Abstract
Case series summary
This study aimed to evaluate the pathologic and diagnostic features of 13 cases of feline glioma diagnosed at two veterinary diagnostic institutions over 16 years. A retrospective search was conducted using the laboratory information system. Selected cases were reviewed, tumors were classified according to the 2007 World Health Organization Classification of Tumours of the Central Nervous System, and tissue sections were submitted to an immunohistochemistry panel for further characterization. Affected cats were adults (average age 8 years), and male neutered (9/13) domestic shorthair cats (12/13) were over-represented. Clinical signs had acute onset and were progressive, lasting from 1–60 days (average course 20.7 days). Euthanasia was elected in all but one case, owing to a poor prognosis or because of a suspicion of rabies. Tumors occurred in the telencephalon (8/13), spinal cord (3/13) and brainstem (2/13). Gross changes in seven cases consisted of well- or poorly demarcated, gray-to-brown, soft, gelatinous masses that often caused secondary changes to the brain. The final diagnosis and classification were made based on histopathology and immunohistochemistry. Diagnoses consisted of oligodendroglioma (six cases), anaplastic astrocytoma (two cases), oligoastrocytoma, anaplastic ependymoma, gliomatosis cerebri, glioblastoma and anaplastic oligodendroglioma (one case each).
Relevance and novel information
This article confirms the need for a full neurologic and neuropathologic examination in cats with clinical neurologic disease. In addition, it highlights and proposes a complete routine immunohistochemical panel for diagnostic confirmation and differentiation between glioma and other morphologically similar central nervous system neoplasms in cats.
Introduction
Primary central nervous system (CNS) neoplasia is uncommon in dogs and cats, with meningioma as the most prevalent tumor in both species.1–3 Glial tumors or gliomas (astrocytoma, oligodendroglioma and ependymoma) are the second most frequent CNS tumors in dogs and the third or fourth most frequent tumors in cats.1,2,4–6 Much of the current knowledge related to canine glioma has arisen from the fact these tumors share striking clinical and pathologic features to their counterpart in humans. In addition, both species have close genomic similarities, tend to share the same environment and, consequently, be exposed to common possible carcinogens, supporting the comparative aspects of canine glioma. 7 However, little information is available on the pathologic and diagnostic features of feline glioma, as most reports concentrate on its clinical aspects or on the description of single cases.1,5,6,8–24 This article describes the clinicopathologic and diagnostic features of 13 cases of feline glioma diagnosed at the University of Georgia Department of Pathology and Athens Veterinary Diagnostic Laboratory, as well as the Cornell University Section of Anatomic Pathology from 2000–2016, and includes a brief review of the veterinary medical literature on feline glioma. We concentrate on the pathology and diagnostic features of these tumors and on the use of a routine immunohistochemical panel for diagnostic confirmation and differentiation between glioma and other morphologically similar CNS neoplasms in cats.
Case series description
Cases were searched for in the Athens Veterinary Diagnostic Laboratory (AVDL) and the New York State Animal Health Diagnostic Center record systems using the following keywords glioma, astrocytoma, glioblastoma, oligodendroglioma, gliomatosis cerebri and epen-dymoma. Relevant data from retrieved cases were reviewed from the submission forms and necropsy reports. Glass slides were evaluated and tumors were classified according to the 2007 World Health Organization (WHO) Classification of Tumours of the Central Nervous System. 25 Archived paraffin-embedded tissues from cases 1–13 were submitted to immunohistochemistry (IHC) for glial fibrillary acidic protein (GFAP; mouse monoclonal, 1:4000 dilution at 60 min [Biogen]), oligodendrocyte lin-eage transcription factor 2 (Olig2; rabbit monoclonal, 1:400 dilution at 60 min [Genetex]), neuron-specific enolase (NSE; mouse monoclonal, 1:2000 dilution at 60 mins [Zymed]), synaptophysin (SYP; mouse monoclonal, 1:600 dilution at 60 mins [Biogen]), Ki-67 (rabbit monoclonal, RTU at 60 mins [Cell Marque]), CD3 (rabbit polyclonal, 1:1000 dilution at 60 mins [Dako]), CD79a (mouse monoclonal, 1:50 dilution at 60 mins [Biocare]), vimentin (VIM; mouse monoclonal, 1:3000 at 60 mins [Biogen]) and pancytokeratin A3/A9 (PCK; mouse monoclonal, 1:100 dilution at 90 mins [Biocare]). Positive control tissues included normal feline spinal cord (GFAP, Olig2, NSE, SYP), lymph node (Ki-67, CD3 and CD79a), small intestine (VIM) and skin (PCK). A review of the veterinary medical literature on feline glioma was conducted using the PubMed/MEDLINE database and searching for the following keywords: feline, cat, cerebral, spinal cord, glioma, astrocytoma, glioblastoma, oligodendroglioma, gliomatosis cerebri and ependymoma.
Signalment, clinical signs and neuroanatomical localization of the tumors are shown in Table 1. In summary, the age of affected cats varied from 2–17 years (median 8 years). Male neutered (9/13) and domestic shorthair cats (12/13) were over-represented. Clinical signs were reported as acute and progressive and lasted from 1–60 days (median 20.7 days). A clinical diagnosis of an intra-axial mass was made based on MRI in cases 1 and 3. Most cats were euthanized because of a poor prognosis at the time of referral (cases 1, 3–5, 7 and 10–12) or because of suspicion of rabies (cases 2, 6, 8 and 9). Cat 13 died spontaneously. Most tumors occurred in the telencephalon (8/13), followed by spinal cord (3/13) and brainstem (2/10). Gross neuropathologic changes were observed in seven cases, and while all tumors were primarily intraparenchymal, two of the spinal cord tumors (cases 7 and 9) also extended to extraparenchymal (intradural) areas.
Signalment, clinical signs, tumor localization and gross pathology in 13 cases of feline glioma diagnosed from 2000–2016

MN = male neutered; DSH = domestic shorthair; DLH = domestic longhair; NA = not available; FS = female spayed
Histologic findings varied among cases. Cases 1 and 2 (anaplastic astrocytoma) were poorly demarcated, highly cellular tumors that were composed of sheets of closely packed, highly pleomorphic round cells with eosinophilic cytoplasm and indistinct cell borders supported by a fine fibrovascular stroma (Figure 1). Nuclei were round or indented, and had finely stippled-to-vesicular chromatin and 1–2 nucleoli. Mitotic activity was 0 (case 1) and 12 (case 2) in ten × 400 fields with extensive areas of necrosis and haemorrhage present throughout the tumors.

Case 2, anaplastic astrocytoma. There is increased cell and nuclear pleomorphism and mitotic activity (arrow), with extensive areas of necrosis (asterisk). Hematoxylin and eosin. Bar = 100 μm
Case 3 (oligoastrocytoma) was a poorly demarcated, moderately cellular tumor with low atypia and nuclear pleomorphism. Neoplastic cells were arranged in loosely packed sheets supported by a fibrovascular stroma (Figure 2). Approximately 50% of neoplastic cells resembled oligodendrocytes, with round, dense nuclei surrounded by a clear cytoplasm. The second neoplastic cell population was morphologically consistent with astrocytes and had prominent, elongated or stellate, eosinophilic cytoplasm and round-to-oval nuclei with finely stippled chromatin and one nucleolus. Mitotic activity was 2 in ten × 400 fields.
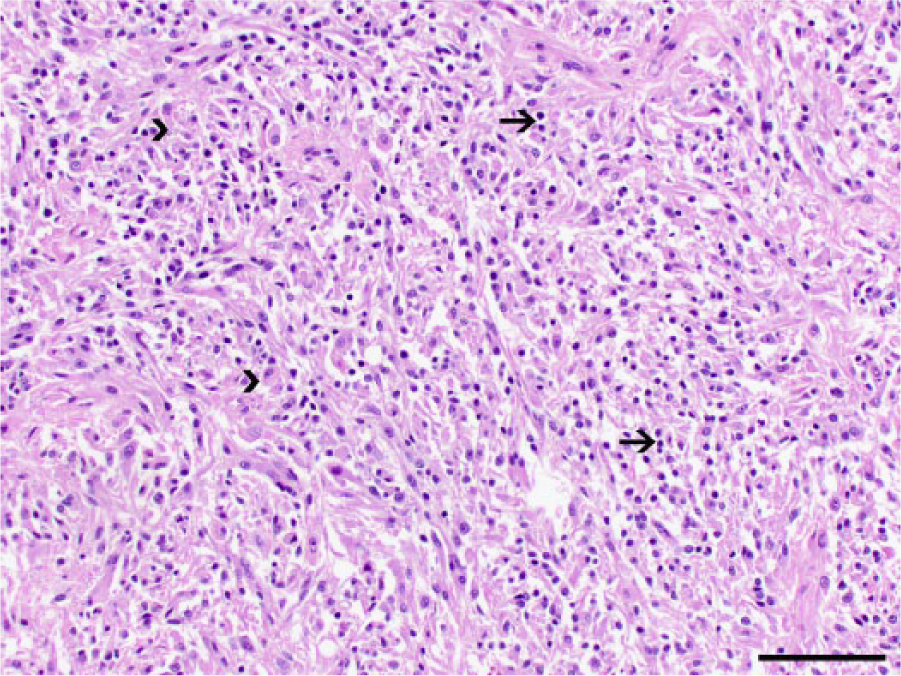
Case 3, oligoastrocytoma. The tumor is composed of two distinct neoplastic cell populations: small, round cells resembling oligodendrocytes (arrows) and large, polygonal cells morphologically consistent with astrocytes (arrowheads). Hematoxylin and eosin. Bar = 200 μm
Cases 4–8 and 13 (oligodendroglioma) were relatively well demarcated, variably cellular tumors with low atypia and nuclear pleomorphism and composed of loosely arranged neoplastic cells on a fine fibrovascular or mucinous stroma (Figure 3). Cells had a moderate amount of eosinophilic, finely granular or clear cytoplasm that often created a perinuclear halo. Nuclei were round and had finely stippled-to-dense chromatin and 1–2 nucleoli. Mitoses were rare or absent. Small areas of necrosis and microvascular proliferation were evident in case 6.

Case 8, oligodendroglioma. Neoplastic cells are morphologically consistent with oligodendrocytes, with a moderate amount of eosinophilic, finely granular or clear cytoplasm that often creates a clear perinuclear halo. Hematoxylin and eosin. Bar = 200 μm
Case 9 (anaplastic ependymoma) was a well-demarcated, highly cellular tumor with moderate atypia and nuclear pleomorphism (Figure 4). Neoplastic cells were arranged in solid areas or palisading around capillaries (pseudorosettes) and supported by a moderate amount of fibrovascular stroma. Cells were polygonal and had abundant, eosinophilic cytoplasm with indistinct borders and round-to-oval nuclei with finely stippled-to-coarse chromatin and 1–3 nucleoli. There were 18 mitoses in ten × 400 fields. Multiple areas of necrosis and haemorrhage were present.

Case 9, anaplastic ependymoma. The tumor is highly cellular, with prominent atypia and nuclear pleomorphism and neoplastic cells arranged in solid areas (asterisk) or palisading around blood capillaries (pseudorosettes) (arrow). Hematoxylin and eosin. Bar = 200 μm
Case 10 (gliomatosis cerebri) was a poorly demarcated, infiltrative neoplasm morphologically similar to cases 4–8. There was no mass formation or disruption of the affected neuroparenchyma. Mitoses were absent.
Case 11 (glioblastoma) was a poorly demarcated, highly cellular tumor with moderate atypia and nuclear pleomorphism. Neoplastic cells were closely arranged on a fine fibrovascular stroma (Figure 5), and had abundant, polygonal or elongate, eosinophilic, fibrillar or vacuolated cytoplasm and round nuclei with finely stippled-to-coarse chromatin and 1–2 nucleoli. Bi- or multinucleate neoplastic cells were scattered throughout. Neoplastic cells frequently palisaded around areas of necrosis. Mitotic rate was 3 in ten × 400 fields.

Case 11, glioblastoma. Neoplastic cells frequently palisade (asterisk) around areas of necrosis (lower right). Hematoxylin and eosin. Bar = 100 μm
Case 12 (anaplastic oligodendroglioma) was a relatively well-demarcated, highly cellular tumor with moderate atypia and nuclear pleomorphism. Neoplastic cells were closely arranged on a fine fibrovascular stroma (Figure 6), and had a moderate amount of eosinophilic, finely granular cytoplasm and round nuclei with coarse chromatin and 1–2 nucleoli. There were four mitoses in ten × 400 fields and extensive areas of necrosis.

Case 12, anaplastic oligodendroglioma. The tumor is highly cellular with extensive areas of necrosis (center). Hematoxylin and eosin. Bar = 100 μm
The IHC findings in all 13 cases are summarized in Table 2 and shown in Figures 7–10. The final diagnosis and tumor grade (Table 2) were based on histopathology and IHC profile, and consisted of oligodendroglioma (six cases), anaplastic astrocytoma (two cases), oligoastrocytoma, anaplastic ependymoma, gliomatosis cerebri, glioblastoma, and anaplastic oligodendroglioma (one case each).
Immunohistochemical findings, diagnosis and grade of 13 cases of feline glioma diagnosed from 2000–2016

Percentage of positive cells
WHO = World Health Organization; GFAP = glial fibrillary acidic protein; Olig2 = oligodendrocytes lineage transcription factor 2; NSE = neuron-specific enolase; SYP = synaptophysin; PCK = pancytokeratin; VIM = vimentin; NA = no classification available
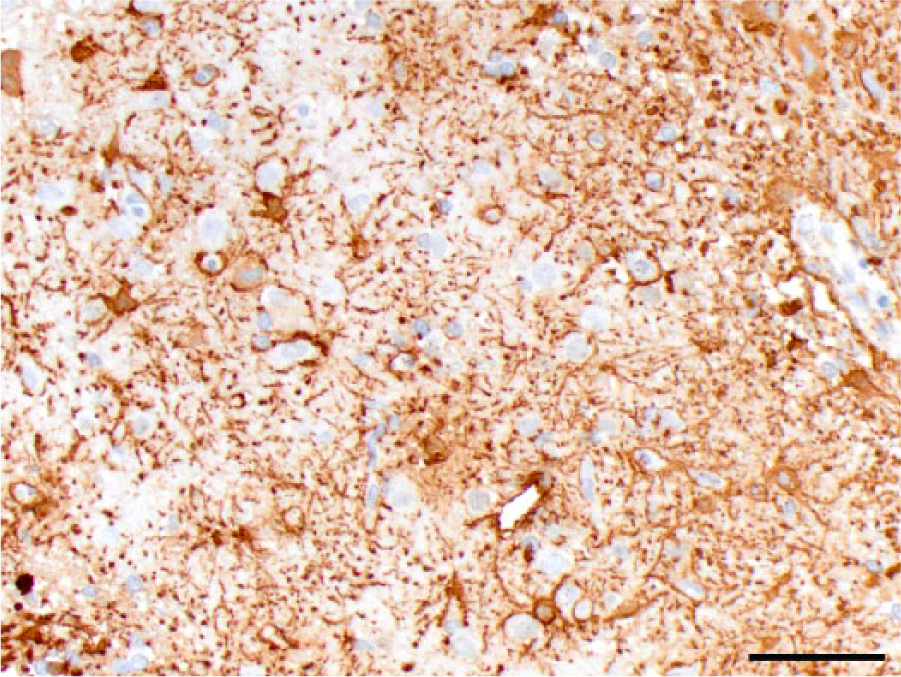
Case 2, anaplastic astrocytoma. Neoplastic cells exhibit strong cytoplasmic immunoreactivity for glial fibrillary acidic protein. Bar = 100 μm
Case 8, oligodendroglioma. Neoplastic cells exhibit nuclear immunoreactivity for oligodendrocyte lineage transcription factor 2. Bar = 100 μm

Case 6, oligodendroglioma. Approximately 60% of neoplastic oligodendrocytes exhibit cytoplasmic immunostaining for synaptophysin. Bar = 200 μm

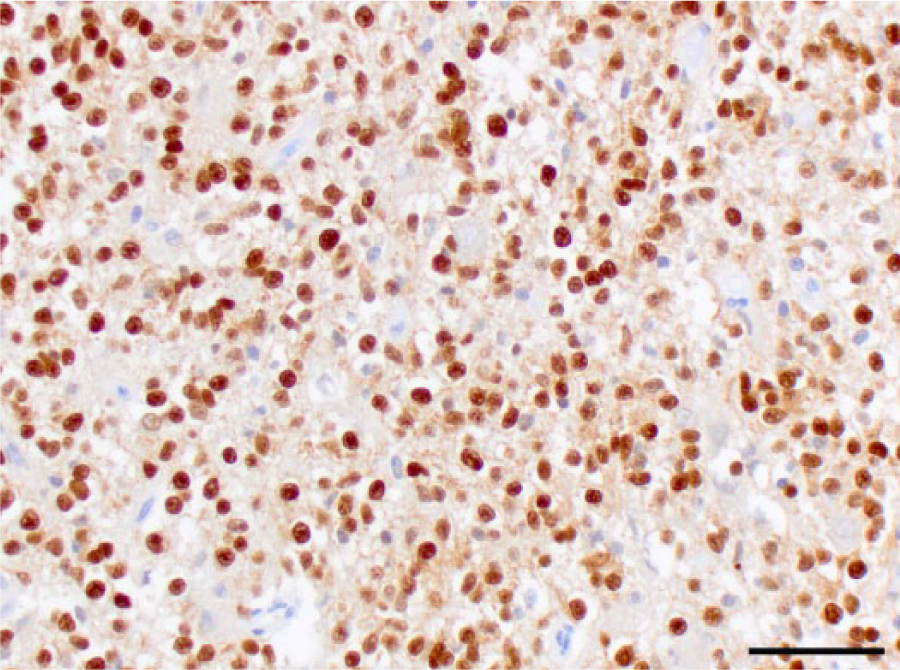
Case 9, anaplastic ependymoma. Numerous neoplastic cells have strong nuclear immunoreactivity for Ki67. Bar = 100 μm
Discussion
Glioma is considered the third or fourth most frequent CNS neoplasm in cats.4–6,26 This article describes the pathological findings of 13 cases of feline glioma diagnosed over a period of 16 years. Tumors were systematically classified and graded according to the 2007 WHO Classification of Tumours of the Central Nervous System and submitted to an IHC panel for diagnostic characterization.
The most frequently reported feline glioma in the veterinary literature is the astrocytoma,3,5,6,8,9,11,15–17,19,21,26–29 followed by ependymoma,3,6,12–14,18,22,24,30,31 oligodendroglioma,5,6,8,10,20,23,26,32 and, less often, mixed glioma, unclassified glioma and gliomatosis cerebri.8,26,27 The most common glioma in the current case series was oligodendroglioma (six cases), followed by anaplastic astrocytoma (two cases), anaplastic ependymoma, gliomatosis cerebri, glioblastoma and anaplastic oligodendroglioma (one case each). A similar search for cases of canine glioma in our archives at the AVDL yielded over 40 cases in the same time period (data not shown), confirming that glial tumors are less commonly diagnosed in cats when compared with dogs.5,6,26,33 Affected cats in the current study were all adult-to-geriatric individuals and no sex or breed predisposition was noted, corroborating data from previous studies.6,8,28 The fact that nine cats were male neutered should be interpreted with caution owing to the low number of cases in this study. Similarly, while 12 cases were domestic shorthair cats, it is likely a reflection of our feline population and not a true breed predisposition. 6
Clinical information is scarce in this study as most of the affected cats were referred to private clinics and did not undergo a complete neurologic examination and diagnostic work-up at the time of evaluation. In contrast, two cases in which an MRI was performed had a clinical diagnosis of intraparenchymal tumor at referral, confirming the importance of neuroimaging in the diagnosis of neurologic diseases in veterinary medicine.4–6 Gliomas are typically intraparenchymal tumors and have ring enhancement and peritumoral edema on MRI. 5 As seen in these two cases, the correlation between the clinical signs and confirmation of a tumor via neuroimaging provides a better understanding of the clinical course and avoids unnecessary biohazard-related efforts when carcasses have to be submitted to the diagnostic laboratory as rabies-suspected cases, as seen with four cases in this study. The clinical course was highly variable in the affected cats, but conclusions cannot be drawn from the clinical data as all individuals were euthanized.
Although a few reports mention a possible predisposition for feline glioma to develop in the brainstem,10,27 the overall distribution of tumors in many publications does not confirm this feature. In fact, these reports reveal that feline glioma occurs predominantly in the telencephalon,5,6,10,11,15–18,20,22–24,28,29,31 followed by spinal cord,8,9,12,19,21,24,26,28 and, less frequently, brainstem and cerebellum.5,6,10,13,14,24,27,28,32 A similar distribution was observed in this study, with eight tumors occurring in the telencephalon, three in the spinal cord, and two in the brainstem and cerebellum.
Although a diagnosis can be made based on routine histological evaluation, glial tumors may share morphologic features that are common to multiple glioma subtypes or with other round cell neoplasms, particularly lymphoma. 4 Therefore, the use of IHC is recommended for diagnostic confirmation in cases of glioma in cats and dogs. 4 While Olig2 and GFAP are not exclusive markers for oligodendrocytes and astrocytes, respectively, widespread expression of Olig2, as observed in the current cases, is a good indicator of the diagnosis of oligodendroglioma.4,8 Similarly, positive GFAP immunostaining does not unequivocally confirm a diagnosis of astrocytoma, but it is highly supportive of such when in combination with negative immunoreactivity for Olig2. 4
Synaptophysin expression in gliomas is unexpected, but it has been reported in subsets of human and canine gliomas, suggesting that subsets of oligodendroglial tumors may share a common progenitor cell with other CNS tumors and exhibit occasional neuronal characteristics.34,35 Three cases of oligodendroglioma in this study were immunopositive for SYP, and the distribution of the immunoreactivity was more suggestive of expression by the neoplastic cells rather than pre-existing neuroparenchyma; however, double-label expression studies would need to be performed to prove this definitively. Moreover, the use of neuronal markers, such as NSE and SYP, may be useful to differentiate gliomas from neuroglial tumors, such as central neurocytoma. Neurocytoma has never been described in cats and has been rarely reported in dogs, where it shares distinct morphologic features with oligodendroglioma. Diagnostic confirmation is achieved based on the consistent immunoreactivity of neoplastic cells for NSE and SYP, with negative immunostaining for Olig2 and GFAP. 36
Given the high frequency of lymphoma in cats, CD3 and CD79a IHC may be indicated in cases where tumor morphology does not suffice for a final diagnosis of glioma and a lymphoma needs to be ruled out. 4 In fact, one case retrieved from our search that was originally diagnosed as oligodendroglioma by routine histopathology exhibited diffuse immunoreactivity to CD79a and VIM, and negative immunoreactivity to all the other immunomarkers; the case was excluded from in the study as it was diagnosed as a B-cell lymphoma based on IHC. All cases included in the current study were consistently negative for the tested lymphocytic immunomarkers.
The diagnosis of oligoastrocytoma in case 3 was supported by the presence of a mixed neoplastic cell population exhibiting morphologic and immunohistochemical features of both oligodendroglial and astrocytic origin. 25 This case was originally diagnosed as an astrocytoma based on histopathology and immunoreactivity for GFAP, supporting the importance of the use an IHC panel with multiple antibodies in the diagnosis of CNS tumors. Glial tumors can exhibit morphologic dualism, where populations of neoplastic cells resemble both oligodendrocytes and astrocytes, but a diagnosis of oligoastrocytoma requires that at least 30% of the tumor be composed of one of the two different neoplastic cell populations. Oligoastrocytoma has been described in cats and dogs,8,37 but the identity of mixed gliomas has been questioned in human medicine, based on molecular studies.38–40 The role of molecular genetic alterations in cases of canine astrocytoma and oligodendroglioma has been explored, but has yielded inconclusive results; similar studies have not been conducted on feline gliomas.41,42
The diagnosis of anaplastic ependymoma in case 9 was made based on the increased cell and nuclear pleomorphism, with formation of pseudorosettes, and on the high mitotic rate, and was supported by the expression of vimentin.24,31 The expression of other IHC markers such as GFAP and cytokeratin may be useful for diagnostic confirmation of ependymoma, but immunoreactivity may be highly variable among cases. 24
An increased number of neoplastic cells immunoreactive for Ki-67 was detected in the two anaplastic tumors in this case series (cases 2 and 9). These findings may suggest that this cell proliferation marker could be useful to support a diagnosis of anaplastic glial tumors when in association with cell and nuclear morphology. However, these results should be interpreted with caution and no definitive conclusion should be drawn, as no immunopositivity for Ki-67 was detected in the glioblastoma, a high-grade tumor.
Conclusions
This article highlights the importance of a complete neurological examination, including neuroimaging, when evaluating cases of neurological disease in cats. It also emphasizes that routine histopathology should be interpreted in association with IHC in cases of feline CNS glioma as atypical or high-grade tumors may need further diagnostic characterization in order to be classified or differentiated from other primary or secondary CNS tumors.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article
