Abstract
Objectives
The objective of this study was to compare the pharmacokinetics of compounded and commercially available aqueous formulations of buprenorphine after a single buccal dose to healthy cats and to evaluate the concentrations of a compounded buprenorphine solution over 21 days when stored at room temperature (RT; 22–24°C) with exposure to light or when refrigerated at 4°C while protected from light.
Methods
Six young healthy male cats were administered single buccal doses of compounded and commercially available formulations of buprenorphine (0.03 mg/kg) using a randomized, blinded, two-period crossover design. Blood samples were obtained over a 24 h period and plasma buprenorphine concentrations were determined using ultra-high-pressure liquid chromatography with mass spectrometry detection. Three batches of the compounded formulation were stored at RT or 4°C and aliquots were evaluated over 21 days for buprenorphine concentration using high-performance liquid chromatography with fluorescence detection.
Results
Plasma concentrations of buprenorphine were above the limit of quantification up to 6 h in some cats and up to 3 h in all cats. The area under the curve was significantly less for the compounded formulation (P = 0.004). A significant difference was not detected between formulations for time to maximum concentration (P = 0.11), maximum concentration (P = 0.06), half-life (P = 0.88) and mean residence time (P = 0.57). Buprenorphine concentration in the compounded formulation was not affected by storage condition or time and remained between 90% and 110% of the target concentration at all time points.
Conclusions and relevance
A buprenorphine solution prepared from sublingual tablets is absorbed after buccal administration in healthy cats. The extent of absorption is significantly less than that of the commercially available formulation. The compounded solution maintains an acceptable buprenorphine concentration for at least 21 days when stored at RT or refrigerated.
Introduction
Pain in cats has historically been undertreated owing to the difficulty in recognizing pain, fear of adverse effects from analgesic drugs and the limited analgesic options for cats. With our growing awareness of the need to treat pain in cats, greater attention has been given to drug development and pain research in this species; however, realistic analgesic options, especially those for chronic use, remain limited. Cat size and temperament, comorbidities, risk of adverse effects, ease and frequency of medication administration, and economic factors further limit the analgesic options available for cats.
Steagall et al recently published a comprehensive review of studies using buprenorphine in cats. 1 This review suggests that the onset, duration and magnitude of antinociceptive effect of buprenorphine is influenced by the route of administration. Multiple pharmacokinetic studies document buprenorphine absorption following buccal administration to cats,2–6 but pharmacodynamic studies have produced inconsistent results regarding the antinociceptive and analgesic efficacy of buprenorphine when given by this route.2,5,7,8 Buccal administration of buprenorphine remains an attractive option because of the ease of administration, non-invasive route of administration, ease of dosing for small patients and high margin of safety.2,9 Because there are no commercially available buprenorphine products labeled for buccal use in cats, extra-label use of an injectable formulation is widely used. However, the expense, inconvenience and waste associated with use of single-dose glass ampules of the commercially available injectable human formulations limits its use in veterinary medicine, especially for chronic use.
A 0.3 mg/ml buprenorphine solution compounded from US Food and Drug Administration (FDA)-approved sublingual tablets has been used at the University of Georgia. However, neither the pharmacokinetics of this compounded preparation nor the stability after compounding have been studied. A recent study by Kirk and Brown demonstrated that a 3 mg/ml buprenorphine solution compounded from buprenorphine active pharmaceutical ingredient powder was stable for 90 days, but the pharmacokinetics of the solution after buccal administration in cats was not evaluated. 10 Additionally, at the time of writing, the use of active pharmaceutical ingredient (bulk) powders is still considered illegal under the Animal Medicinal Drug Use Clarification Act, and use of the approved sublingual tablets to compound represents a more legally correct veterinary compounding scenario. 11
Therefore, the objectives of this study were to compare the pharmacokinetics of compounded and commercially available aqueous formulations of buprenorphine after a single buccal dose to healthy cats and to evaluate the concentrations of the compounded buprenorphine solution over 21 days when stored at room temperature (22–24°C) with light exposure or when refrigerated at 4°C and protected from light. We hypothesized that the extent of systemic absorption of the compounded buprenorphine would be less than that of the injectable formulation, and there would be no significant effect of storage condition or time on buprenorphine concentration in the compounded formulation.
Materials and methods
Preparation of buprenorphine solutions for buccal administration
The buprenorphine solution used in this study was prepared from commercially available 2 mg buprenorphine hydrochloride tablets (Roxane Laboratories) approved by the FDA for sublingual use in humans. Tablets were dissolved in deionized water, and Simple Syrup NF (Humco) was added to bring the solution to the desired concentration of 0.3 mg/ml (Table 1). Each batch of the solution was prepared by a licensed pharmacist and stored in an amber plastic vial at room temperature for no more than 24 h prior to administration.
Formula for compounded buprenorphine solution (0.3 mg/ml)

Buprenorphine strength-over-time testing
Six batches of the compounded buprenorphine formulation were prepared for stability testing. Half were stored at room temperature (22–24°C) with light exposure, and half were refrigerated at 4°C while protected from light. All batches were stored in amber plastic vials. Immediately after preparation and after 1, 4, 7, 10, 14 and 21 days of storage, 2 ml aliquots were obtained from each batch and stored at −80°C until assayed. At the time of sampling, each batch was observed for a change in appearance (color, opacity, consistency, uniformity) and pH was measured.
Animals
Six male domestic shorthair cats (two neutered, four intact) from a research colony were used in this study. The cats were 17–26 months old and weighed 4.8 ± 0.9 kg (mean ± SD). All cats were deemed healthy based on the results of general physical examinations, complete blood counts and serum biochemistry analyses. They were housed individually during the experiments. The Institutional Animal Care and Use Committee at the University of Georgia approved the study protocol.
Catheter placement
On the day prior to each study, food was withheld in the morning and the cats were anesthetized with ketamine (7 mg/kg) and dexmedetomidine (5 μg/kg IM). As these drugs took effect, salivary pH was measured by placing a pH strip into a cheek pouch until moist and then comparing the indicator strip color to the reference chart. Supplemental oxygen was provided via a facemask, and isoflurane was administered as needed to maintain anesthesia for catheter placement. A 20 G, 12 cm polyurethane catheter (Arrow International) was placed into a medial saphenous vein, capped with a needle-free male adaptor plug, and secured to the skin with nylon suture and tape.
Drug administration
Food was withheld overnight and water was withheld for 20 mins prior to buprenorphine administration. Both food and water were offered 90 mins after dosing. Cats were randomly assigned using an online program (random.org) to receive a commercially available aqueous formulation (0.3 mg/ml Buprenex; Reckitt Benckiser Pharmaceuticals) or the compounded buprenorphine solution at a dose of 0.03 mg/kg (0.4–0.6 ml/cat), both administered in the buccal pouch. The drug was administered to each cheek pouch using a 1 ml syringe. The investigator who administered the drug was blinded to which formulation was given. The ease of administration was noted and the cats were observed for adverse effects (eg, salivation, vomiting, ataxia) throughout the study period. All cats received both treatments with a 14 day washout between doses.
Blood sample collection and handling
Blood samples were collected via the medial saphenous catheter immediately before and at 5, 10, 15, 22, 45, 60, 90, 180, 360, 720 and 1440 mins after drug administration. Prior to the collection of each sample, the catheter was scavenged by withdrawing 1.5 ml blood into a syringe containing 0.5 ml heparinized saline (1 USP unit/ml). The volume of blood collected (1.7–2.5 ml per sample) was adjusted for each cat such that no more than 5 ml/kg was collected over the 24 h study period. After each sample, the scavenged blood was returned and the catheter was flushed with saline equal to the volume of blood withdrawn. Samples were transferred to lithium heparin tubes, temporarily stored on ice and centrifuged for 10 mins at 2000 g and 4°C. Plasma was harvested within 1 h of sample collection and was stored at −80°C until assayed.
Buprenorphine analysis in feline plasma
Feline plasma samples were analyzed by ultra-high-pressure liquid chromatography (UPLC) followed by tandem mass spectrometry detection using a method developed in the authors’ laboratory. Calibration curves and quality controls were prepared by fortifying blank plasma with stock solutions of buprenorphine hydrochloride (Sigma-Aldrich) dissolved in 100% methanol. Plasma samples, standards and quality controls were then prepared by adding a 250 µl sample to 250 µl 2% ammonium hydroxide in water in a glass tube, and vortexing for 15 s. The sample mixture was then added to 2 ml supported liquid extraction cartridges (Isolute SLE+; Biotage) and a light vacuum was applied to initiate absorption. Two aliquots of 2.5 ml methyl tert-butyl ether (Fisher Scientific) were added to the cartridges, allowed to sit for 5 mins and then slowly eluted through under light vacuum. The resulting eluate was then placed in an evaporator and dried under a 20 psi stream of nitrogen for 20 mins at 45°C. Samples were reconstituted in 250 µl 50:50 methanol:water (v/v). All samples were filtered into injection vials using 0.2 µm nylon syringe filters (4 mm Nalgene filters) attached to a 3 ml syringe. Volumes of 8 µl for samples and standards were injected on an Acquity Classic UPLC with Acquity I-Class UPLC with an Acquity Xevo TQD mass spectrometer (MS/MS) (Waters) using a flow rate of 0.3 ml/min. Separation was achieved using an Acquity UPLC HSS T3 column (1.8 µm, 2.1 × 100 mm) and Vanguard column (Waters). A gradient was used and the initial mobile phase was 0.1% formic acid in water:0.1% formic acid in acetonitrile (85:15 v/v) for the first 2.5 mins. The mobile phase then switched to 10:90 v/v from 2.5 mins to 4 mins. In the last 1 min of the run, the mobile phase was 85:15 v/v. The Xevo TQD was run in ESI+ mode. The quantification MRM transition was 468.39 →100.89. Column temperature was maintained at 30°C and sample temperature was maintained at 10°C.
The retention time observed for buprenorphine was 1.48 mins. Standard curves were linear over a concentration range of 0.1–10 ng/ml with an r2 ⩾0.99. The lower limit of quantification was 0.1 ng/ml, which was determined from the lowest point on a linear calibration curve that met our acceptance criteria and using guidelines published by the United States Pharmacopeia. 12 Accuracy (percent nominal concentration) and precision (relative SD) were determined from quality-control samples fortified with buprenorphine at concentrations of 0.25, 0.75, 2.5 and 7.5 ng/ml (n = 6 for each concentration). Accuracy ranged from 90–104%, while precision ranged from 5–11% over the concentrations tested.
Buprenorphine analysis in compounded syrup
Buprenorphine was quantified in syrup using a modified method to account for the high concentrations (target concentration of 0.3 mg/ml). High-pressure liquid chromatography (HPLC) followed by fluorescence detection was used for quantification. Calibration curves and quality controls were prepared by fortifying blank NF Simple Syrup with stock solutions of buprenorphine hydrochloride (Sigma-Aldrich) dissolved in 100% methanol. Samples, standards and quality controls were then prepared by adding a 10 µl sample to 990 µl 83:17 of acetonitrile:water in a glass tube and vortexing for 10 s. All samples were filtered into HPLC injection vials using 0.2 µm nylon syringe filters (4 mm Nalgene filters) attached to a 3 ml syringe. Volumes of 100 µl for samples and standards were injected on an Alliance HPLC with fluorescence detector (Waters) set at an excitation wavelength of 210 nm and emission wavelength of 338 nm, and using a flow rate of 1.2 ml/min. The mobile phase consisted of 83:17 acetonitrile:10 mM ammonium acetate in water. Column temperature was maintained at 30°C and sample temperature was maintained at 10°C. Separation was achieved using an XBridge C8 column (3.5 µm, 4.6 × 150 mm) and guard column (Waters).
The retention time observed for buprenorphine was 4.5 mins and the total run time was 8 mins. Standard curves were linear over a concentration range of 100–500 µg/ml with an r2 ⩾0.99. The lower limit of quantification was 100 µg/ml. Accuracy and precision were determined from quality-control samples fortified with buprenorphine at concentrations of 100, 200 and 400 µg/ml (n = 6 for each concentration). Accuracy (% nominal concentration) and precision (relative SD) ranged from 88–98%, and precision was 1%.
Pharmacokinetic analysis
Pharmacokinetic analysis was performed using commercially available software (Phoenix WinNonLin version 6.4 [Certara]). Pharmacokinetic parameter values were estimated using non-compartmental methods. The area under the concentration vs time curve extrapolated to infinity (AUC(0–∞)) was calculated using the linear-log trapezoidal rule. A minimum of three time points were used in the estimation of the elimination rate constant (λz). The maximum plasma concentration (Cmax) and time to maximum plasma concentration (Tmax) were taken directly from the data.
Statistical analysis
The AUC(0–∞), λz, half-life (T1/2), mean residence time (MRT), Cmax and Tmax were compared between drug formulations using a Mann–Whitney test for non-parametric data or paired Student’s t-test for parametric data. A two-way repeated measures ANOVA was used to evaluate the effect of storage condition and time on drug concentration in the compounded formulation. Significance was set at P <0.05 for all analyses.
Results
All cats showed some resistance to restraint and drug administration, and some drug loss was observed in 5/6 attempts to administer the commercial formulation and 3/6 attempts to administer the compounded formulation. Substantial drug loss was observed during administration of the compounded formulation to one cat after it bit the syringe and caused the drug to fall on its Elizabethan collar. Plasma buprenorphine concentrations in this cat were <2 SD from the mean at all time points and were excluded from pharmacokinetic analyses. Evidence of nausea, vomiting, discomfort and aversion to taste were not observed in any cat. The salivary pH was 8.5 in two cats and 9.0 in four cats. The mean pH of the compounded formulations was 3.9 (range 3.7–4.0).
Plasma concentrations of buprenorphine were above the limit of quantification of the assay (0.1 ng/ml) up to 6 h in some cats and up to 3 h in all cats. Plasma concentration vs time curves for each formulation are shown in Figure 1. The pharmacokinetic parameter values for the AUC(0–∞), λz, T1/2, MRT, Cmax and Tmax for the commercial and compounded buprenorphine formulations are shown in Table 2. The fraction of the AUC extrapolated to infinity was <15% for both formulations.

Mean ± SD of plasma concentrations of buprenorphine in six healthy adult male cats following buccal administration of commercially available (solid line, circles) and compounded (dashed line, squares) formulations at a dose of 0.03 mg/kg
Pharmacokinetic parameter estimates following buccal administration of commercially available and compounded formulations of buprenorphine at a dose of 0.03 mg/kg to healthy cats
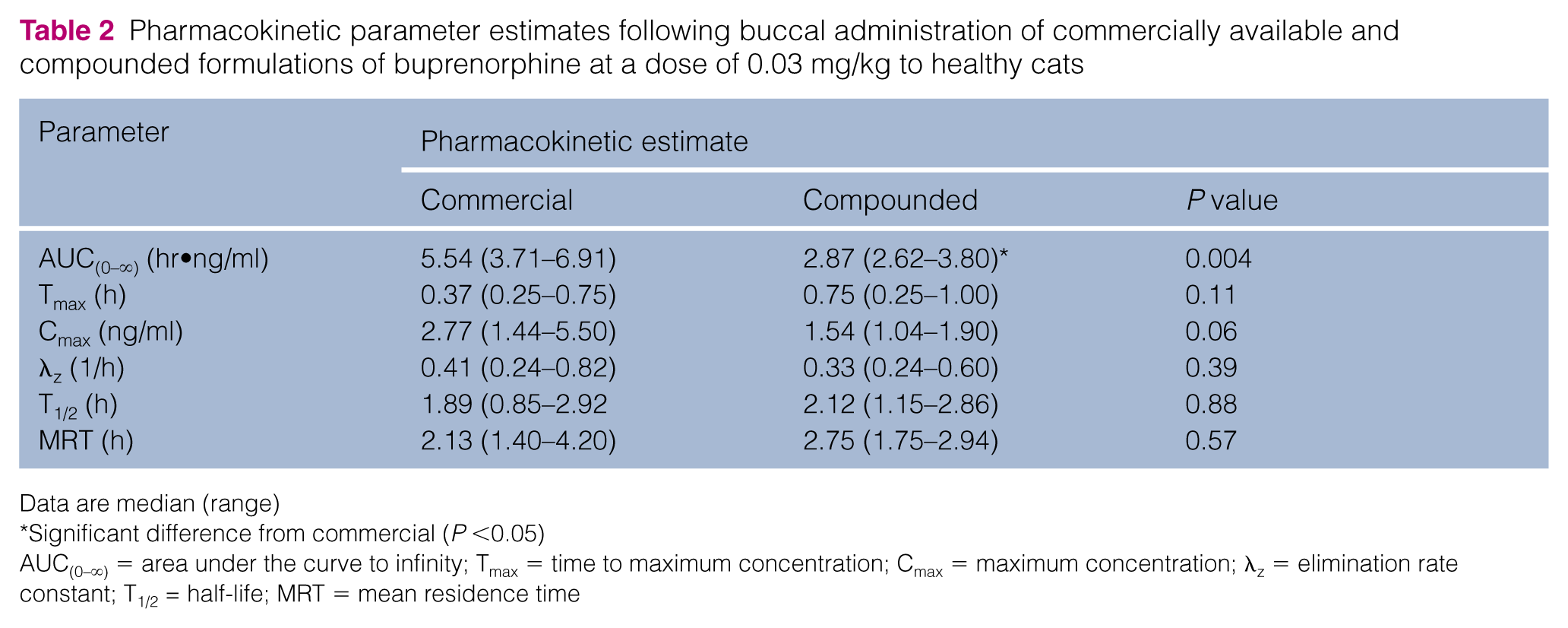
Data are median (range)
Significant difference from commercial (P <0.05)
AUC(0–∞) = area under the curve to infinity; Tmax = time to maximum concentration; Cmax = maximum concentration; λz = elimination rate constant; T1/2 = half-life; MRT = mean residence time
Drug concentrations in the compounded formulation are shown over time and under both storage conditions in Figure 2. There was no appreciable change in color or clarity of the formulation over time under either storage condition. However, crystals formed around the rim of the bottle by day 7 for those batches stored at room temperature. This did not occur at any time point for the refrigerated batches.

Mean ± SD buprenorphine concentration in buprenorphine solution (0.3 mg/ml) compounded from sublingual tablets stored at room temperature (22–24°C) with exposure to light (dashed line, circles) or refrigerated at 4°C and protected from light (solid line, squares) for 21 days. Aliquots from each storage condition were evaluated over time. There was no effect of storage condition (P = 0.42) or time (P = 0.22) on the drug concentration. The buprenorphine concentration was within 90–110% (dashed horizontal lines) of the theoretical concentration (solid horizontal line) at all times as required by United States Pharmacopeia standards for pharmaceutical compounding
Discussion
The results of the present study show that a buprenorphine solution prepared from sublingual buprenorphine hydrochloride tablets is absorbed after buccal administration in healthy cats with no adverse effects. The extent of absorption, indicated by the area under the plasma concentration curve, was significantly less than that of the commercially available formulation. The average relative bioavailability (%) of the compounded formulation compared with the commercial formulation calculated in this study is 57.25%. Multiple factors could have contributed to the lower relative bioavailability of the compounded formulation.
The pharmacokinetics of buprenorphine in this study are different from the reported values in similar studies.3–5 Generally, the results from our study report a lower Cmax and shorter elimination T1/2 than previously described. We attribute some of these differences to differences in study design and methodologies.3,5 For example, in the 2005 report by Robertson et al, 3 the Cmax was reported to be 7.5 ng/ml, but samples were obtained from a jugular catheter, which overestimates the actual plasma concentrations of buprenorphine.4,13 The dose-adjusted Cmax reported by Hedges et al (0.13 ng/ml) 5 was slightly greater than our finding of 0.09 ng/ml. These authors also reported detectable buprenorphine in feline plasma up to 24 h after administration, which was longer than our study (6 h). Upon further inspection of the data of the previous study, the mean buprenorphine concentration at 24 h appears to be lower than the lower limit of quantification of the assay (0.05 ng/ml), making the data difficult to interpret. 5 In our study, we may have detected buprenorphine levels for longer in some cats with a more intensive sampling strategy, although we were unable to replicate the limit of quantification of 0.05 ng/ml in the study reported by Hedges et al. 5
Buccal administration of buprenorphine is well-tolerated by most cats,2,3,7–9 and was the preferred route of drug administration by cat owners in one study.
2
The compounded formulation described in this study is considered by the federally enforceable compounding standard, the United States Pharmacopeia, to be a Simple compound. USP <795> defines a Simple compound as:
Making a preparation that has a United States Pharmacopeia (USP) compounding monograph or that appears in a peer-reviewed journal article that contains specific quantities of all components, compounding procedure and equipment, and stability data for that formulation with appropriate beyond use dates (BUDs); or reconstituting or manipulating commercial products that may require the addition of one or more ingredients as directed by the manufacturer.
By definition, this compound can be easily prepared in a pharmaceutical setting that complies with applicable laws and regulations, without the need for specialized equipment or bulk powders.10,14
The buprenorphine concentration in the compounded solution remained within 90–110% of the target concentration and did not change significantly over a 21 day period, regardless if the solution was stored at room temperature on a benchtop or if it was refrigerated. Kirk and Brown reported similar findings in their study, 10 which demonstrated that a buprenorphine solution compounded from buprenorphine powder was stable for 90 days when refrigerated or stored at room temperature. In the present study, refrigeration prevented the formation of crystals around the rim of the vial that formed by day 7 when stored at room temperature. Crystals form similarly when the compounding syrup alone is stored under these conditions, thus they seem to result from drying of the syrup rather than crystallization of the buprenorphine product used in the compounded solution. Kirk and Brown also noted formation of white particulate matter in the threads of the container bottles on day 21 when stored at room temperature but not with refrigeration. 10 A limitation of the present study was that we did not perform a stability-indicating assay as did Kirk and Brown, 10 which established a 90 day stability for compounded buprenorphine; therefore, a compounding pharmacy may elect to utilize a standard beyond-use date of 14 days refrigerated, as suggested by the United States Pharmacopeia, for the formulation described in this study.10,14
One limitation of this study was the small population of cats, which may have increased the chance of a type II statistical error. A second limitation was the potential influence of fasting on salivary pH, which could affect drug absorption. Although the effect of fasting on salivary pH in cats and its effect on buprenorphine absorption have not been specifically evaluated to our knowledge, previous studies have reported similar salivary pH measurements in cats that were fasted3,9,15 and those that were not.4,16 The salivary pH was also measured under anesthesia the day prior to buprenorphine administration in our study, which may not have accurately represented the salivary pH at the time of buprenorphine administration. However, the salivary pH measured in our study (8.5–9.0) was similar to that reported in fasted, awake cats (8.7–9.0) in another study. 9 A third limitation was drug loss during administration of buprenorphine, which may have affected the results.
Conclusions
A buprenorphine solution prepared from sublingual tablets is absorbed after buccal administration in healthy cats, but to a lesser extent than a commercially available injectable product administered via the same route. Patients should be monitored carefully for evidence of pain due to variations in drug absorption and analgesic effect, regardless of the drug formulation used. The compounded buprenorphine solution in this study is not associated with adverse effects and maintains an acceptable buprenorphine concentration for 21 days when stored at room temperature or refrigerated.
Footnotes
Acknowledgements
We acknowledge Kathleen Hoover, Ethan Karstedt, Lynn Reece and Lisa Reno for their assistance with sample collection and processing, and Alex Taylor for his assistance with sample analyses.
Author note
This study was presented, in part, at the 25th Annual Scientific Meeting of the European College of Veterinary Surgeons.
Conflict of interest
CWS has received funding from Elanco. KMM has received funding and/or honoraria from Zoetis, Bayer, Piedmont Animal Health and Jurox. All funding was for purposes unrelated to the present study. The authors have no other potential conflicts to declare related to this study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article
