Abstract
Objectives
The objective of this study was to examine the density and distribution of goblet cells (GCs) in the feline conjunctiva and to investigate a potential effect of age and sex on GC density (GCD).
Methods
Thirty-nine eyes of 21 cats euthanased for reasons unrelated to this study were used. Fixed upper and lower eyelid and bulbar conjunctiva were divided into nasal and temporal regions. The third eyelid was excised and investigated separately. Samples were embedded in paraffin wax; sections were stained with periodic acid–Schiff reaction and analysed with light microscopy. To determine the topographic distribution of GCs, each region was subdivided into the marginal, palpebral and bulbar zone. In each zone 200 epithelial cells, including GCs, were counted. Goblet cell index was defined as a percentage of the epithelial cells.
Results
The palpebral zone of both eyelids contained significantly (P <0.001) more GCs (27.5–32.0%) than the marginal or bulbar areas. The highest GCD was found in the nasal palpebral zone of the upper eyelid (32.0%). Marginal and bulbar sites contained fewer numbers of GCs (2.6–10.0%). The lowest GCD was detected in the nasal bulbar zone of the lower eyelid (2.6%). Overall the nasal region contained significantly (P = 0.036) more GCs than the temporal region, but there was no significant difference in GCD between the upper and lower eyelids. Correlation analysis did not show any effect of age or sex on GC counts.
Conclusions and relevance
GCD in the palpebral zones and on the anterior surface of the third eyelid was highest; the lowest density was found in the bulbar zones of the lower eyelid and in the marginal zones of both eyelids. Overall, higher GCD was found in the cat than in other species. Age and sex have no effect on GCD.
Introduction
Mucin plays an important role in the preocular tear film. In addition to lubricating the corneal epithelium, it stabilises the tear film and delivers an optically smooth surface overlying the cornea. It also provides a physical barrier against pathogens and debris, and contributes to local immunity.1–5 Tear film break-up time has been defined as a non-invasive, indirect measurement of preocular mucin. However, the lack of standardisation and a wide variety of results obtained in cats are questioning its usefulness as a research tool.6–8
Ocular surface mucin is primarily produced by the conjunctival goblet cells (GCs). Studies in physician ophthalmology suggest that goblet cell density (GCD) is a good indicator of ocular surface health.1,9
The localisation and density of GCs varies between species.2,10–14 From our clinical observations and from a previous study, we know that cats do not tolerate ocular ointments well. 6 The question arose as to whether this was due to a different mucin layer compared with other species.
According to the publication by Moore et al, 2 dogs have few GCs in the temporal bulbar area, the very same site where we took our samples with the Biopore membrane device.2,6 In our impression cytology samples we could not find any GCs. 6
A few publications have described GCD in various areas of the feline conjunctiva.8,15,16 However, by the time we started this research (end of 2014) no comprehensive morphological study on the feline conjunctiva had been published. Therefore, our aim was to describe the anatomical distribution and density of the GCs in feline conjunctiva. Furthermore, we investigated if age and sex have an effect on GCD.
Recently, Sebbag et al published a study on the density and distribution of GCs in the feline conjunctiva. 17 Their results and findings are compared and discussed.
Materials and methods
The eyes of 21 cats (39 eyes; nine spayed females, 10 castrated males, two intact females [mean ± SD age 140.4 ± 70.3 months (range 6–216)]) euthanased for reasons unrelated to this study were used. Prior to euthanasia no ophthalmic examination could be performed, but none of the cats had a history of any acute or chronic ocular surface disease.
One cat had what was presumed to be a feline diffuse iris melanoma in one eye; another cat had one-sided exophthalmos, and a third cat already had one eye enucleated. In these cases the other clinically healthy eye served as the sample material.
All eyes were enucleated transpalpebrally, leaving the conjunctival sac intact and attached to the globe. The globe was transected approximately 3–5 mm behind the limbus. By cutting the cornea horizontally, eyelids and conjunctiva were divided into lower and upper halves. Each half was spread out on a piece of cardboard and put into a cassette. The third eyelid was dissected carefully from the lower half and also spread out on a cardboard sheet and put into a separate cassette. Care was taken not to damage or stretch the conjunctiva. All samples were immersed into 4% buffered formaldehyde solution (Formaldehydlösung SAV neutral gepuffert 4%; SAV LP) and stored at room temperature until further processing.
Fixed halves were divided into the nasal and temporal parts by removing a 2 mm central strip. Afterwards, samples were dehydrated (Shandon Excelsior ES Tissue Processor; Thermo Fisher Scientific).
Nasal and temporal parts were embedded into paraffin wax and 3–4 µm sections were cut parasagitally with a sliding microtome (Reichert Optische Werke AG). Sections were mounted on glass slides and stained with periodic acid–Schiff (PAS). All samples were examined with a light microscope using × 4, × 10, × 20 and × 40 magnification.
To determine the distribution of GCs the conjunctiva was divided into three different zones according to their histopathological appearance. Slides were scanned using a digital slide scanner (Aperio Scanscope CS2; Leica Biosystems) (Figure 1) and cells were counted onscreen using Photoshop CS5-Extended (Adobe Systems).

Histological structure of the feline conjunctiva from the eyelid margin to the bulbar edge
GCs were identified by positive PAS staining within the conjunctival epithelium. GCD was defined by counting the GCs per 200 epithelial cells (goblet cell index [GCI]). In areas containing fewer than 200 epithelial cells all cells were counted. Damaged conjunctival areas were excluded from the study. If the quality of the specimen was not sufficient, one or more additional sections were made.
Regions of the conjunctiva were identified with a special code. This included the examined eyelid, region and zone. For example ULNM stands for the marginal (M) zone of the nasal (N) region of the upper eyelid (UL).
Statistical analysis
Statistical analysis was performed using SPSS version 19 (IBM). For each area data for the right and left eyes were pooled. Differences in GCD among the various sampling sites were analysed with a general linear model using eyelid, region and zone as within-subject factors. Post-hoc analysis was performed using least significant difference procedures. The assumption of normal distribution was tested using Kolmogorov–Smirnov test. The impact of age on GCD was investigated using Pearson’s correlation analysis. To evaluate the differences in GCD between males and females, t-tests for independent samples were used. For all analyses a P value of <0.05 was considered significant.
Results
Conjunctiva was divided into three zones based on their histological appearance.
Marginal zone (M): begins where the multilayered keratinised epithelium of the eyelid skin turns into a double- or triple-layered squamous or cuboidal epithelium. The epithelium contains melanocytes and very few GCs (Figure 2).
Palpebral zone (P): follows the marginal area. The epithelium is non-keratinised, cuboidal or columnar, double- or triple-layered, usually folded and contains numerous GCs (Figure 3). Occasionally, melanocytes are interspersed between epithelial cells.
Bulbar zone (B): the epithelium is thin, double- or simple-layered, columnar-to-cuboidal with few GCs. GCs are placed singly between epithelial cells (Figure 4). This zone is easy to damage as it has almost no subconjunctival tissue.
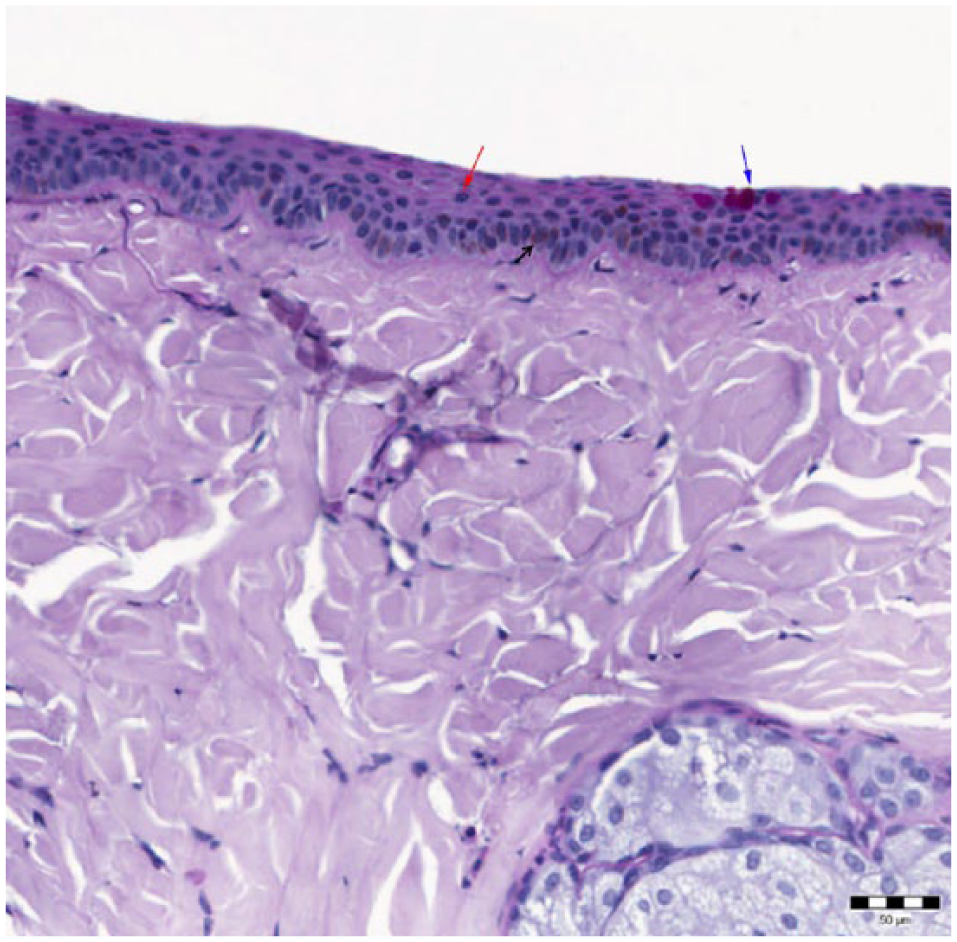
Image shows the marginal zone of the conjunctiva. The epithelium is multilayered (red arrow), contains melanocytes (black arrow) and few goblet cells (blue arrow); periodic acid–Schiff reaction

Folded palpebral conjunctiva with goblet cells (black arrow). The epithelium is double- or triple-layered (red arrow)

Bulbar conjunctiva with scattered goblet cells (black arrow). Note that the epithelium is simple- or double-layered (red arrow) and this area is almost lacking in subconjunctival tissue
The inner site of the third eyelid was histologically similar to the bulbar conjunctiva, whereas the outer part was like the palpebral area (Figure 5). The limbus of the third eyelid was covered with stratified, non-keratinised epithelia and usually contained melanocytes.

Third eyelid. Note that the folded palpebral side has numerous goblet cells (black arrow), but on the bulbar side (red arrow) only few goblet cells are seen. The blue arrow indicates the cartilage of the third eyelid
A marked difference in GCD among regions was noted. The palpebral zone of both eyelids contained significantly more GCs (mean GCI: 27.5–32.0%) than the marginal or the bulbar regions (P ⩽0.001). The highest number of GCs was identified in the upper lid nasal palpebral zone (ULNP) (mean GCI: 32.0%), whereas the lowest number of GCs was found in the lower lid nasal bulbar zone (LLNB) (mean GCI: 2.6%). Furthermore, a significant difference was found between the nasal and temporal regions (P = 0.036). The nasal region contained significantly more GCs than the temporal region. However, there was no significant difference in mean GCD between the upper and the lower eyelids (P = 0.062). Mean GCI for each region and distribution of GCs are shown in Table 1 and Figures 6–8.
Goblet cell indices – descriptive statistic for each region

UL = upper eyelid; N = nasal; M = marginal; P = palpebral; B = bulbar; T = temporal; LL = lower eyelid; 3EL = third eyelid

Distribution of goblet cells in the conjunctiva. Note that the highest densities were found in the palpebral zone of the nasal upper and lower eyelid. The lowest density was found in the bulbar zone of the nasal lower lid, followed by the marginal zones of both eyelids

Mean goblet cell densities for the three zones of the upper eyelid

Mean goblet cell densities for the lower eyelid
The palpebral surface of the third eyelid contained significantly (P = 0.029) higher numbers of GCs (mean GCI: 27.1 %) than the bulbar area (mean GCI: 8.7%).
Correlation analysis showed no significant effect of age on GC counts in any region and there was no significant difference in GCD between male and female cats.
Discussion
Similarly to the study by Sebbag et al, 17 GCD showed a wide variation among sites. Although the publication by Sebbag et al is on the same topic, the numerical data are not – only the rates are comparable because of the differences listed below. 17 In our study each conjunctival quadrant (ie, dorsonasal, dorsotemporal, ventronasal and ventrotemporal) was divided into three zones, from the eyelid margin to the corneoscleral limbus, based on their unique histological appearance. Sebbag et al also investigated three zones, but they used different descriptions. 17 They defined the palpebral conjunctiva as beginning where the very first GC appears after the eyelid margin. This is followed by the forniceal conjunctiva, where the epithelium is folded. Their definition of the bulbar conjunctiva was similar to ours. Furthermore, cell counting was also different in the study by Sebbag et al. 17 First, 200 consecutive basal epithelial cells were counted, and then GCs that were among these basal cells were counted. Afterwards, GCD was given as a percentage of the basal epithelial cells. 17 We counted 200 epithelial cells, including GCs, and the GCI was given as a percentage of the epithelial cells.
We believe that our data are more accurate and easier to use than those from this recent publication. 17 First, there is an inter-individual difference in the distribution of GCs, therefore a zone should be clearly defined by its histological features and not just by the appearance of GCs. Second, with stretching of the sample the folding of the conjunctiva may disappear and the fornix can no longer be identified. Lastly, counting the basal epithelial cells is hardly applicable on a daily basis because for a clinician it is not easy to differentiate between epithelial and basal epithelial cells, and a larger tissue sample is required to gain a result.
Three studies have used conjunctival biopsies to define the density of GCs in different regions of the feline conjunctiva. However, comparison is again hard to do with different sampling sites. Cullen et al took the samples from the palpebral conjunctiva, 15 Grahn et al from the forniceal conjunctiva 16 and Lim et al from the ventronasal fornix. 8 The exact description of the sampling sites is missing. These studies used a very low number of animals, whereas in our study 39 eyes from 21 cats were used. The distribution of GCs is not homogenous in any region; therefore, a single conjunctival biopsy may not display the actual numbers. They also applied a different measurement of GCD. In the studies by Cullen et al and Lim et al 50 consecutive epithelial cells were counted,15,16 and in the study by Grahn et al 50 basal epithelial cells were counted. 8 Then, the number of GCs was counted among the 50 epithelial/basal epithelial cells and the final data given as a ratio.8,15,16
Overall, a standardised method of describing GCD in feline ophthalmology is needed. The highest number of GCs in the present study was found in the ULNP and in the nasal palpebral zone of the LLNP. It shows proximity to the data published in dogs and chinchillas, although the cat seems to have a higher number of GCs, especially in the nasal areas.2,11 In dogs, the highest GCI was detected in the ventronasal fornix in cats in the ULNP zone. The marginal zones – like in other species – had a low number of GCs. The obtained results are closest to those observed in guinea pigs. 12 Low GCDs were found in the bulbar areas, in agreement with the study by Sebbag et al, but the numbers here were also higher than in other species.2,11,12,17 This resembles the equine bulbar conjunctiva. 13
A study that examined the feline nictitating membrane was published in 1994. 18 It found that the palpebral surface is folded and contains more GCs than the bulbar surface, but no further quantification was made. Sebbag et al also found higher GCD on the anterior surface. 17 GCD has been described for the canine nictitating membrane. Samples were taken from the palpebral surface by impression cytology. Mean GCD was 34.6% for dogs. 3 In cats we found slightly lower values; mean GCD of the palpebral surface was 27.1%, which was significantly higher than on the bulbar side.
We did not detect any effect of age on GC numbers in cats. This is in contrast with a study in mice where age-related loss of GCs was noted. 19 In physician ophthalmology the results are controversial. Some studies could not establish a correlation between GC count and age, whereas other studies state that the GC population decreases with age.20–22
Conclusions
Mucin, secreted by the conjunctival GCs, is important for the ocular surface. Loss of GCs could result in different ocular surface diseases and other ocular anomalies can result in decreased GCD. Our study established baseline data of density and distribution of GCs, which could help to diagnose and monitor different ocular anomalies. Furthermore, these data will be useful when the in vivo effect of topical mucinomimetic agents will be tested on the feline eye. For sampling, either the palpebral conjunctiva or the anterior surface of the third eyelid should be used because they are rich in GCs. Additionally, according to our study, age and sex have no effect on GCD.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
