Abstract
Objectives
The purpose of this study was to assess efficacy of deslorelin, a gonadotropin-releasing hormone (GnRH) agonist marketed in Europe for the control of male dog reproduction, for the postponement of puberty in queens.
Methods
Nine prepubertal queens aged 3–9 months were selected for this study; their general and reproductive health was checked through clinical, haematological, vaginal cytology and hormonal tests. Following treatment with a 4.7 mg deslorelin implant, each cat received a monthly clinical examination and blood was collected for hormonal assay every third month. Cats were monitored for 14.1 ± 5.2 (range 7–23) months.
Results
All cats were in good body condition and normal health prior to treatment. Their health status remained unchanged throughout the study and no significant variation was observed with regard to serum progesterone or oestradiol. Seven days post-treatment, 1/9 queens showed signs of heat, and one other queen showed complete vaginal keratinisation. No other signs of heat were subsequently observed in any other queen. Five queens were lost during the study after 7, 7, 16, 17 and 18 months of observation (during which time they did not show signs of heat). By the end of the study, no sign of puberty was observed in the four remaining queens at 21–36 months of age.
Conclusions and relevance
A 4.7 mg deslorelin implant was able to suppress the feline pituitary–gonadal axis, leading to postponement of puberty for up to 21–36 months in the four queens that completed the study. Deslorelin can be considered as a safe method to postpone puberty in queens.
Introduction
The control of cat reproduction is a common problem in households, which is usually approached with reproductive surgery. Surgical gonadectomy has historically been the preferred approach to control reproduction in small animals, and particularly in cats. However, over recent decades there has been increased attention to animal welfare, which has resulted in pet cat owners and particularly cat breeders being increasingly interested in the use of non-invasive, reversible ways to control reproduction in their animals, particularly in Southern European countries.
Gonadotropin-releasing hormone (GnRH) agonists, such as deslorelin, are commercially available products currently marketed for non-surgical sterilisation in male dogs. Their prolonged action causes pituitary downregulation and blocks gonadal activity. Deslorelin implants are increasingly being used off-label in adult cats to stop roaming, make tomcats temporarily sterile and keep queens out of heat.1–9 In prepubertal male and female dogs the use of a 4.7 or 9.4 mg deslorelin implant is capable of delaying puberty for several months (8–30 weeks).10–12 A 9 month delay was obtained in experimental queens kept in a laboratory environment with a photoperiod of 14 h of light (14L:10D).13,14 Delaying puberty might be helpful in practice as it could give a hesitant owner more time to take a decision on what approach to use to control reproduction in her/his queen.
The objective of this study was to assess efficacy of the 4.7 mg deslorelin implant to postpone puberty in privately owned prepubertal queens kept in a house-type environment.
Materials and methods
Nine domestic shorthair European prepubertal queens were used for this study with the owners’ consent. Queens were 3–9 months of age and 1.4–3.5 kg body weight at the beginning of the study. The nine queens were living at home with their owners (each queen belonging to a different owner) and were under continuous observation, although they all had free access to an outside backyard where they could come in contact with other cats and could also roam free. General and reproductive health conditions as well as prepubertal status were assessed at the beginning of the study through: (a) reproductive history; (b) clinical and reproductive exami-nation, which included inspection of external genitalia, vaginal cytology and palpation of mammary glands; (c) urine collection by cystocentesis; and (d) blood collection for haematology and hormonal assay. Queens were sedated for blood and urine collection using butorfanol IM (Dolorex; Intervet) 0.3 mg/kg, medetomidine IM (Sedator; Eurovet Animal Health) 10 µg/kg and ketamine IM (Ketavet; Parke-Davis) 3 mg/kg.
A 4.7 mg deslorelin implant (Suprelorin; Virbac) was administered inbetween the shoulder blades, after which queens were checked for items (a) and (b), as above, 7 days after implantation. From day 30 post-implantation onwards monthly checks were performed (for 18 months from the treatment of the first queen), which included items (a) through (d) (including sedation for urine and blood collection). Clinical examination included a visual and palpation check of the injection site to detect side effects. Haematology was performed using Advia 120 equipment (Siemens, Milan, Italy). Urinalysis was performed using Aution Sticks 10 PA (Arkay, Shiga, Japan). Serum concentrations of oestradiol (E2) and progesterone (P4) were measured using chemiluminescence (Immulite, Medical Systems, Genova, Italy) with a minimum detectable concentration of 0.2 ng/ml for P4 and 20 pg/ml for E2. Puberty was defined as the time when persistent, long-lasting oestrous behaviour was initially displayed, along with the presence of a fully keratinised vaginal smear and with interest in and breeding attempts by tomcats. In order to avoid missing a weak or short-lived oestrous period, cat owners were given a detailed explanation of feline reproductive behaviour at the beginning of the study.
Queens were enrolled into the study during a 13 month period starting in January 2008 and were followed up until puberty was displayed as defined above. The study was intended to last for a total time of 18 months from the treatment of the first queen. Queens not showing puberty after that time would be followed up by monthly telephone consultations with their owners; the onset of puberty was determined by the owner reporting classical heat-related behaviour (continuous calling, rolling and showing a lordotic position) being observed in their queens. Data were analysed using one-way analysis of variance (ANOVA, GLM procedure, Sigmastat 2.0), considering time as an independent variable and hormonal values, vaginal cytology and weight as dependent variables. The day of implantation was considered as day 0 and the time post-implantation was divided into five periods of 60 days (period 1 = day 1–60; period 2 = day 61–120; period 3 = day 121–180; period 4 = day 181–240; period 5 = day >240). Correlations among different parameters were checked using the Pearson’s correlation index.
Results
The total duration of the study from enrolment of the first queen to the last clinical check and blood sampling was 19 months (January 2008 to July 2009) (Table 1). Between July 2009 and April 2010 cat owners were contacted regularly by telephone to check time of puberty onset in the queens that had not shown puberty earlier. All cats were in good clinical condition and normal health prior to treatment. Haematological and urinalysis parameters were available for 8/9 queens (Tables 2 and 3) and were normal in all tested animals. The health status of all queens remained unchanged throughout the study except for queen 4 which died at 27 months of age (18 months after being implanted, without having shown puberty) because of a feline leukaemia virus (FeLV) infection. Queens 2 and 3 disappeared from their homes during the seventh month of the study (both at the age of 15 months, without having shown puberty) and never came back; queen 5 died at 19 months of age (without having shown puberty) following a car trauma; and queen 8 was ovariectomised (at the owner’s request) while still prepubertal at 24 months of age. In the remaining four queens puberty was reported by telephone consultation with the owner to have occurred between 21 and 36 months of age.
Identification, breed, age at study onset and total duration of observation (months from study onset) in nine queens administered a 4.7 mg deslorelin implant (Suprelorin; Virbac) prior to onset of puberty

Lost to follow-up
Died from a feline leukaemia virus infection
Died following a car trauma
Ovariectomised at owner’s request
Haematological values in eight prepubertal queens prior to treatment with a subcutaneous 4.7 mg deslorelin implant (Suprelorin; Virbac)
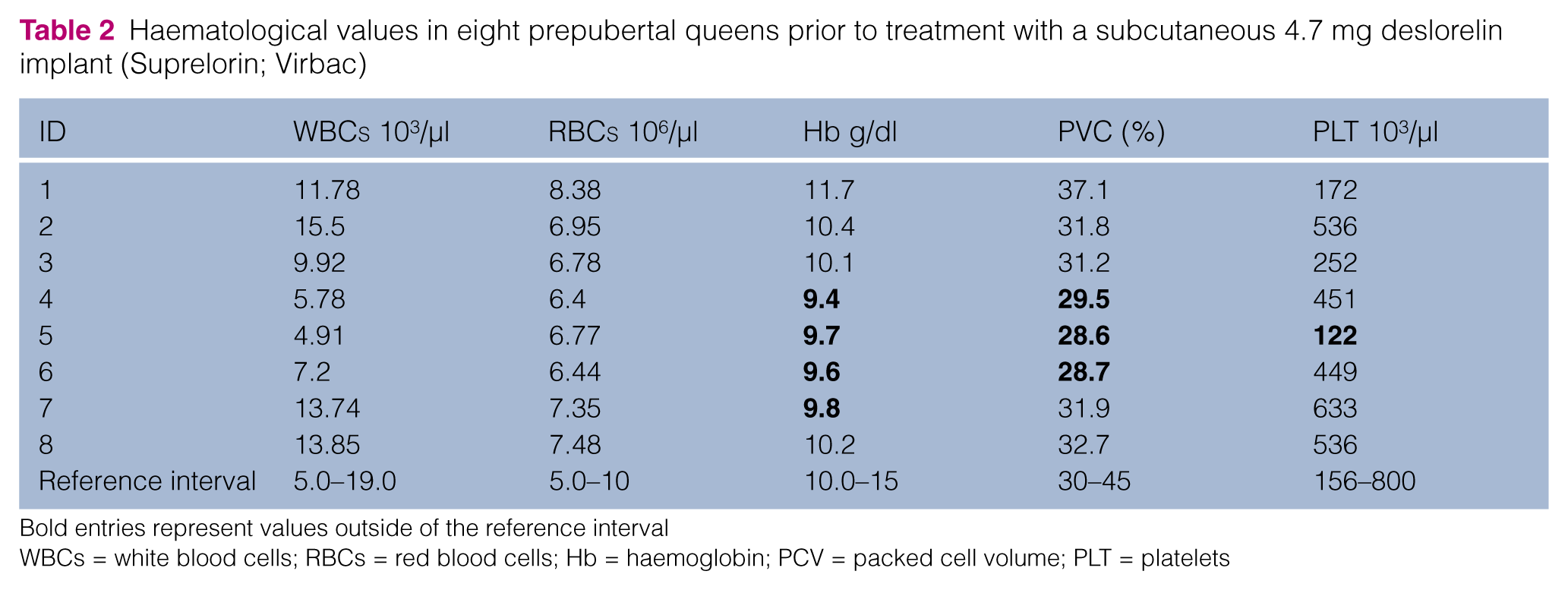
Bold entries represent values outside of the reference interval
WBCs = white blood cells; RBCs = red blood cells; Hb = haemoglobin; PCV = packed cell volume; PLT = platelets
Urine specific gravity, pH, proteins, ketone bodies, bilirubin and red blood cells (RBCs) of eight prepubertal queens prior to treatment with a subcutaneous 4.7 mg deslorelin implant (Suprelorin; Virbac)

Bold entry represents value outside the normal reference interval
A full-fledged heat lasting approximately 8 days with normal display of oestrous behaviour (continuous calling, rolling and showing a lordotic position) was observed 7 days after treatment in queen 2. No other signs of behavioural heat were observed in any other queen for the entire duration of the study.
Data on vaginal cytology are shown in Table 4. Post-treatment vaginal cytology results were characterised by a significantly higher percentage of keratinised epithelial cells during the first 2 months when compared with the rest of the study (Figures 1 and 2). For instance, queen 2 had 100% keratinised vaginal epithelial cells at day 7 post-treatment (coinciding with her heat on day 8 post-implantation, see above). Queen 3 had 95% keratinised vaginal epithelial cells at day 8 post-treatment. Queen 4 had 100% keratinised vaginal epithelial cells at day 33 post-treatment. The percentage of keratinised vaginal epithelial cells was <5% in these five queens for the rest of the study and fluctuated between 0 and 25% in the remaining four queens throughout the entire duration of the study. Statistical analysis showed that average values of percent keratinised vaginal epithelial cells in period 1 (days 1–60) was significantly higher than periods 2 and 3 (P <0.05) and period 4 (P <0.01). Pearson correlation index showed a negative correlation between days post-treatment and percentage of keratinised vaginal epithelial cells.
Body weight, keratinised cells, concentration of serum progesterone and oestradiol in nine prepubertal queens treated with a subcutaneous 4.7 mg deslorelin implant (Suprelorin; Virbac)

Day 0 = day of treatment; Period 1 = 1–60 days post-treatment; Period 2 = 61–120 days post-treatment; Period 3 = 121–180 days post-treatment; Period 4 = 181–240 days post-treatment; Period 5 = >240 days post-treatment; PT = post-treatment; P4 = progesterone; E2 = oestradiol

Variation of percent keratinised vaginal epithelial cells in nine domestic European shorthair queens from before (day 0) to 580 days following treatment with a 4.7 mg deslorelin implant (Suprelorin; Virbac). Day 0 = day of treatment; period 1 = 1–60 days post-treatment; period 2 = 61–120 days post-treatment; period 3 = 121–180 days post-treatment; period 4 = 181–240 days post-treatment; period 5 = >240 days post-treatment

Average percentage of keratinised cells in nine prepubertal queens treated with a subcutaneous 4.7 mg deslorelin implant. Day 0 = day of treatment; period 1 = 1–60 days post-treatment; period 2 = 61–120 days post-treatment; period 3 =121–180 days post-treatment; period 4 = 181–240 days post-treatment; period 5 = >240 days post-treatment. The insert shows average (± standard deviation) values of keratinised cells (KC), concentration of serum progesterone (P4) and oestradiol (E2) in all animals throughout the study
Following treatment with deslorelin and throughout the study, hormonal values remained basal or very close to basal concentration (Table 4). Serum P4 was observed to fluctuate between 0.2 and 1.2 ng/ml (n = 64). The majority of serum samples had values between 0.2 and 0.99 ng/ml; in only four of the 64 samples was there a value between 1.0 and 1.2 ng/ml. Serum E2 was observed to fluctuate between <20 and 120 pg/ml (n = 57). The majority of serum samples had values of <20 pg/ml; 19/57 samples had values between 20.7 and 66.5 pg/ml. Values >20 pg/ml were not found in association with either signs of behavioural heat or a keratinised vaginal smear.
Body weight increased in all queens from 2.43 ± 0.66 kg (period 0) to 3.1 ± 0.44 (period 2) and to 3.4 ± 0.43 kg (period 5) (Table 4). There was a positive correlation between body weight and time (Pearson index 0.542). Cat 4 showed a regular increase in body weight until day 244 post-treatment, after which its body weight started to decrease, presumably due to the FeLV infection which was responsible for the cat’s death on day 499 post-treatment.
In the four queens that were followed up for a prolonged period of time, puberty was confirmed by the owner as occurring at the ages of 36 (queen 1), 24 (queen 6), 25 (queen 7) and 21 months (queen 9) (Table 1). Queen 8 was spayed at 24 months of age while still prepubertal.
There were no side effects at the injection site in any of the nine treated queens.
Discussion
A 4.7 mg deslorelin implant allowed the queens in our study to remain prepubertal until the age of 22.5 ± 6 months. Considering the different times at which some of our patients were lost during the study, puberty was delayed at least until the age of 15 months in all nine queens, at least until 19 months in 7/9 queens, at least until 21 months in 6/9 queens, at least until 24 months in 5/9 queens and until 25–36 months in the last three queens. This is a significantly longer than normal period when considering that the reported average age at puberty for domestic cats is 4–12 months.15,16
The interval between treatment and onset of puberty observed in our cats was longer than that previously reported following treatment with a 4.7 mg deslorelin implant. 13 Risso et al 13 treated 100-day-old queens with a 4.7 mg deslorelin implant and observed puberty between the age of 6 and 14 months in 28/30 cats. The reason for a longer interval between treatment and puberty in our cats is difficult to explain. Environment and latitude could play a role in this difference. Risso et al 13 had a very homogeneous population of cats maintained in a laboratory environment with a strictly followed 14L:10D photoperiod. In our case, the various cat breeds and the different environmental conditions may have played a role. A natural photoperiod may be particularly relevant when causing a reversible block of cyclicity in seasonal species: the treatment effect may cease at a time when the season exerts a negative feedback on the hypothalamic–pituitary axis (HPA) heat, or puberty may be further delayed until the photoperiod stimulates the HPA again. Our study was conducted at a latitude of 45.24 N, while the study of Risso et al 13 was conducted at a latitude of 34.5 S. Proximity to the equator influences photoperiod and may stimulate resumption of cyclicity. Sample size can also influence outcome, as we ended up with five queens while the results of Risso et al are based on 28 queens that completed the study. 13 However, our results highlight the fact that the postponement of puberty in queens treated with a 4.7 mg deslorelin implant may occur well beyond 15 and as late as 21–36 months of age.
In 3/9 cats (queens 2, 3 and 4) fully keratinised vaginal smears were found during the first month post-treatment. These queens, as well as all other queens in our study, were initially considered as prepubertal based on history, clinical and reproductive examination as well as hormonal assay. These three queens were treated at the age of 8, 8 and 9 months, respectively, all in January. At such an age and considering their weights (2.7, 2.3 and 3.5 kg, respectively) and the photoperiod, it is possible that they were very close to their first oestrus. Therefore, it is likely that the deslorelin implant may have caused puberty in queen 2. The other two cats (queens 3 and 4) showed only a high degree of keratinisation of the vaginal epithelium without any other behavioural signs indicative of puberty. This is probably due to GnRH causing minor luteinising hormone (LH) pulses, which are capable of causing follicular growth (with secretion of oestradiol and vaginal epithelial keratinisation) but not ovulation. Such minor LH pulses are frequently observed in females towards the end of anoestrus, and bitches are known to experience late anoestrus increases in oestradiol concentrations that are associated with vaginal keratinisation but not vulvar swelling or serosanguineous vulvar discharge. 17 Therefore, it is possible that what queens 3 and 4 of our study experienced following treatment may actually have been not puberty but only a minor LH pulse eliciting vaginal keratinisation, while pubertal heat instead might have occurred once the effect of deslorelin vanished. This is an aspect that deserves further study. Although puberty in domestic cats can occur as late as 12 months,15,16 when considering the administration of deslorelin to a queen to prevent puberty this should ideally be carried out well before 8 months of age.
Deslorelin treatment did not affect the general health condition of the cats. All queens were in good health prior to treatment onset, and all but one queen remained healthy during the study: queen 4 died at 27 months of age due to FeLV. All the queens in our study had free access to a backyard from where they could come in contact with other cats. Since none of our study cats were tested for FeLV prior to and during the study and none of them were vaccinated against this virus, it is not possible to determine when infection in queen 4 occurred, although looking at its weight data the infection probably occurred after day 244.
Fertility after treatment was not checked in this study. This is an area that deserves further investigation as owners who require a reversible sterilisation for their cats are certainly interested in being reassured about the fertility potential of their animals being maintained. Body weight, on average, increased during the study. The increase in body weight is probably due to the animals still being in a growing phase of their lives. Although our results highlight the fact that deslorelin does not block body growth in prepubertal animals, this was not conclusively shown by our study due to the lack of a control group treated with a sham implant.
Conclusions
Deslorelin can be considered as a safe method to postpone puberty in queens, devoid of side effects and effective up to 21–36 months of age provided that it is administered well before 8 months of age and, ideally, around 3–5 months of age in order to avoid inducing puberty. Although the rate of growth could not be compared with that of a control group, body growth was not prevented by the deslorelin treatment. Further studies are necessary to better understand the duration of effect as well as post-treatment fertility following the use of deslorelin in prepubertal cats.
Footnotes
Author note
Preliminary results of this study were presented as a poster at the 2010 Congress of the the European Veterinary Society for Small Animal Reproduction. Romagnoli et al. Postponement of puberty in male and female prepuberal cats with deslorelin: a preliminary study. Proceedings 7th EVSSAR Congress, Louvain-La-Neuve, Belgium, on May 14–15, 2010, p 145.
Conflict of interest
The last author of this paper received a grant in 2010 from Virbac to study the effects of deslorelin in cats, which is the drug investigated in this study. At the time of the study (2008–2009) there was no conflict of interest.
Funding
This study was supported by a 2008 University of Padova grant called Ricerca Scientifica ex quota ex 60% grant. 60A08-1998/08.
