Abstract
Objectives
The purpose of this study was to describe the perioperative and postoperative complications as well as short-term and long-term outcomes in cats with ureteral obstructions treated by placement of a subcutaneous ureteral bypass (SUB) device without imaging control. The second objective of this study was to compare cats treated by SUB device with cats treated by traditional surgical intervention.
Methods
Data were obtained retrospectively from the medical records (2014–2016) of cats that underwent SUB placement (SUB cats) and cats that underwent traditional ureteral surgery (C cats).
Results
Nineteen SUB devices were placed without fluoroscopic, radiographic or ultrasonographic guidance in 13 cats. Fifteen traditional interventions (ureterotomy and neoureterocystostomy) were performed in 11 cats. Successful placement of the SUB device was achieved in all cats with only one major intraoperative complication (kinking of the kidney catheter) and one minor intraoperative complication (misplacement of the kidney catheter). Eleven SUB cats recovered from the surgical procedure; two SUB cats and three C cats died during the anaesthesia recovery period. Postoperative SUB complications included anaemia (n = 2), urinary tract infection (UTI) (n = 4), non-infectious cystitis (n = 5) and SUB device obstruction (n = 1). Postoperative traditional surgery complications included anaemia (n = 7), UTIs (n = 6), non-infectious cystitis (n = 1), re-obstruction (n = 4) and ureteral stricture (n = 1). Median postoperative duration of hospitalisation (3 days) was significantly shorter for SUB cats than for C cats (P = 0.013). Ten SUB cats (76.9%) and four C cats (40%) were still alive at a median follow-up of 225 days and 260 days, respectively. Owners were completely (90%) or mostly (10%) satisfied with the SUB device placement.
Conclusions and relevance
SUB device placement appears to be an effective and safe option for treating ureteral obstruction in cats, and this study has shown that fluoroscopic guidance is not essential in all cases.
Introduction
Ureteral obstruction is an increasingly commonly diagnosed disease and is a challenging condition to manage in cats. 1 Ureterolithiasis is the most common cause reported, with greater than 98% being calcium oxalates. 2 Historically, options for treatment were restricted to either medical management or surgical correction via a ureterotomy, ureteral resection and anastomosis, ureteroneocystostomy or ureteronephrectomy. 3 Medical management alone has been shown to be effective in only a minority of cats with stones (17%).2,4 Traditional interventions are associated with a high rate of major complications such as urinary leakage, recurrence or ureteral stricture (over 30%), and mortality rates range from 18% to over 30%.2–6 More recently, the placement of feline double pigtail ureteral stents (Vet Stent; Infiniti Medical LLC) or a subcutaneous ureteral bypass (SUB) device (Norfolk Vet Products) has allowed novel treatment options. Ureteral stent placement in veterinary medicine was first reported in 2007. 7 The largest study (69 cats) reported successful short-term outcomes, but stent replacement was subsequently required in 27% of ureters and temporary dysuria was a problem in 37.7% of cats. The use of a SUB device was first described in 2010. 8 This has also been shown to be an effective treatment option and a significant advantage of this device is the presence of a subcutaneous port which allows flushing of the device every 3 months to prevent occlusion and to allow urine sampling for culture.
In human urology, the use of a subcutaneous nephrovesical bypass has been reported in approximately 100 patients and is considered to be a salvage procedure reserved for patients where other medical and/or surgical therapies have failed.9–15 Few complications were noted and long-term patency rates were reportedly excellent compared with ureteral stents and other more aggressive ureteral surgery. In veterinary medicine, only two studies, conducted by the creator of the SUB device, have reported its use.16,17 The first evaluated the pre-, peri- and postoperative parameters for the prediction of outcome and renal recovery in cats treated with ureteral stent or SUB device. The second studied cats with circumcaval ureters associated with a ureteral obstruction treated with ureteral stent or SUB device.
The purpose of this study was to evaluate the perioperative and postoperative complications as well as short-term and long-term outcomes in cats with ureteral obstructions treated by placement of a SUB without fluoroscopic guidance. The second objective of this study was to compare cats treated by SUB device with cats treated by traditional surgical intervention. We hypothesised that morbidity and mortality would be lower in cats with ureteral obstruction treated by SUB device than in cats with ureteral obstruction treated by traditional surgery.
Materials and methods
Case selection
The medical records of all cats treated at VetAgro Sup, Campus Vétérinaire de Lyon with a SUB device for a ureteral obstruction from January 2014 to January 2016 were retrospectively included in the study. Cats were included in the SUB group (SUB cats) if one or more SUB devices were placed at surgery following diagnosis of ureteral obstruction. Case information included signalment (breed, sex and age), clinical signs, duration of clinical signs, physical examination findings, initial biochemical values of urea, creatinine and potassium, packed cell volume (PCV), urine specific gravity (USG), urine culture (cystocentesis or pyelocentesis), methods of diagnosis, renal pelvic dilation, surgical procedure performed and time to discharge. Cats were excluded if they had an incomplete medical record. For comparison purposes, an unmatched control population of cats with ureteral obstruction treated by traditional surgeries (C cats) during the same period was selected. Traditional surgery included ureterotomy and ureteroneocystostomy. The surgical procedure performed was selected on the basis of the owner (cost issue) and clinical preferences.
Preoperative case management
Following diagnosis of ureteral obstruction, medical treatment was instituted in all cases before surgery. Medical management consisted of various modalities, including intravenous (IV) fluid therapy, oxygen therapy and analgesia as needed. Electrolyte imbalances were also managed before surgery.
Surgical procedure
Surgery was performed as soon as the patient was judged stable enough for anaesthesia. Time between presentation and surgery was recorded. Cats were administered amoxicillin with clavulanic acid (20 mg/kg IV) after induction of anaesthesia and repeated every 90 mins until the end of the surgery. Analgesia included premedication with methadone (0.3 mg/kg IV) or fentanyl (4 µg/kg IV) and maintenance with a fentanyl constant rate infusion (1–8 µg/kg/h IV). Anaesthetised cats were positioned in dorsal recumbency and surgically prepared for a midline celiotomy. The SUB was placed according to the surgical guide provided by Norfolk vet 18 on the affected side. However, the kidney catheter was placed without imaging control. The hydrophilic guide wire was inserted into the renal pelvis and coiled up blindly. The kidney catheter was advanced until the radiopaque marker was inside the kidney. Finally, the locking string was blindly pulled to tighten. A urine sample was taken from the renal pelvis for bacteriological culture for each cat. At the end of the procedure, the Huber needle was inserted through the silicone diaphragm of the port for leak and patency testing. Postoperative plain film radiographs were performed to ensure the correct position of all devices and to check for the creation of the loop into the renal pelvis. For C cats, traditional interventions were performed as previously described with the use of a surgical microscope. 19
All patients were carefully monitored during the postoperative period. Pain control was administered according to clinician preference. All patients received an antimicrobial treatment (amoxicillin with clavulanic acid 15 mg/kg PO q12h) until reception of bacterial culture results. Antibiotherapy was stopped if bacterial culture was negative and continued if positive, with the same antibiotic or with another one according to antibiogram. The SUB device was flushed 1 month after surgery, then every 3 months during the first year and every 3–6 months later.
Surgical data
Intraoperative data included unilateral or bilateral SUB placement, the cause of ureteral obstruction based on surgical inspection, anaesthesia time, procedure time and intraoperative complications. Immediate postoperative data (prior to discharge) included complications, serum biochemical data (urea, creatinine, and potassium, PCV and USG obtained in the immediate postoperative period and daily until discharge), the number of days of hospitalisation and whether the patient survived to discharge. Complications were categorised as catastrophic (resulting in death), major (necessitating revision surgery or requiring important medical intervention) or minor (resolved intraoperatively, managed with simple medical treatment or not requiring intervention).
Follow-up
Postoperative data obtained included short-term (between 7 and 30 days after the procedure) and long-term (>30 days after the procedure) complications (same categorisation as intraoperative complications), ultrasound follow-up, serum biochemical values of urea, creatinine, and potassium, USG, urine culture and overall survival time. Final outcome was assessed by owner interviews. Owners were also asked to rate their level of subjective satisfaction as: completely satisfied, mostly satisfied, somewhat unsatisfied, or completely unsatisfied of the SUB device placement.
Recorded data and statistical analysis
Numerical data distribution’s normality was tested by visual approach and the D’Agostino and Pearson omnibus normality test. Numerical data were not normally distributed and were expressed as median and range. The Mann–Whitney U-test was used to compare quantitative parameters. Qualitative data were compared by the Fisher’s exact test. Kaplan–Meier curves were compiled to represent survival in SUB and C cats. The log rank test was used to compare the Kaplan–Meier curves. A P value of less than 0.05 was considered significant. All analyses were performed using commercial software (Prism 6.0e; GraphPad Software).
Results
Twenty-four cats met the inclusion criteria for the study, with 13 SUB cats and 11 C cats.
Signalment and clinical signs
Median age of the SUB cats was 4.5 years (range 2.5–11 years) and 4.0 years (range 1.5–9 years) for C cats. There was no significant difference in age of cats between groups (P = 0.60). Domestic shorthair cats (6/13 SUB cats and 6/11 C cats) and Birmans (4/13 SUB cats and 2/11 C cats) were the most represented breeds in both groups; Persian (1/13 SUB cat and 2/11 C cats), Oriental Shorthairs (1/13 SUB cat), Maine Coons (1/13 SUB cat) and Norwegian Forest cats (1/11 C cat) were the other breeds represented. There were six spayed females and one intact female in each group. The remainders were castrated males. Median body weight was 3.4 kg (range 2.4–6.9 kg) for SUB cats and 3.2 kg (range 1.4–4.5 kg) for C cats. There was no significant difference in body weight of cats between groups (P = 0.14).
The most common clinical signs included weakness (11/13 SUB cats; 9/11 C cats), anorexia or inappetence (11/13 SUB cats; 11/11 C cats), vomiting (8/13 SUB cats; 5/11 C cats) and weight loss (3/13 SUB cats; 7/11 C cats). Median duration of clinical signs before presentation was 8 days (range 2–101 days) for SUB cats and 8.5 days (range 1–90 days) for C cats. There was no significant difference in duration of clinical signs between groups (P = 0.69).
Physical examination and diagnostic test results
Renal asymmetry was present in 4/13 SUB cats and 4/11 C cats. Other clinical abnormalities included bradycardia (7/13 SUB cats; 2/11 C cats), hypothermia (6/13 SUB cats; 3/11 C cats) and dehydration (5/13 SUB cats; 7/11 C cats).
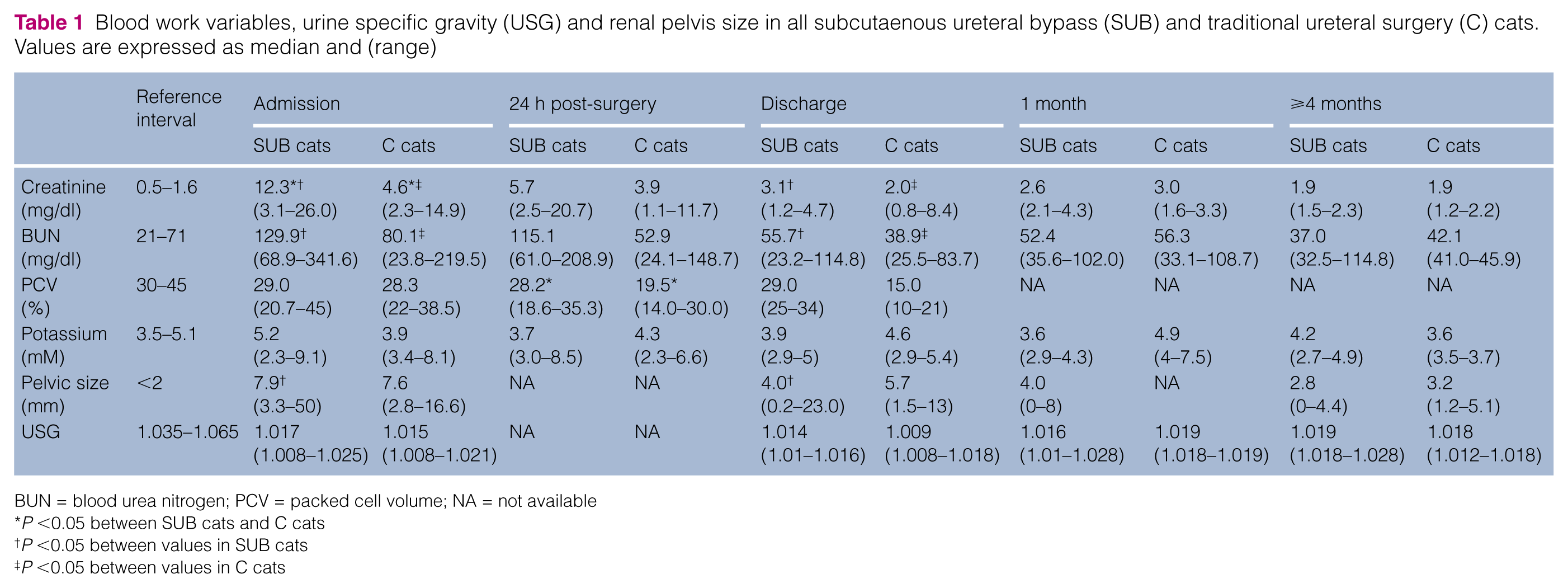
Preoperative laboratory values are summarised in Table 1. All cats had creatinine values above the reference interval and all cats had a USG <1.035. Seven SUB cats (53.8%) and three C cats (27.3%) had hyperkalaemia. The median serum creatinine at presentation was 12.3 mg/dl (range 3.1–26.0 mg/dl) for SUB cats and 4.6 mg/dl (range 2.3–14.9 mg/dl) for C cats. The median serum potassium concentration was 5.2 mmol/l (range 2.3–9.1 mmol/l) for SUB cats and 3.9 mmol/l (range 3.4–8.1 mmol/l) for C cats. The median PCV at presentation was 29.0% (range 20.7–45%) for SUB cats and 28.3% (range 22–38.5%) for C cats. None of the cats had positive urine culture at presentation or crystalluria on urine sediment examination. The serum creatinine concentrations were significantly higher for SUB cats than for C cats (P = 0.02). There were no significant differences in serum potassium concentration (P = 0.23) and PCV (P = 0.56) between groups.
Blood work variables, urine specific gravity (USG) and renal pelvis size in all subcutaenous ureteral bypass (SUB) and traditional ureteral surgery (C) cats. Values are expressed as median and (range)
BUN = blood urea nitrogen; PCV = packed cell volume; NA = not available
P <0.05 between SUB cats and C cats
P <0.05 between values in SUB cats
P <0.05 between values in C cats
Diagnostic imaging
Abdominal ultrasound and plain film radiographs (lateral and ventrodorsal views) were performed in all cats for diagnosis. All obstructions were due to lithiasis. Seven SUB cats had unilateral ureteral obstruction and six had bilateral ureteral obstruction. Seven C cats had unilateral ureteral obstruction and four had bilateral ureteral obstruction. Pyelectasis was systematically present on obstructed kidneys with a median pyelectasis of 7.9 mm (range 3.3–50 mm) for SUB cats and 7.6 mm (range 2.8–16.6 mm) in C cats (reference <2 mm). 20 There was no significant difference in pyelectasis between groups (P = 0.36).
Surgical period
Median time between presentation and surgery was 1 day (range 1–4 days) for SUB cats and 1 day (range 1–4 days) for C cats. There was no significant difference between groups (P = 0.06).
Seven of 13 cats received a unilateral SUB device, and 6/13 received bilateral SUB devices (Figure 1). Median duration of anaesthesia was 105 mins (range 92–122 mins) for unilateral SUB devices and 150 mins (range 129–165 mins) for bilateral SUB devices. Median duration of surgery was 68 mins (range 60–85 mins) for unilateral SUB devices and 98 mins (range 87–105 mins) for bilateral SUB devices.

Intraoperative views of a subcutaenous ureteral bypass device placement in a cat. (a) Placement of the hydrophilic guide wire in the renal pelvis through the intravenous catheter at the caudal pole of the kidney. (b) Placement of the locking loop kidney catheter and adhesion of the silicone/dacron cuff to the renal capsule with sterile tissue glue. (c) Placement of the bladder catheter. (d) Placement of the subcutaneous port sutured to the body wall
Of the C cats, 5/11 cats underwent unilateral ureterotomy, 4/11 cats had bilateral ureterotomy and 2/11 cats had unilateral neoureterocystostomy. Median duration of anaesthesia for unilateral traditional surgery was 169 mins (range 165–220 mins) and 229 mins (range 207–231 mins) for bilateral traditional surgery. Median duration of surgery was 130 mins (range 125–153 mins) for unilateral traditional surgery and 171 mins (range 160–183 mins) for bilateral surgery. Duration of anaesthesia and surgery were highly significantly longer for unilateral (P = 0.002 each) and significantly longer for bilateral (P = 0.01 each) traditional surgery than for unilateral or bilateral SUB placement, respectively.
Two intraoperative complications occurred in the SUB cats: a major one with a kink in the kidney catheter during placement and a minor one with a misplaced kidney catheter into the renal pelvis. The kink was observed on postoperative radiographs in the subcutaneous part of the kidney catheter and immediately treated by another puncture through the body wall further from the subcutaneous port during re-intervention. Entry point of the misplaced catheter was placed too dorsally in the kidney, which prevented correct replacement of the kidney into the retroperitoneal cavity. The kidney catheter was removed and the procedure was restarted without other difficulties (the first puncture was repaired by a cruciate suture on the renal capsule). Postoperative radiographs confirmed the correct position of 17/19 SUB devices (Figure 2a) and showed two locking loop catheters not tightened enough to create the loop into the renal pelvis (Figure 2b). The untightened loops were not corrected during re-intervention. No intraoperative complications occurred in the C cats.
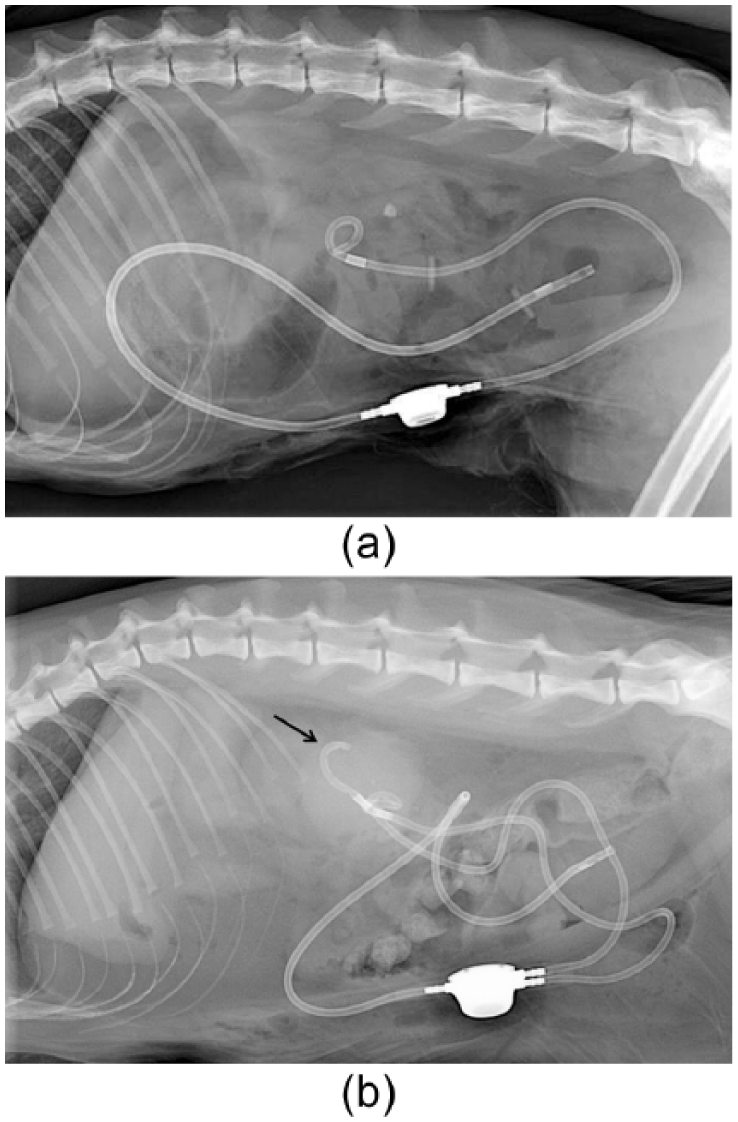
Lateral radiographic views of a subcutaneous ureteral bypass (SUB) device. (a) Correct placement of a unilateral SUB device. (b) Locking loop kidney catheter not tightened enough to create the loop (black arrow) in a bilateral SUB device
Immediate postoperative follow-up
Eleven of 13 cats recovered from the SUB surgical procedure. Two cats had catastrophic complications and died during the anaesthesia recovery period. One developed hypotension and seizures and died 2 days after surgery from cardiopulmonary arrest. This cat had the misplaced kidney catheter. The other cat developed hypotension and was euthanased the day after surgery.
Three C cats died during the immediate postoperative period. The first one underwent a bilateral ureterotomy and developed bilateral re-obstruction 1 day after surgery and was euthanased. The two other cats developed severe anaemia and refractory hypotension and were euthanased.
Eleven of 13 SUB cats and 8/11 C cats were discharged from the hospital, indicating a perioperative mortality rate of, respectively, 15.4% and 27.3%. Median postoperative duration of hospitalisation was 3 days (range 2–7 days) in SUB cats and 5 days (range 3–20 days) in C cats. Median postoperative duration of hospitalisation was significantly shorter in SUB cats than in C cats (P = 0.013).
Two SUB cats and seven C cats developed severe anaemia (PCV ⩽20%). A blood transfusion was required in one SUB cat and two C cats, leading to one major and one minor immediate postoperative complication in SUB cats and two major and five minor immediate postoperative complications in C cats. The median PCV 24 h after surgery was significantly lower in C cats than in SUB cats (P = 0.004) (Table 1). There was no significant difference in occurrence of severe anaemia between groups (P = 0.13).
All SUB cats had an improvement in azotaemia after surgery (Table 1). Serum creatinine decreased on average by 77.3% and the decrease was significant between admission and discharge in SUB cats (P <0.001). Six of eight survival C cats had an improvement in azotaemia (Table 1). Serum creatinine decreased on average by 46.4% and the decrease was significant between admission and the discharge in C cats (P = 0.023).
Between admission and discharge, pyelectasis decreased in SUB cats on average by 43.2% (Table 1) and the decrease was significant (P = 0.014). Pyelectasis decreased in C cats on average by 23.2% (Table 1) but the decrease was not significant during the same period (P = 0.38).
Short-term follow-up
One major short-term complication (urethral obstruction by lithiasis) occurred in each group. A perineal urethrostomy was performed after failure of medical treatment and both cats recovered uneventfully.
Seven of 11 SUB cats underwent minor short-term complications. Three cats developed urinary tract infections (UTIs) due to Escherichia coli, coagulase-negative Staphylococcus species and Acinetobacter baumannii, respectively. Two were successfully treated with antibiotics and one was not cured due to a multidrug-resistant organism (A baumannii), but was in good health at the time of follow-up (540 days post-surgery). Three cats presented with non-infectious cystitis and recovered with a 1 week analgesic treatment. One cat developed an obstruction of the left SUB device 2 weeks after placement, which resolved by flushing of the device.
The eight surviving C cats suffered nine short-term complications: one for seven cats and two for one cat. Complications were: UTIs (n = 6), which resolved with antibiotic therapy, non-infectious cystitis (n = 1), ureteral re-obstruction (n = 1), which resolved with fluid therapy, and non-obstructive ureteral stricture (n = 1).
Long-term follow-up
Ten SUB cats were still alive at a median follow-up of 225.0 days (range 60–600 days). One of 11 discharged SUB cats died from chronic kidney disease (CKD) 5 months after surgery. The survival rates at 1, 3 and 6 months were 84.6%, 83.3% (n = 12) and 72.7% (n = 11), respectively. Serum creatinine continued to decrease after discharge but remained slightly increased for 7/10 cats, with a median of 1.9 mg/dl (range 1.5–2.3 mg/dl) approximately 4–6 months after surgery. Ultrasound examination at the same period showed a good placement of the device and median minor pyelectasis of 2.8 mm (range 0–4.4 mm).
Four C cats were still alive at a median follow-up of 260 days (range 150–600 days). There was no significant difference in follow-up duration between groups (P = 1). One cat was lost to follow-up. Three of eight discharged C cats died. Two cats had re-obstruction of the same ureter 2 months after the first surgery. One suffered cardiopulmonary arrest during re-intervention and the other one was euthanased. The third cat died from chronic CKD 29 months after surgery. The survival rates at 1, 3 and 6 months were 70.0%, 50.0%, and 37.5% (n = 8), respectively (the cat lost to follow-up was excluded).
The mortality rate was not significantly different in C cats compared with SUB cats at 1, 3 and 6 months postoperatively (P = 0.62, P = 0.17 and P = 0.18, respectively). Kaplan–Meier curves were used to estimate the survival function at 1, 3 and 6 months postoperatively (Figure 3). The log rank test showed no statistical difference between the two curves at 1, 3 and 6 months (P = 0.39, P = 0.11 and P = 0.12, respectively).

Kaplan–Meier survival curves at 1 (a), 3 (b) and 6 (c) months postoperatively for cats that underwent traditional ureteral surgery (C) and cats treated by placement of a subcutaneous ureteral bypass (SUB)
Three of 10 surviving SUB cats underwent minor complications. One cat developed a UTI due to Enterococcus faecalis. After a 2 month antibiotic treatment, culture still showed the presence of the same strain. The cat was asymptomatic and antibiotics were stopped. Two cats presented with recurrent signs of non-infectious cystitis with haematuria, stranguria, pollakiuria and bladder wall thickening on ultrasonography. Bacteriological analysis failed to show any bacteria. Clinical signs resolved with short-term analgesic treatment. No minor complications occurred in C cats. There were no significant differences in UTIs and non-infectious cystitis occurrences between groups (P = 0.71 and P = 0.36, respectively).
Owners of the 10/13 surviving SUB cats were contacted by phone; 9/10 (90%) reported full satisfaction and 1/10 (10%) were mostly satisfied.
Discussion
In this study, SUB devices were successfully placed in all cats. We showed that implantation of the SUB device without fluoroscopic guidance is effective, safe and feasible, with few cats experiencing significant postoperative complications and an overall mortality rate of 15.4%. To our knowledge, no mortality rate is available with this technique in the literature and this study is the first to describe SUB device placement without fluoroscopic imaging guidance. Access to fluoroscopy is limited and this novel approach could allow more specialist surgeons to use SUB devices for feline ureteral obstruction. Fluoroscopy procedures expose the surgeon to radiation-induced risks. There are, however, likely to be some limitations to this technique. Insertion of the nephrostomy catheter into the proximal ureter, such as is performed in cases with very minor pyelectasis (<2 mm) would be very challenging/impossible without fluoroscopic guidance. In our study, minimum pyelectasis was 3.3 mm. It may also be challenging to use this technique in cats with nephroliths in the renal pelvis, reducing the space for nephrostomy placement; however, this was achieved without complication in some cats in this study. Only three minor complications occurred in our cases, likely directly due to the absence of imaging guidance: one misplaced kidney catheter and two locking loop catheters not adequately tightened to create the loop (2/19, 10.5%); nevertheless, these complications had no major consequence.
Immediate perioperative replacement was necessary in only two cats because of misplaced and kinked catheters. Misplacement of the kidney catheter has never been reported during SUB placement, probably due to the systematic use of fluoroscopy. 18 Prevalence of a per-surgical kink of the catheter is unknown in the literature. 18 In our study, this complication appeared in 1/19 SUB placements (5.3%). SUB device placement allowed shorter median surgery time (85 mins) compared with stent placement (105–182 mins).21,22 The anaesthesia time should also be as short as possible in these cats already suffering from CKD, in order to minimise potential hypotension which could lead to renal hypoperfusion. The two SUB cats that died in the immediate postoperative period were particularly clinically debilitated. In C cats, no intraoperative complication occurred, but traditional surgeries require manipulation of ureters which can cause postoperative ureteral strictures.2,5 In the C cats of our study, one ureteral stricture occurred without associated re-obstruction but five re-obstructions occurred in four cats (one cat 24 h after surgery, one cat during the short-term follow-up and two cats during the long-term follow-up) with possible non-diagnosed stricture.
Another important result of this study is that median postoperative duration of hospitalisation was shorter (3 days) for SUB cats than for C cats (5 days, P = 0.018). In addition, hospitalisation duration after SUB placement in our study was shorter compared with that published for other surgical treatments such as the use of ureteral stents (8 days). 21 A shorter hospitalisation time showed a more rapid recovery and led to reduced stress in cats and enhanced client satisfaction.
In the present study, two major postoperative complications occurred in SUB cats, with one urethral obstruction (1/13, 7.7%) and one anaemia necessitating a blood transfusion (1/13, 7.7%). Eight days after the surgery one cat presented a urethral obstruction due to lithiasis, leading to a perineal urethrostomy. This complication was not related to the SUB device placement. Moreover, the same complication occurred in a C cat. The major complications in C cats were anaemia necessitating a blood transfusion (2/11 cats) and ureteral re-obstruction (five recurrences in four cases) leading to the death of three cats (27.3%). Several major mid- and long-term complications have been reported with ureteral stenting including device migration (5–5.9%), re-obstruction of ureters (10–19%) and pyelonephritis (1.9–2%) requiring stent replacement or the use of a SUB device in 27% of cases.22,23 Those complications were not encountered in the present study. SUB device replacement is an extremely rare complication compared with ureteral stenting, for which obstructions and migrations are the more frequent causes of stent replacement. 24 One of the most important advantages of the SUB device is the ability to flush the system and prevent re-obstruction. Moreover, the inner diameter of the SUB device is larger than the stent’s inner diameter. As the SUB device totally shunts the ureters, the obstructions linked to ureteral adhesions or strictures reported with stents do not apply. In addition, unlike the stent, this system is securely fixed to the kidney, subcutaneous tissues and to the bladder, therefore preventing migration.
Cats are often anaemic at diagnosis of ureteral obstruction, either due to concurrent CKD or from excessive blood sampling during previous hospitalisations.4,25 A study showed that obstructive uropathy was associated with clinically relevant effects on erythropoiesis in human patients. 26 Moreover, anaemia seems to be common after ureteral surgery due to haemorrhage during the procedure, erythropoietin production defect after kidney injury due to ongoing CKD and prolonged obstruction or surgical procedures.25,27 Finally, fluid resuscitation is also associated with hospital-acquired anaemia, and institution of fluid therapy lowers PCV. 25 In the present study, the median PCV 24 h after surgery was significantly lower in C cats than in SUB cats and more severe anaemia occurred in C cats (7/11, 63.6%) than in SUB cats (2/13, 15.4%). In C cats, anaesthesia time was significantly longer, as was hospitalisation, which could predispose to hospital-acquired anaemia compared with SUB cats. However, there were no differences in the occurrence of severe anaemia and need of transfusion between groups. This absence of difference could be due to the small size of both groups. Two C cats required transfusions and two others died but also met transfusion criteria. Kulendra et al (2014) also reported a high transfusion rate with ureteral stenting (11.5%). 21
Ten minor complications occurred in SUB cats during the short- and long-term postoperative period: obstructed SUB device (1/19, 5.3%), UTIs (4/13, 30.8%), and non-infectious cystitis (5/13, 38.5%).
An obstructed SUB device seems to be a less common complication compared with stent obstruction (10%), 23 with just one case in the present study (1/19, 5.3%). Reported patency of the SUB device has been evident in over 90% of cats over a median of 2.5 years. 24 Berent, who is the creator of the device, reports six obstructions with stone debris leading to four re-interventions (SUB replacements), two resolutions with serial flushing among a series of 100 SUB device placements. 24 The reported high rate (4/6, 66.7%) of surgical re-intervention after obstruction of the device showed the importance of preventing device obstruction by flushing the subcutaneous shunting port every 3 months. In the present study, obstruction of the sole obstructed device was resolved with simple flushing. For those reasons, the first SUB device flushing was made 1 month after surgery for every cat.
UTIs are reported in 15% of SUB placement cases.16,18 In the present study, the UTI rate was higher (30.8%). All SUB cats received antibiotics until reception of the bacterial culture but Berent’s protocol of 2 weeks of marbofloxacin was not followed.17,22 This could lead to more bacterial infections. The main concern is the presence of multidrug-resistant organisms, causing a non-resolution of two infections (2/4 cats). Surprisingly, these two cats did not exhibit any clinical signs at the time of follow-up. UTIs were also frequent in C cats (54.5%) but all resolved after antibiotic treatment. With ureteral stenting, UTIs were reported in 26–31.7% of animals but resolved with antibiotics in 94.7% of cats.22,23 In our study, there was no significant difference in the occurrence of UTIs between SUB and C cats. However, SUB cats seem to be more prone to bacterial resistance. Infections are a problematic complication due to the presence of an exogenous implant. Infections are often difficult to treat when implants are placed into the animal’s body since their surfaces are substrates for bacterial biofilm formation. 28 As a result, bacteria are more prone to evade the host immune response and antimicrobial therapy, leading to inadequate elimination of infection.28,29 The SUB device seems to act like any implant, and the present study showed that healing of UTIs appears to be difficult. A recent study showed that orally administered antibiotics excreted in the urine can reduce the risk of biofilm formation and persistent infection for ureteral devices. 30 Despite this complication, long-term follow-up did not show any difference between SUB and C cats, and the satisfaction of SUB cats owners was high.
Non-infectious cystitis is reported in 5/13 (38.5%) SUB cats in the present study. Similar rates are reported for cats with ureteral stenting (25–37.7%).21–23,31 The most likely explanation is a chronic irritation of the bladder because of the presence of the bladder catheter. In parallel, non-infectious cystitis was an uncommon complication in C cats (1/11, 9.1%). However, there was no significant difference in the occurrence of non-infectious cystitis between groups. All cats in the present study required only a short-term analgesic treatment for resolution of clinical signs. Owners should be warned of this minor complication that can reoccur multiple times in the life of their animal. Replacing a stiff stent with a soft one was reported as necessary to resolve stranguria and pollakiuria in 6% of cases with ureteral stenting. 22 To our knowledge, SUB device replacement for chronic non-infectious cystitis has never been reported.
Serum creatinine decreased on average by 77.3% between admission and discharge. In the C group, serum creatinine decreased on average by 46.4%. However, it is difficult to state that the SUB device allows a better renal function recovery as serum creatinine of the SUB cases was significantly higher than that of the C cats, which may represent a bias in the study.
The main limitations of our study were its retrospective nature and the small sample size of the two groups. Also, the follow-up period was too short to clearly establish long-term outcomes of the surgery and, in particular, to compare it with the ureteral stenting technique for which median survival time was established. Moreover, even if there was no significant difference in follow-up duration between groups, there was generally longer follow-up for C cats than for SUB cats, which may have falsely improved the long-term outcome of the latter. As a retrospective study, cats’ characteristics and follow-up parameters, as well as time, were not perfectly homogeneous. A comparison with ureteral stenting, which is a recent advance in the treatment of ureteral obstruction, would have been interesting. A prospective study evaluating the surgery’s outcome on cats with homogenous clinical status is warranted to establish long-term follow-up and prognosis.
Conclusions
Recently, the SUB device has become the first choice for all causes of feline ureteral obstructions. This study shows that this procedure can be achieved without fluoroscopic guidance and without major difficulties, allowing a shorter time of hospitalisation compared with the traditional approach, with fewer short- and long-term complications. There are far fewer long-term problems with the SUB device than with ureteral stenting or traditional surgery in cats, when appropriately managed. However, median survival time and long-term complication monitoring is warranted.
Footnotes
Acknowledgements
The authors want to thank Mathieu Magnin for his assistance with the statistical results and Charlotte Thorneloe for improving the use of the English language in the manuscript.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
