Abstract
Practical relevance:
Hyperadrenocorticism (HAC) is a relatively uncommon endocrinopathy of older cats, with a mean age at diagnosis of 10 years. In addition to pituitary-dependent and adrenal-dependent hypercortisolism, clinical signs of HAC can result from adrenal sex steroid-producing tumours.
Clinical challenges:
While HAC in cats has many similarities to canine HAC, there are key differences in presentation, diagnosis and response to therapy. Most, but not all, cats with HAC have concurrent diabetes mellitus, which is often insulin resistant. Up to a third of cats with HAC have extreme skin fragility and are at high risk of debilitating iatrogenic skin tears during diagnostic or therapeutic interventions. Infections of the skin and nail beds, and urinary, respiratory and gastrointestinal tract, secondary to cortisol-induced immune suppression, are also common. Cats respond differently to dogs to adrenal function tests including adrenocorticotropic hormone (ACTH) stimulation and dexamethasone suppression tests; a 10-fold higher dose of dexamethasone is recommended in cats to screen for HAC. Curative treatment options include adrenalectomy or transsphenoidal hypophysectomy. Radiation or medical treatment may improve clinical signs. The response to mitotane therapy is poor. While trilostane is the medical treatment of choice based on retrospective studies, investigations into the pharmacokinetics of this drug in cats are lacking.
Global importance:
Feline HAC occurs worldwide and is not associated with any purebreed predisposition. Although uncommon, adrenal sex steroid-producing tumours have a higher prevalence in cats than in dogs.
Evidence base:
The information in this review is drawn from over 180 reported cases of feline HAC. Reports investigating clinical presentation, clinicopathological findings and treatment outcomes are observational, retrospective multiple case series (EBM grade III) or single case reports (EBM grade IV). While most endocrine testing studies for diagnosis are cohort controlled analytical studies (EBM grade III), prospective, randomised, placebo-controlled studies have been performed (EBM grade I).
Introduction
While the term ‘hyperadrenocorticism’ (HAC) refers to overproduction of any adrenocortical hormone, in veterinary medicine it is typically used to describe hypercortisolism (Cushing’s syndrome). HAC may also occur secondarily to pituitary or adrenal tumours that secrete other hormones with glucocorticoid-like activity; for example, alpha-melanocyte-stimulating hormone (α-MSH), progesterone, oestradiol, testosterone, androstenedione and 17α-hydroxyprogesterone.
This review discusses the signalment, history, clinical findings, diagnosis, treatment and prognosis for cats with spontaneous HAC (hypercortisolism), iatrogenic HAC and adrenal sex steroid-producing tumours. For information on hyperaldosteronism (not covered here), readers are referred to an earlier review by Djajadiningrat-Laanen et al. 1

Aetiopathogenesis
In contrast to canine HAC, HAC in cats is a relatively uncommon endocrine disorder, with a little over 180 cases reported in the veterinary literature.2–40 A large UK study estimated the prevalence of HAC in UK dogs attending primary care practices to be 0.28%. 41 Approximately 80% of feline cases are pituitary-dependent hyperadrenocorticism (PDH). Carcinomas comprise a minority of cases overall, since at least 90% of PDH cases and 50–60% of adrenal-dependent hyperadrenocorticism (ADH) cases are caused by adenomas.2–40
PDH results from autonomous secretion of ACTH by tumours arising from corticotroph cells of the pars distalis (more commonly) or pars intermedia. This stimulates excessive production of cortisol from the adrenal cortex and causes cortical hyperplasia. ADH is usually secondary to autonomous cortisol production by tumours arising from the adrenal cortex.
Clinical signs
Commonly reported clinical signs and physical examination findings, collated from reports of 167 cats with naturally occurring HAC published between 1973 and 2015, are listed in Table 1. Clinical signs are typically present for several months before presentation to a veterinarian, although time to presentation ranges from weeks to more than a year.2,5,6,11–19,22–33,35,36,38–40 Clinical signs overlap with other diseases (eg, diabetes mellitus), and early signs may be subtle.
Clinical signs and physical examination findings collated from reports of 167 cats with hyperadrenocorticism *


Whether hypercortisolism can cause secondary central or nephrogenic diabetes insipidus in cats has not been established. 35 Polydipsia and polyuria may result from comorbidities; for example, diabetes mellitus, chronic kidney disease (CKD) or hyperthyroidism. Polyphagia has been reported in non-diabetic cats with HAC.9,10,19,42
HAC causes skin atrophy and reduced skin elasticity, resulting in prominent subcutaneous veins and fragile, thin, papery skin (Figure 1). Comedones, a common feature of canine HAC, can sometimes be detected on ventral abdominal skin (Figure 1). Skin tears following minor trauma, routine handling or even grooming are common complications (Figure 2). Footpad excoriations can also occur. Extreme care should be taken when handling cats with suspected HAC to avoid iatrogenic trauma. Alopecia is typically bilaterally symmetrical, may involve the thoracic, ventral abdominal, flank and limb regions, and may be patchy or generalised. An ’unkempt’ haircoat may result from growth of brittle and sparse hair, skin scaling and seborrhoea. Failure of hair regrowth after clipping is common (Figures 1–4).9,18,35,42

Nine-year-old female neutered Devon Rex cat with diabetes mellitus and pituitary-dependent hyperadrenocorticism (PDH). The cat had abdominal distension (‘pot-belly’), alopecia of the ventral abdomen with comedones (a and b), and bilaterally symmetrical alopecia of the pelvic limbs with thin, papery skin and excoriations (c)

(a)This 9-year-old male neutered British Blue cat with PDH had extreme skin fragility and tore a large piece of skin as it squeezed between two bars of a security grill. (b) Nail bed infection and (c) fragile skin from the head that was inadvertently torn loose during grooming in the same cat
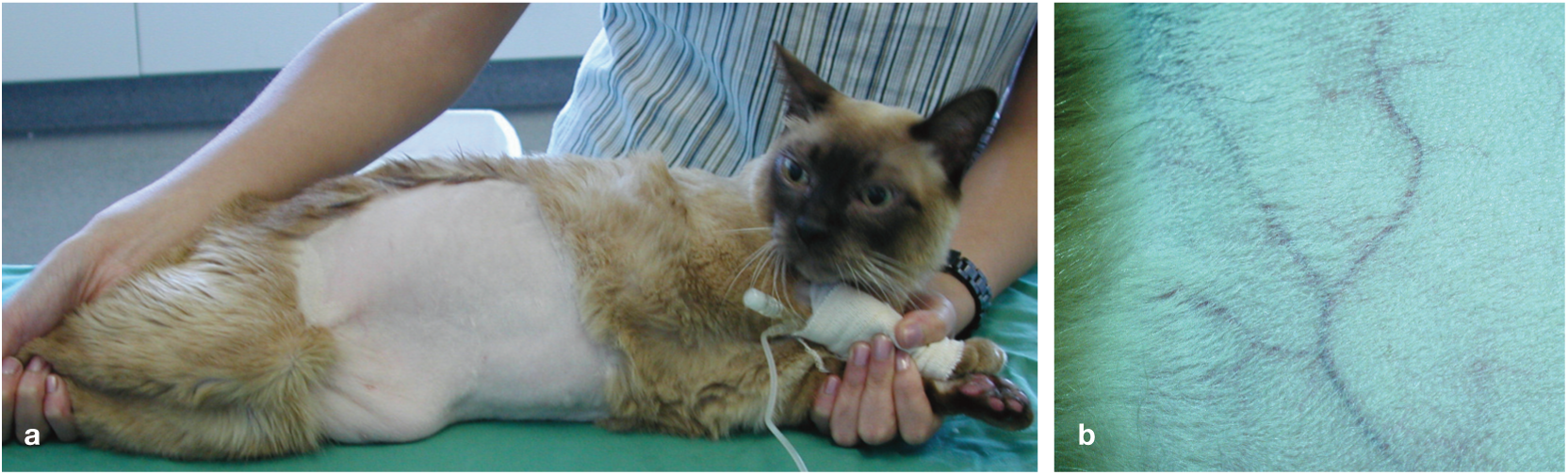
Seven-year-old male neutered Burmese cat with PDH. 18 Four months after clipping for an abdominal ultrasound examination to investigate the cause of its hypertensive retinopathy, the cat re-presented with severe polydipsia and polyuria. The hair on the ventral abdomen had not regrown (a), and the skin was thin and papery with prominent subcutaneous vessels (b)
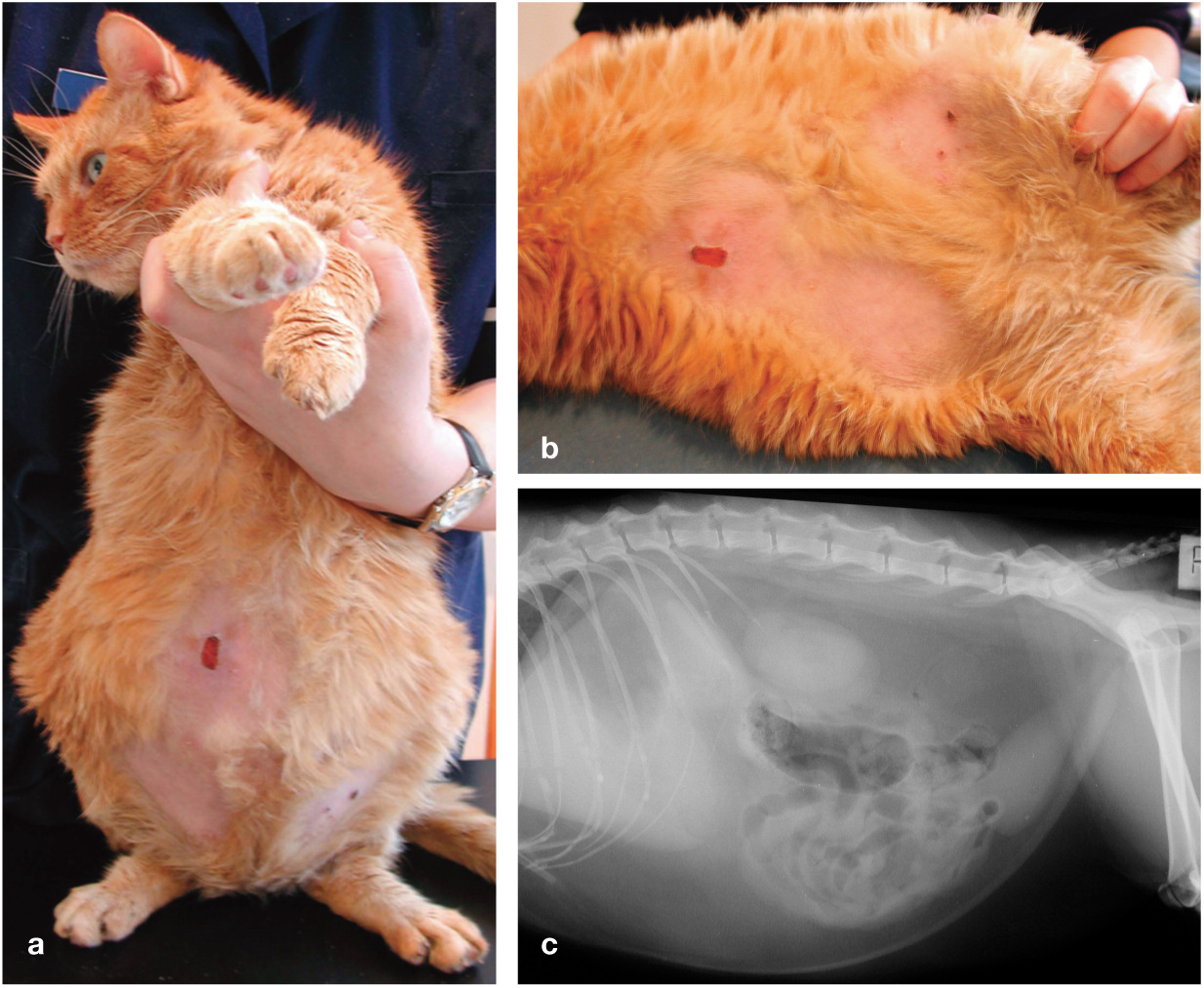
Nine-year-old male neutered domestic shorthair cat with adrenal-dependent hyperadrenocorticism (ADH). Note the ventral abdominal alopecia and skin excoriations resulting from thin skin (a,b). The cat had a ’pot-bellied’ appearance on physical examination (a) and abdominal distension on a lateral abdominal radiograph (c). In cats with hyperadrenocorticism (HAC), these signs result from hepatomegaly, abdominal fat deposition and abdominal muscle wasting
Hypercortisolism also leads to blood vessel friability and reduced fibrous response to injury which, in turn, can produce excessive bruising, delayed wound healing and reduced ligament and tendon strength.3,35,37 Weight loss from concurrent diabetes mellitus is common. Muscle wasting, due to increased protein catabolism, is generalised or involves the lumbar musculature and contributes to weight loss. Although common, weight loss may not be apparent to owners because of abdominal distension (‘pot-belly’) resulting from hepatomegaly, abdominal fat deposition and abdominal muscle wasting (Figure 4). Depending on the balance of these factors, cats with HAC occasionally gain, rather than lose, weight. 9
Generalised weakness and/or a plantigrade hindlimb stance are less common signs in feline HAC. Where present, contributing factors may include peripheral diabetic neuropathy, muscle wasting and hypokalaemia.
Uncommon clinical signs include gastrointestinal signs (eg, inappetence, vomiting and diarrhoea), systemic hypertension, neurological signs from a pituitary macroadenoma, or a palpable adrenal mass.8–10,13,15,16,18,19,28,31,33,35,39
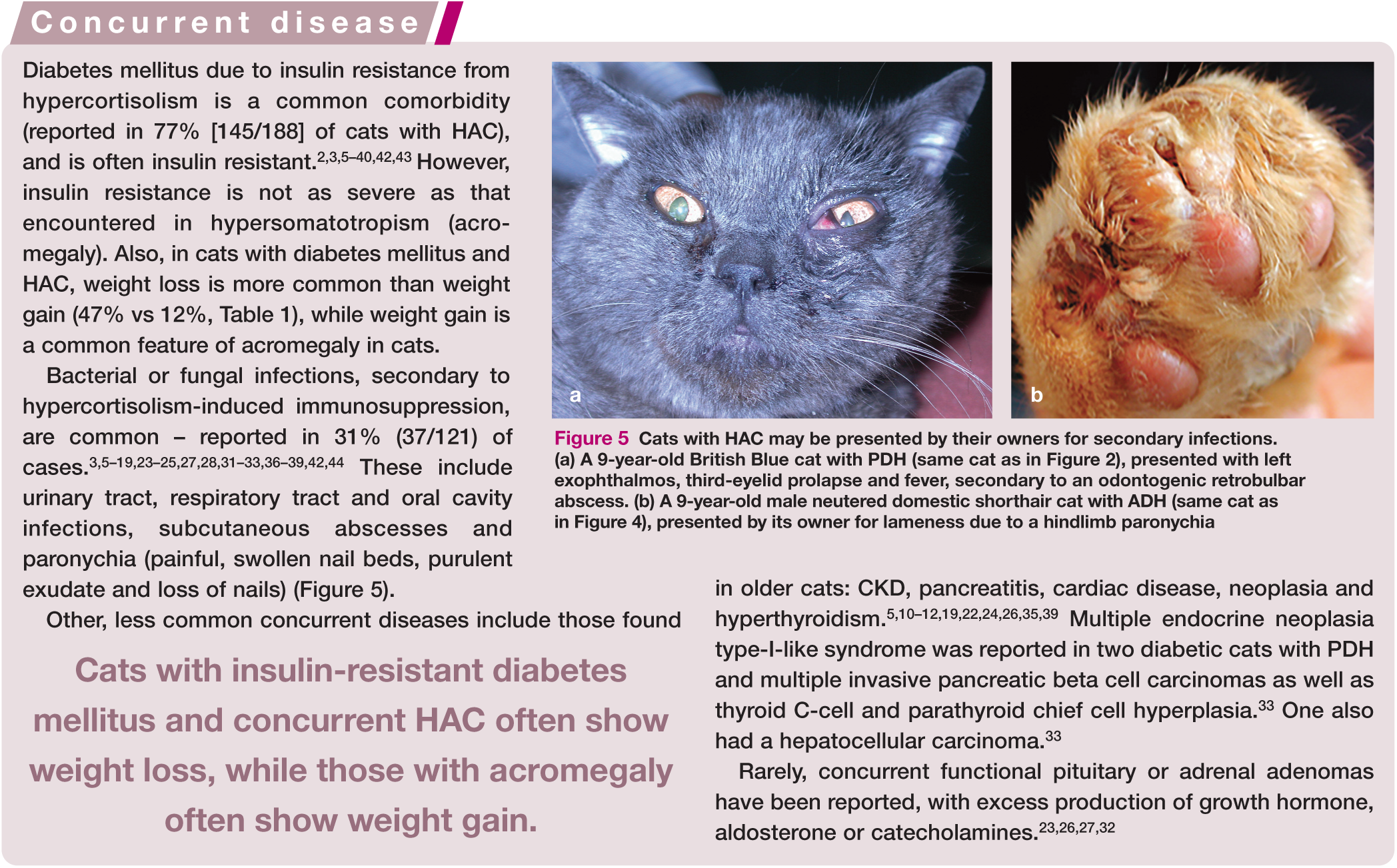

Cats with HAC may be presented by their owners for secondary infections. (a) A 9-year-old British Blue cat with PDH (same cat as in Figure 2), presented with left exophthalmos, third-eyelid prolapse and fever, secondary to an odontogenic retrobulbar abscess. (b) A 9-year-old male neutered domestic shorthair cat with ADH (same cat as in Figure 4), presented by its owner for lameness due to a hindlimb paronychia
Diagnostic tests
Clinical pathology
Haematological findings in cats with HAC may be unremarkable or may reveal components of a ‘stress’ leucogram (mild-to-moderate mature neutrophilia, lymphopenia, eosinopenia and/or monocytosis).5,6,8–13,15,16,21,22,24,25,35,37,38 a mild-to-moderate anaemia is occasionally present.10,19,35 Serum biochemical abnormalities are usually nonspecific; Table 2 summarises biochemistry data collated from reports of 138 cats with naturally occurring HAC published between 1975 and 2015 where laboratory findings were documented.
Abnormalities in serum biochemistry collated from reports of 138 cats with HAC *


Lithium heparin blood sample from the cat pictured in Figure 1. This was grossly lipaemic despite fasting, due to hypertriglyceridaemia, which occurs in approximately 7% of cats with HAC
Although hyperglycaemia secondary to concurrent diabetes mellitus is common, it is important to note that some cats will have transient diabetes mellitus or ‘stress’ hyperglycaemia. 35 In contrast to dogs with HAC, elevations in alanine aminotransferase (ALT) and alkaline phosphatase (ALP) are much less frequent in affected cats. Since cats lack a glucocorticoid-induced isoenzyme of ALP, increases in ALP activity occur secondarily to diabetes mellitus or other concurrent diseases. Hypercholesterolaemia is common, but not as frequent as in canine HAC (Table 2).
Total thyroxine is usually within the reference interval; however, some cats may have decreased concentrations secondary to nonthyroidal illness.5,7,10,19,23,35 Feline immunodeficiency virus and feline leukaemia virus testing is usually negative.5,7,17,26,38
Urinalysis has typically revealed glucosuria in cats with concurrent diabetes mellitus. Unlike dogs, the majority of cats with HAC have a urine specific gravity (USG) of >1.020. However, USG may range from isosthenuria to well concentrated and is influenced by comorbidities such as CKD or hyperthyroidism. Glucosuria may also cause a mild artefactual increase in USG. Proteinuria (not quantitated) has been reported in some cats.6–8,10,13,15,17–19,22–25,31,32,35,37
Endocrine testing
Endocrine testing should be performed to support the initial diagnosis in cats suspected to have HAC. Signalment, history, clinical signs and laboratory findings suggestive of HAC will increase the positive predictive value of endocrine testing.
Urine cortisol to creatinine ratio
The urine cortisol to creatinine ratio (UCCR) can be used as an initial screening test. Similar to its use in the investigation of canine HAC, this test has high sensitivity but low specificity for diagnosis of HAC in cats. 42 It is used as a rule-out test due to the high predictive value of a negative result.
UCCR values will increase with stress (eg, hospitalisation) and illness, and home-collected urine samples should be submitted for analysis several days after a veterinary visit.45,46 UCCR values also show day-to-day variation and so a minimum of two morning urine samples collected on consecutive days is advised. UCCR results do not vary with age, gender, neuter status or breed, but hyperthyroid cats have significantly higher values.42,45–47 The median UCCR was 13–16 x 10−6 for home-collected urine samples from 87 healthy cats in two reports.42,47 Thirteen cats with HAC had median UCCR values of 80–122 x 10−6 (note reference ranges are laboratory specific).3,42
Low dose dexamethasone suppression test
Dexamethasone suppression tests are used to assess the appropriateness of the physiological negative feedback response to exogenous glucocorticoid administration. In healthy animals, hypothalamic secretion of corticotropinreleasing hormone is suppressed for at least 24 h, thereby suppressing pituitary ACTH secretion and adrenal cortisol secretion.
The low dose dexamethasone suppression test (LDDST) is arguably the best initial screening test for HAC due to its high sensitivity. However, when using the same protocol as for dogs (0.01 mg/kg IV and serum cortisol measurements at 0, 4 and 8 h), 10–20% of healthy cats will fail to demonstrate suppression.5,19,34,48–50 Thus, to increase test specificity, the LDDST protocol in cats involves administration of a 10-fold higher dose of dexamethasone (0.1 mg/kg IV), with serum cortisol measurements at 0, 4 and 8 h.5,7,51 Using this protocol, 4 and 8 h serum cortisol levels in healthy cats will be suppressed to <~40 nmol/1 (or similar reference interval, depending on the laboratory) (Figure 7).7,50 Cats with HAC are resistant to pituitary suppression from dexamethasone administration and usually have an 8 h cortisol above the reference interval (Figure 7).

Expected results of a low-dose dexamethasone suppression test (LDDST) (0.1 mg/kg dexamethasone IV) in a healthy cat (a) and in a cat with HAC (b)
Overall, the LDDST in cats has high sensitivity and moderate specificity for the diagnosis of HAC. 35 However, occasionally cats with HAC return normal test results on an LDDST using 0.1 mg/kg dexamethasone. Diagnosis should be pursued using alternative tests in cats with clinical signs that are highly suggestive of HAC but that return normal LDDST results (eg, combined UCCR and LDDST test and adrenal sex steroid panels [see below]). Cats with diabetes mellitus (well or poorly controlled), and without HAC, have comparable LDDST results to healthy cats. 34

Adrenocorticotropic stimulation test
The ACTH stimulation test is typically performed by administering either 5 pg/kg or 125 pg/cat of tetracosactide or co-syntropin (synthetic ACTH) IV, with serum cortisol measurements at 0 and 60 mins.52–58 The intramuscular route can also be used; however, peak cortisol levels are lower and occur earlier, at 30–45 mins.50,52,59,60 The time to peak cortisol levels following administration of ACTH depends variously on the dose and route of administration and use of synthetic vs natural ACTH.50,52,54–56,58
Healthy cats do not show cortisol level increases above the reference interval. However, as few as one-third of cats with HAC show positive test results.5,6,8,9,15,19,35,50,53 Thus, with its poor sensitivity and only moderate specificity (due to positive results in some cats with non-adrenal illness), the ACTH stimulation test is not well suited as an initial diagnostic screening test. 19
Alpha-melanocyte-stimulating hormone
Pituitary tumours have been reported in cats that secrete excessive amounts of α-MSH together with ACTH or that show positive immunohistochemical staining for α-MSH.3,4,22,23,27 These cats have high basal α-MSH and may also show partial or lack of suppression with dexamethasone suppression tests. α-MSH may have weak glucocorticotropic effects. 4 A case of a melanotroph pituitary adenoma that was only secreting α-MSH has been reported; the cat showed insulinresistant diabetes mellitus and clinical signs of HAC. 4

Cytology of adrenal masses
Fine-needle aspirate cytology of adrenal masses has been described in cats and dogs, 61 and was found to assist in differentiation of adrenocortical tumours from phaeochromocytomas. Cytology did not differentiate between benign and malignant tumours and also cannot identify functional adrenocortical tumours.
Differentiation of ADH from PDH
Once HAC has been diagnosed, further investigation may be required to differentiate ADH from PDH.
Endocrine testing
Low dose dexamethasone suppression test
In almost half of cats diagnosed with HAC using an LDDST, discrimination between ADH and PDH is possible on the basis of the results if the:
4 h cortisol value is <~40 nmol/1; or
4 h or 8 h cortisol value is >~40 nmol/1 but is <50% of the 0 h value.5,16,35
These results can occur in cats with PDH because suppression of the hypothalamic-pituitary axis with PDH can be transient (Figure 8).

Results of a low-dose dexamethasone suppression test (LDDST) (0.1 mg/kg IV) differentiate PDH from ADH in some situations. (a) The 8 h cortisol measurement is elevated, indicating escape from suppression and likely HAC. The 4 h cortisol value is within the reference interval, consistent with a diagnosis of PDH. (b) The 4 h cortisol is elevated but is <50% of the value of the 0 h cortisol, consistent with PDH. (c) The 8 h cortisol is both elevated and <50% of the value of the 0 h cortisol, consistent with PDH
High dose dexamethasone suppression test
The high dose dexamethasone suppression test (HDDST) involves administration of 1 mg/kg dexamethasone IV and serum cortisol measurements at 0, 4 and 8 h. Suppression to >50% of baseline cortisol at 4 h or 8 h is suggestive of PDH but only occurs in approximately 40–50% of PDH cats.11,13,35,37
Combined UCCR and low dose oral dexamethasone suppression test
This test – a modification of the UCCR – involves home collection of two morning urine samples on consecutive days.3,27,42 Immediately following collection of the second sample, 0.1 mg/kg dexamethasone is administered orally at 8 h intervals three times (8 am, 4 pm and 12 am), with a third urine sample collected on the morning of the third day. Suppression occurs if the UCCR of the third urine sample is <50% of the mean of the baseline values, and this result is consistent with PDH. If there is less than 50% suppression, differentiation between ADH and PDH cannot be determined. 42 This test correctly differentiated 10/13 (77%) cats with PDH and can be used as a combined screening and differentiation test.3,42 If the UCCR of the first two urine samples is within the reference interval then HAC is excluded.35,42
Endogenous ACTH measurement
Plasma endogenous ACTH levels are expected to be increased in cats with PDH and decreased in cats with ADH.3,5,7,11,13,18,19,35,37,50 However, since plasma ACTH concentrations in healthy cats can be below the detection limit of ACTH assays, the test cannot be used as a screening test. 50 Also, although most cats with PDH have markedly increased endogenous ACTH levels, some fall to within the reference interval (10–60 pg/ml).3,5–7,18,19,35,50 Many commercial ACTH assays are not validated for use in cats, are expensive and require sample transport on dry ice to the laboratory. 19
Endogenous ACTH precursor measurement
A study of a small number of cats measuring plasma ACTH precursors found that 8/9 cats with PDH had levels ⩾229 pmol/l, while healthy cats, cats with acromegaly and diabetes or cats with diabetes mellitus alone had levels <100 pmol/l. 2 To the authors’ knowledge, this test is not commercially available.
Diagnostic imaging
Diagnostic imaging of the adrenal and pituitary glands is used for differentiation of PDH from ADH rather than for initial diagnosis of HAC. Identification of adrenal or pituitary enlargement does not indicate increased function.
Abdominal ultrasonography or radiography
Abdominal radiographs typically identify abdominal distension, hepatomegaly and increased amounts of intra-abdominal fat in cats with HAC (Figure 4c).5–9,16,21,22,24,37 In some ADH cases a large adrenal mass may be visible.5–7,9,24,31 Occasionally one or both adrenal glands may be mineralised; however, this is also a common incidental finding in healthy cats.16,31,39,51 Thoracic radiographs are unremarkable unless concurrent intrathoracic disease is present.7–9,21,22
Several ultrasonographic studies have evaluated adrenal gland dimensions in cats (Table 3). The sensitivity of ultrasonography for discrimination testing is dependent on the technical expertise of the operator. Valentin et al (2014) reported a 93% sensitivity of ultrasonography for differentiation of ADH from PDH. 19 Combes et al (2014) found there was least inter- and intra-observer variability in adrenal height measurements. 62 Bilateral measurements of adrenal length and height of cranial and caudal poles should be recorded during ultrasonographic investigation of adrenal gland size (Figure 9). Most cats with PDH have bilaterally symmetrical adrenal gland enlargement; however, in some, adrenal size is within the reference interval, and rarely unilateral adrenomegaly is identified.6–9,13,15,18,19,21,33,34,37,39
Ultrasonographic adrenal gland size in sick and healthy cats

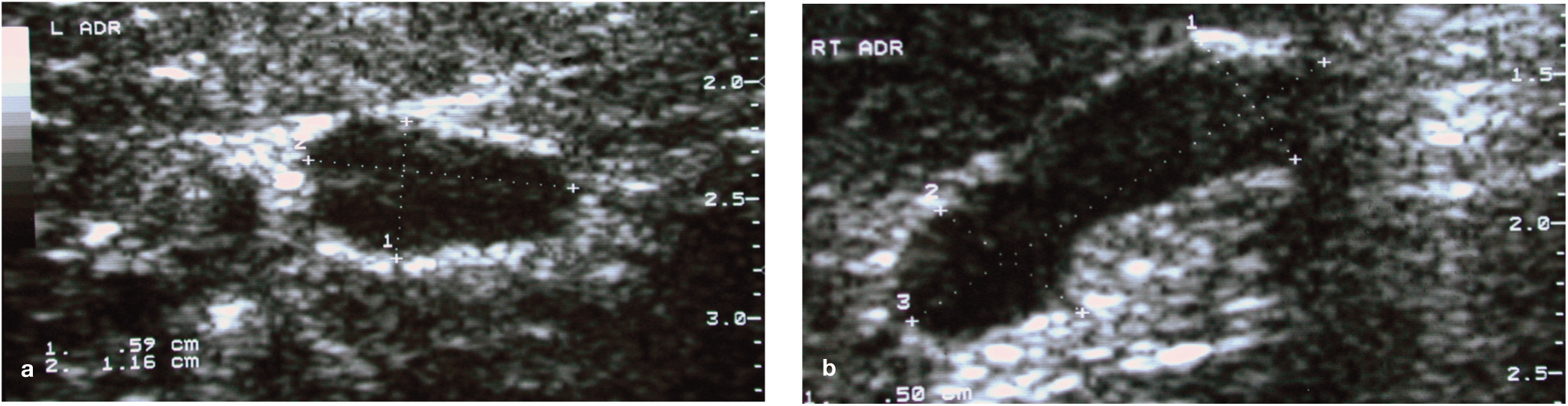
Ultrasonographic measurements of the adrenal glands of the cat with PDH pictured in Figure 3. (a) Left adrenal cranial pole height 5.9 mm, length 11.6 mm. (b) Right adrenal cranial pole height 5.0 mm, caudal pole height 5.6 mm, length 16 mm
Other diseases associated with adrenomegaly include hyperthyroidism, acromegaly and hyperaldosteronism.63–65 The adrenal glands of hyperthyroid cats are approximately 20% larger than those of healthy cats and may contain hyperechoic foci. 65 Importantly, diabetes mellitus does not cause adrenal gland enlargement in cats. 34

Other ultrasonographic changes detected in cats with HAC include hepatomegaly and increased amounts of intra-abdominal fat. The liver may appear diffusely hyper- or hypo-echoic.7,8,15,16,19
Cats with ADH have a unilateral adrenal mass or adrenomegaly with atrophy of the contralateral gland. However, in some cases the unaffected adrenal may be within the reference interval.5,6,9,10,19,24,31,34 Approximately equal numbers of right- and left-sided cases are reported.5,9,10,19,24,26,31,32,34,36 The presence of vascular invasion or evidence of metastases is consistent with a carcinoma. Bilateral adrenocortical adenomas (occurring in the same cat at different time points) and bilateral tumours of different functional type (eg, phaeochromocytoma and adrenocortical adenoma) occur occasionally.5,6,32
Brain computed tomography or magnetic resonance imaging
Approximately 50% of PDH cases are thought to have pituitary macroadenomas that can be detected using computed tomography (CT) or magnetic resonance imaging (MRI) (Figure 10).3,5,13,15,19,23,25,27,30,35 Pituitary gland size reference intervals for healthy cats are described. 67 Pituitary imaging is important in cats with PDH. The identification of a pituitary macroadenoma has implications for therapy, since medical treatment will not prevent progressive tumour enlargement or the development of associated neurological signs.

Advanced diagnostic imaging of the cat with PDH pictured in Figure 1. (a) Post-contrast transverse CT showing a heterogeneously enhancing pituitary macroadenoma (arrow). The lesion is shown also on sagittal (b) and coronal (c) post-contrast magnetic resonance images

CT or MRI may also be used to obtain relevant information regarding adrenal tumours.
Treatment
Several treatment modalities have been used for cats with HAC, including medical management, adrenalectomy, transsphenoidal hypophysectomy and irradiation of the pituitary gland. Only adrenalectomy or transsphenoidal hypophysectomy are potentially curative.
Medical management
Trilostane
Trilostane is a synthetic steroid analogue that competitively inhibits 3-β-hydroxysteroid dehydrogenase. This enzyme allows conversion of pregnenolone and 17-α-hydroxy-pregnenolone to progesterone and 17-α-hydroxyprogesterone.
Most trilostane-treated cats have shown a reduction in, but not resolution of, clinical signs (and insulin doses, if diabetic), as well as improved ACTH stimulation test results. Reported doses used for treatment of feline HAC range from 10–30 mg/cat PO q12–24h; however, lower starting doses of 1–2 mg/kg/day have been suggested more recently.5,7,8,18,19,23,35 The optimal time to perform an ACTH stimulation test in dogs is 2 h after trilostane administration, but is not known in cats.5,7,8,68 It has been suggested to aim for a 2–4 h post-trilostane ACTH stimulation cortisol measurement of 50–150 nmol/l in cats. 35 Use of compounded trilostane is not recommended; in one study, 38% of tested compounded trilostane products were not the prescribed strength or had variable dissolution characteristics. 69
Adverse effects of trilostane in cats include anorexia, lethargy, weight loss, pancreatitis and hypoadrenocorticism.5,7,18,19,23 The median survival time in one study of 15 trilostane-treated cats was 617 days (range 80–1278 days). 5 Although the pharmacokinetics of trilostane in cats are yet to be established, the drug is mostly well tolerated and is the most efficacious medical treatment currently available for feline HAC.5,7,8,19
Mitotane
Mitotane is an adrenocortical cytotoxic agent that targets the zona fasciculata and reticularis. In cats, it is not recommended for firstline therapy since it is far less effective than trilostane, clinical signs often progress and ACTH stimulation test results remain unchanged.5,6,12,16,19 Reported doses range from 25–50 mg/kg/day PO for induction and 25–50 mg/kg/week for maintenance therapy.6,12,16 Adverse effects including anorexia, lethargy and vomiting are common, even in the absence of an effect on cortisol concentrations. Lethargy and gastrointestinal signs can also occur secondarily to hypoadrenocorticism. 12 Reported survival times range from less than 2 months to more than 63 months.6,19
Other medical therapies
Other steroid hormone synthesis inhibitors that have been used in a small number of feline HAC cases include metyrapone, ketoconazole and aminoglutethimide.5,19,21,24,35,37,70 These drugs are not recommended due to adverse effects, lack of efficacy and/or difficulty sourcing them.24,35,37,71
Adrenalectomy
Although potentially curative, unilateral adrenalectomy (for ADH) or bilateral adrenalectomy (for PDH) is associated with a relatively high complication rate due to poor wound healing, immunocompromise and skin fragility. Careful patient selection, preoperative planning, and intra- and postoperative management are essential to minimise complications and achieve the best outcomes (Figure 11). In cats wtih ADH, abdominal CT examination before surgery facilitates identification of tumour invasion (phrenicoabdominal vein, caudal vena cava and renal vein) and thrombosis.

Hydrocortisone continuous rate infusions should be administered intra- and postoperatively for 24-48 h (0.3–0.5 mg/kg/h IV), followed by oral prednisolone (0.1–0.3 mg/kg/day PO), which is then is tapered off over 6–8 weeks in the case of a unilateral adrenalectomy. After bilateral adrenalectomy, ongoing glucocortcoid therapy as well as mineralocorticoid supplementation with fludrocortisone (0.1–0.3 mg/cat PO q24h) or desoxycorticosterone pivalate (2.2 mg/kg SC q25 days) is required.
Complications of adrenalectomy include haemorrhage, thromboembolism, wound dehiscence, skin lacerations, sepsis and pancreatitis, as well as hypoglycaemia and iatrogenic hypoadrenocorticism (eg, gastrointestinal signs, electrolyte derangements).5,6,9,18,20,21,32 Laparoscopic adrenalectomy for resection of non-invasive unilateral adrenocortical tumours has been described, but complications including haemorrhage and the necessity to convert to open surgery were common.26,72
Approximately 50–70% of cats achieve a greater than 1 year survival time after adrenalectomy, with most mortality occurring in the first week postoperatively.5,6,9,18,20,21,26,32,35,38 Clinical signs resolve 1–3 months after successful surgery (Figure 11). Successful long-term outcome following bilateral adrenalectomy is largely dependent on client compliance with corticoid supplementation. Case series of cats undergoing adrenalectomy for various types of adrenal tumours identified that anaesthesia time greater than 4 h was a negative prognostic factor; male gender and preoperative hypokalaemia were positive prognostic factors (ie, presence of an aldosterone-rather than cortisol-secreting tumour); and adrenal tumour size and the side the tumour was located on were not associated with outcome.20,73
Hypophysectomy
Microsurgical transsphenoidal hypophysectomy for PDH has been described in nine cats with pituitary adenomas.3,4,27 Complications included oronasal fistula, soft palate dehiscence, transient reduction in tear production and recurrence of HAC due to pituitary remnants.
Six cats showed resolution of HAC and three achieved diabetic remission. However, three cats died from concurrent disease 2 weeks to 6 months postoperatively, one died after 8 months from surgical complications and two died unexpectedly after 2 weeks and 5 months, respectively. Only three cats survived for more than 15 months.3,4,27
Careful peri- and postoperative management is required to avoid complications including hypoadrenocorticism, hypothyroidism and diabetes insipidus. Perioperative management involves frequent monitoring of electrolytes and osmolality, and treatment with hydrocortisone (1 mg/kg IV q6h) and desmopressin (one ocular drop q8h of 0.01% solution). 3 Postoperative management involves monitoring of tear production and treatment with cortisone acetate (1 mg/kg PO q12h), levothyroxine (10 pg/kg PO q12h) and desmopressin. 3 The cortisone dose is reduced to 0.25 mg/kg PO q12h over several weeks. Desmopression is tapered off over several weeks, and monitoring of biochemistry, total thyroxine and UCCR is scheduled every 4–6 months.
Although hypophysectomy can be curative, availability of surgical expertise is limited in many countries and successful long-term outcome is dependent on client compliance with postoperative treatment and monitoring.
Radiation
Radiation therapy for PDH generally involves multiple fractionated treatments. Availability of a gamma knife or a stereotactic capable linear accelerator can reduce this to a single or low number of treatments. Small numbers of cats with HAC treated with radiation therapy have been reported, often as part of larger case series involving non-functional or other types of functional pituitary tumours such as those causing acromegaly.6,19,29,35,40
Some cats have shown improvements in clinical signs of HAC after radiation therapy. 40 Cats with concurrent diabetes mellitus may have reduced insulin requirements or enter diabetic remission following treatment.19,35,40 Cats with neurological signs secondary to a pituitary mass typically show an improvement in neurological signs over weeks to months.29,40
Radiation side effects can be early or late in onset including epilation, fur depigmentation, otitis externa, brain necrosis, cataract development and hearing loss.29,40 Side effects may be manageable with anti-inflammatory doses of prednisolone but some, such as late-onset brain necrosis, are irreversible.

Cats that respond to treatment can have prolonged survival times of 1–2 years.6,29,35,40 Access to radiation therapy is currently limited in many countries and use of fractionated radiotherapy involves multiple general anaesthetics with potentially increased risk in debilitated patients.
Iatrogenic hyperadrenocorticism
Although considered uncommon, cases of clinically significant iatrogenic HAC are documented in cats and may be under-recognised.74–78 Most reports involve cats treated with long-term or high dose corticosteroids including prednisolone, methylprednisolone acetate, triamcinolone and dexamethasone. Clinical signs and clinical pathology findings are the same as for spontaneous HAC.74–77
ACTH stimulation testing is diagnostic, with lack of stimulation seen. Endogenous ACTH levels would be expected to be low. Hepatic histopathology is consistent with steroid hepatopathy with hepatocyte swelling from glycogen accumulation, and the adrenal glands atrophy.44,76 Gradual tapering off of corticosteroids is recommended, with resolution of abnormalities typically occurring over a few months.44,75,76

Sex steroid-producing adrenocortical tumours
HAC can occur secondarily to adrenal tumours that secrete other hormones, or their intermediates, with glucocorticoid activity (eg, progesterone), either alone or in combination with other sex steroids and/or aldosterone.20,35,70,79–88 Although uncommon overall, sex steroid-producing tumours appear to be relatively more common in cats than in dogs.
Paraneoplastic aberrant steroid synthesis pathways or enzyme deficiencies result in proximal hormone precursors accumulating or being redirected to other pathways.80,81,83 Progesterone is a precursor hormone for androgens, oestrogens, aldosterone and cortisol. A tumour secreting aldosterone may also secrete increased levels of aldosterone intermediates such as progesterone due to an enhanced biosynthetic pathway (see box above).
Progesterone competes with cortisol for cortisol-binding protein and with androgens and oestrogens for sex hormone-binding protein. High progesterone can therefore result in high levels of unbound cortisol and sex hormones. Progestogens are also glucocorticoid agonists and have long-lasting suppressive effects on the hypothalamic-pituitary-adrenal axis. Their intrinsic insulin-antagonistic activity may contribute to concurrent diabetes mellitus. Other adrenal gland hormones may also have intrinsic glucocorticoid activity and affinity for glucocorticoid receptors and cortisol-binding protein.
Depending on the hormones secreted by the tumour, affected cats may develop clinical signs similar to hypercortisolism and some may have concurrent clinical signs of hyperaldosteronism; for example, hypokalaemic polymyopathy and hypertension.35,70,79–81,83 In some cats, clinical signs may be predominantly attributed to one hormone despite hypersecretion of multiple hormones. 83 Overproduction of sex steroids without glucocorticoid effects has also been reported, resulting in clinical signs of oestrus or development of male secondary sexual physical characteristics in neutered animals; for example, urine spraying, aggression, skin thickening and penile spines.72,82,84–86
Reported feline sex steroid-producing adrenal tumours are listed in Table 4. In some cases complete sex steroid panels were not performed and tumours may have been hypersecreting additional hormones.70,72,80,83 Affected cats usually have low serum cortisol levels on ACTH stimulation tests despite clinical signs suggestive of hypercortisolism.70,79,81–83 Possible causes include a lack of enzymes within the adrenal tumour to convert progesterone to cortisol, or hypothalamic-pituitary-adrenal axis suppression by other hormones. Cats with sex steroid-producing adrenal tumours typically have high basal hormone levels and post-ACTH stimulation measurements may not be required for diagnosis.20,70,79–85,88
Feline sex steroid-producing adrenal tumours reported in the veterinary literature

y = years old; MN = male neutered; DSH = domestic shorthair; FN = female neutered; NR = not recorded
Sex steroid-producing adrenocortical tumours can be challenging to diagnose, especially when clinical signs are due to multiple hormone types, and thus may be underrecognised. One study detected marked elevations in corticosterone and progesterone in some cats with marked elevations of aldosterone (>3000 pmol/l) on sera submitted for aldosterone assay. 89 An increased clinician index of suspicion and use of adrenal hormone panels is required for diagnosis. Limitations include access to reference laboratories and lack of hormone reference intervals for cats. Adrenalectomy is the treatment of choice and has led to resolution of clinical signs and return of sex steroid hormone concentrations to within reference intervals in weeks to months.70,79,81–85
Key Points
While HAC in cats has many similarities to canine HAC, awareness of key differences is important for correct diagnosis and management.
Concurrent diabetes mellitus, which is often insulin resistant, is present in most but not all cats with HAC. Unlike insulin-resistant diabetic cats with acromegaly, HAC is more often associated with weight loss rather than weight gain.
The effects of hypercortisolism on the integument of cats with HAC are dramatic, resulting in extremely fragile skin that is prone to tearing (tears reported in a third of cases). This can impact on the choice of screening tests and therapy, especially in uncooperative cats, since restraint for even relatively non-invasive procedures, such as venepuncture, may be problematic.
Secondary infections are common in cats with HAC, and can be severe.
The prognosis for cats with HAC left untreated is poor. Unfortunately, the response of feline HAC to medical therapy is inferior compared with canine HAC.
Adrenalectomy and transsphenoidal hypophysectomy are the only curative treatment options, but appropriate patient selection is essential to achieve the best outcomes.
Adrenal sex steroid-producing tumours are relatively more common in cats than dogs, and these tumours may co-secrete multiple hormones.
Footnotes
Acknowledgements
The authors wish to thank Dr Peter Bennett and Dr Christine Griebsch for review of the draft article.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
