Abstract
Objectives
The aim was to retrospectively evaluate the effects of acid-suppressant therapy in a population of cats with chronic kidney disease (CKD). The study objectives were to evaluate the effects of acid-suppressant therapy on clinicopathologic variables and progression of CKD over time.
Methods
The databases of two institutions were searched over an 11 year time span for cats fitting inclusion criteria for CKD. A total of 89 cats met the criteria for inclusion and were grouped according to either early (ie, stages 1–2) or advanced (ie, stages 3–4) CKD. Variables were statistically analyzed before and after treatment with either: (1) proton pump inhibitors (PPIs; n = 17), (2) histamine-2 receptor antagonists (H2RAs; n = 30), (3) combined acid-suppressant therapy (PPI + H2RA; n = 6) or (4) no acid-suppressant therapy (n = 36). Shapiro–Wilk testing and Q-Q plots were used to assess normality and variance, respectively. A complete randomized design with a mixed-effects repeated measures ANOVA was used to evaluate for differences in stage, treatment and time, as well as the interaction between these effects.
Results
A significant increase in blood creatinine concentration was found over time independent of severity of CKD and treatment group (P = 0.0087). A significant increase in blood sodium concentration (change of 3.12 mmol/l) was found independent of stage in cats receiving PPI therapy (P = 0.0109). A significant decrease in total blood magnesium (change of 0.15 mmol/l) was detected in two cats with early CKD receiving combined acid suppressants (P = 0.0025).
Conclusions and relevance
Results of this retrospective study suggest that cats with CKD receiving PPI therapy may develop alterations in blood sodium concentrations but do not experience more rapid progression of CKD.
Introduction
Proton pump inhibitors (PPIs; eg, omeprazole, pantoprazole) are superior to histamine-2 receptor antagonists (H2RAs; eg, famotidine, ranitidine) for the treatment of acid-related disorders such as gastroesophageal erosion, ulceration and hyperacidity in humans,1,2 and presumably, in cats.3,4 Short-term PPI administration is generally well tolerated; however, chronic PPI administration has been associated with severe electrolyte derangements (eg, hypomagnesemia, hypokalemia, hyponatremia),5–11 hypocobalaminemia,12,13 and alterations in blood calcium and magnesium homeostasis in humans.14–16 Recently, an association between chronic PPI use and increased risk of development or progression of chronic kidney disease (CKD) and increased incidence of acute on chronic renal insufficiency has been reported. 17 The development of any of these sequela would be concerning for our feline patients.
The effects of chronic omeprazole administration at doses suggested for the treatment of acid-related disorders in cats have only been evaluated in one pilot study. 18 Eight weeks of omeprazole administration resulted in serum hypergastrinemia and abrupt drug withdrawal-associated rebound gastric hyperacidity in cats. Despite this, clinical signs associated with these changes were not readily apparent. Moreover, no significant differences, compared with placebo, were found in blood concentrations of electrolytes, calcium, magnesium or bone homeostatic parameters when cats received omeprazole. While omeprazole was relatively well tolerated in this healthy cat population, no studies have evaluated an association of chronic PPI administration with adverse effects in cats with systemic disease. Cats with CKD represent a particularly important cohort to evaluate for several reasons. First, acid suppressants are a top medication administered to cats with CKD. 19 Additionally, CKD can induce electrolyte (eg, potassium, sodium, phosphorus and calcium) and parathyroid (eg, renal secondary hyperparathyroidism) alterations in cats. Thus, cats with CKD may experience more severe adverse effects as a result of chronic PPI administration compared with healthy cats.
The central objective of this study was to describe and compare the effect of PPIs, H2RAs, combined acid-suppressant therapy or no acid-suppressant therapy on clinicopathologic variables in cats with CKD over time. Our hypothesis was that cats receiving PPI therapy would have no changes in clinicopathologic variables vs those receiving H2RAs or no acid suppressants. This study is the first to retrospectively evaluate the effects of acid-suppressant therapy in a large population of cats with naturally occurring CKD.
Materials and methods
The electronic databases from the University of Tennessee College of Veterinary Medicine (UTCVM) and North Carolina State University College of Veterinary Medicine (NCSU CVM) were used to identify all cases of feline CKD from January 2005 to January 2016. The following terms were used to identify possible cases for study inclusion: anorexia, arterial hypertension, azotemia, CKD, chronic nephritis, dilatation renal pelvis, hypercalcemia, hypocalcemia, hyperparathyroidism, hyper- or hypokalemia, infarct kidney, mineralization, polydipsia, polyuria, proteinuria, protein-losing nephropathy, renal failure and vomiting.
Cats were classified as either CKD stage 1, 2, 3 or 4 based on the IRIS guidelines (http://iris-kidney.com/pdf/treatment-recommendation-cats.pdf), last modified in 2015 (ie, stage 1 [creatinine <1.6 mg/dl], stage 2 [creatinine 1.6–2.8 mg/dl], stage 3 [creatinine 2.9–5.0 mg/dl] and stage 4 [creatinine >5.0 mg/dl]). Initial creatinine concentrations only were used for staging purposes. Cats were not further sub-staged by hypertension or proteinuria. Cats were also classified according to the type of acid suppressant they were receiving (ie, group 1, PPI; group 2, H2RA; group 3, PPI + H2RA; group 4, no acid-suppressant therapy).
Data recorded for each cat included age, sex, type and duration of acid-suppressant administration (ie, less or more than 2 months of therapy), two sets of clinicopathologic values (eg, blood creatinine [University of Tennessee reference interval {RI} 0.9–2.0 mg/dl, NC State RI 0.7–1.5 mg/dl], potassium [University of Tennessee RI 2.5–4.6 mmol/l, NC State RI 3.5–5.1 mmol/l], sodium [University of Tennessee RI 141–147 mmol/l, NC State RI 148–155 mmol/l], chloride [University of Tennessee RI 114–124 mmol/l, NC State RI 114–124 mmol/l], total calcium [University of Tennessee RI 9.0–10.8 mg/dl, NC State RI 8.8–11.2 mg/dl], magnesium [University of Tennessee RI 0.69-0.97 mmol/l, NC State RI 0.78-1.0 mmol/l], platelet count, albumin and urine specific gravity [USG]), the presence or absence of hypertension, proteinuria and existing comorbidities. Clinicopathologic data collected from both academic institutions were collected on one analyzer per institution. For patients receiving acid-suppressant therapy, the initial set of clinicopathologic data represented baseline (ie, just prior to initiation of therapy from either the institution or referring veterinarian), whereas the second set was the last date available with complete data while still receiving therapy (groups 1–3) or not receiving acid-suppressant therapy (group 4). The presence of any other therapy that may have interfered with serum electrolytes or USG (eg, diuretics, angiotensin-converting enzyme inhibitors, beta-blockers, steroids and non-steroidal anti-inflammatory drugs), as well as the type of diet, therapeutic renal diet or other, were also recorded.
Cats were excluded if there was failure to document a minimum of two consecutive sets of USGs <1.035 and concurrent serum creatinine concentrations collected at least 2 weeks apart in accordance with IRIS staging guidelines. Cats were also excluded if they were receiving an acid suppressant but initiation and cessation of therapy could not be clearly determined from the record. Lastly, cats were excluded and eliminated from statistical analyses if they had two initial USGs <1.035 but subsequent USGs were ⩾1.035.
Statistical analysis
A mixed-effects repeated-measures ANOVA was performed in order to analyze subjects for changes in blood potassium, sodium, total magnesium, total calcium and creatinine concentrations. Rather than analyzing each stage separately, stages were grouped by severity of disease because not every treatment was represented in each stage. Subjects were classified as having either early (ie, stages 1–2) or advanced (eg, stages 3–4) CKD. Each dependent variable was analyzed for differences in stage, treatment and time, as well as the interaction between these effects in both groups. Stage and treatment were treated as between-subject factors and time was treated as a within-subject factor. In order to meet all statistical assumptions regarding normality or the residuals and equality of variances, a log transformation was necessary for all dependent variables except total calcium and sodium. Post-hoc analyses were performed to further investigate differences between significant effects. A Shapiro–Wilk W and Q-Q normality plots were used to evaluate normality of ANOVA residuals. Levene’s equality of variances test was used to evaluate each model for equal variances among treatment groups. A P value of <0.05 was considered significant for all analyses.
Results
Sample population
From January 2005 to January 2016, medical records of cats with suspect CKD from both institutions were analyzed. A total of 358 cases met the initial search criteria; 269 were eliminated as a result of incomplete records for the variables of interest or the lack of criteria for CKD (ie, cats were determined to have acute kidney injury based on a subsequent decrease in creatinine from initial diagnosis to the last time point). A total of 89 cats (62 cats in the IRIS stages 1–2 group and 27 cats in the IRIS stages 3–4 group) were included in the study. Stages 1, 2, 3 and 4 included nine, 53, 22 and five cats, respectively. Subjects from the UTCVM included nine, 43, four and one cats categorized as stages 1, 2, 3 and 4, respectively. The remaining cases were from the NCSU CVM and included zero, 11, 17 and four cats categorized as stages 1–4, respectively. A total of 40 (45%) cats were males and 49 (55%) were females. Cats were grouped as either <7 or ⩾7 years of age. Eighty-one (91%) of the cats were ⩾7 years of age and eight (9%) were <7 years of age.
Concurrent diseases
A total of 88 cats (98%) were documented as having one or more concurrent diseases. Comorbidities documented in this population of cats included cardiac disease (n = 36), gastrointestinal disease (eg, inflammatory bowel disease, small cell lymphoma; n = 17), concurrent upper and lower urinary tract disease (eg, nephroliths, ureteroliths, bladder calculi; n = 13), hyperthyroidism (n = 10), diabetes mellitus (n = 8), neoplasia (eg, splenic hemangiosarcoma, small-cell lymphoma, primary lung tumor, nasal tumor, transitional cell carcinoma; n = 6), idiopathic hypercalcemia (n = 5), chronic rhinosinusitis (n = 4), pancreatitis (n = 4), periodontal disease (eg, stomatitis, resorptive lesions, n = 4), allergic bronchitis (n = 3), dermatitis (n = 3), neurologic abnormalities (ie, vestibular disease, seizures, idiopathic epilepsy, suspect Guillain Barré syndrome; n = 3), osteoarthritis (n = 3), cholecystitis (n = 2), constipation (n = 2), corneal ulceration (n = 2), hepatic lipidosis (n = 2), obesity (n = 2), primary hyperparathyroidism (n = 2) and obstipation (n = 1). One cat each had multiple myeloma, megaesophagus, a mediastinal mass, a peri-renal pseudocyst and abdominal hernia.
Treatment groups
The number of cats within each stage receiving acid-suppressant or no therapy is outlined in Table 1. Of cats receiving some form of acid-suppressant therapy, a total of 34 (n = 34/53; 64%) were treated for a period of ⩾2 months’ duration, while 19 cats (n = 19/53; 36%) were treated for <2 months’ duration. Of cats receiving PPI therapy, 65% (n = 11/17) were treated for ⩾2 months’ time, with a median duration of 24 weeks (range 2–240 weeks). Nine of thirty cats (30%) were receiving H2RA therapy for ⩾2 months, with a median duration of 4 weeks (range 4–240 weeks). For cats receiving combined acid suppressants, 80% (n = 4/6) received therapy for ⩾2 months’ duration, with a median of 8 weeks (range 2–96 weeks). The type of PPI administered included oral omeprazole in all cats (n = 23) and intravenous pantoprazole in one cat with stage 3 CKD during hospitalization. The dose and frequency of PPI therapy, recorded in 19 cats, ranged from 1.0 mg/kg orally or intravenously q24h (n = 3) to q12h (n = 16). Omeprazole formulation, recorded in 20 cats receiving PPI therapy, included compounded omeprazole capsule (5.5 mg; n = 1) and fractionated omeprazole tablet (n = 19). The majority of cats receiving an H2RA were receiving famotidine (n = 32). Four cats with stage 2 CKD were receiving ranitidine. The dose and frequency of H2RA therapy ranged from 0.5–1.0 mg/kg orally or intravenously q12h. No specific formulations were recorded for H2RA therapy.
Cats in each IRIS stage receiving either proton pump inhibitor (PPI), histamine-2 receptor antagonist (H2RA), combination acid suppressant or no acid-suppressant therapy

Data are n (%)
Concurrent medications
A total of 26 (29%) cats were documented as receiving concurrent medications. Concurrent medications administered included atenolol (n = 9), angiotensin-converting enzyme inhibitors (eg, enalapril, benazepril; n = 11), furosemide (n = 2), prednisolone (n = 3) and Epikatin (Vetoquinol) (n = 1). Figure 1 illustrates concurrent therapies and IRIS stages of cats within each treatment group.

Concurrent therapies and IRIS stages of cats within each treatment group. ACE = angiotensin-converting enzyme; PPI = proton pump inhibitor; tx = treatment; H2RA = histamine-2 receptor antagonist
Dietary therapy
A total of 42 cats (47%) were documented as receiving a diet formulated for CKD (ie, therapeutic renal diet), with the remainder of cats either noted as receiving a non-renal diet (n = 6/89; 7%) or lacking information about diet in the records (n = 41/89; 46%). Of the cats receiving a renal diet, 24 (57%) were classified as having early stage CKD, whereas the remaining 18 (43%) were classified as advanced. In the 42 cats known to be receiving a renal diet, four cats were not receiving an acid suppressant, 11 cats were receiving a PPI, 23 cats were receiving a H2RA and four cats were receiving a combination of both. Commercial renal diets recorded included Purina NF (Nestlé Purina), Hill’s k/d (Hill’s Science Diet; Hill’s Nutrition) and Royal Canin LP (Royal Canin USA).
Variables statistically analyzed
For blood sodium, a marginally significant treatment by time interaction was observed, thereby indicating that sodium concentrations of cats change differently over time under the effects of each treatment (P = 0.0497). This was attributed to a change in sodium concentrations over time for cats receiving PPI therapy, which occurred regardless of CKD stage (Figure 2). Reported mean sodium values for cats receiving PPI therapy were 147.7 mmol/l at time 1 to 150.8 mmol/l at time 2 (P = 0.0109), which resulted in a time 2 value outside the RI for sodium for some cats. This represents a ~2% change over time in sodium concentrations for cats receiving PPI therapy. Four of 17 cats receiving PPI therapy were defined as hypernatremic according to their institution’s RI (n = 3/16 from NC State and 1/1 from the University of Tennessee). No additional changes over time were observed for cats receiving other treatments.

Mean sodium concentration increased over time for all cats receiving proton pump inhibitor (PPI) therapy, independent of stage. Time is represented on the x-axis as either time 1 (initial data) or time 2 (data collected following therapy), and the sodium concentration represented on the y-axis. Treatment groups are represented by the following symbols: PPI (circles), histamine-2 receptor antagonist (H2RA; upward pointing triangles), PPI and H2RA therapy (downward pointing triangles) and no acid-suppressant therapy (boxes). Symbols and error bars represent the mean ± SD. **P = 0.0109
A significant stage by treatment by time interaction was observed for blood total magnesium concentrations (P = 0.046). Post-hoc analysis demonstrated that total magnesium concentrations changed significantly over time in cats with stage 1–2 CKD that received both acid suppressants (n = 2 cats; P = 0.0025). This resulted in an untransformed mean blood total magnesium concentration of 1.0 mmol/l at time 1 vs 0.5 mmol/l at time 2. The mean time 2 concentration resulted in a value below the standard RI for total magnesium. Stage 3–4 cats receiving both acid suppressants did not show a significant change over time. Figure 3 depicts the change in total magnesium over time in cats receiving combined acid-suppressant therapy.

Total magnesium concentration decreased significantly over time for only early stage (1–2) cats with chronic kidney disease (CKD) receiving concurrent proton pump inhibitor and histamine-2 receptor antagonist therapy. No significant change in magnesium over time was seen in advanced-stage (3–4) CKD cats. CKD stage is represented on the x-axis by early CKD (n = 2) or advanced CKD (n = 4), and time 1 (initial data) and time 2 (data collected following therapy) as the first and second circles or squares, respectively. Plasma total magnesium concentrations are represented as untransformed means ± SEM. **P = 0.0025
Blood creatinine concentrations of cats with stage 1–2 CKD significantly differed from cats with stage 3–4 CKD (P <0.0001). Untransformed mean creatinine concentrations of cats with stage 1–2 and stage 3–4 CKD were 2.2 and 4.4 mg/dl, respectively. Overall differences in creatinine concentrations between treatments were observed (P = 0.048). This can be primarily attributed to differences between PPI and H2RA groups (P = 0.0086). Untransformed pooled mean creatinine concentrations were 2.9 mg/dl for cats receiving PPI vs 4.0 mg/dl for cats receiving H2RA. The untransformed mean creatinine concentrations of cats that received both acid suppressants and no acid-suppressant therapy were 3.6 mg/dl and 3.0 mg/dl, respectively. These values were not significantly different. Lastly, blood creatinine concentrations changed significantly over time (P <0.009), regardless of treatment, with untransformed means of 3.0 mg/dl for time 1 and 3.7 mg/dl for time 2.
There were no significant differences in blood total calcium or potassium concentrations over time between treatment or CKD groups.
Other clinicopathologic variables
Clinicopathologic variables recorded when available but not statistically analyzed included blood chloride, USG, the presence or absence of hypertension and gross proteinuria. These values are reported in Tables 2 and 3 for cats in which these variables were recorded.
Mean or median chloride (Cl−) concentrations and urine specific gravity (USG) for cats within each treatment group at time 1 and time 2
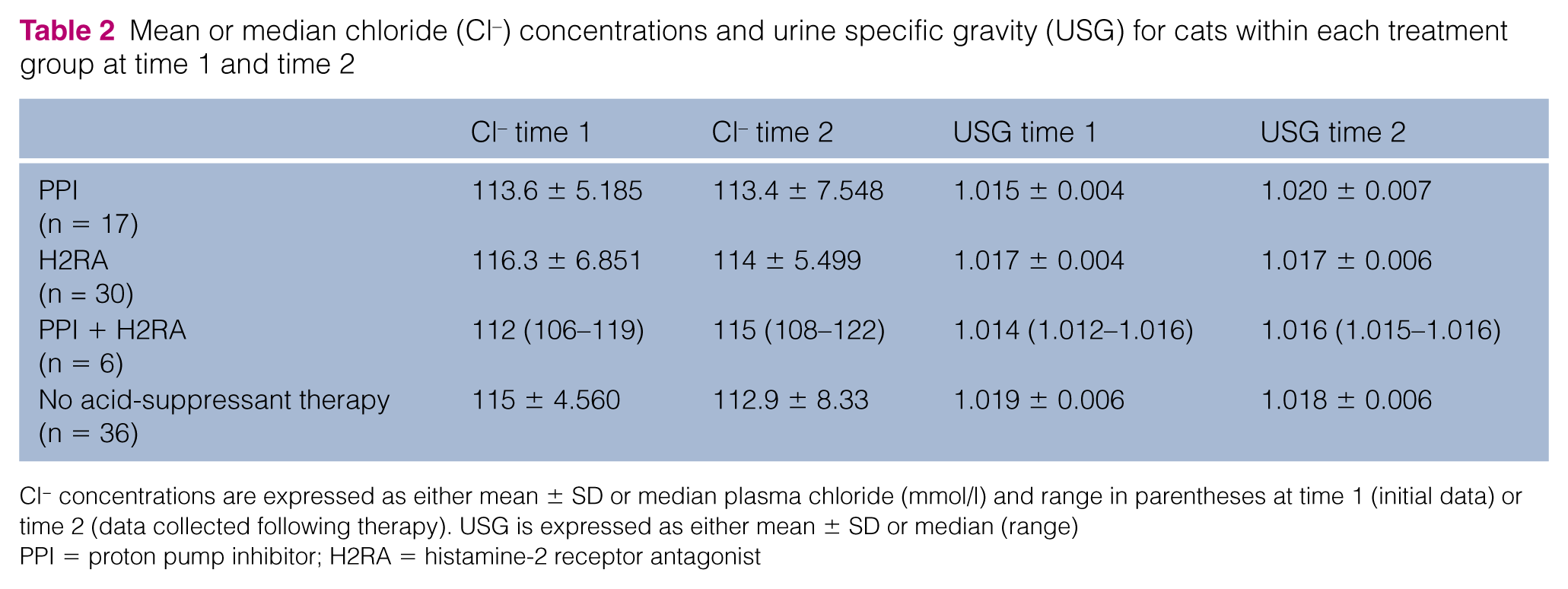
Cl− concentrations are expressed as either mean ± SD or median plasma chloride (mmol/l) and range in parentheses at time 1 (initial data) or time 2 (data collected following therapy). USG is expressed as either mean ± SD or median (range)
PPI = proton pump inhibitor; H2RA = histamine-2 receptor antagonist
Number of cats with gross proteinuria and systemic hypertension within each of the four treatment groups, as well as the duration of therapy for that treatment

The total number of cats within each group is denoted as n in the first column, and the number out of that total for the variable of interest is expressed in the second and third column. The duration of therapy is reported as the median number of weeks, with the range in parentheses.
PPI = proton pump inhibitor; H2RA = histamine-2 receptor antagonist
Discussion
There are no published veterinary studies that have evaluated the effects of chronic acid-suppressant therapy in companion animals with metabolic diseases that could potentiate the adverse effects, including clinicopathologic changes, of such drugs.
As some dietary vitamins and minerals (eg, cobalamin 12 , magnesium5,16) require an acidic pH for intestinal absorption, decreased absorption of these compounds might occur secondary to chronically increased gastric pH. In this study, we found a significant increase in blood sodium concentrations over time for all cats receiving PPI therapy, independent of the severity of CKD, as well as a decrease in total magnesium in early-stage CKD cats receiving both a PPI and H2RA. The increase in blood sodium concentrations in cats treated with PPI therapy suggests that cats with CKD of any stage might be at an increased risk for developing an increase in sodium concentration over time. Sodium disturbances, most commonly reported to be a decrease in serum sodium, also occur in humans receiving PPI therapy. These disturbances are most commonly reported in elderly individuals with comorbidities who are receiving multiple other medications,9,10 or pediatric patients with genetic defects (ie, Klippel–Feil syndrome). 20 No studies describe sodium disturbances secondary to PPI therapy in humans patients with renal disease. Increased sodium and its effect on the renin angiotensin aldosterone system (RAAS) activation in cats with CKD would be detrimental.
The reason for sodium increases in cats receiving PPI therapy is unknown. Diet or drugs might have contributed to the change in blood sodium over time, given that cats were not receiving a standardized diet across treatment groups and cats were also receiving concurrent medications. However, cats receiving an H2RA or no acid-suppressant therapy were also not standardized to diet; thus, we believe that diet alone was unlikely to be a contributing factor. An increased percentage of cats receiving only H2RA therapy were also receiving concurrent therapies in comparison with cats in the PPI-only group. Given this finding, it is unlikely that the administration of concurrent drugs was responsible for the increase in plasma sodium concentration seen in the PPI group. Changes in water consumption or hydration status may have affected sodium concentrations, but owing to the retrospective design, this variable could not be evaluated in this study. That being said, one would expect this to occur with equal chance within each treatment group. Administration of subcutaneous fluids might influence blood sodium concentrations. As subcutaneous fluid therapy was not recorded as a treatment here, it is unknown if this was a factor contributing to the change in sodium in this population of cats. Lastly, it should be noted that the starting sodium concentration of the PPI group was slightly lower than that of the other groups, but this change was not statistically evaluated. It is unknown how this might influence interpretation of the sodium concentrations in this study. Prospective, randomized, placebo-controlled studies to evaluate the potential causative role of PPI therapy on increased sodium concentrations in cats with CKD are warranted.
A significant decrease in total blood magnesium concentrations was observed in cats with early (ie, stage 1–2) CKD receiving combined acid-suppressant therapy (ie, PPI + H2RA) but not with either acid suppressant alone. However, the same effect was not observed in cats with advanced CKD receiving combined acid-suppressant therapy. Hypomagnesemia is a recognized adverse effect secondary to PPI therapy in humans. In one large, cross-sectional study, hypomagnesemia secondary to PPI therapy was identified in human patients receiving concurrent diuretic therapy. 21 This same study found that H2RA use was not associated with development of hypomagnesemia, with or without diuretics. In the current study, only 1/6 cats receiving combined acid-suppressant therapy was reported as receiving diuretic therapy, and this cat had stage 3 CKD (ie, advanced stage), which was not in the group in which significant hypomagnesemia was seen. Therefore, concurrent diuretic therapy was not a contributing factor to the development of hypomagnesemia in this study. Unfortunately, a low number of cats received combined acid-suppressant treatment (n = 6), only two of which were in the early CKD group. Additionally, only total magnesium concentrations are reported in this study owing to the lack of cats with measured ionized magnesium. Assessing total blood magnesium rather than ionized magnesium might overestimate the incidence of hypomagenesemia if not accounting for subjects with hypoalbuminemia. These contributing factors make drawing definitive conclusions regarding an association of combined acid-suppressant therapy and hypomagnesemia in cats with early CKD difficult. Thus, additional prospective studies are required to determine if combined acid-suppressant therapy causes ionized hypomagnesemia in cats with early CKD.
While the primary site of action for PPIs are the H+/K+ ATPases in the gastric parietal cell, PPIs may also affect H+/K+ ATPases in the kidney 22 and osteoclasts in bone. 23 For example, PPI administration has been reported to induce renal tubular acidosis with subsequent hypokalemia and hypomagnesemia. 22 In a recent study, a significant risk for the development of end-stage renal disease (ESRD) over time was observed for humans with renal disease receiving a PPI compared with those receiving either an H2RA or no acid-suppressant therapy. 24 The odds ratio for risk of ESRD in patients receiving a PPI was 1.83 compared with the control population. Additional studies also suggest that new PPI users are at an increased risk of developing incident CKD over a 10 year period,17,25 experience faster progression of CKD and are more likely to develop ESRD. 25 While some of these odds ratios do not implicate a large risk, investigation of an association and, if indicated, a causal relationship, between PPIs and progression of CKD in companion animals is warranted. In the current study, a significant increase in plasma creatinine over time was seen independent of treatment or severity of CKD (ie, early vs advanced). Therefore, these results do not suggest that PPI therapy significantly increased creatinine over time in comparison with H2RAs or lack of acid-suppressant therapy. While a significantly lower creatinine was reported in cats receiving PPI therapy compared with H2RA therapy, this is felt to be a reflection of a small number of cats in the H2RA group (n = 3/30) that had a rapid increase in blood creatinine at the time 2. This was not observed in any of the cats receiving a PPI. The inability to detect a significant effect of PPI use on progression of CKD in cats, despite findings in humans that suggests an association, may be because PPI use does not cause progression of CKD in cats, a longer duration of therapy is needed to observe such an effect or may be an artifact of an insufficient sample size.
As a result of the retrospective nature of the study, we were unable to evaluate the effects of certain variables, such as duration of therapy and formulation of acid suppressant, on the development of adverse effects. We were also unable to control for the influence of additional drug therapy or subcutaneous fluids on the development of adverse effects. The nature of the design also made it impossible to ensure that all measurements were recorded on the same analyzer machine. Because it was impossible to standardize measurements to one machine, minor fluctuations were deemed acceptable if at least two consecutive values were obtained during initial staging and the creatinine was within 0.3 mg/dl of the lower limit for that IRIS stage. Lastly, older cats with CKD evaluated at tertiary care facilities often have concurrent disease. This study was no exception with 88 cats (ie, 98% of the study population) documented to have comorbidities. Thus, we cannot discount an effect of concurrent disease on the development of adverse effects of acid-suppressant therapy.
It is also of interest that many cats with IRIS stage 2 CKD received acid-suppressant therapy. According to current IRIS guidelines, only stage 3 and 4 cats that are experiencing vomiting, hyporexia or clinical signs of nausea should be treated with an acid suppressant. In this study, >50% of cats receiving either a PPI or H2RA had stage 2 CKD. One study investigating the prevalence of uremic gastropathy in cats with CKD was unable to identify gastric erosion or ulceration in any cats with CKD regardless of stage. 26 These data might indicate that clinicians are over-prescribing acid suppressants to patients with CKD and are not in compliance with recommended guidelines. However, it should be noted that some cats with stage 2 CKD exhibit more clinical signs, and, therefore, therapy should be targeted towards treatment of clinical signs rather than stage of CKD alone.
Conclusions
This study is the first to describe the effect of chronic acid-suppressant therapy in cats with CKD. The results suggest that both classes of acid suppressants are relatively well tolerated in feline patients with CKD. Acid-suppressant therapy was not associated with a significantly worsened progression of CKD over time in this population of cats. However, blood sodium should be monitored closely in cats with CKD receiving chronic PPI therapy. Prospective, placebo-controlled, randomized clinical trials with particular attention focused on the effects of chronic acid-suppressant therapy on blood sodium and magnesium are warranted.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
