Abstract
Objectives
The aim of the study was to determine the effect of a common clinical practice, the transmucosal (TM) application of corn syrup, on postoperative blood glucose concentrations in kittens.
Methods
This study was designed as a randomized controlled trial. Seventy-five kittens between the age of 8 and 16 weeks scheduled for surgical sterilization by gonadectomy at an animal shelter were enrolled. Kittens were randomly assigned to either a routine treatment group that received the shelter’s typical postoperative application of corn syrup immediately following gonadectomy or to a control group that did not receive typical treatment. Three blood glucose measurements were obtained per kitten: baseline reading prior to preoperative examination, immediately postoperatively and 20 mins postoperatively. The median values of the 20 min postoperative blood glucose reading for the control and treatment groups were compared using the Wilcoxon rank-sum test.
Results
Postoperative application of corn syrup to the oral mucosa of kittens did not result in significant elevations in blood glucose compared with controls. No kitten was hypoglycemic (⩽60 mg/dl) at any point during the study.
Conclusions and relevance
TM application of corn syrup did not affect postoperative blood glucose concentrations in kittens. Protocols that use routine TM administration of corn syrup in kittens should be re-evaluated.
Introduction
Numerous studies within the past two decades have shown few adverse physiologic effects and low complication rates when performing pediatric gonadectomy procedures.1–7 In fact, surgical time is significantly shorter, healing is more rapid and pre-adoptive sterilization in shelters eliminates the need for adopter compliance.2,4,8 Thus, acceptance of this practice is growing and it is commonplace within animal shelters and ‘high quality high volume’ (HQHV) surgical sterilization clinics.
Despite the growing popularity of early-age gonadectomy, concerns remain about the appropriate management of pediatric patients. One common concern is the increased risk of hypoglycemia due to a lack of glycogen stores within the immature liver.9–16 The potential for developing hypoglycemia is likely increased during periods of fasting, stress and anesthesia, and can result in complications even during elective and uncomplicated surgery and recovery.17,18
In human medicine, there is some evidence supporting the transmucosal (TM) administration of dextrose to treat hypoglycemia in infants.19,20 Similarly, veterinary surgical guidelines have suggested routine swabbing of the gums of pediatric patients with corn syrup postoperatively in order to prevent or treat hypoglycemia. 11 Despite the lack of evidence, this practice is a routine component of the recovery protocol for pediatric patients in many animal shelters and HQHV sterilization clinics. The aim of this study was to evaluate the effect of routine oral TM application of corn syrup on postoperative blood glucose concentration in kittens.
Materials and methods
Animals
Kittens undergoing elective gonadectomy at a local animal shelter were included in the study if they were between the ages of 8 and 16 weeks and apparently healthy on physical examination (American Society of Anesthesiologists [ASA] status 1). Age was initially reported by shelter staff and then confirmed by weight and dentition on physical examination by a veterinarian (REK). Kittens were housed with littermates if present or singly if not. Kittens had food removed no later than midnight prior to surgery. Water remained freely available. All study procedures were approved by the Institutional Animal Care and Use Committee of Midwestern University.
Defining hypoglycemia and hyperglycemia
The determination of a blood glucose cut-off for hypoglycemia in human neonatal and pediatric patients is controversial despite significant study. 21 In human pediatric patients, a hypoglycemic threshold of 65–68 mg/dl has been established. 14 In small animal patients, including adult cats specifically, hypoglycemia has been defined as a blood glucose of ⩽60 mg/dl.22,23 Hypoglycemia was defined as ⩽60 mg/dl for the purposes of this study.
Hyperglycemia is defined as a blood glucose concentration >150 mg/dl in human pediatric studies and is the value above which postoperative complications in human patients are associated.13,14 This concentration is just slightly above the upper range of euglycemia in non-fasted 7-week-old kittens (105–145 mg/dl). 24 Hyperglycemia was defined in the present study as a blood glucose concentration >150 mg/dl.
Determination of corn syrup dose
Cotton-tip applicators soaked in corn syrup are the typical vehicle used for routine postoperative application of corn syrup. The maximum possible volume of corn syrup conveyed by a cotton-tip applicator was calculated in order to standardize the application for the study. To achieve this, five dry swabs from three different brands of cotton-tip applicators were labeled and weighed on a microgram scale, then soaked in corn syrup for 24 h. The loaded swabs were then weighed, and the original dry weight was subtracted from the loaded weight to calculate swab capacity. Next, three swabs from the brand of cotton-tip applicator with the greatest capacity were soaked in corn syrup (Karo Light Corn Syrup; ACH Food Company) for 24 h, weighed and then the contents transferred to the oral mucosa of a 12-week-old kitten. The discharged swabs were weighed and the discharged weight subtracted from the loaded weight. The volume of corn syrup that could most optimistically be expected to transfer was calculated using the greatest difference (0.47 g) and dividing by the density of light corn syrup (1.33 mg/ml) to arrive at a volume of 0.35 ml. There is 1000 mg/ml sugar, mainly dextrose, in corn syrup, so that a 0.35 ml volume of corn syrup equates to a 350 mg dose of dextrose per kitten.
Blood sample collection
A handheld glucometer (AlphaTRAK 2; Abbott Laboratories) was used to measure blood glucose concentration. The same unit was used for all sample readings to eliminate inter-glucometer variability. Each batch of test strips was calibrated with control solution, as per the manufacturer’s guidelines, prior to use. Blood samples were collected from the metacarpal and metatarsal paw pads of each kitten using the lancet that was provided with the glucometer. All researchers were trained on use of the glucometer from the manufacturer’s training literature. Three samples were taken per kitten: (1) baseline reading prior to performing the preoperative examination; (2) immediately postoperatively; (3) 20 mins postoperatively. A different paw pad was used to acquire each sample to avoid sampling from sites of prior collection trauma.
Perioperative protocols
Kittens scheduled for surgery that qualified for enrollment in the study were entered into a Microsoft Excel spreadsheet and assigned a random decimal number between 0 and 1. The kittens were sorted by this number in order to create a random surgical order, then the number was rounded to an integer value to determine routine treatment (1) and control (0) groups.
All kittens were induced using a single intramuscular administration of a combination of dexmedetomidine (0.02 mg/kg [0.04 mg/lb]), ketamine (2.75 mg/kg [6.06 mg/lb]) and butorphanol (0.275 mg/kg [0.606 mg/lb]) (DKT). Female kittens were intubated and maintained at a surgical plane of anesthesia throughout the surgery on isoflurane. Additional postoperative analgesia beyond the DKT was available on a rescue basis. Sterilization procedures were performed by the same veterinarian (REK), using common HQHV pediatric surgical techniques. The median surgical time for spays was 5 mins (range 4–19 mins) and 1 min (range 1–15 mins) for neuters. Surgical times were skin to skin and rounded to the nearest minute.
Kittens in the routine treatment group received an oral mucosal administration of corn syrup immediately following their postoperative blood sampling. A 1 ml syringe was used to apply 0.35 ml corn syrup evenly along the left and right sides of the maxillary buccal mucosa, with care taken to avoid extra-oral application.
Kittens were recovered following the animal shelter’s protocol. Temperature, pulse and respiration measurements were taken immediately upon entry to the recovery area and kittens with temperatures <37.2°C (99°F) were actively warmed until reaching normothermia. After completion of the 20 min postoperative blood draw, kittens that were not ambulatory were administered a single reversal dose of atipamezole (0.01 mg/kg [0.02 mg/lb] IM). Kittens continued to be monitored until fully sternal. The researcher in the recovery area was blind to patient treatment group. Metrics recorded during the recovery period included immediate and 20 min postoperative temperatures, time of extubation and minutes to sternal recumbency.
Statistical analysis
A non-parametric Wilcoxon rank-sum test was used to compare age, weight and body condition score between groups to ensure comparability. Blood glucose values were tested for normality using the Shapiro–Wilk normality test, and were found to be non-normally distributed. A non-parametric Wilcoxon rank-sum test was used to compare 20 min postoperative blood glucose values between treatment groups. A Wilcoxon matched-pairs signed-ranks test was used to compare intra-group measures between immediate postoperative and 20 min postoperative readings.
Results
Animals
Seventy-five kittens (40 male, 35 female) were enrolled in the study. The control group consisted of 37 kittens (20 males, 17 females). The median age of the control group was 10 weeks (range 8–16 weeks) with a median weight of 1.1 kg (range 0.9–1.6 kg) and median body condition score (BCS) on a nine-point scale of 4 (range 4–5). 25 The treatment group consisted of 38 kittens comprised of 20 males and 18 females. The median age of the treatment group was 10 weeks (range 8–16 weeks) with a median weight of 1.1 kg (range 0.9–1.5 kg) and median BCS on a nine-point scale of 4 (range 3–5). The median age, weight and BCS did not differ significantly between the two groups.
Blood glucose concentrations
At baseline, the median blood glucose concentration of kittens in the control group was 96.0 mg/dl (range 62–161 mg/dl) and that of the routine treatment group was 105.5 mg/dl (range 61–148 mg/dl). A second measurement was obtained immediately postoperatively, in which the control group had a median concentration of 144.0 mg/dl (range 85–283 mg/dl) and the treatment group’s median concentration was 143.5 mg/dl (range 74–241 mg/dl). A final measurement was obtained 20 mins postoperatively, in which the control group’s median concentration was 159.0 mg/dl (range 77–337 mg/dl) and the routine treatment group was 157.0 mg/dl (range 61–279 mg/dl). The medians of the blood glucose concentrations at each time point were not significantly different between the control and treatment groups. Median blood glucose increased in both the control (P = 0.0439) and treatment (P = 0.0102) groups between the immediate postoperative and 20 min readings.
The blood glucose concentrations of both groups increased over time (Figure 1). From the baseline pre-examination blood glucose reading following an overnight fast, none of the kittens were hypoglycemic based upon blood glucose concentrations and no kittens showed clinical evidence of hypoglycemia. One percent (1/75) of kittens were hyperglycemic. At the immediate postoperative reading, no kittens were hypoglycemic, 56% (42/75) had blood glucose concentrations between 60 and 150 mg/dl and 44% (33/75) were hyperglycemic. The final blood glucose measurement, taken 20 mins postoperatively, showed both the control and treatment group having a similar distribution and spread, with no kittens from either group being hypoglycemic (Figure 2). At this point, over half of the kittens (55% [41/75]) were hyperglycemic.
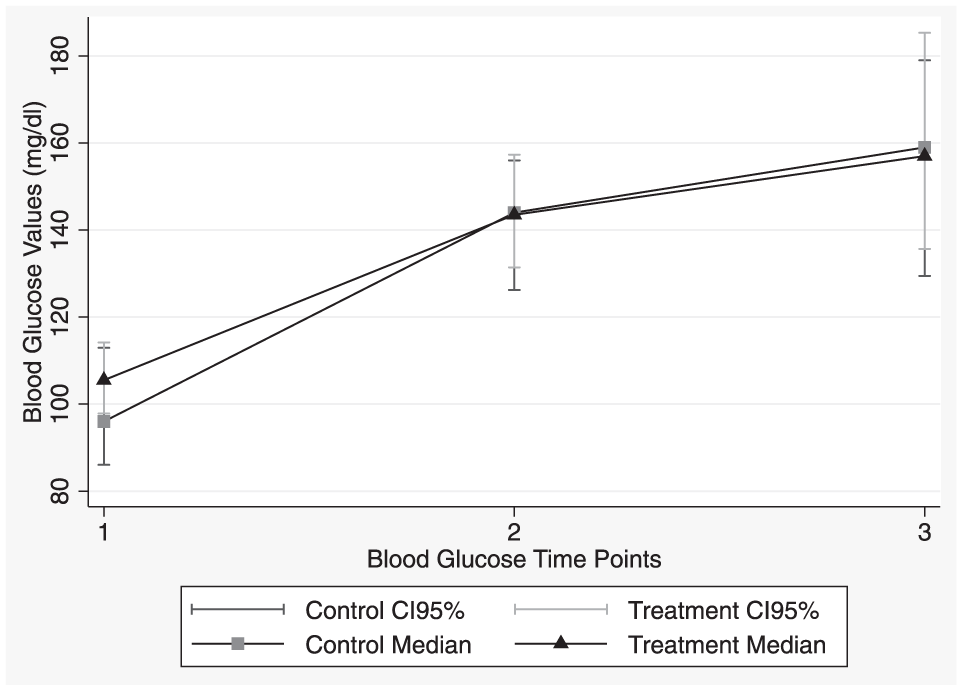
Plot of the medians with 95% confidence intervals (CIs) for the treatment and control groups at: (1) baseline; (2) immediately after surgery; and (3) 20 mins after surgery

Histograms of the 20 min postoperative blood glucose readings for the control (left) and treatment (right) groups with lines at hypoglycemia (60) and hyperglycemia (150)
Discussion
In this study, the common protocol of postoperative TM application of corn syrup did not induce a significant increase in blood glucose concentration compared with controls. This may be due to several factors, such as timing, route or dose. The aim of postoperative TM corn syrup is to prevent hypoglycemia in the early postoperative period prior to a patient regaining the ability to swallow. A 20 min postoperative measurement therefore seemed likely to capture any effect within the desired window of time. In addition, plasma glucose concentrations from sublingual administration of dextrose in children have been shown to peak at 20 mins, but it is unknown whether the same timing would be true for kittens.26–28
The TM route of administration may be a reason for failure of treatment to cause a significant change in blood glucose. Much of the information regarding oral glucose transport is based upon research in human patients; however, unlike humans, cats are obligate carnivores that lack expression of a gene needed to detect sweet tastes and do not exhibit a dietary preference for sugars.29,30 It is unknown if cats possess glucose transporters within the oral cavity similar to other species. If not, the lack of a significant effect of corn syrup application may be due to the inefficient nature of buccal application.
The dose of corn syrup in this study was used to mimic existing protocols in shelters and HQHV clinics when administered by cotton swab. The corn syrup was administered via syringe in this study to standardize the amount between study patients. The dose of dextrose given was 350 mg, which represents a dose per kitten between 233 and 389 mg/kg based on the weight of treatment group kittens; this is comparable to recommended intravenous doses in veterinary and human medicine.22,31 If kittens potentially have poor oral absorption, the dose used may be insufficient; however, this volume of corn syrup approaches the largest realistic dose that could safely be administered in clinical practice, owing to concern over providing a large volume of an oral solution to a patient without airway control.
All study kittens, independent of treatment with glucose, were able to maintain a blood glucose concentration above the hypoglycemic threshold of 60 mg/dl after being subjected to an overnight fast and routine gonadectomy procedure. This finding may be specific to an HQHV or similar clinical scenario, and we urge caution in over-interpretation of the results. All kittens were ASA status 1 and surgical times were very short; perioperative blood glucose may be different in debilitated animals or those undergoing lengthier anesthetic and surgical procedures.
Interestingly, even though the traditional concern is for pediatric patients to develop hypoglycemia postoperatively, the majority of kittens (55% [41/75]) in the present study were found to be hyperglycemic following the 20 min recovery period. One of the factors that may be driving the increase is stress-induced hyperglycemia. Stress hyperglycemia is routinely seen in cats and can present a challenge to obtaining accurate blood glucose concentrations within a veterinary clinic setting.32–35 In the present study, obtaining blood glucose readings, physical examination, induction drugs and handling during recovery could all contribute to a stress response.
Surgery and anesthesia are also factors that may contribute to hyperglycemia. Surgical trauma triggers the hypothalamic–pituitary–adrenal axis to release cortisol, which promotes gluconeogenesis in the liver. 36 One study found that in 10 adult cats presented for ovariohysterectomy, all cats were euglycemic preoperatively and hyperglycemic (>150 mg/dl) postoperatively. 37 Besides the surgical stress response, the anesthetic drugs used in this study may also contribute to hyperglycemia. No studies have demonstrated alterations in blood glucose secondary to dexmedetomedine administration in cats; however, other alpha-2 agonists such as medetomidine have been linked to an increase in glucose concentrations.38–40 An increase in plasma glucose concentration is seen within 15 mins of medetomidine administration to adult cats; however, a statistically significant increase from baseline values is not seen until 1 h post-administration for nearly all concentrations administered.39,40 All blood glucose readings from our study were conducted prior to this 1 h time point, but future studies might be performed to evaluate alternate drug combinations. Additionally, compared with dogs, cats have been shown to exhibit a higher degree of drug- and surgical-induced hyperglycemia, and differing liver enzyme activity may allow cat livers to produce more glucose.37,40,41 Thus, future studies using pediatric canine patients are warranted, as physiologic responses between cats and dogs are likely different.
Additionally of note, hyperglycemia has been shown in human medicine to have detrimental effects on surgical outcomes, including pediatric patients specifically.13,14,36,42 Further studies are needed in veterinary medicine to determine if the same hyperglycemic adverse effects seen in human patients, such as delayed healing and increased risk of infection, also arise from transient hyperglycemia during the perioperative period in animals. If hyperglycemia has deleterious effects, the routine attempt to increase the blood glucose of kittens perioperatively may be undesirable in this patient population.
Conclusions
This study provides evidence for clinics that routinely perform pediatric spay-and-neuter procedures to reference when developing perioperative management protocols. Fasted pediatric kittens undergoing gonadectomy procedures did not become hypoglycemic in this study, suggesting that routine supplementation of dextrose is unwarranted. Additionally, those kittens that are truly hypoglycemic may require an alternative route of administration as no significant increase in blood glucose concentration was seen with TM application of corn syrup.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
