Abstract
Objectives:
This paper, the second in a series of three on ‘feline leprosy’, provides a detailed description of disease referable to Mycobacterium lepraemurium, the most common cause of feline leprosy worldwide.
Methods:
Cases were sourced retrospectively and prospectively for this observational study, describing clinical, geographical and molecular microbiological data for cats definitively diagnosed with M lepraemurium infection.
Results:
A total of 145 cases of feline leprosy were scrutinised; 114 ‘new’ cases were sourced from the Victorian Infectious Diseases Reference Laboratory records, veterinary pathology laboratories or veterinarians, and 31 cases were derived from six published studies. Sixty-five cats were definitively diagnosed with M lepraemurium infection. Typically, cats were 1–3 years of age when first infected, with a male gender predilection. Affected cats were generally systemically well. All had outdoor access. Lesions tended to consist of one or more cutaneous/subcutaneous nodules, typically located on the head and/or forelimbs, possibly reflecting the most likely locations for a rodent bite as the site of inoculation for organisms. Nodules had the propensity to ulcerate at some stage in the clinical course. The cytological and histological picture varied from tuberculoid, with relatively low bacterial numbers, to lepromatous with moderate to high bacterial numbers. Treatment was varied, although most cats underwent surgical resection of lesions with adjunctive medical therapy, most often using a combination of oral clarithromycin and rifampicin. Prognosis for recovery was generally good, and in two cases there was spontaneous remission without the requirement for medical intervention. Untreated cats continued to enjoy an acceptable quality of life despite persistence of the disease, which extended locally but had no apparent tendency to disseminate to internal organs.
Conclusions and relevance:
M lepraemurium causes high bacterial index (lepromatous) or low bacterial index (tuberculoid) feline leprosy. The infection typically causes nodules of the skin and/or subcutis (which tend towards ulceration) on the head and/or forelimbs. The disease usually has an indolent clinical course and infected cats have a generally favourable response to therapeutic interventions, with rare cases undergoing spontaneous resolution. Genomic analysis may yield clues as to the environmental niche and culture requirements of this elusive organism. Prospective treatment trials and/or additional drug susceptibility testing in specialised systems would further inform treatment recommendations.
Introduction
Initial reports of ‘feline leprosy’ were likely dominated by Mycobacterium lepraemurium infections, and cat-to-cat inoculation studies fulfilled ‘Koch’s postulates’ for disease causation. Lesions had a propensity to be sited on the head and/or forelimbs, and were nodular in nature involving the skin and subcutis, with a tendency to ulcerate as the disease progressed. The histological appearance varied from tuberculoid disease, comprising a mixture of lymphocytes and macrophages, usually associated with relatively small numbers of acid-fast bacilli (AFB), to lepromatous, comprising predominantly epithelioid macrophages or giant cells associated with numerous, generally intracellular, AFB. There was a tendency for tuberculous lesions to develop central caseous necrosis, which grossly was correlated with an ulcerated appearance.
Many different approaches to treatment have been tried over the years, with the anti-leprosy agent clofazamine perhaps having the best efficacy in vivo, while rifampicin has documented efficacy in vitro. M lepraemurium is extremely difficult to grow in the laboratory, although a few specialised laboratories in Japan1,2 and Belgium 3 have managed to propagate this organism in vitro, permitting limited susceptibility testing and other studies.

This paper, the second in a series of three, concerns patients with skin lesions proven to be caused by M lepraemurium and represents the largest collection of such cases reported to date. Feline leprosy case series published prior to the turn of the century could not determine if a mixture of different aetiological agents, including M lepraemurium, but also other mycobacterial patho gens such as Myco bacterium visibile, Candidatus ‘Myco bacterium tarwinense’ and the novel species Candidatus ‘Myco bac terium lepraefelis’, were involved in these cutaneous feline mycobacterial infections.
Materials and methods
Case recruitment, demographics, data retrieval, cytological and histological assessment, molecular microbiological methods, genetic analysis, mycobacterial culture and statistical analysis were essentially the same as described in the first paper in this series, and interested readers are referred to that publication for information. 4 different primers were used to partially sequence the sodA gene of M lepraemurium: ‘AvsodAF’ 5’-ACATCTCGGGGCAGATCA-AC-3’ (forward) and ‘AvsodAR’ 5’-GTAGTCCGCCTTGACGTTCT-3’ (reverse).
Results
Clinical data
A total of 145 cases of feline leprosy were analysed; 114 were sourced from the Victorian Infectious diseases Reference Laboratory (VIdRL) records, veterinary pathology laboratories or veterinarians, with an additional 31 cases derived from six published studies.5–10 Of this total, 65 had M lepraemurium infection, comprising 42 ‘new’ cases and 23 cases recorded previously in the literature (Table 1).
Detailed case data for 65 cats infected with Mycobacterium lepraemurium

F = entire female; FN = female spayed; M = entire male; MN = male castrated; NR = not recorded; ? = neuter status unknown; DSH = domestic shorthair; DMH = domestic mediumhair; DLH = domestic longhair; VIC = Victoria; QLD = Queensland; NSW = New South Wales; TAS = Tasmania; WA = Western Australia; BI = bacterial index; FIV = feline immunodeficiency virus; FeLV = feline leukaemia virus; +ve = positive; –ve = negative; CLFZ = clofazamine; RIF = rifampicin; CLM = clarithromycin; DOXY = doxycycline; ENFLX = enrofloxacin; MARBFLX = marbofloxacin; MOXIFLX = moxifloxacin; PRDFLX = pradofloxacin; SC = subcutaneous
Although not directly stated, bacterial numbers were presumed from the pathological description of ‘tuberculoid’ as likely being low bacterial index (BI), with ‘lepromatous’ likely being high BI by definition
Cats were generally young, with a median age of 2 years, although older cats (up to 14 years) were occasionally infected. The preponderance of young cats was striking, with the great majority of affected cats being less than 4 years of age. Where gender was recorded, 43/62 (69%) M lepraemurium infections were in male cats, which is substantially more than would be expected based on the age/gender pyramid for Australian cats. 12 Where breed was recorded, 44/54 were domestic shorthair, 2/54 were domestic longhair, two were Ragdoll, and there was one each of Burmese, Manx cross, Maine Coon, Siamese, Persian and British Shorthair; the crossbreed to pedigree ratio was similar to that of the Australian feline population. 12
Geographical location data were recorded for 63 cats; 16 from Australia, 31 from New Zealand (Figure 1), seven from Canada, seven Zealand (Figure 1), seven from Canada, seven from France, one from the Greek Island of Kythira and one from New Caledonia. M lepraemurium infection was documented to have a wider geographical distribution than any of the other species responsible for feline leprosy, and was the only causative species of this condition in the Australian island state of Tasmania. Although they could be encountered in almost any geographical location, M lepraemurium infections appeared to be overrepresented in coastal cities and rural areas.

Geographical distribution of feline infections caused by M lepraemurium in the Australasian region, where accurate location data was known. Note the prominence of cases in coastal areas
Where lifestyle was recorded, all cats had unsupervised outdoor access and many were known hunters and/or fighters. Feline immunodeficiency virus (FIV) status was only recorded in nine cats, with just one cat being antibody-positive. None of the nine cats tested for feline leukaemia virus were seropositive.

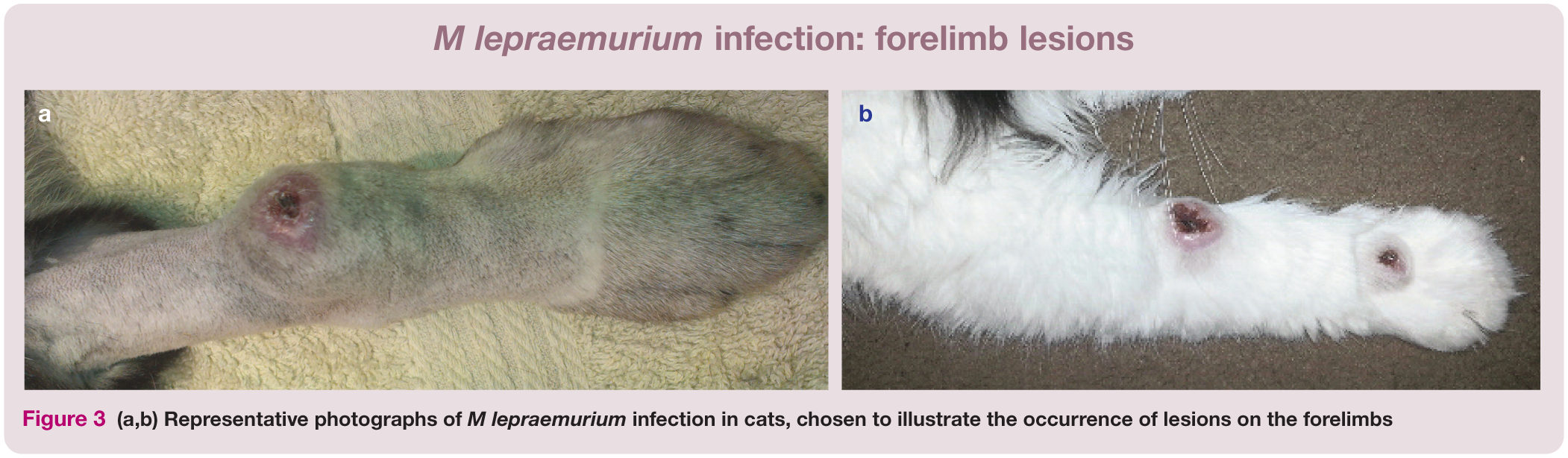

Where clinical features and clinical course were well documented, 34/49 (69%) cats had a few lesions, with only 4/49 cases having generalised nodular skin disease; the remainder had multiple lesions at disparate anatomical sites. Lesions were mainly on the head (21/49 [43%] cases) and forelimbs (23/49 [47%]) (Figures 2 and 3). No cats with this aetiology underwent necropsy examination. Of the 40 cases where cytology or histology was available for review, a high bacterial index (BI) was observed in 23/40 cases (58%), while the remainder had a low BI. In many cases where a low BI was recorded, the lesions were ulcerated; however, ulceration was also occasionally observed as a clinical feature of high BI lesions.

(a–c) Representative photographs of M lepraemurium infection in cats, chosen to illustrate the occurrence of lesions on the head

(a,b) Representative photographs of M lepraemurium infection in cats, chosen to illustrate the occurrence of lesions on the forelimbs
Treatment and outcome data
Treatment protocols used in cats in this study varied widely and often were influenced by owner finances and commitment, and also the patient’s temperament.
Where it was possible to implement medical therapy, protocols included several months of treatment with one or more of the following drugs: rifampicin, clarithromycin, clofazimine, a fluoroquinolone (enrofloxacin, marbofloxacin, moxifloxacin or pradofloxacin) and doxycycline. A combination of rifampicin and clarithromycin was used most commonly, mainly due to favourable cost and drug availability. Many cats were treated with surgical resection alone (often with incomplete margins due to the anatomical location of the lesions; eg, eyelids, distal limbs) or, more commonly, a combination of surgical resection and adjunctive medical therapy.
The outcome was generally favourable for cats with M lepraemurium infections, with resolution in 24/27 cases (89%) where sufficient follow-up was available to make an assessment, even with multiple or generalised lesions. disease burden did not appear to influence outcome for this infection, nor did the BI of the lesions. Two cats were euthanased due to progressive or persistent cutaneous lesions, rather than evidence of systemic disease (cases 17 and 43). Spontaneous resolution was observed in two cats infected with M lepraemurium (cases 23 and 24).
A summary of data concerning age, gender, anatomical distribution of lesions and outcome for cats infected by M lepraemurium is provided in Table 2.
Summary data concerning age, gender, anatomical distribution of lesions and outcome for cats infected by M lepraemurium

BI = bacterial index
Microbiological and molecular data
Fresh tissue, and fixed cytological and paraffin-embedded samples from 37 cases underwent dNA extraction, PCR amplification and sequencing (see supplementary material). Known acquired mutations in the rpoB gene conferring rifampicin resistance in other mycobacterial species13–15 were not found in any of the isolates in this study (data not shown).
Mycobacterial dNA derived from fresh biopsy tissue from case 36 underwent draft genome sequencing during the course of the study. The methods and results of these investigations will be published separately (C o’Brien et al, manuscript in preparation).
Discussion
This study greatly extends the body of knowledge concerning M lepraemurium infections in cats.
Anatomical predilection
This organism has a predilection to involve structures about the head and/or forelimbs, but generally without involvement of ocular tissues or periocular skin. This would be consistent with the theory that this organism is likely a pathogen inoculated into a feline predator via the bite of a rodent suffering from systemic mycobacteriosis. 16 This is in contrast to Candidatus ‘M tarwinense’ infections, where lesions often involve the ocular tissues (conjunctiva, cornea, eyelids, nictitating membrane), and nasal and periocular skin and subcutis as a result, perhaps, of inoculation via inter-cat aggression or selfinoculation during grooming.
Signalment
The age range of affected cats is wide (<1 to 14 years); however, the vast majority of cases occurred in young adult cats (<4 years old), presumably because cats of this age group are more likely to roam, explore and exploit territory for hunting. Additionally, older cats may acquire resistance to the organism after asymptomatic exposure at a younger age. Similar factors may also influence the male predilection, as this group, even if castrated, generally roam over a larger area and are more aggressive than female cats. The breakdown of domestic crossbreeds vs pedigree cats is in accord with their ratio in the overall Australian cat population, without any suggestion of immunocompromise in certain pedigree cats playing a role in predisposing to infection.
There were too few cats subjected to retroviral testing to draw any firm conclusions, but with only 1/9 cats tested being FIV-positive, it appears unlikely that acquired immunodeficiency is an underlying predisposition, especially considering the young age and apparent good health of the majority of cats. Indeed, the epidemiology suggests a localised disease of a low-grade pathogen in an immunocompetent patient, with a mycobacterial species somewhat adapted to mammalian hosts, but much more likely to produce systemic disease in rodents than in Felidae.
Clinical course
The clinical course of M lepraemurium in the majority of cats could be perhaps described as indolent, with no suggestion of internal organ involvement in any case based on clinical criteria. Occasional cases have widespread, generalised cutaneous nodules, suggestive of haematogenous spread of the organism. The authors have also observed a limited number of cases in which there has been what appears to be sporotrichoid spread (ie, via the lymphatics from the primary focus of infection).
Organism biology
This biological behaviour is consistent with an environmental saprophyte adapted to a rodent host. The pathogen is less adept at living in a feline patient, and infected cats thus represent an incidental ‘dead end’ host.
Analysis of five gene targets in M lepraemurium demonstrated that all clinical isolates were remarkably clonal at the five loci examined. If this homogeneity was carried over the entire genome, the variation in clinical severity from cat to cat would most likely reflect the number of organisms inoculated and/or variability in the innate and acquired immunological response of the feline host. The apparent genetic homogeneity of M lepraemurium is reminiscent of two other fastidious mycobacterial species – the unnamed agent of the canine leproid granuloma syndrome, which appears to be clonal over an extensive worldwide geographical range, and Candidatus ‘M tarwinense’, which appears to be clonal, but over a far more restricted region. 17
Treatment and outcome

As is typical of an observational study, treatment regimens were not consistent. However, cats tended to respond favourably, especially to surgical excision and long-term use of a combination of two or more antimicrobials usually effective against slow-growing mycobacteria (see box above).
Risk of contagion
To date, there has been no instance of direct spread from cat to cat. Although it is theoretically possible to effect this using infectious material in a contrived experimental setting, in the authors’ view the risk of contagion from cats is slight. It is fascinating that, to date, M lepraemurium infections have not been recorded in dogs, ferrets or native wildlife.
Key Points
M lepraemurium, a fastidious mycobacterium related to M avium, is capable of causing both high BI (lepromatous) or low BI (tuberculoid) ‘feline leprosy’. It is likely that infections follow an altercation with rodents that inoculate infectious propagules into the dermis or subcutis of the feline patient.
Lesions generally consist of nodules (which tend towards ulceration) present on the head and/or forelimbs, with or without local lymph node involvement.
Cases tend to have an indolent clinical course and a generally favourable response to therapeutic interventions.
The best management approach consists of complete surgical resection of easily accessible lesions, and/or debulking of lesions in difficult anatomical locations, with adjunctive medical therapy using two or more antimicrobial agents with known activity against slow-growing mycobacteria.
The inability of the organism to grow using both traditional and unconventional mycobacteriological media remains a mystery, although it is hoped that detailed examination of the draft genome will yield some clues.
Supplemental Material
Click here for Supplementary Table S1
Multi-locus sequence data concerning Mycobacterium lepraemurium
Footnotes
Acknowledgements
Thanks go to the staff of the Mycobacterium Reference Laboratory, VIdRL, especially Caroline Lavender, for technical assistance, and the many veterinarians and veterinary pathologists who contributed case material and other assistance to this study, especially Laura Brandt, Bronwyn Smits, Catherine Harvey, Rob Fairley, Richard McCoy and Sergio Sanchez Picado of Gribbles Pathology, and Adrienne French, Catherine Williamson, dawn Seddon, Geoff orbell and Cathy Harvey of New Zealand Veterinary Pathology.
Date accepted: 7 March 2017
Supplementary material
Table S1: Multilocus sequence data concerning M lepraemurium.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Carolyn o’Brien was supported by an Australian Postgraduate Award scholarship. Richard Malik was supported by the Valentine Charlton Bequest administered by the Centre for Veterinary Education, The University of Sydney.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
