Abstract
Objectives
Owing to its highly infiltrative growth, feline injection-site sarcoma (FISS) carries a significant risk of local tumour recurrence. Parameters of possible prognostic significance (eg, tumour size and location, resection of de novo vs recurrent tumours, and achievement of tumour-free surgical margins) were examined with regard to their influence on recurrence rate (RR), disease-free interval (DFI) and survival time (ST).
Methods
This was a retrospective analysis of cats with FISSs located on the chest or abdominal wall or the interscapular region treated in a single institution using a standardised radical resection technique with 3 cm lateral margins and full-thickness body wall resection (tumours over chest/abdominal wall) or a minimum of two fascial planes (interscapular tumours).
Results
Median postoperative DFI and ST of 131 cats with FISSs was 21 and 24 months, respectively. Patients operated on for recurrent tumours were significantly more likely to die from tumour-related reasons compared with patients with de novo tumours (P <0.001). RR and DFI in the different tumour locations were comparable (P = 0.544 and P = 0.17, respectively). Local tumour recurrence occurred in 38.1% of the cats. Cats operated on for tumour recurrences had a significantly higher chance of another recurrence (RR 55.5% vs 33.3%; P = 0.005). Completeness of excision was determined by taking tumour bed biopsies. Tumour bed biopsies that did not contain tumour cells were associated with a significantly lower RR compared with those with tumour cells (30.5% vs 76.2%).
Conclusions and relevance
Depending on prognostic factors such as surgery for primary vs recurrent tumour, tumour-free resection margins and tumour location, the RR in FISS ranges from 33–55%, despite curative intent radical surgery. This study may help in identifying patients at risk for recurrence.
Introduction
Feline injection-site sarcomas (FISSs) are among the most commonly diagnosed malignant skin neoplasms in the cat,1–3 and a tumour incidence between 0.63/10,000 and 1/1000 injected cats has been reported.3–9 The most accepted hypothesis concerning the aetiology and pathogenesis of these tumours suggests that in genetically susceptible cats the local inflammatory reaction that occurs after vaccinations or other injections induces a stepwise malignant transformation of connective tissue cells into a sarcoma.4,10–12 Histologically, these tumours are mostly fibrosarcomas, but other sarcoma subtypes have also been described. 13 The histological features of FISS differ from those of fibrosarcomas of non-vaccination sites and are typically characterised by peritumoral perivascular infiltration of lymphocytes, intratumoral giant cells and/or macrophages, necrotic foci and a high tendency of local infiltration.13–16 In addition, many cats have a history of previous injections at the site of the tumour. FISSs are highly invasive tumours, and microscopic tumour extensions and satellites may be found several centimetres distant from the palpable borders of the mass. The rate of metastasis appears to be low and reportedly ranges only between 10% and 28%.10,17,18
Early and radical surgery currently remains the first-choice therapy. Additional therapies such as radiation, chemotherapy and immunotherapy have been utilised using various protocols. Adjuvant and neoadjuvant radiotherapy reportedly prolongs the time to tumour recurrence in cases with incomplete resections.19–22 Recently, immunotherapy using local injections of a transfected canarypox virus, locally expressing interleukin-2, has been shown to significantly improve the disease-free interval (DFI) in cats when combined with surgery and radiation therapy.23–25 Although the efficacy of chemotherapy has not been fully proven, its use has commonly been reported.9,26–29
Owing to the highly invasive growth of FISS, incomplete resections are common, and patients carry a high risk of local recurrence.17,19,20,30–35 In earlier studies using conservative resection techniques recurrence rates (RRs) of up to 70% were reported.6,13,32,36,37 Radical en bloc resections with at least 3 cm lateral margins and at least two fascial planes for deep margins have been advocated as the gold standard of treatment.8,9,31,38,39 With ‘radical surgery’, RRs ranging between 14% and 40% have been reported; however, many studies lack details on tumour size, tumour location, width and depth of resection, number of primary vs recurrent tumours, method of evaluation of resection margins, and duration of postoperative follow-up. Furthermore, some studies include adjuvant therapies such as radiation and chemotherapy.17,19,20,26,31,32,34,35,37,39
The goal of the present study was to determine the RR in a large series of cats with a FISS located exclusively on the chest, abdominal wall or the interscapular region, treated in a single institution using a standardised radical resection technique. Parameters of presumed prognostic significance such as tumour size and location, resection of a primary vs recurrent tumour, and the presence of tumour cells at the surgical margins were evaluated statistically.
Materials and methods
Cats with a FISS treated surgically at Hofheim Small Animal Clinic (Germany) between 1998 and 2012 were evaluated retrospectively. Inclusion criteria were cats with a FISS of either the chest or abdominal wall or the interscapular region in which a radical surgery with curative intent was deemed possible, based on subjective clinical evaluation (palpation) of the tumour by the surgeon. Cats were not included if they had inadequate preoperative data on the size and location of their tumours, less than 12 months of follow-up, radiographic signs of metastasis or were not operated on with curative intention. All patients were operated on by the same surgeon (MK).
The diagnosis of a FISS was based on the documentation of a soft tissue sarcoma in a location commonly used for injections with typical histological characteristics, such as presence of peri-tumoral inflammation dominated by lymphocytes, intratumoral macrophages and multinucleated giant cells, marked cellular pleomorphism, central necrosis and highly infiltrative growth.
The cats were divided into four groups according to anatomical location of the FISS and whether they had a de novo tumour (not previously operated on, primary tumour), or a local recurrence after a previous surgical resection at a first opinion practice. Group 1 were de novo chest and abdominal tumours, group 2 de novo interscapular tumours, group 3 recurrent chest and abdominal tumours, and group 4 recurrent interscapular tumours.
All patients underwent standard diagnostic and staging procedures prior to surgery, including fine-needle aspiration biopsy of the mass, two-view thoracic radiographs (right and left lateral projections), as well as haematology and clinical chemistry evaluation. Tumours were divided into three size groups (largest diameter <2 cm, diameter 2–4 cm and diameter >4 cm).
Tumours of the chest or abdominal wall were excised en bloc with a lateral margin of at least 3 cm to the palpable tumour, including the overlying skin. The deep resection margin included a full-thickness chest or abdominal wall resection. If tumours partially overlapped with the long dorsal musculature, a resection of the lumbar fascia entering deep into the long lumbar musculature was performed. In cases presenting with recurrent tumours, the palpable tumour mass and the entire scar of previous surgeries were included in the resection using the same margins. Interscapular tumours were resected with a 3 cm minimum lateral margin and at least two fascial planes deep, including interscapular musculature, dorsal spinous processes or, if necessary, dorsal parts of the scapulae. Defect closure was performed by either direct apposition of neighbouring tissue using muscle flaps (mainly M latissimus dorsi, M obliquus externus or M sartorius muscle flaps), or by surgical mesh in cases where muscle flaps could not be performed.
Completeness of tumour excision was determined by sampling 10–12 small (5 mm × 5 mm up to 10 mm × 10 mm) biopsies from the in vivo tissue adjacent to the excised specimen. In body wall tumours this was the border of the resection defect. In interscapular tumours samples were taken from both the lateral margins and the deep tumour bed of the resection site. The samples were taken immediately after tumour resection, prior to defect closure and preferably from locations deemed by the surgeon to carry the highest risk for residual tumour (eg, subcutaneous tissue closest to the tumour, fascial interfaces, the area between the scapula and the axial musculature, musculature between dorsal spinous processes). If any one of the tumour bed biopsies contained tumour cells on histopathology the resection was considered incomplete.
Clinical re-evaluation including physical examination and palpation of the resection site, as well as thoracic radiographs at 3 month intervals, was recommended for all patients following surgery, but was subject to the owners’ compliance. Local recurrence was suspected in any case when a firm mass was palpated in the surgical field. Depending on the owners’ compliance, suspected recurrences were confirmed cytologically or histologically. Additional follow-up was performed by clinical examination at Hofheim Small Animal Clinic and/or telephone investigation with the owner or referring veterinarian. Only patients with a minimal follow-up time of 12 months were included in the study.
The DFI was defined as the time between the definitive surgery and any suspected or confirmed local tumour recurrence and was calculated only for those patients that actually experienced a recurrence. The survival time (ST) was defined as the time between the definitive surgery and death. Cats that were alive at the end of the study or died of non-tumour-related reasons were excluded from analysis at the time of study closure, or at time of death, respectively. Cause of death was classified as tumour-related, non-tumour-related, postoperative complication and unknown.
Statistical analysis
Descriptive statistic data are reported as median ± SD or average size (range minimum–maximum size). Kaplan–Meier survival analysis was performed using the Gehan–Breslow statistic for calculation of median ST and DFI. A 95% confidence interval was used for the median DFI and ST, and the level of significance for all analyses was set at P <0.05.
Comparison of ST and DFI between the different groups was carried out using the Mann–Whitney U-test; the Kruskal–Wallis and ANOVA tests were performed for comparison of the DFI and tumour size in different tumour locations. The probability of tumour recurrence with remaining tumour cells in the tumour bed and the dependence of a positive tumour bed on the tumour location were tested by Pearson’s χ2 test. The influence of tumour size on ST, DFI and positive tumour bed was evaluated using the Kruskal–Wallis test. Distribution of cause of death and RR was assessed by Pearson’s χ2 test. Cats that were lost to follow-up and alive at the end of the study were excluded from analysis at the time of their last recheck. Cats that died from postoperative complications while in the clinic were excluded from analysis of DFI.
Results
Epidemiology and tumour characteristics
A total of 131 cats met the inclusion criteria. There was an equal sex distribution (64 male castrated, 67 female spayed). The mean ± SD age at time of presentation to Hofheim Veterinary Clinic was 9.7 ± 2.99 years (range 4–19 years). There were 123 domestic shorthair cats (93.9%), three Maine Coons (2.3%) and one each (0.8%) of Persian, Norwegian Forest Cat, Siamese, British Shorthair and Russian Blue.
Seventy-six cats (group 1) had a primary (de novo) tumour on the lateral chest or abdominal wall (n = 40 and n = 36, respectively). In cats where the resection field included (parts of) the chest wall, an average of 3.5 ribs (range 1–5 ribs) were resected en bloc with the tumour. In this group the median tumour size (largest diameter) of the chest and abdominal wall tumours was 3.0 cm (range 1–6 cm) and 2.0 cm (range 0.5–6 cm), respectively.
Twenty-eight cats had a primary (de novo) interscapular tumour (group 2). In this group of cats the median tumour size was 2.4 cm (range 0.3–6 cm). There was no significant difference in tumour size between cats from group 1 and 2 (P = 0.217).
Nineteen cats were presented with a recurrence of a tumour previously excised from the chest or abdominal wall at a first-opinion practice (group 3). Of these, 17 presented for the first recurrence; two had already been operated on twice. At presentation, the median diameter of the palpable mass of the recurrent tumour was 1 cm (range 1–4 cm) for recurrences of chest wall tumours and 2 cm (range 1–7 cm) for those of the abdominal wall. Eight patients had involvement of the chest wall and were treated with a chest wall resection with an average of 4.4 ribs resected (range 2–7 ribs).
Eight patients were presented with a recurrent interscapular tumour previously excised at a first-opinion practice (group 4). The median tumour size at presentation was 1 cm (range 0.5–6 cm). Five patients were presented at time of first recurrence, three had already had two previous excisions.
Defect closure
In 29/95 patients with chest or abdominal wall tumours (groups 1 and 3), a primary closure of the defect by re-adaptation of the local musculature was possible. One or more muscle flaps were used in 58 patients. In eight cats (three abdomen, five chest) a Prolene mesh had to be utilised to cover the defect. In all patients with interscapular tumours, primary defect closure was possible after undermining the skin and subcutis adjacent to the resection defect. When necessary, a re-adaptation of the scapula was performed using PDS suture (USP 2-0). All cats with interscapular tumours were released from the clinic and were fully ambulatory within 2–4 weeks.
Survival times and cause of death
Five cats with a primary resection died during hospitalisation in the postoperative phase from complications, four with a chest tumour and one with an abdominal wall tumour. In all cases with chest wall tumours four ribs had been resected and death occurred 2, 3, 3, and 4 days postoperatively, respectively. The cat with the abdominal wall resection died 3 days postoperatively. All cats had been closely monitored during hospitalisation. Cardiorespiratory arrest was the presumed cause of death in all cases. In two cases pulmonary infiltrates suggestive of pulmonary oedema were present on thoracic radiographs, although these cats had no clinical evidence of pre-existing cardiac disease. Unfortunately, none of these cases were released for necropsy by the owners.
Thirty-eight cats died for tumour-related reasons (tumour recurrence). Forty cats were euthanased or died from non-tumour-related diseases (chronic kidney disease n = 8; cardiac disease n = 8; senile decay n = 6; car accident n = 4; seizure activity of unknown aetiology n = 3; unrelated neoplastic disease n = 3; death following surgery for non-tumour-related disease n = 2; chronic dermatological disorder n = 2; myiasis n = 1; orthopaedic problems n = 1; respiratory disease n = 1; bite injury n = 1). In three cats the cause of death was unknown.
Median ± SD follow-up time for the remaining 126 cats was 25.5 ± 24.35 months (range 2–129 months). Eighty-six cats had died by the end of the study and 45 were alive at the end of the study after a median ± SD follow-up time of 34 months (range 13–82 months).
The overall median ± SD postoperative ST was 24 ± 25.3 months (range 0–
When only the 126 cats released from the clinic were evaluated, the median ± SD ST was 25.5 ± 24.3 months (range 2–
Cause of death in 131 cats with feline injection-site sarcoma

Data within brackets are %.There was a significant chance of dying of tumour-related reasons if the animal was operated on for a recurrent tumour (P <0.001). CKD = chronic kidney disease

Kaplan–Meier plot depicting survival time (ST) in all 126 cats released from the hospital. There was a significant difference in ST in cats that experienced a recurrence compared with those that remained free of disease (P <0.001)
Cats without residual tumour cells in the tumour bed (n = 105) had a significantly longer median ST compared with those with the presence of tumour cells in the tumour bed (n = 21) (median ± SD ST 28 ± 26.19 months vs 16.0 ± 13.47 months; P = 0.026) (Figure 2). Tumour size did not significantly affect the median ST.
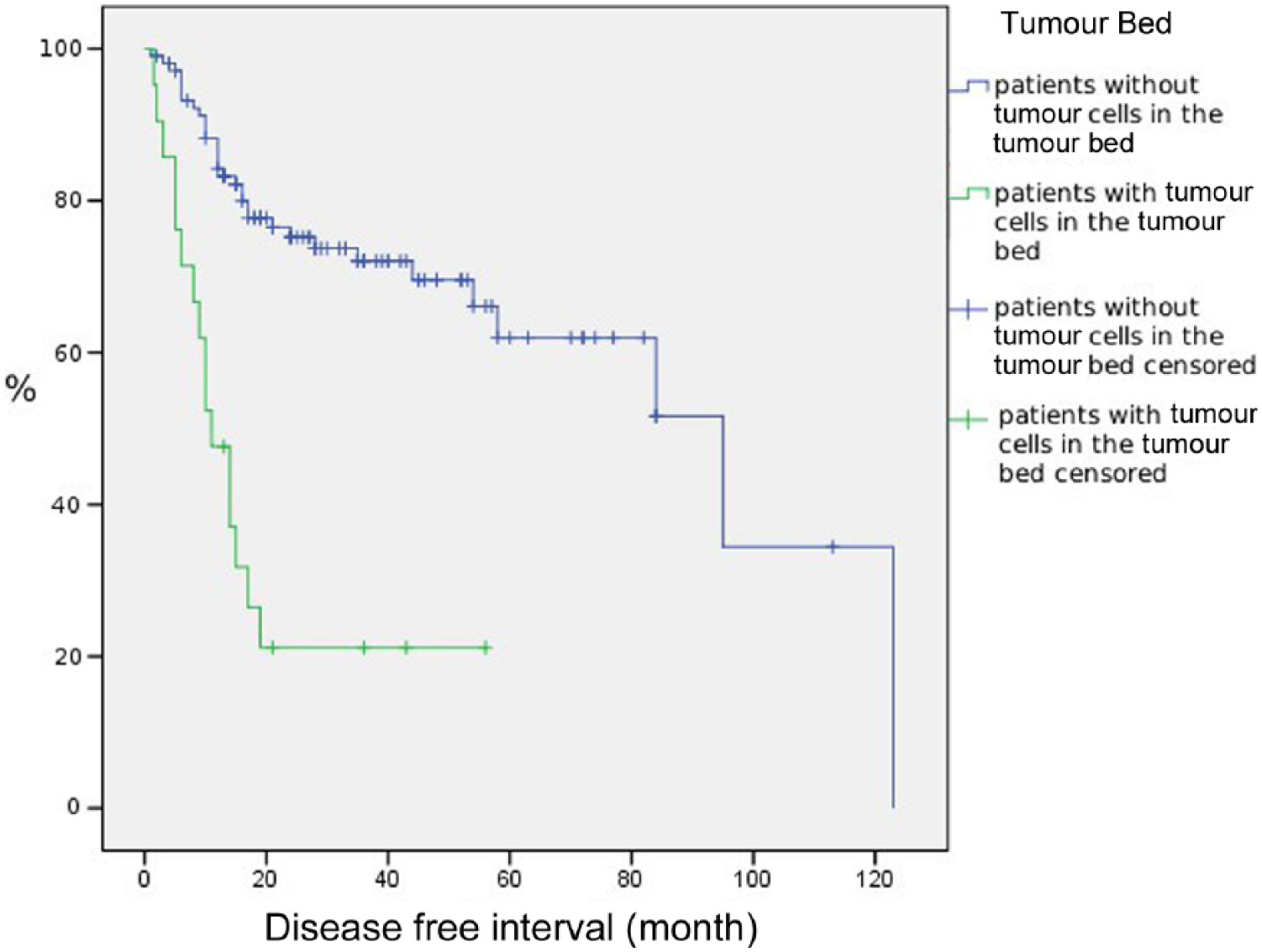
Kaplan–Meier plot depicting the disease-free interval of cats with (n = 21; green line) and without (n = 105, blue line) tumour cells detected in the tumour bed biopsies (P = 0.026)
Disease-free interval
The median ± SD DFI of all 126 cats released from the clinic was 21 ± 24.5 months (range 1–
Survival time (ST) and disease-free interval (DFI) in relation to tumour location in the 99 cats with de novo tumours (groups 1 and 2)

Data are median ± SD
Forty-eight of the 126 (38.1%) cats that survived the postoperative period experienced local tumour recurrence. Thirty-three of these 48 cats belonged to groups 1 and 2 (surgery for a primary tumour) and reached a median ± SD DFI of 12 ± 28.1 months (range 1–123 months). The median ± SD DFI of the 15 cats that had recurrence after surgery for a recurrent tumour (groups 3 and 4) was 11 ± 15.1 months (range 3–
Recurrence rate
In 21/131 (16%) cats the tumour bed biopsies contained tumour cells and resection was therefore considered incomplete. Eight of 76 cats (10%) in group 1, 4/28 (14%) in group 2, 4/19 (21%) in group 3 and 5/8 (62%) in group 4 had tumour cells in tumour bed biopsies. As 5/76 patients in group 1 died perioperatively, only 126 cats could be evaluated for the rate and time of tumour recurrence. Sixteen of the 21 patients (76.2%) with positive tumour bed biopsies and 32/105 patients (30.5%) with negative tumour bed biopsies experienced a tumour recurrence. The median follow-up time for cats with and without recurrence was 17.1 and 32.5 months, respectively. The probability of tumour recurrence with positive tumour bed was significantly higher compared with negative tumour bed biopsies (P <0.001). The relative risk of developing a recurrence with a positive tumour bed was 2.6 (odds ratio [OR] 7.8). Cats presenting with recurrent tumours (groups 3 and 4) had a significantly higher risk of residual tumour cells in the tumour bed compared with patients from groups 1 and 2 (OR 3.833; P = 0.006).
According to this, cats in groups 3 and 4 had a significantly higher chance of recurrence (RR 55.5%; n = 15/27) compared with patients with primary tumours (RR 33.3%; n = 33/99) (P = 0.005). The relative risk of a previously operated on cat of developing another recurrence was 1.8 (OR 2.7). Receiver operating (ROC) analysis (ROC area 0.739) resulted in a 65% probability of a recurrence after 10 months, 21% after 20 months and 15% after 30 months.
There was a trend towards a higher likelihood of a presence of tumour cells in the tumour bed in interscapular tumours (n = 9/36) compared with chest (n = 6/48) and abdominal wall tumours (n = 6/47) (P = 0.085), but the RR in the different tumour locations was not statistically different (P = 0.544).
There was no significant association between the tumour diameter and the location of the tumour. On multivariant analysis the likelihood of the tumour bed biopsies to contain tumour cells was not associated with tumour size (P = 0.726). In addition, tumour size did not significantly affect the RR; however, there was a trend towards higher recurrence rates with increasing tumour size (P = 0.084) (Table 3).
Recurrence rate of tumours and survival times (STs) dependent on size of tumour at presentation in the 99 patients with de novo tumours (group 1 and 2, median follow-up 24 months)

Discussion
This study confirms previously reported US,17,19,20,27,31,39,40 New Zealand 26 and European21,28,34,35 data concerning age, breed and sex distribution of cats affected by FISSs. In US reports, a significant proportion of FISSs affecting the (hind)limbs have been reported in addition to chest/abdominal wall and interscapular locations.17,19,20,31,32,40 It is noteworthy that in contrast to reports from the USA, FISSs on the limbs is virtually non-existent in the German cat population,10,41 which most likely reflects different preferences for injection/vaccination sites. Therefore, in the present study, only tumours on the chest or abdominal wall and in the interscapular location were included.
The study population included a higher number of chest/abdominal wall tumours than interscapular ones, but this may represent a selection bias, because only tumours considered operable with a curative intent were included in the study. In a previous study by one of the authors in >2000 cases of FISSs in Germany, an equal distribution was reported between interscapular and body wall locations. 42 Similarly, no conclusions on the rate of metastasis at time of diagnosis can be drawn from this study, as cats with radiographic lesions suggestive of pulmonary metastasis were not considered candidates for curative resections and therefore not included in the study.
In most studies, the histopathological subtypes of FISS influenced neither the biological behaviour of the tumours nor the treatment outcome.28,35 Therefore, for the purpose of this study, all histological variants of FISS were treated in the same manner. Histological grading of FISS into three malignancy grades has been performed by some authors using various canine soft tissue sarcoma grading systems, which are based on cellular differentiation, presence of necrosis and mitotic activity.16,21,28,35,40,43 Tumour grade did not prove to have prognostic significance in any of these studies and owing to the lack of a universally accepted grading system, the tumours in the present case series were not graded.
Most published studies on FISSs either include tumours of various locations17,19,20,31,32,40 or do not specify the tumour locations.21,27 Few studies give details on ST and DFI in regard to tumour location. In the study by Davidson et al, 31 cats with tumours on the limbs had a higher median ST and DFI than patients with tumours on the trunk (limb: median ST and DFI of 15 months; trunk: median DFI and ST of 9 and 11 months, respectively). This was confirmed by Hershey et al, 17 who showed that appendicular tumours treated with limb amputation were associated with a significantly longer DFI in comparison with other tumour locations (DFI median 325 days vs 66 days; P = 0.007). Kobayashi et al included six cats with tumours in the hindlimb in a series of 92 cats. 19 They reported that tumour location did not have a significant influence on ST or DFI. No further details were given, but considering that their study was designed to evaluate the effects of radiation on recurrence, it could be assumed that these cats had received limb-sparing operations and were treated for residual disease. As cats with appendicular FISSs can frequently be treated curatively by amputation, it is conceivable that an appendicular tumour location influences DFI and ST. 31
In most previous studies no further information is given on the prognosis (recurrences or STs) of patients with body wall vs interscapular tumours. Only Romanelli et al reported no significant association between prognosis and anatomical tumour location in a series of 57 cases. 28 However, almost half of the patients included in their study had been operated on for recurrent tumours and 15 had received adjuvant radiation in addition to surgery, which may limit the interpretation of their data. According to the present study, cats with tumours of the abdominal wall had a significantly longer ST compared with patients with chest wall tumours. Abdominal tumours tended to be smaller than chest wall and interscapular tumours (median diameter 2.0 cm vs 3.0 cm vs 2.4 cm, respectively). In addition, there was a significantly higher proportion of unclean resections in interscapular vs body wall tumours (25% vs 12.6%). These factors combined with the relative ease of resection compared with other locations may have contributed to the longer DFI of abdominal wall tumours (median DFI 29.5 months vs chest wall 20.5 months vs interscapular 16.0 months) and, as a result, may have led to improved STs of tumours in this location.
Most previous reports do not give details on the width of resection19,31,32,34 and/or include various adjuvant therapies, which may affect the outcome.17,19–21,26,31,32,34 Therefore, most studies are not comparable with the present study in which a standardised surgical approach was used with only one surgeon involved. Giudice et al reported on 48 cases with chest/abdominal or interscapular FISSs treated by surgery alone with resection margins of at least 3 cm. 35 Seventeen of the 48 cats (35.4%) in their study experienced a tumour recurrence; however, follow-up in some cases was only 6 months and no information was given on median ST or DFI. Romanelli et al 28 reported a 36% RR in 42 cats treated with surgery alone, using 4–5 cm lateral margins and one facial plane deep ‘where possible’. Phelps et al reported radical en bloc resections of primary or recurrent tumours with 5 cm margins in 91 patients with tumours in various locations, including limbs. 40 With this approach the authors achieved a 14% RR with a median recurrence-free interval of 309 days. However, in their study 32/91 cats (35%) were lost to follow-up, including 4/10 cases with chest or abdominal wall resections. In addition, the postoperative follow-up time was as low as 80 days with no further details provided in the study.
After a median follow-up time of 34 months, 86 cats in our study had died, and in all 38 tumour-related deaths massive local tumour recurrence was documented. Forty cats died of non-tumour-related diseases (Table 1). Euthanasia in almost all cases was performed at the referring veterinarian’s practice and no systematic restaging and/or necropsy was performed at this time. Therefore, owing to the retrospective nature of the study, it remains unknown how many of the cats developed metastasis during the course of the disease. It is possible that, especially in cases of death classified as non-tumour-related, undiagnosed tumour metastasis may have contributed to the condition of the animal thus leading to the decision to euthanase the cat.
According to our study, >20% of the cats developed a recurrence >20 months after surgery. Several other authors reported recurrences of the tumours after long time intervals (1 to several years).26,35,40 Studies using a more conservative (marginal) surgical approach demonstrated a significantly shorter DFI compared with a radical surgery. 17 This was confirmed in the 15 cats of groups 3 and 4 in our study, which had significantly shorter median DFI after the initial surgery compared with the DFI after a more radical approach (DFI 7 months vs 12 months, respectively; P = 0.018). Some of the studies in the literature report fairly short follow-up times, which may affect the RR (Davidson et al: 31 60 days, RR 25.6%; Romanelli et al: 28 129 days, RR 30–48%; Phelps et al: 40 80 days, RR 14 %). Martano et al, 34 who followed all 20 cats until death, reported an RR of 35%. The reason for the sometimes surprisingly long time interval between radical resection and tumour recurrence remains unclear. Cancer dormancy is a poorly understood phenomenon, and various mechanisms have been reported, including angiogenic dormancy, cellular dormancy and immune system surveillance.44,45 However, surgical stress may actually promote the transition of dormant tumour cells into active proliferation. 46 Therefore, it may also be speculated that the new tumour formation ⩾2 years after the first surgery may not be a true recurrence originating from residual tumour cells but actually may represent a new and independent tumour formation originating from fibroblasts that experienced malignant transformation in the inflammatory, pro-angiogenic and therefore tumor permissive microenvironment of the postoperative field.45,46 The sometimes long time intervals between the surgery and the ‘recurrence’ makes it desirable for future studies to extend the follow-up time beyond 1 or even 2 years.
According to our results, cats presenting with a recurrent tumour had a significantly higher risk of another recurrence compared with cats with tumours that had not previously been operated on (RR 55.5% vs 31.7%; P = 0.004). Almost identical results were achieved by Cohen et al, 32 who reported a 55% vs 31% RR in recurrent vs not previously operated patients. In another study including 41 cats, 68% of the cats experienced tumour recurrence within 2 years of surgery, but cats that had surgery for recurrent tumours had a significantly shorter tumour-free period. 37 Phelps et al reported the lowest RR of all studies. 40 However, 72 (79%) of their 91 cats had primary tumours removed, and only 19 (21%) recurrent tumours removed, which, among other variables, could have positively influenced the results of their study.
The histopathological assessment of surgical margins is considered one of the best determinants of adequate surgical treatment and for many cancers has been proven to be a strong predictor of treatment outcome.47,48 Unfortunately, until today, there have been no universally accepted standards regarding the evaluation and reporting of surgical margins in both human and veterinary oncology.48,49 It was not until recently that a large panel of veterinary pathologists published recommendations in an attempt to standardise the processing of submitted tumour tissues and the reporting of margins in resection specimens in veterinary patients. 50 Several methods such as the assessment of the surgical margin on two perpendicular cross sections, by serial lamellar sectioning, by tangential sectioning (shave biopsies) and by taking tumour bed biopsies have been reported.48,50 Inking of the cut surface or ‘critical areas’ by the surgeon has been used to aid the pathologist in identifying areas of interest on the submitted specimen.48,50,51 All techniques have inherent advantages and disadvantages, and, regardless of method used, there are limitations in sensitivity.48,52 In addition, post-removal (artefactual) changes in the dimensions of a sample, sliding of tissue planes and insufficient fixation, especially in large tissue blocks, may affect the processing and interpretation of a sample.52–54
For FISSs, several studies have proven the value of margin determination in predicting DFI and ST. Studies discriminating ‘positive’ (containing tumour cells; ‘dirty’) from ‘negative’ (no tumour cells; ‘clean’) margins consistently found a prognostic significance, regardless of histopathological sampling technique used (Table 4). With the tumour bed biopsy technique used in the present study, any identification of neoplastic cells in sections of the biopsies is considered consistent with an incomplete surgical excision. 50 Besides higher costs, the potential disadvantage of this technique is that it is dependent on the surgeon taking the appropriate tissue samples and no objective margin measurement can be performed. However, considering the lack of standardisation in veterinary pathology, taking multiple samples and specifically sampling ‘critical areas’ of the resection bed by the surgeon may increase the sensitivity of margin evaluation. To our knowledge systematic studies concerning the techniques of margin assessment are lacking in veterinary medicine. In the present study cats without tumour cells in tumour bed biopsies had a significantly lower RR compared with cats in which the tumour bed biopsies contained tumour cells (30.5 vs 76.2 %; P <0.001). Our study demonstrated a significantly higher risk of finding tumour cells in the tumour bed in surgeries for recurrent tumours (P = 0.006) and a trend towards a higher proportion of tumour-containing tumour bed biopsies in interscapular vs body wall tumours (25% vs 12.6%; P = 0.085).
Literature review regarding the influence of completeness of excision on recurrence rate and prognosis in feline injection-site sarcoma
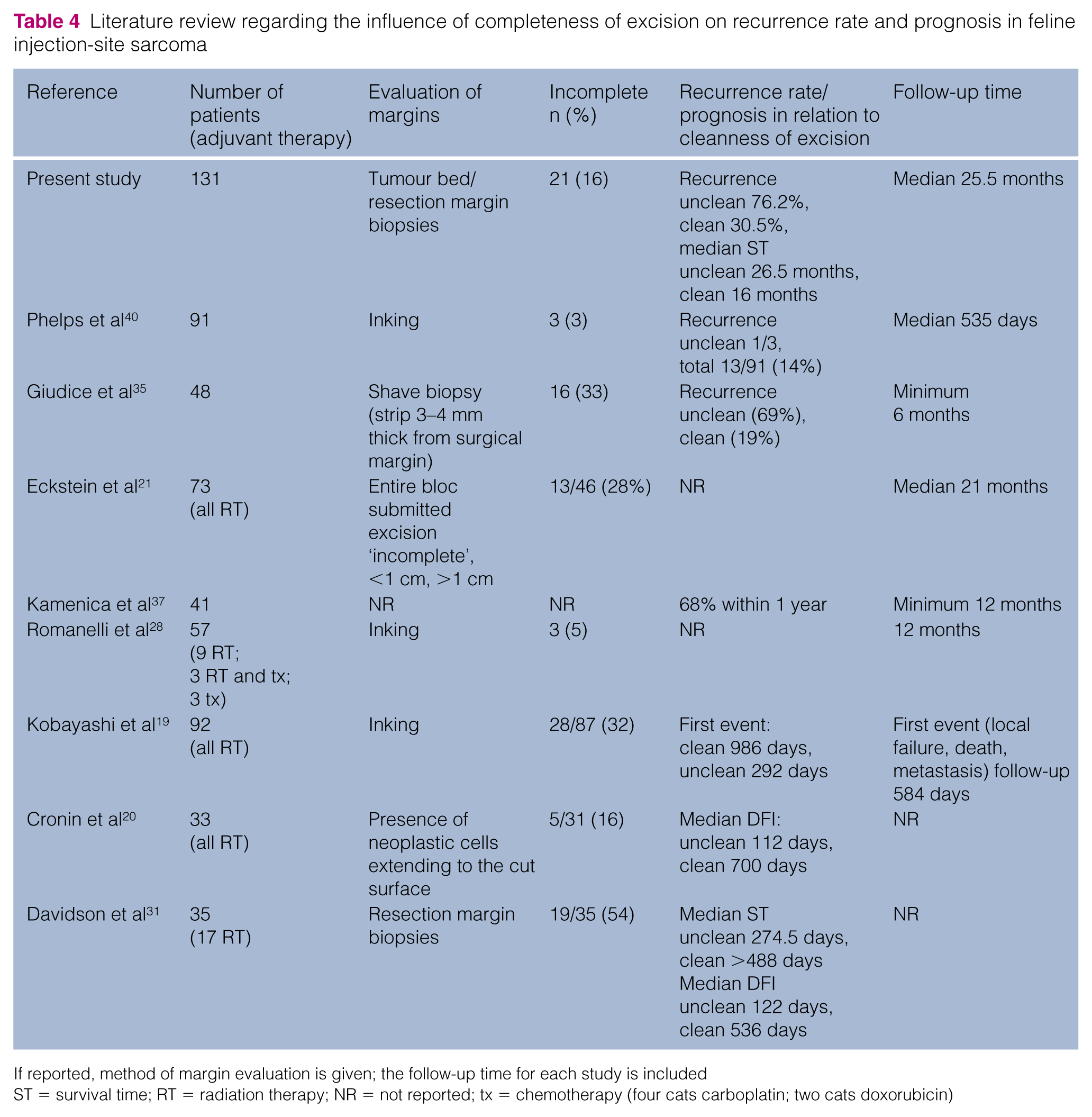
If reported, method of margin evaluation is given; the follow-up time for each study is included
ST = survival time; RT = radiation therapy; NR = not reported; tx = chemotherapy (four cats carboplatin; two cats doxorubicin)
In the study by Dillon et al,
55
cats with tumours
A 4.6% perioperative mortality rate was reported in a study including 153 cats, 56 which is similar to the rate of 3.8% (n = 5/131) in the present study. Considering the extent of the surgical intervention, many pet owners may find the fatal complication rate acceptable in the context of treating an otherwise terminal disease.
Conclusions
Radical surgical excision of FISSs can be curative or at least lead to durable remissions but still results in recurrence rates ranging from 30–>50%, dependent primarily on factors such as surgery for primary vs recurrent tumour, complete excision with the absence of tumour cells in the tumour bed and tumour location. The results of this study may help in identifying cats at risk of recurrence.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
