
Two studies have claimed an almost constant pituitary size in domestic shorthair cats.21,22 However, differences in skull morphology of different cat breeds should be taken into account, and these results should be questioned. In this study, the pituitary size of brachycephalic and mesocephalic cats was examined using MRI. The influence of age and body weight on the size of the pituitary gland was investigated, as well as the effect of sex and neutering status on hypophyseal dimensions.
Materials and methods
Brachycephaly and mesocephaly were defined as a qualitative characteristic according to the phenotypic appearance of the cats and breed specific skeletal features. To form the mesocephalic group, we retrospectively searched the medical records of the Justus Liebig University in Giessen to identify cats that had undergone MRI diagnostics of the brain without any evidence of pituitary gland disease. Only cats without signs of polyuria/polydipsia, diabetes mellitus or alopecia were accepted. Blood analysis and urinalysis were not available in these cats. In addition, no cats that had been pretreated with steroids or insulin were included. Presence of cardiomegaly or intracranial neoplasia led to exclusion from the study. Cats with incomplete MRI sequences were also excluded.
For the brachycephalic group, Persian cats were evaluated prospectively, considering the same exclusion criteria as for the mesocephalic group. Blood work and urinalysis were not performed in any of the cats. Detailed overview of the study groups is given in the supplementary table. MRI scans and anaesthesia were approved by the Ethics Committee of Animal Experiments of Justus Liebig University and the local Hessian government (reference number GI18/17-Nr.A20/2013). Anaesthesia was induced with diazepam (0.5 mg/kg IV Diazepam-Ratiopharm; Ratiopharm) and propofol (2–4 mg/kg IV Vetofol; Bayer Vital), and maintained by isoflurane (1.5–2.0% Isofluran CP; CP-Pharma) after intubation.
Imaging
The MRI studies were performed with a 1.0 Tesla Gyroscan NT Philips Medical System with NT-Intera 1.0T Standard R11 software (MR Systems Intera Release 11.1.4.6 2006-03-10) using a Philips C4 surface coil. The pituitary gland was measured from sagittal and transverse MRI images. All images were evaluated retrospectively, and were acquired at the Clinic for Small Animal Surgery at the Department of Veterinary Clinical Sciences of Justus Liebig University in Giessen, Germany. In the brachycephalic population, the sagittal T2 turbo spin echo (TSE)-weighted and transverse T1 3D fast field echo-weighted images were evaluated (Figure 1a,b). For the mesocephalic cats, T2 TSE-weighted sagittal and T1 SE-weighted transverse post-contrast images were available (Figure 2a,b). In these studies, gadopentate dimeglumine (0.5 mmol/ml) was intravenously administered at a dose of 0.4 ml/kg body weight. The scanning protocols are shown in Tables 1 and 2.
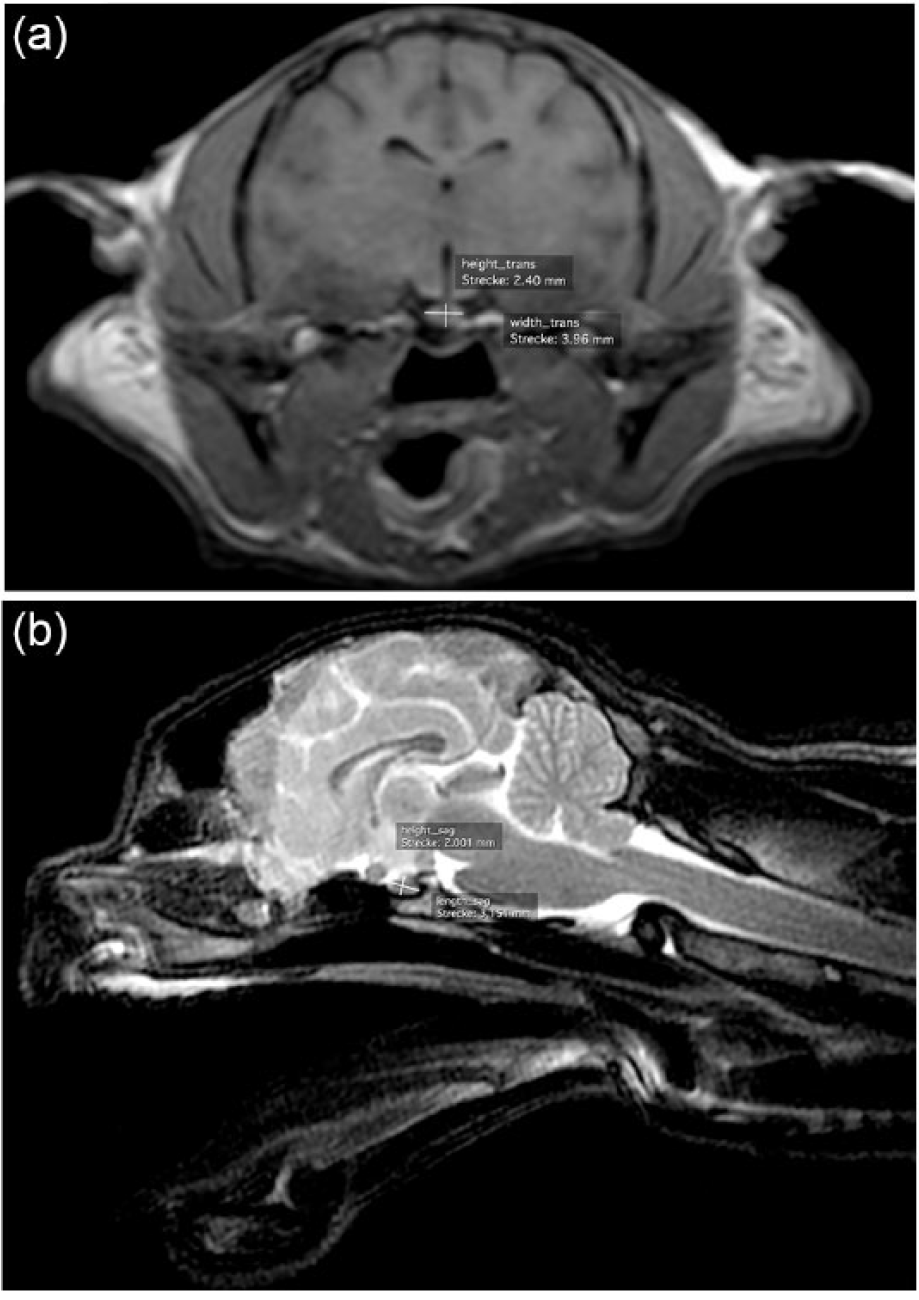
Example of pituitary gland measurement in (a) a transverse T1 3D fast field echo-weighted MRI image and (b) a sagittal T2 turbo spin echo-weighted MRI image of a brachycephalic cat

Example of pituitary gland measurement in (a) a transverse T1 spin echo-weighted post-contrast MRI image and (b) a sagittal T2 turbo spin echo-weighted MRI image of a mesocephalic cat
MRI sequence parameters in the mesocephalic cats

The non-homogeneity of data was a result of the retrospective nature of this study
MRI sequence parameters in the brachycephalic cats

FFE = fast field echo
Morphometric procedures
The length and height of the pituitary gland were measured from MRI images taken in the sagittal plane, and width and height were measured in the transverse plane. Measurements were performed by the first author (TCH), with a second measurement performed by a blinded co-author (MJS) to check the inter-observer reproducibility. The images were analysed using OsiriX Imaging Software, Advanced Open-Source PACS Workstation, DICOM Viewer. It was ensured that the length and width were perpendicular and parallel to the presphenoid or basisphenoid bone. Mean measurements and SDs were calculated for each group. Residues were calculated with BMDP1R software, and the standard distribution was checked with Q-Q-Plot. Data were evaluated by univariate and bivariate ANCOVA if the pituitary gland measurements were found to be related to cranial shape, sex, neutering status, age or body weight. A P value <0.05 was considered to be statistically significant, and a P value <0.001 was considered highly significant. Inter-observer reproducibility was assessed in three steps of statistical analysis. In a first step a comparison of mean values between the two observers was realised by means of the t-test for dependent variables giving mean differences and SDs. In a second and third step data were evaluated by Bland–Altman analysis with calculation of the correlation coefficient and evaluation if there was a trend between the differences of mean values between the observers (Table 3). 23
Statistical analysis of inter-observer reliability*

MD = mean inter-observer difference; PGSH = pituitary gland sagittal height; PGSL = pituitary gland sagittal length; PGTH = pituitary gland transverse height; PGTW = pituitary gland transverse width
Two observers
Results
All 59 cats met the inclusion criteria, and 32 brachycephalic and 27 mesocephalic cats were included in this study (Table 4). Of these cats, 30 were male (eight intact and 22 neutered) and 29 were female (11 intact and 18 neutered). The mean age of the brachycephalic cats was 4.1 ± 3.8 years (range 0.6–13.5 years) and the mean age of cats in the mesocephalic group was 8.9 ± 4.4 years (range 1.5–15.9 years). The brachycephalic group had a mean body weight of 2.93 ± 0.72 kg (range 1.84–4.30 kg) and mesocephalic cats had a mean body weight of 4.69 ± 1.02 kg (range 3.0–6.60 kg). From the MRI images, cats were diagnosed with either no abnormal findings or one of the following disorders, unrelated to pituitary gland pathology: otitis media, otitis externa, nasopharyngeal polyp, retinal detachment or behavioural changes. The mean pituitary gland measurements in brachycephalic cats were pituitary gland sagittal height (PGSH) of 2.15 ± 0.15 mm, pituitary gland transverse height (PGTH) of 2.42 ± 0.21 mm, pituitary gland transverse width (PGTW) of 4.44 ± 0.27 mm and pituitary gland sagittal length (PGSL) of 3.14 ± 0.30 mm. In mesocephalic cats, the pituitary gland measurements were PGSH of 2.94 ± 0.16 mm, PGTH of 3.09 ± 0.26 mm, PGTW of 4.73 ± 0.31 mm and PGSL of 4.88 ± 0.30 mm. Measurements ranged from 1.9–2.5 mm for PGSH, from 2.5–3.8 mm for PGSL, from 2.0–3.0 mm for PGTH and from 4.0–4.9 mm for PGTW in the brachycephalic group, and from 2.8–3.5 mm for PGSH, from 4.3–5.5 mm for PGSL, from 2.7–3.5 mm for PGTH and from 4.3–5.4 mm for PGTH in the mesocephalic group (Table 5).
Overview of study groups regarding sex, neutering status, age and body weight (BW)

BC = brachycephalic cats; MC = mesocephalic cats
Comparison of pituitary gland measurements in brachycephalic and mesocephalic cats

PGSH = pituitary gland sagittal height; PGSL = pituitary gland sagittal length; PGTH = pituitary gland transverse height; PGTW = pituitary gland transverse width; BC = brachycephalic cats; MC = mesocephalic cats
There was a highly significant correlation between cranial shape (brachycephalic vs mesocephalic) and pituitary gland height (sagittal and transversal planes) and pituitary gland sagittal length (P <0.0001). There was no statistically significant influence of cranial shape on pituitary gland width (Table 6). Sex had a significant influence on pituitary gland width in brachycephalic cats (P = 0.022). When both mesocephalic and brachycephalic cats were considered, there was a remarkable correlation between sex and pituitary gland width (P = 0.066), but this did not reach statistical significance (Table 7). In general, male cats showed a greater pituitary gland width than female cats, although this was not statistically significant. The effect of neutering status was only evaluated in the brachycephalic group, as neutered cats were not distributed homogeneously in the mesocephalic group (one intact male and two intact females; Table 4), with neutering status found to have no significant influence on pituitary measurements. In the brachycephalic group, age showed a significant influence on pituitary gland height (P = 0.012), width (P = 0.044) and length (P = 0.012) indicating an allometric correlation between age and pituitary gland size. Body weight showed no significant influence on pituitary gland measurements in the brachycephalic group. The analysis of the inter-observer reproducibility yielded excellent results regarding the correlation coefficients all close to 1.0 for every measurement. In no case was a statistical significant trend regarding the measurement differences shown. Student’s t-test showed no significant mean differences for PGSL and PGTW. For PGSH (P = 0.04) and PGTH (P = 0.0002) mean differences of the two observers were statistically significant (Table 3).
Statistical influence of brachycephaly vs mesocephaly on measurement of the normal feline pituitary gland (n = 59 cats)

Univariate analysis of covariance. Covariates age and body weight
PGSH = pituitary gland sagittal height; PGTH = pituitary gland transverse height; PGSL = pituitary gland sagittal length; PGTW = pituitary gland transverse width
Statistical influence of age, sex, neutering status and body weight on pituitary gland measurements

Bivariate analysis of covariance. Covariates age and body weight for the evaluation of sex. Covariates sex and neutering status for the evaluation of age and body weight
PGSH = pituitary gland sagittal height; PGTH = pituitary gland transverse height; PGSL = pituitary gland sagittal length; PGTW = pituitary gland transverse width
Discussion
In this study, we evaluated from MRI images whether there were differences in pituitary gland dimensions in mesocephalic and brachycephalic cats. In general, brachycephalic cats showed smaller pituitary glands than mesocephalic cats. Therefore, it cannot be assumed that pituitary gland sizes are relatively constant among cats, as proposed by two studies that used MRI and CT images to measure the pituitary gland.21,22 This should be considered if cryohypophysectomy is planned. Owing to the range of pituitary dimensions present in the mesocephalic cat population, precise measurement of the pituitary gland is crucial before cryosurgical intervention. Furthermore, for interpretation of the pituitary gland size in regard to the diagnosis of pathologies, such as acromegaly or Cushing’s disease, other clinical findings and laboratory tests should be considered.
The pituitary gland could be easily defined in all available MRI images, regardless of the chosen magnetic resonance tomography sequence. The easiest delineation was achieved in post-contrast T1-weighted images, consistent with a previous report. 22 Unfortunately, these images were not available for every cat. For all 27 mesocephalic cats T1-weighted spin echo post-contrast images were available for transverse sections. Transverse sections of the pituitary gland in the 32 brachycephalic cats were available as T1 3D fast field echo images without contrast agent administration. In both groups, sagittal sections of the pituitary glands were evaluated in T2 TSE images. The inconsistency of the MRI sequences is one of the major limitations of this study. Ideally, we would have chosen solely T1 spin echo post-contrast images in all planes. This was not possible owing to the retrospective nature of the study regarding the mesocephalic population and the fact that the owners of the cats in the brachycephalic group declined the administration of contrast medium owing to possible side effects. Nevertheless, pituitary glands could be delineated successfully in all cases. This result is supported by the inter-observer reproducibility, which showed almost perfect agreement (Table 3). When compared with previous studies,21,22 the main improvement of the current investigation is the use of a larger sample size and the inclusion of two different groups, including brachycephalic cats. A comparison of the pituitary gland measurements obtained by two recent studies with the data of mesocephalic cats reported in the current study is presented in Table 8.21,22 Our study used high-field MRI (1.0 Tesla [T]) for image acquisition. Wallack et al 22 performed pituitary gland measurement with a low-field 0.4 T MRI. A stronger magnetic field strength leads to an improved image quality and spatial resolution. 24 A recent study in people showed that MRI examinations with more T made preoperative localisation of pituitary microadenomas relatively better. 25 MRI might be superior to CT examination of the pituitary gland. Guy et al 26 compared the assessment of the pituitary gland and parasellar region with CT and MRI. The pituitary gland height could be assessed more accurately on MRI images. Moreover, the posterior pituitary was only identifiable with MRI and the pituitary stalk was more readily seen on MRI images than CT images. 26 We did not perform a comparison between MRI systems of different magnetic field strength, or a comparison with CT measurements. Therefore, we can only assume that our measurements obtained with a 1.0 T system might be more accurate than those of Wallack et al. 22 For the measurement of normal pituitary gland sizes MRI might be superior to CT. 26 However, in cases of pituitary microadenomas CT had similar abilities for evaluation of shape, location and size as MRI. 27
Comparison of pituitary gland measurements in mesocephalic cats with previously published data

Mean measurements in mm ± SD (range)
T = Tesla; PGSH = pituitary gland sagittal height; PGTH = pituitary gland transverse height; PGTW = pituitary gland transverse width; PGSL = pituitary gland sagittal length
In brachycephalic breeds, skeletal growth is diminished as a result of hormonal deficits or epiphyseal dysfunction of bones. However, the brain growth of these breeds is not affected.28–30 It is known that the brain parenchyma increases with growth of the individual, but cranial capacity does not increase to the same extent. 31 In brachycephalic cats, an increase in skull width and height can be observed with progressive shortening of the skull, which is dependent on the degree of brachycephaly. 32 To our knowledge, no studies have determined what happens to the pituitary gland during ontogeny or ageing of individual cats, especially in brachycephalic breeds such as Persian cats, which have reduced skull growth. For the evaluation of correlations between neutering status, age, body weight and pituitary gland size only the brachycephalic group was considered. There are two reasons for this decision. First, the inhomogeneous distribution of sex and neutering status among the mesocephalic cats; and, second, as the mean age in the mesocephalic and brachycephalic groups was 8.9 ± 4.4 and 4.1 ± 3.8 years, respectively, and the mean body weight was 4.69 ± 1.02 and 2.93 ± 0.72 kg, respectively, a reliable correlation could not be derived (Table 4). This is because one would inevitably come to the statistical conclusion that the pituitary gland increases in volume with increasing body weight and age, with a generally smaller pituitary gland in brachycephalic cats (Table 5). When only the brachycephalic population was considered, age had a statistically significant influence on the pituitary gland, with increased height, width and length (Table 7). This may be explained by the lower median age of the brachycephalic group, as there were eight cats (25% of all brachycephalic cats) aged <15 months at the time of evaluation and therefore not all cats were fully mature (Table 9). Although a correlation between pituitary volume and patient age has not yet been described in cats, 22 a correlation is well known in humans and proven in sheep.33,34 Langen ascribed the increase in size to an increase in activity of the pituitary gland during adolescence. 34 As the brachycephalic population showed a range in age between 7.66 and 161.9 months this might be a possible explanation. We did not further investigate whether the increase in pituitary gland size was due to hormonal activity during adolescence. A larger population and a histopathological examination would be necessary to describe a possible relationship between pituitary gland size and age. A statistical significant influence of body weight on pituitary gland size could not be verified in this study (Table 7). This finding consents to the result of a previous study. 22
Comparison of immature and adult brachycephalic cats
Percentage of brachycephalic group
BW = body weight; PGSH = pituitary gland sagittal height; PGSL = pituitary gland sagittal length; PGTH = pituitary gland transverse height; PGTW = pituitary gland transverse width; immature = mean age <15 months; BC = brachycephalic cats; adult = mean age >15 months
Sex had a remarkable influence on pituitary gland width in neutered cats. For this evaluation only neutered individuals of the mesocephalic and brachycephalic group were considered. This is owing to the lack of intact female and male individuals among the mesocephalic cats. A more homogeneous distribution of neutering status was not possible owing to the retrospective data collection regarding the mesocephalic group. Nowadays, most client-owned cats are neutered. In the brachycephalic group neutering status was distributed more homogeneously because there were younger cats and cats used as breeding animals. Evaluating the influence of sex on pituitary gland size solely in the brachycephalic group the same correlation could be derived. There was a statistically significant correlation between sex and pituitary gland width (Table 7). Male cats showed a greater pituitary gland width than female cats, which reflects the findings of Tyson et al. 21 In general, male cats are larger and have a higher body weight than female cats, in addition to a larger cerebral volume.35,36 From an allometric point of view, this could explain the larger pituitary gland size. Neutering status could only be evaluated in the brachycephalic group owing to a lack of intact individuals in the mesocephalic group (Table 4). Additionally, 25% of cats among the brachycephalic population were immature. It is known that neutering leads to a hypertrophic gonadotropic cell population in the pituitary gland, this appeared to have no influence on pituitary gland size in this study. But, for the stated reasons, results could be biased and, consequently, the effects of neutering could not be reliably studied.
The question of why brachycephalic cats showed smaller pituitary glands in this study is not easily answered. Skeletal growth has been shown to be reduced in brachycephalic dogs, 30 and this could possibly be applied to brachycephalic cats. We did not evaluate whether the pituitary gland in brachycephalic breeds is smaller as a result of reduced synthesis of growth hormones and factors, and further studies examining and comparing plasma levels of insulin-like growth factor 1 and growth hormone in addition to body weight and body size are required to make a qualified statement. In human medicine, children and juveniles with an isolated lack of growth hormone had a smaller hypophyseal volume than a healthy reference group, 37 indicating a correlation between hormone plasma levels and pituitary gland size. There is good-to-excellent inter-observer reproducibility. All correlation coefficients are close to 1.0 (Table 3) and mean differences between the observers regarding PGSL and PGTW are not statistically significant. The significance for PGSH and PGTH is statistically evident but practically irrelevant owing to their dimension (one-hundredth of 1 mm).
There are several limitations of the study. First, the selection of the study population. The mesocephalic group was evaluated retrospectively. Mean age and body weight were higher than in the brachycephalic group (Table 7), which made a statistical examination of both groups combined impossible. Further, we assumed normal functional and structural integrity of the pituitary gland in the study population. None of the cats underwent histopathological examination, blood analysis or urinalysis, and therefore neoplastic changes and endocrine disorders of the gland could not be ruled out. It is possible that we could have missed hyperglycaemia, glucosuria or other laboratory findings related to acromegaly or Cushing’s disease. However, none of the cats presented with any overt clinical signs associated with pituitary disease.
In this study, all brachycephalic MRI images had been acquired for another study that evaluated intracranial abnormalities unrelated to, or that did not affect, the pituitary gland. The mesocephalic cats had been scanned owing to central nervous abnormalities or pathologies that did not affect the pituitary gland, for example external and medial otitis, idiopathic epilepsy or behavioural changes. Ideally, only healthy cats with previous laboratory blood and urinalysis and a homogenous age and sex distribution would have been chosen. In addition, a universal MRI protocol would have been favourable. In particular, the consequent use of contrast medium for easier pituitary gland delineation might be preferable. 22 The exclusion of cats with lacking laboratory work-up and immature cats regarding the brachycephalic group would have substantially decreased the number of cases. Within the limits of this study, we showed that a uniform pituitary gland size cannot be expected in cats. Furthermore, there was a general trend for brachycephalic cats to have smaller pituitary glands than mesocephalic cats.
Conclusions
Precise individual measurement of the pituitary gland is mandatory if a cryosurgical intervention is considered. Unfortunately, there is still a lack of studies that have focused on the cryosurgical properties of pituitary gland tissue and neoplasia of the pituitary gland. Further research on hypophyseal cryosurgery in cats is required to achieve a more favourable outcome than that previously described.16,17 Even so, this treatment option has considerable advantages over the conventional surgical method, giving it great potential for this application.
Supplemental Material
Click here for Supplementary Material
Overview of all study cats including breed, sex, age, body weight and pituitary gland measurement
Footnotes
Supplementary material
Overview of all study cats including breed, sex, age, body weight and pituitary gland measurement.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
