Abstract
Objectives
Hypertrophic cardiomyopathy, a primary disorder of the myocardium, is the most common cardiac disease in cats. However, determination of myocardial deformation with two-dimensional speckle-tracking echocardiography in cats with various stages of hypertrophic cardiomyopathy has not yet been reported. This study was designed to measure quantitatively multidirectional myocardial deformations of cats with hypertrophic cardiomyopathy.
Methods
Thirty-two client-owned cats with hypertrophic cardiomyopathy and 14 healthy cats serving as controls were enrolled and underwent assessment of myocardial deformation (peak systolic strain and strain rate) in the longitudinal, radial and circumferential directions.
Results
Longitudinal and radial deformations were reduced in cats with hypertrophic cardiomyopathy, despite normal systolic function determined by conventional echocardiography. Cats with severely symptomatic hypertrophic cardiomyopathy also had lower peak systolic circumferential strain, in addition to longitudinal and radial strain.
Conclusions and relevance
Longitudinal and radial deformation may be helpful in the diagnosis of hypertrophic cardiomyopathy. Additionally, the lower circumferential deformation in cats with severe hypertrophic cardiomyopathy may contribute to clinical findings of decompensation, and seems to be related to severe cardiac clinical signs. Indices of multidirectional myocardial deformations by two-dimensional speckle-tracking echocardiography may be useful markers and help to distinguish between cats with hypertrophic cardiomyopathy and healthy cats. Additionally, they may provide more detailed assessment of contractile function in cats with hypertrophic cardiomyopathy.
Introduction
Hypertrophic cardiomyopathy (HCM), a primary disorder of the myocardium, is the most common cardiac disease in cats.1,2 Histopathologically, it is associated with myocardial hypertrophy, fibre disarray, increased loose connective tissue, intramural coronary arterial narrowing, myocardial ischaemia and fibrosis, all of which are believed to interfere with the generation of force and relaxation of the cardiac muscle. 3 However, conventional echocardiography has revealed that systolic function in HCM cats is hyperdynamic, with a high ejection fraction and fractional shortening (FS).4,5
Recently, two-dimensional speckle-tracking echocardiography (2D-STE) has been used for the diagnosis and assessment of HCM in humans and cats.6–10 This technique has enabled the assessment of myocardial variables, such as strain and strain rate, which provide better quantification of regional and global myocardial deformations and might have higher sensitivity than conventional echocardiographic parameters for detecting subtle myocardial function abnormalities.11–13 Furthermore, 2D-STE enables the assessment of myocardial function in three directions (longitudinal, circumferential, radial).7,11,14 Previous feline 2D-STE studies demonstrated that systolic segmental radial deformations, 8 torsional assessments 10 and diastolic deformations8,9 were helpful to distinguish between cats with HCM and healthy cats. However, this multidirectional assessment of myocardial function with 2D-STE has not been previously reported in cats with various stages of HCM. This study was designed to measure quantitatively multidirectional myocardial deformations in cats with HCM. We hypothesised that myocardial deformations assessed with 2D-STE could be used to detect HCM and would be sensitive markers of systolic dysfunction in cats with HCM.
Materials and methods
Animals
Our study population consisted of 32 client-owned cats with HCM and 14 healthy cats serving as controls. These cats were presented for cardiac screening at the Veterinary Medical Hospital of Nippon Veterinary and Life Science University in Japan, and their clinical findings were analysed retrospectively. The breeds of the HCM cats were Scottish Fold (n = 14), Ragdoll (n = 4), domestic shorthair (n = 3), Norwegian Forest Cat (n = 3), Maine Coon (n = 2), Russian Blue (n = 2), Munchkin (n = 2), Bengal (n = 1) and Exotic Shorthair (n = 1). Cats with HCM were diagnosed if the echocardiographic left ventricular (LV) wall thickness was 6 mm or more, measured by 2D methods. LV thickness was measured from the short-axis view and the mean values of three consecutive cardiac cycles of the thickest segment were used. We excluded cats that had systolic blood pressure >160 mmHg (non-invasive oscillometric methods) or systemic and other cardiovascular diseases known to cause LV hypertrophy. There was no evidence of dehydration. Cats with LV outflow obstruction were also included in the study. LV outflow obstruction was defined as turbulent LV outflow with high velocity (>2.0 m/s) by using continuous-wave Doppler ultrasound.9,10 Cats that had already been treated by a referral hospital were included in the study.
HCM cats were divided into three groups according to their clinical signs: asymptomatic, symptomatic without congestive heart failure (CHF–) and symptomatic with CHF (CHF+). These cats were categorised based on a combination of owner responses to detailed questioning, physical examination, and thoracic radiographic and echocardiographic findings. Asymptomatic cats had echocardiographic HCM findings but no clinical signs of heart failure. Symptomatic cats without CHF (CHF–) had echocardiographic HCM findings and mild clinical signs (exercise intolerance, dyspnoea and tachypnoea). Symptomatic cats with CHF (CHF+) had echocardiographic HCM findings, severe clinical signs (exercise intolerance, dyspnoea, tachypnoea at rest or with mild exercise) and past or current radiographic signs of CHF (pulmonary oedema, pericardial effusion and pleural effusion). Symptomatic cats were managed with medications and monitored carefully during examinations. After examination at our hospital, the medications were reconsidered in accordance with HCM severity. None of the cats required hospital treatment. The control cats were healthy, with normal findings on complete physical examination, electrocardiography, thoracic radiography and transthoracic echocardiography. None of the control cats had a history of clinical signs of cardiac disease.
Standard echocardiography
Conventional 2D and Doppler examinations were performed by a single trained investigator using a Vivid 7 echocardiographic system (GE Healthcare) and 7S transducer, and simultaneous ECG limb lead II was recorded and displayed on the images. All data were obtained for at least three consecutive cardiac cycles in sinus rhythm from non-sedated cats that were manually restrained in right and left lateral recumbent positions. A single trained observer analysed the images using an off-line workstation (EchoPac PC, version 108.1.4; GE Healthcare). The left atrial to aortic root ratio was obtained from the right parasternal short-axis view using the B-mode method. 15 End-diastolic interventricular septal thickness (IVSd), end-diastolic LV free-wall thickness (LVFWd), end-diastolic LV internal diameter (LVIDd), end-systolic left ventricular internal diameter (LVIDs) and fractional shortening were measured using the B-mode method from the right parasternal short-axis view of the LV. As indicators of LV hypertrophy, the relative wall thickness was calculated as the ratio of the sum of the IVSd and LVFWd to the LVIDd. 16 Transmitral flow was obtained from the left apical four-chamber view, and the peak velocity of the early diastolic wave (E-wave), deceleration time of the E-wave and peak velocity of the late diastolic wave (A-wave) were measured. In cats in which the E and A waves were fused, values for those waves were not used. For all analyses, the mean values of three consecutive cardiac cycles in sinus rhythm from high-quality images were used.
2D-STE
2D-STE protocols previously published for dogs and cats were applied.10,12–14,17,18 High-quality images for 2D-STE analysis were carefully obtained by the same investigator. To evaluate radial and circumferential deformation by 2D-STE, we used a right parasternal short-axis view of the LV at the level of the papillary muscles. A left apical four-chamber view was used to analyse longitudinal deformation. Images were again analysed by a single observer using an offline EchoPAC workstation as described previously.10,12–14,17,18 We selected one cardiac cycle (from one QRS complex to the next QRS complex) from the high-quality images and manually traced the endocardial borders of the myocardium in end-diastole to select the appropriate region of interest. Each region of interest was then adjusted to incorporate the entire myocardial thickness and checked by the observer to ensure that it was visually synchronised with the cardiac movement throughout the entire cardiac cycle. The computer software automatically traced the myocardium, created six segments in each image and evaluated whether it reliably followed the myocardial motion. If the initial evaluation failed as a result of an inability to trace the region of interest during a myocardial movement, the endocardial borders were retraced and manually corrected as needed. If multiple evaluation attempts failed, the failed segment was excluded from the analysis. Finally, we measured the peak systolic strain and the peak systolic strain rate in the longitudinal, circumferential and radial directions. The mean of all six segmental values was used for all variables. The mean values of the measurements from three consecutive cardiac cycles from high-quality images were used in all analyses.
Statistical analysis
Data are expressed as medians and interquartile ranges. All statistical analyses were performed with SPSS software version 15.0J for Windows and R software version 2.8.1. The Kruskal–Wallis test was used to compare the distribution of value of age, body weight, body surface area, heart rate, systolic blood pressure and all echocardiographic variables among the groups (controls, asymptomatic, CHF– and CHF+). Parameters that demonstrated significant differences among groups using the Kruskal–Wallis test were then subjected to multiple comparisons using the Steel–Dwass test. Linear regression analysis was performed to examine correlations of 2D-STE variables with IVSd and LVFWd. Values of P <0.05 were considered significant. Intraobserver reproducibility was assessed by having the observer repeat measurements three times for three randomly selected cats on different days and determining the coefficients of variation.
Results
Feline profiles and standard echocardiography
The clinical characteristics and conventional echocardiographic data of healthy controls and cats with HCM are summarised in Table 1. Body weight, heart rate and systolic blood pressure did not differ significantly among the four groups. The asymptomatic cats were significantly older than the controls (P = 0.046). Twelve percent of the asymptomatic cats (2/17), 44% of the CHF– cats (4/9) and 100% of the CHF+ cats were receiving medical treatment from the referral hospital at the time of the examination. Angiotensin-converting enzyme inhibitors were given to cats in the asymptomatic (2/17; 12%), CHF– (3/9; 33%) and CHF+ (3/6; 50%) groups. A calcium channel blocker was given to cats in the CHF– (1/9; 11%) and CHF+ groups (2/6; 33%). A beta-blocker was given to cats in the CHF– (2/9; 22%) and CHF+ groups (2/6; 33%). Isosorbide dinitrate was given to cats in the CHF– (2/9; 22%) and CHF+ groups (1/6; 17%). Furosemide was given to cats in the CHF+ group (4/6; 67%). Thromboprophylaxis was given to cats in the CHF– (3/9; 33%) and CHF+ groups (2/6; 33%).
Characteristics and conventional echocardiographic data for healthy cats and cats with hypertrophic cardiomyopathy

Data are expressed as medians (25–75% interquartile range) unless otherwise indicated
CHF– indicates cats without congestive heart failure findings
CHF+ indicates cats with congestive heart failure findings
Within a row, values differed significantly (P <0.05) from controls
HR = heart rate; bpm = beats per min; SBP = systolic blood pressure; LVOTO = left ventricular outflow tract obstruction; LA/Ao = left atrial to aortic root ratio; LVIDd = end-diastolic left ventricular internal diameter; LVIDs = end-systolic left ventricular internal diameter; RWT = relative wall thickness; FS = fractional shortening
2D-STE
All views for the analysis of 2D-STE were recorded at average rates of 155 (116–214) frames/s, which is adequate for feline evaluation.8,9,19 After analysis using an off-line workstation, 777/792 (98.1%) segments for long-axis images and 792/792 (100%) segments for short-axis images were included for statistical analysis. The average coefficients of variation for intraobserver reliability for longitudinal, circumferential and radial deformations were 4.1%, 3.7% and 8.3%, respectively.
The 2D-STE data for healthy cats and cats with HCM are summarised in Table 2. Cats with asymptomatic CHF and CHF+ groups had significantly lower peak systolic longitudinal strain values (P = 0.014 and P = 0.02, respectively). Cats with asymptomatic HCM and CHF+ groups had significantly lower peak systolic radial strain values (P = 0.004 and P = 0.015, respectively). HCM cats with CHF had also significantly lower peak systolic radial strain rate and circumferential strain values (P = 0.015 and P = 0.04, respectively). Other deformations did not significantly differ between the classes of HCM and controls.
Peak systolic two-dimensional speckle-tracking echocardiography data for healthy cats and cats with hypertrophic cardiomyopathy
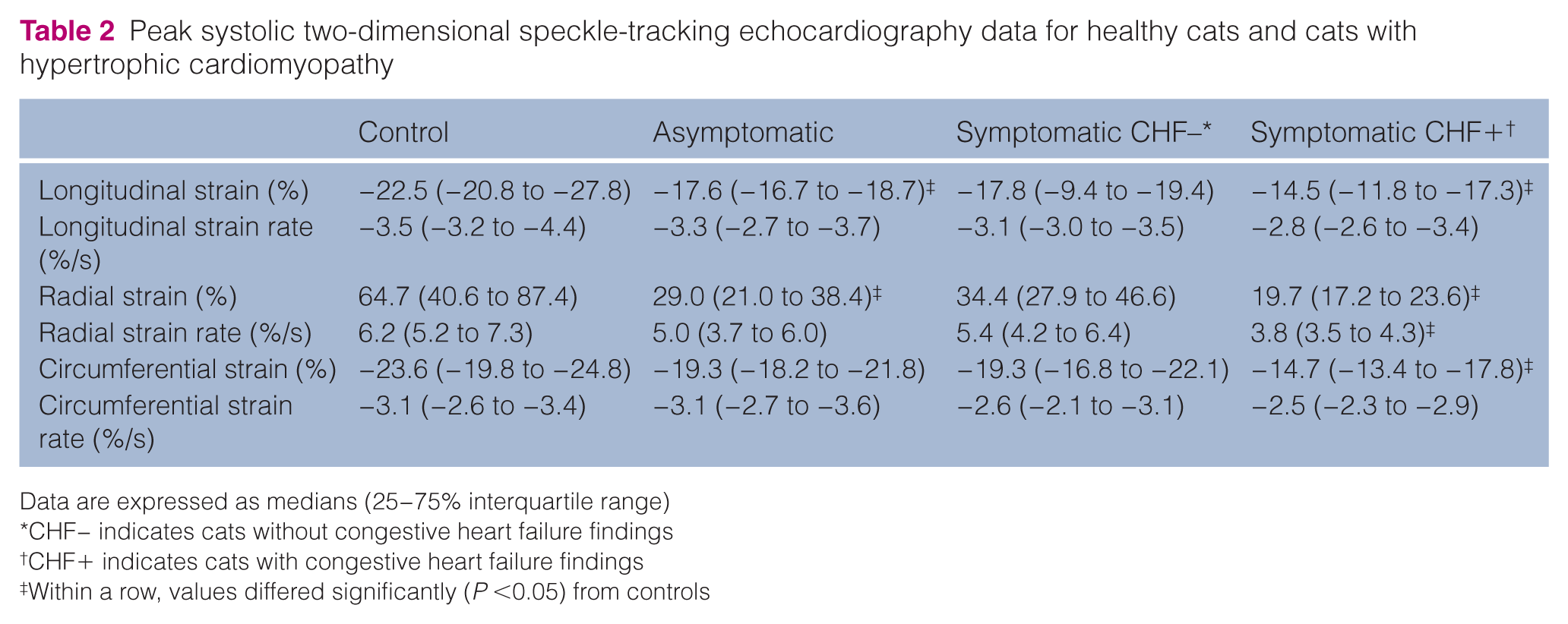
Data are expressed as medians (25−75% interquartile range)
CHF− indicates cats without congestive heart failure findings
CHF+ indicates cats with congestive heart failure findings
Within a row, values differed significantly (P <0.05) from controls
Peak systolic longitudinal strain was significantly correlated with IVSd and LVFWd (ρ = −0.726, P <0.001; and ρ = −0.576, P <0.001, respectively). Peak systolic radial and circumferential strains were correlated with IVSd and LVFWd (IVSd: ρ = −0.396, P = 0.007 and ρ = −0.425, P = 0.004; LVFWd: ρ = −0.434, P = 0.003 and ρ = −0.348, P= 0.019). Radial and circumferential strain rates were also correlated with LVFWd (ρ = −0.335, P = 0.023 and ρ = −0.322, P= 0.029, respectively).
Discussion
We found that multidirectional myocardial deformations could be assessed with 2D-STE to evaluate the myocardial function of cats, with adequate repeatability. The present study also demonstrated that 2D-STE analysis allows non-invasive detection of abnormal systolic deformations in cats with HCM, despite an apparently normal or supernormal contractile state of the LV based on conventional echocardiography. These deformations may have been altered by myocardial compensatory mechanisms and myocardial pathological changes. In the clinical progression of HCM in cats, myocardial deformations differed according to the direction of myocardial contraction. Therefore, the determination of multidirectional myocardial deformations may facilitate detailed assessment of systolic function in cats with HCM.
Previous studies demonstrated that myocardial functional variables derived from tissue Doppler techniques were useful for detecting myocardial systolic impairment in cats with HCM.4,5,20 Wess et al reported that longitudinal strain assessed by tissue Doppler imaging was decreased in cats with HCM, 5 and decreases more in more severe HCM stages. Likewise, we showed that depressed longitudinal strain was observed in cats with HCM. However, measurements obtained using tissue Doppler technique are affected by the translation of the heart itself, the tethering of surrounding myocardial motions and Doppler angle dependency.21,22 Furthermore, tissue Doppler measurement is weakly associated with myocardial contractile function in human patients with LV dilation. 21 A newly developed echocardiographic tool, 2D-STE, tracks grey-scale B-mode images of unique speckle patterns in the myocardium and is therefore relatively independent of the cardiac translation, tethering and angle parameters. 23 Also, the 2D-STE enables the assessment of myocardial function in three directions (longitudinal, circumferential, radial).7,11,14 As the diagnosis of HCM in cats is sometimes difficult, assessment of the myocardium using 2D-STE may be useful to distinguish between HCM and healthy cats.
In the present study, cats with HCM had adequate systolic function compared with controls, as indicated by similar LVIDs and fractional shortening values. However, cats with HCM had decreased longitudinal and radial deformations compared with controls, as assessed by 2D-STE. These findings may suggest occult systolic dysfunction in cats with HCM, which is consistent with previous studies.5,7,24 Histopathological changes and alterations in myocardial fibre orientation may be related to these functional abnormalities. 25 A previous study demonstrated that force-length relations are reduced in humans with HCM, indicating that myocardial contractility is depressed in some regions. 26 Furthermore, Zimmer et al found increased myocardial fibre diameter to be a determinant of impaired LV function, regardless of the underlying disease. 27 These myocardial abnormalities, as assessed by 2D-STE, may help the detection of HCM disease.
In this study, some myocardial deformations were significantly related to LV wall thickness. Similarly, a previous feline study demonstrated that tissue Doppler-derived longitudinal strain was decreased with increasing left ventricular concentric hypertrophy. 5 These relationships may be explained by an association of the degree of myocardial changes and ischaemia with the severity of wall thickening. 28 However, Maron et al found no correlation between LV wall thickness and the amount of disordered tissue, 29 with many thickened regions containing large proportions of organised fibres and regions of normal thickness containing disordered architecture. Myocardial deformations derived by 2D-STE may reflect subtle functional abnormalities independent of morphological changes of the myocardium. 30 These relationships among myocardial deformations, systolic function and histopathological characteristics in the myocardial wall in cats with HCM should be investigated in future studies.
Circumferential strain in severely symptomatic cats with HCM was lower than that of controls. Circumferential deformations play an important role in cardiac pump function in people with cardiac disease,31,32 and LV myocardial contractions that are impaired in the longitudinal direction are compensated for by circumferential shortening to preserve pump function in subclinical patients with cardiovascular risk factors. 33 Circumferential deformations may indicate a more severe degree of myocardial damage and subsequent progression of cardiac symptoms. Accordingly, the lower circumferential deformation in cats with severe HCM may contribute to clinical findings of decompensation and seems to be related to severe cardiac clinical signs. Impaired myocardial functions are associated with the progression of clinical signs and adverse cardiovascular outcomes in patients with HCM.34,35 Although clinical outcome could not be assessed in this study, myocardial deformation derived by 2D-STE may be related to the cardiac clinical signs of cats with HCM.
This study has several limitations. First, because our study was a non-invasive clinical investigation, we could not definitively measure myocardial contractility using the invasive assessment that is the gold standard for assessing LV function. Second, diagnosis of HCM was based on morphological rather than pathological criteria. Third, we did not longitudinally assess myocardial deformation using 2D-STE in the same cats. We were therefore unable to relate changes in myocardial deformation to deterioration or improvement of HCM. Fourth, the enrolled cats were not matched for age. However, systolic myocardial deformations were similar between young and aged dogs. 18 Fifth, the small number of cats in our study may have had an influence on the statistical power and limit the extrapolation of these findings to larger populations. Sixth, using three cardiac cycles for the 2D-STE analysis may be too small for this novel assessment. However, we tried to correct these high-quality views carefully, to ensure that they represented conditions that cats were as calm as possible. Also, our results suggest that 2D-STE repeatability was clinically adequate. Finally, we did not consider the influences of medications on the measured parameters. These medications may affect myocardial function, thereby complicating the interpretation of our results. These limitations should be addressed in future investigations.
Conclusions
Myocardial deformations assessed by 2D-STE differed in cats with HCM compared with controls, despite their apparently normal systolic function, as determined by conventional echocardiography. Longitudinal and radial deformations may be markers for the detection of HCM disease. Furthermore, the lower circumferential strain in cats with severe HCM may contribute to clinical findings of decompensation and seems to be related to severe cardiac clinical signs. Measurement of multidirectional myocardial deformations using 2D-STE may provide more detailed assessment of contractile function in cats with HCM. Nevertheless, the clinical importance of these deformations and their relationship to survival time requires further investigation.
Footnotes
Acknowledgements
The authors wish to thank Takahiro Saito, Asahi Takiguchi, Saori Masuda, Yuma Iwase and Takeshi Shintani for their technical assistance.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
