Abstract
Objectives
The primary objective of this study was to retrospectively assess the diagnostic utility of feline renal fine-needle aspiration cytology by assessing diagnostic yield, cytologic characteristics and diagnostic accuracy. The secondary objective was to characterize ultrasonographic features of sampled kidneys to determine if they influenced diagnostic yield.
Methods
Slides, images and patient data were collected from the University of Minnesota Veterinary Medical Center database. Slides were designated as diagnostic or non-diagnostic. Non-diagnostic slides were used in calculating diagnostic yield and excluded from other analysis. Slides were evaluated for cytologic characteristics and assigned a single primary diagnosis. Ultrasound still images were evaluated for descriptive characteristics and characteristics of specific lesions were described. Cases with confirmatory testing were used to determine diagnostic sensitivity, specificity and positive and negative predictive values for detecting neoplasia.
Results
Of 96 cytologic submissions available for review, diagnostic yield was 68%; 48% of samples were at least moderately cellular. Of 87 cases with ultrasound data, kidneys showing subcapsular renal infiltrate, diffuse renal enlargement without pelvic dilation and infiltrative/nodular change were more likely to yield diagnostic samples. Of 12 confirmed cases, cytology was 100% sensitive and specific for the detection of neoplasia (four round-cell tumors and two carcinomas). Three cases with non-neoplastic histologic diagnoses were considered cytologically normal, two incorrectly diagnosed the pathology present, and one correctly diagnosed the pathology. While some imaging characteristics were more commonly seen in neoplastic vs non-neoplastic lesions, the sample size was insufficient for definitive correlation.
Conclusions and relevance
This is the first major analysis of feline ultrasound-guided renal fine-needle aspiration cytology. This technique generates adequate samples for interpretation at rates comparable to other soft tissues and is most useful in the diagnosis of neoplasia. Some imaging characteristics are indicative of the likelihood of obtaining an adequate sample for cytologic interpretation.
Introduction
Renal disease is common in cats with a range of causes and modes of progression. Urinalysis and biochemical indicators are often used to screen for renal disease but are not specific for particular etiologies. Chronic kidney disease (CKD) is most common in geriatric cats, with one retrospective study showing that 53% of cases occurred in animals over 7 years of age. 1 It is usually characterized by small, irregular kidneys with histopathologic evidence of tubulointerstitial inflammation and fibrosis. 2 Although cytology is generally considered to be useful in the evaluation of many organ systems, the value of tissue microscopy, including both cytology and histopathology, in cats with CKD has been questioned based on assertions that the results are unlikely to alter prognosis, treatment or outcome, nor to identify an underlying cause in most cases.3,4 In addition, cats with renal disease may have comorbidities predisposing to hemorrhagic complications, including coagulopathy and hypertension, although reported rates of complications are relatively low.3,5 Cytology has been invoked as a less invasive and rapid alternative to kidney biopsy, with suggested indications including renal mass lesion, renomegaly and specifically to evaluate for renal lymphoma, but supporting data in the literature are sparse.6,7
Imaging, especially ultrasound, can be important in the evaluation of feline renal disease and is routinely utilized for the collection of both cytology and histopathology samples.8,9 Ultrasound facilitates identification of focal, multifocal and diffuse infiltrative disease (lymphoma, amyloidosis, lymphoplasmacytic infiltrate, etc), the underlying pathology of which can be ranked in terms of relative likelihood based on ultrasonographic interpretation and pre-test probability of the various diseases; it is also used to guide tissue sampling.10–13 Ultrasound can help define whether disease originates in the renal pelvis, parenchyma or in the intervening interface (tissue space between the outer margin of the pelvic epithelium and the renal parenchyma).8,9,11,12 In cats with kidneys lacking detectable architectural abnormalities but with abnormal echointensity, ultrasound also provides information about parenchymal infiltrates ranging from round-cell neoplasia through inflammatory disease and fibrosis – keeping in mind that hyperechoic cortices can be a result of pathology, or a normal aging change in cats due to fat deposition.9,10 However, while ultrasound abnormalities may be recognized, there are only a limited number of diagnoses achievable using ultrasound alone, including hydronephrosis, polycystic renal disease and end-stage kidneys. All other causes of renal disease require microscopy to permit a specific diagnosis. It is at this point this study is focused.
The current literature on feline renal cytology lacks large data-based studies evaluating its clinical use, especially in the context of imaging findings. Therefore, the primary purpose of this study was to evaluate the diagnostic features of feline renal ultrasound-guided fine-needle aspiration (FNA) cytology. To this end, we calculated the overall diagnostic yield (percentage of slides with adequate material of adequate quality to yield a cytologic diagnosis) and described the common cytologic characteristics of ultrasound-guided FNA renal samples in cats. The second objective was to characterize the ultrasonographic features of sampled kidneys to determine if specific characteristics influenced the diagnostic yield and what imaging features were seen in round cell vs carcinoma vs non-neoplastic lesions. Finally, we evaluated the diagnostic accuracy of cytology for the detection of neoplasia and non-neoplastic lesions as compared with histology or other supportive testing. To ensure diagnostic reporting of sufficient quality, we used the Standards for Reporting of Diagnostic Accuracy (STARD). 14 This 25-item checklist helps assure the internal and external validity of data while avoiding bias, and is meant to assist readers in assessing these parameters. It is also meant to improve the accuracy and completeness of reporting.
Materials and methods
Case selection and medical records review
The medical record database of the University of Minnesota Veterinary Medical Center was searched to identify all cats with renal cytology evaluated between January 2005 and June 2014. Cases were excluded if cytology slides were not available for review. Medical records were reviewed by one of the authors (CAM) for kidney sampled, breed, sex and age. If available, histopathology reports were reviewed and the reported histopathologic diagnoses recorded. Cases with non-diagnostic histopathology reports or cytology results were excluded from cytology:histopathology correlation analysis.
Review of cytology samples
All available archived Wright–Giemsa-stained cytology slides were reviewed for a consensus diagnosis by two authors (CAM and LCS). One author (LCS) is a board-certified veterinary clinical pathologist. Both reviewers were aware of signalment and imaging findings but were blinded to histopathology results. All slides were evaluated for total nucleated cellularity, blood contamination and numbers of inflammatory, epithelial, round and mesenchymal cells, as well as the vacuolization of renal tubular cells. Each feature was scored according to a standardized semi-quantitative method in which 0 = not seen, 1 = rare or mild, 2 = moderate, 3 = marked and 4 = severe. Anisocytosis and anisokaryosis were scored as normal or increased; the nuclear:cytoplasmic ratio was described as normal or increased; and the average number and numerical range of nuclei and nucleoli per cell were recorded. Background material (proteinaceous fluid, necrotic debris, ultrasound gel, cytoplasmic fragments, extracellular matrix and cholesterol crystals), hemorrhage (erythrophagocytosis, iron pigment, hematoidin), mitotic figures, infectious agents and glomeruli were noted.
Using the quantity and quality of cytologic elements (including cellular and non-cellular components), slides were categorized as diagnostic or non-diagnostic depending on whether there was sufficient material to render a cytologic interpretation. Diagnostic slides were assigned one of the following diagnoses: inflammation (suppurative or mixed), neoplasia (epithelial, mesenchymal, round cell, neuroendocrine or anaplastic), hyperplasia, necrosis, cyst, hemorrhage or no cytologic abnormalities. Inflammation was categorized as suppurative when >80% of inflammatory cells were neutrophils, while mixed inflammation displayed <80% neutrophils accompanied by other inflammatory cells. Neoplasia was categorized as epithelial, mesenchymal, round cell, neuroendocrine or anaplastic, and as malignant or benign based on the presence or absence of criteria of malignancy (anisocytosis, anisokaryosis, variable nuclear:cytoplasmic [N:C] ratio, nucleoli, bizarre mitotic figures), consistent with routine diagnostic cytopathology standards.
For the purposes of calculating diagnostic accuracy, cytology results were compared with histopathology or other confirmatory diagnostic results. Histology was performed on samples collected via necropsy, ureteronephrectomy or biopsy of other tumors deemed to be disseminated and thus representative of the renal mass.
Review of ultrasound studies
Archived still ultrasound images were reviewed by two authors (CAM and DAF). One author (DAF) is a board-certified veterinary radiologist. The time between FNA sampling and imaging was recorded in each case. The typical protocol for renal aspiration at the Veterinary Medical Center at the University of Minnesota is as follows: full bilateral renal ultrasound as part of a complete abdominal scan, followed by skin and transducer aseptic preparation. Real-time guidance of a 22 G 1.5 inch (or spinal) needle through the path of least tissue resistance is executed followed by rapid forward and reverse thrusts of the needle through the region of interest, with or without negative syringe pressure on the needle while in the region of interest or both depending on the radiologist. If more than one aspirate was performed, color Doppler was used to assess for hemorrhage prior to and following any subsequent aspiration, or at the discretion of the radiologist for single aspirate procedures. If sedation is required one of the following protocols is used, per the clinician’s discretion: butorphanol (0.2 mg/kg IV) alone, or butorphanol (0.1–0.2 mg/kg IV) and midazolam (0.1–0.2 mg/kg IV).
When examining ultrasound still images, authors were only aware of patient signalment, and were blinded to both cytologic findings and histopathologic diagnoses. Kidney ultrasound images were evaluated for size, fluid surrounding the kidney, renal border shape (irregular/regular, symmetric/asymmetric), cortex echogenicity compared with liver and spleen, medullary echogenicity compared with cortex, renal pelvis echogenicity (shadowing, complex fluid, stones), corticomedullary interface (defined as abnormal if unclear due to increased medullary echogenicity), the presence of renal pelvis dilation (only dilation ⩾0.3 cm noted), the presence of masses, description of any masses present (size, echogenicity) and other abnormalities or comments, such as subcapsular renal infiltrate. If a mass on one or both kidneys obscured them on ultrasound, a note was made and no further analysis was performed.
The ultrasound findings were evaluated in the context of categorization of the cytology sample as diagnostic vs non-diagnostic to determine the diagnostic yield correlated with each imaging characteristic. Ultrasound features and renal cytology diagnoses were compared with available histopathologic diagnoses or other supportive diagnostic data (ie, monoclonal gammopathy diagnosed via electrophoresis) in the cases where such data were available to determine imaging findings characterizing round cell vs carcinoma vs non-neoplastic lesions (Figure 1). When comparing cytology and histology, histologic diagnoses were considered definitive.

Flow diagram of participants, illustrating the number of cases (individual animals) and submissions (samples from an individual animal) available for each type of data analyzed in the study
Statistical analysis
A χ2 analysis was used to determine if there were significant differences in diagnostic yield based upon the kidney sampled. The proportion of adequate cytology samples was determined by dividing the number of cases for which a cytologic diagnosis could be determined by the total number of cases with representative samples that were reviewed. The proportion of adequate samples was also calculated specifically for kidneys with ultrasound features that were identified in ⩾5 cats. Because of the small numbers of cases with histologic or molecular confirmation, the calculations for diagnostic accuracy of cytology were restricted to the detection of neoplasia of any type vs a non-neoplastic process. Diagnostic sensitivity was determined as follows: number of true positives/(true positives + false negatives). Diagnostic specificity was determined using the following formula: number of true negatives/(true negatives + false positives). Positive predictive value (PPV) was determined using the following formula: number of true positives/(true positives + false positives). Negative predictive value (NPV) was determined using the following formula: number of true negatives/(true negatives + false negatives). Descriptive characterization of the ultrasound appearance of round-cell tumors, carcinoma and non-neoplastic lesions was performed by calculating the percentage of cases with a particular ultrasonographic feature evaluated in the study.
Results
Study population
In total, 101 patients yielding 104 cytology submissions were identified: one patient had four submissions over the course of 1 day in sampling the left kidney, the right kidney, the left renal pelvis and a renal cyst. Eight cases were excluded owing to an inability to retrieve cytology samples, leaving a total of 93 patients with 96 submissions. Between one and 10 slides were available for review for each case. Non-diagnostic samples were included for the calculation of diagnostic yield, but were not used for other analyses. Thirteen breeds were represented in this study, including: 64 domestic shorthairs, nine domestic longhairs, seven domestic mediumhairs, four Siamese and 12 other purebreds. One cat was an intact male, 37 were spayed females and 58 were neutered males.
Ultrasonographic findings and diagnostic yield
Of the 96 cytology submissions, 87 had renal ultrasound data available: six cats did not have ultrasound data available and the cat with four submissions had ultrasound performed only once. Of the 87 ultrasound studies, 83 were performed the same day as the FNA, whereas two were performed 1 and 2 days prior to cytologic sampling, respectively. All 87 ultrasounds had still images for review. In one, the right kidney was sampled and it was visible as only a large central cavitation, so only the presence/absence of surrounding fluid and the renal boarder shape were assessed. In five cases, both kidneys were sampled so the ultrasound characteristics of both kidneys were included. The number of cases with each ultrasound characteristic varied from five to 44. When stratified according to the presence of specific ultrasonographic abnormalities, diagnostic yield varied from 50–100% (Table 1); the highest yield was in kidneys with subcapsular renal infiltrate (100%), diffuse renal enlargement (any echogenicity) without pelvic dilation (100%), and normal or enlarged infiltrative/nodular kidney (84%).
Diagnostic cytology yield (proportion of adequate samples) by ultrasound characteristic in 87 cats with both renal aspirates and ultrasound studies

Table is sorted by frequency of observation; only features with n ⩾5 are listed. Cases may be counted in more than one ‘ultrasound characteristic’ category. ‘Enlargement’ means renal length was three times the length of the second lumbar vertebra. Infiltrative/nodular kidney is defined as changes in the renal parenchyma that are non-uniform, may cause an irregular surface and could not be defined as specific masses or measurable nodules
Cytologic findings
Of the 96 cytology submissions, 64 were diagnostic, giving a diagnostic yield of 68%. The aspirated kidney was specifically noted for 79/96 cytology submissions. The left kidney (52/79) was sampled significantly more frequently (P <0.0005) than the right kidney (20/79). Both kidneys were sampled in seven cases. Of diagnostic cases (n = 64), the cytology samples from 12.5% were extremely (4+) cellular, 26.6% were markedly (3+) cellular, 32.8% were moderately (2+) cellular, 26.6% were minimally (1+) cellular and 1.6% (one case) was acellular but classified as diagnostic owing to the presence of sufficient necrotic debris to classify the pathologic process as ‘necrosis’. Of the non-diagnostic cases (n = 32), all were acellular. All but two diagnostic samples had some degree of blood contamination; however, indicators of mild hemorrhage (erythrophagocytosis, hemosiderin, hematoidin, etc) were only observed in a single case. Renal tubular epithelial cells were noted in 46 cases, typically in mild (19%) to moderate (18%) numbers. Severe (4+) numbers of renal tubular cells were only seen in 3% of cases and marked (3+) numbers in 14% of cases. Tubular cells occurred individually and in clusters as round-to-polyhedral cells characterized by moderately abundant basophilic cytoplasm often containing lipid droplets and a single, round, eccentrically oriented nucleus (Figure 2). No glomeruli were observed. Rod-shaped bacteria were identified in one case, which had concurrent mixed inflammation; Escherichia coli grew in a urine culture. Background material was seen in a small percentage of cases and included necrotic debris (14%), extracellular matrix (8%), ultrasound gel (3%), and proteinaceous material, mucin and collagen fibers (2% each).
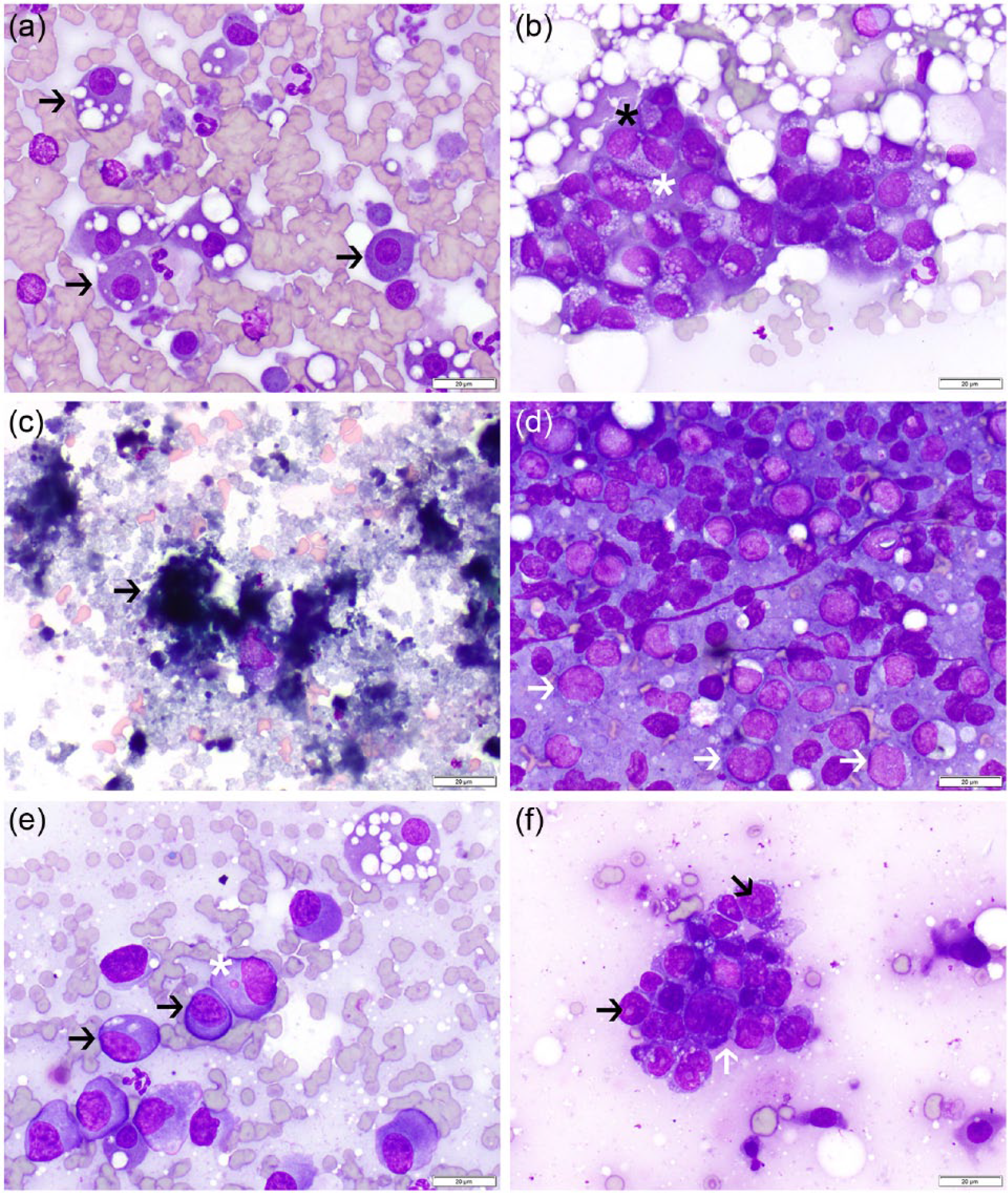
(a) Normal feline renal tubular epithelial (black arrows) cells are often individualized cells with a moderate amount of basophilic cytoplasm and single, round, polarized nuclei. Lipid droplets are frequent and may distend the cytoplasm. Dark granules are present in some cells. Cytoplasmic fragments can be noted in the background. (b) Atypical renal tubular epithelial cells in a histologically confirmed case of feline infectious peritonitis. Cells are disorganized and exhibit moderate anisocytosis, anisokaryosis (white asterisk), high nuclear:cytoplasmic (N:C) ratios, binucleation (black asterisk) and an atypical vacuolar pattern. (c) Amorphous basophilic debris (black arrow) consistent with necrosis in the kidney of a cat histologically diagnosed with subcapsular hemorrhage and necrosis. (d) A predominance of large atypical lymphocytes (white arrows) is seen in smears from a histologically confirmed case of renal lymphoma. (e) Large, atypical round cells (black arrows) characterized by abundant amphophilic cytoplasm with eosinophilic margins and round-to-oval nuclei with stippled chromatin in a cat with a monoclonal gammopathy. One cell contains a microsatellite nucleus (white asterisk). Two normal renal tubular cells contain variable numbers of lipid droplets, and scant lipid is present in the background. (f) A cohesive cluster of disorganized atypical epithelial cells in a cat with histologically confirmed renal carcinoma. Cells exhibit marked anisocytosis and anisokaryosis with a high N:C ratio and scant vacuolated basophilic cytoplasm (white arrows). One or two large, round nuclei are present in each cell (black arrows) (Wright–Giemsa, × 500 oil immersion)
Of the 64 diagnostic FNAs, 21 (33%) were cytologically diagnosed as neoplasia, including 12 cases of round-cell tumors, six carcinomas, two sarcomas and one anaplastic malignancy. Of these cases, four round-cell tumor cases (three lymphomas and one plasma-cell tumor) and two carcinomas had histologic or clinical confirmation. The definitive round-cell tumors were characterized by marked (3+) to severe (4+) cellularity, with mild (1+) to marked (3+) blood contamination. One case had concurrent mild (1+) mixed inflammation. Two of the cases had normal renal tubular cells present, whereas two were composed entirely of neoplastic cells. One had mitotic figures. The plasma cell tumor was characterized by marked nuclear pleomorphism (Figure 2). The carcinomas showed mild (1+) to marked (3+) cellularity, moderate (2+) blood contamination and necrotic debris. One also had extracellular matrix material. One case showed mild (1+) mixed inflammation concurrently present. Both cases had only neoplastic cells, of which displayed marked (3+) to severe (4+) anisocytosis, moderate (2+) to severe (4+) anisokaryosis, normal to increased N:C ratio, single nuclei and nucleoli.
Of the remaining 43 diagnostic quality samples, renal tubular hyperplasia (n = 13; 21%) was the most common finding. A primary diagnosis of inflammation was made in four (6%) cases. Mixed inflammation was identified in three of these, one of which was associated with renal tubular hyperplasia and necrosis and one case had 1+ bacteria (rods) present. One case was diagnosed with suppurative inflammation only with no cytologic evidence of etiology. Necrosis was diagnosed as the primary process in one case and no cytological abnormalities were found in 25 cases. Figure 2 shows the cytologic features of four confirmed diagnoses.
Diagnostic accuracy of cytology
Sixteen cases had confirmatory testing, 15 of which had correlate histology reports (Figure 1). One cat had monoclonal gammopathy diagnosed via electrophoresis and a chemotherapy responsive subcutaneous round-cell tumor, which were considered sufficiently clinically supportive of the cytologic diagnosis of round-cell tumor in the kidney to be included in cytology:histopathology correlation. Of the 15 histology cases, four had non-diagnostic cytologies that were excluded from analysis, resulting in 12 cases with confirmatory testing that were used in evaluation of cytology:histopathology correlation. Of these 12, 11 had biopsy samples that were collected via necropsy (n = 9), ureteronephrectomy (n = 1) or biopsy of other tumors clinically deemed to be disseminated and thus representative of the renal mass (n = 1). Biopsies were collected between 24 h and 227 days from FNA with 4/11 collected within 48 h (Table 2).
Cytologic and histologic diagnoses for cases that had diagnostic cytology samples and confirmatory histology or sufficient clinical follow-up

Cases in which disseminated disease was present and histologic diagnosis was inferred via biopsy of other tissue
NA = not available
The sensitivity, specificity, PPV and NPV of cytology for the diagnosis of neoplasia was 100%. When specific tumor subtypes were evaluated, the sensitivity, specificity, PPV and NPV for lymphoma and carcinoma were also all 100% (confirmatory testing for sarcoma and anaplastic malignancy were not available). For non-neoplastic processes the sensitivity was 16.7%, the specificity was 100%, the PPV was 100% and the NPV was 54.5%.
Although not formally included in calculations of diagnostic accuracy, urine culture was positive for E coli for one case in which a fine-needle aspirate of the renal pelvis was cytologically diagnosed as mixed inflammation. However, no etiologic agents were identified in the cytology sample.
Renal ultrasound characteristics by histologic diagnosis
The frequency of ultrasound findings in cases with confirmatory testing (n = 14) were recorded and examined in order to determine if specific ultrasound findings were characteristic of round-cell tumor, carcinoma or a non-neoplastic process (Table 3). Thirteen were histology cases (three of which had non-diagnostic cytology samples) and one was a case with sufficient clinical follow-up (Figure 1).
Ultrasonographic features of round-cell tumor, carcinoma and ‘non-neoplastic’ cases confirmed histologically or by clinical follow-up, listing only observed features

Data are %. ‘Enlargement’ means renal length was more than three times the length of the second lumbar vetebra. Infiltrative/nodular kidney is defined as changes in the renal parenchyma that are non-uniform, may cause an irregular surface, and could not be defined as specific masses or measurable nodules
Of the three cases diagnosed as round-cell tumor that had corresponding imaging, two were hypoechoic (less echogenic than the liver) and two were infiltrative/nodular in appearance. One or more of the following ultrasound findings were seen in each of the three cases, not necessarily simultaneously: bilateral hypoechoic masses, subcapsular renal infiltrate, diffuse renal enlargement (confirmed by radiographs) with pelvic dilation, pelvic dilation, unclear corticomedullary interface and perinephric fluid.
Of the two cases histologically diagnosed as carcinoma that had corresponding imaging performed, both had renal enlargement (confirmed by radiographs) without pelvic dilation and unclear corticomedullary interface. One or more of the following ultrasound findings were seen in each of the two cases, not necessarily simultaneously: unilateral mixed echogenicity mass, hypoechoic (less echogenic than the liver), hyperechoic (iso- or hyperechoic to the spleen) and subcapsular renal infiltrate.
Non-neoplastic kidneys represented a wider variety of disease processes and therefore showed greater variety in ultrasound findings. No non-neoplastic kidneys showed enlargement, while 20% (1/5) of neoplastic cases showed it in conjunction with pelvic dilation and 40% (2/5) of neoplastic cases showed it without pelvic dilation. Twenty-two percent (2/9) of non-neoplastic kidneys had unilateral or bilateral masses, while 40% (2/5) of neoplastic kidneys had unilateral had bilateral masses. Only 22% (2/9) of non-neoplastic kidneys were hypoechoic, while 60% (3/5) of neoplastic kidneys were hypoechoic.
Discussion
The overall diagnostic yield of feline renal FNA cytology was 68%, which is comparable to FNA cytology of canine kidneys (71%), feline non-mammary cutaneous and subcutaneous masses (83%), and feline lymph nodes (85%).15–17 Previous publications have suggested that a major limitation of renal FNA cytology is the high frequency of minimally cellular samples. 6 Our study found that only 33 cases (34%) were acellular and of those 32 (33%) were interpreted as non-diagnostic. Furthermore, of all slides examined, 8% were markedly cellular, 22% were moderately cellular and 18% were minimally cellular. Although pre-screening submission bias and variations in aspirator expertise cannot be excluded, these data show that renal cytology has the potential to provide diagnostic information at rates comparable to other tissues routinely undergoing cytologic evaluation.
Thus far no evidence-based guidelines have been published regarding radiology-based indications for the practice of feline renal FNA cytology. A previous report has suggested that FNA is most useful for solitary or multifocal mass lesions, marked echotexture changes or, in cases of renomegaly, absent hydronephrosis. 6 The literature has also suggested that FNA is least useful for small or shrunken kidneys, although no definitive data were provided to support this. 6 Our data show that diagnostic yield varies greatly across ultrasonographic findings, with subcapsular infiltrate, diffuse renal enlargement (any echogenicity) without pelvic dilation and normal or enlarged infiltrative/nodular kidneys providing the highest diagnostic yield (100%). Somewhat unexpectedly, the ultrasonographic lesion with the lowest diagnostic yield (50%) was the presence of a mass. The presence of pelvic dilation also had a low diagnostic yield at 56%.
In comparison to feline renal FNA, a study of canine renal FNA showed 27% of samples to be acellular and non-diagnostic, with 32% severely cellular and 35% moderately cellular. 15 Furthermore, canine ultrasonographic findings associated with the highest diagnostic yield were found to be unclear corticomedullary interface (90%), pelvic dilation (90%) and an infiltrative or nodular appearance (86%). The ultrasound findings associated with the lowest diagnostic yield in the dog were iso- or hyperechoic medulla compared with cortex (60%), hyperechoic kidney (iso- or hyperechoic to spleen, normal or enlarged; (71%) and bilateral isoechoic/mixed echogenicity mass (71%). Taken together, these data suggest that renal FNA has a slightly higher diagnostic utility in the dog. Additionally, these findings may reflect that dogs and cats are predisposed to different kinds of renal pathology, considering that different ultrasound appearances have different diagnostic yields in the dog vs the cat. However, more study, including prospective work, is necessary before any definitive conclusions can be made.
Although numbers of confirmed diagnoses were low, our preliminary results suggest that renal FNA is most useful in diagnosing neoplasia: we calculated 100% sensitivity, specificity, PPV and NPV for the neoplastic lesions in our study. We also found that renal FNA is poor at ruling out non-neoplastic processes, with 16.7% sensitivity and 54.5% NPV; three cases were considered cytologically normal but given non-neoplastic histologic diagnoses in our study. However, we found that renal FNA may be more effective at ruling in non-neoplastic processes than ruling them out, with 100% specificity and 100% PPV in our sample set. Given the propensity of cats to develop fibrotic kidneys in response to a variety of insults it is not surprising that cytologic diagnosis of non-neoplastic lesions is less reliable; we also found that all four of the cases that had histologic diagnoses but non-diagnostic cytologies had non-neoplastic histologic diagnoses.1,2,5 Nonetheless, these findings indicate that a combination of cytologic and ultrasonographic characteristics provides a basis for determining which patients will likely benefit from histologic sampling vs clinical and imaging follow-up.
Regarding standard neoplastic cytologic diagnostic criteria, all cases of histologically confirmed carcinoma had marked (3+) to severe (4+) anisocytosis and moderate (2+) to severe (4+) anisokaryosis, with one case showing an increased N:C ratio. Criteria of malignancy were not assessed in round-cell tumors, which were instead diagnosed by the presence of abnormally high numbers of a uniform population of round cells in the sample, usually of large size, indicative of immaturity. There was variation in ultrasound appearance among cases of histologically confirmed neoplastic (round-cell tumors and carcinomas) and non-neoplastic processes. While these data suggest some ultrasonographic findings may be more prevalent in some specific diagnoses than others, the small sample size limited any definitive correlation between ultrasound findings and histologic diagnosis and further study is indicated.
Previous study of canine renal FNA found it to have a 78% sensitivity and PPV for neoplastic lesions, but a 50% specificity and NPV. 15 Aspirates from non-neoplastic lesions had a sensitivity of 50%, PPV of 29%, specificity of 77% and NPV of 89%. These data were calculated using 19 cases with histopathology confirmation and nine with PCR for antigen receptor rearrangement (PARR) confirmation, making the data set more robust than our feline data. Of additional note, two canine cases histologically diagnosed as carcinoma were incorrectly cytologically diagnosed as non-neoplastic, whereas two cases histologically diagnosed as non-neoplastic were incorrectly cytologically diagnosed as carcinoma. In the dog, all histopathologically diagnosed cases of carcinoma had a mass present, whereas no non-neoplastic cases had a mass. However, no such relationship was found in our feline data, but it is notable that the feline sample size was much smaller.
The calculation of diagnostic accuracy in our study was limited by the small numbers of cytology cases with confirmatory testing. To increase this latter population, we considered using the PARR assay as a surrogate lymphoma diagnostic. However, in the light of the potentially low diagnostic yield with archived renal aspirates and the comparatively low sensitivity and specificity of this test, we elected to forgo it.15,18 Our study population also includes a case of feline multiple myeloma, which lacks a traditional tissue-based confirmatory diagnosis. Our decision to include this case was based upon ancillary diagnostics, which revealed a monoclonal serum protein and plasma-cell-rich cutaneous mass, and a positive response to systemic chemotherapy (ie, prednisone and cyclophosphamide). This approach was further supported by previous work reporting that lymphoproliferative disease is the most common cause of a monoclonal gammopathy. 19
Evaluation of the diagnostic accuracy of cytology in retrospective studies can be influenced by selection bias regarding which cases evaluated by cytology are then later evaluated by histopathology. 20 Of the 15 cases with histologic diagnoses, 13 were obtained at necropsy, one was based upon by the biopsy of tissues involved in a disseminated presumed neoplastic process, and only one was determined by direct renal biopsy. In future prospective studies, it would be ideal to recruit cases in which cytologic and histologic examinations were coincident and to include equal numbers of neoplastic and non-neoplastic lesions. Evaluation of kidney size as determined on abdominal radiographs and cytologic diagnostic yield would also be useful. Nonetheless, this is the largest study to date of cytology and imaging in cats documenting diagnostic recovery rate, common cytologic features and diagnostic accuracy of cytology, as well as how information from imaging studies informs cytologic interpretation.
Conclusions
Feline renal FNA and cytology is a useful diagnostic tool for assessing the cause of kidney disease. It yields diagnostic samples at a comparable rate to renal FNA in the dog and other soft tissue FNA in the cat. It is most sensitive for diagnosing neoplastic lesions (100%) and most specific for neoplastic (100%) and non-neoplastic lesions (100%). However, the sample size for these statistics was limited (n = 12) and further study is indicated.
By utilizing ultrasonographic findings in an individual case, clinicians can approximate the likelihood of acquiring a diagnostic renal FNA sample based on the ultrasound lesions seen. Specifically, kidneys with the following lesions resulted in a higher diagnostic yield compared with the average diagnostic yield of all renal FNAs: subcapsular renal infiltrate, diffuse renal enlargement without pelvic dilation, infiltrative/nodular appearance, hypoechoic kidney (less echogenic than liver) and unclear corticomedullary interface.
Footnotes
Author note
This article was presented, in part, in poster form at the Merial Veterinary Scholars Program, Ithaca, NY, USA, July 2014.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This project was funded in part by the Summer Scholars Program, internal funds from the College of Veterinary Medicine, University of Minnesota.
