Abstract
Objectives
The aim of this study was to determine the plasma pharmacokinetic profile, tissue concentrations and urine elimination of cefazolin in cats under surgical conditions after a single intravenous dose of 20 mg/kg.
Methods
Intravenous cefazolin (20 mg/kg) was administered to nine young mixed-breed cats 30 mins before they underwent surgical procedures (ovariectomy or orchiectomy). After antibiotic administration, samples from blood, some tissues and urine were taken. Cefazolin concentrations were determined in all biological matrices and pharmacokinetic parameters were estimated.
Results
Initial plasma concentrations were high (Cp(0), 134.80 ± 40.54 µg/ml), with fast and moderately wide distribution (distribution half-life [t½(d)] 0.16 ± 0.15 h; volume of distribution at steady state [V(d[ss])] 0.29 ± 0.10 l/kg) and rapid elimination (body clearance [ClB], 0.21 ± 0.06 l/h/kg; elimination half-life [t½], 1.18 ± 0.27 h; mean residence time 1.42 ± 0.36 h). Thirty to 60 mins after intravenous administration, cefazolin tissue concentrations ranged from 9.24 µg/ml (subcutaneous tissue) to 26.44 µg/ml (ovary). The tissue/plasma concentration ratio ranged from 0.18 (muscle) to 0.58 (ovary). Cefazolin urine concentrations were high with 84.2% of the administered dose being eliminated in the first 6 h postadministration.
Conclusions and relevance
Cefazolin plasma concentrations remained above a minimum inhibitory concentration of ⩽2 µg/ml up to 4 h in all the studied cats. This suggests that a single intravenous dose of 20 mg/kg cefazolin would be adequate for perioperative prophylactic use in cats.
Introduction
Cefazolin is a parenteral first-generation cephalosporin with good activity against gram-positive cocci (Staphylococcus species, including beta (β)-lactamase producing strains, and Streptococcus species) and many Enterobacteriaceae (Escherichia coli, Klebsiella species, Proteus mirabilis), Pasteurella species and anaerobes.1,2 A breakpoint for bacteria isolated from animals has been set at ⩽2 μg/ml. 3 Cefazolin, as with other β-lactams, is characterized as having time-dependent killing antibiotic activity with minimal persistent effects. Therefore, antibiotic plasma concentrations should be kept above the minimum inhibitory concentration (MIC) for as long as possible during the antibiotic therapy (where this length of time is T >MIC). It has been established that a T >MIC of 40–50% of the dosing interval is appropriate for a successful outcome.4,5
Parenteral first-generation cephalosporins (cephalothin and cefazolin) are usually recommended for prophylaxis of surgical wound infections. Although they are very similar, cefazolin surpasses cephalothin in some important aspects. Cefazolin, unlike cephalothin, has no nephrotoxic effects, 6 or adverse effects on blood clotting and platelet function (platelet aggregation, bleeding time, platelet count, platelet size, prothrombin time or activated partial thromboplastin time). 7 Additionally, cefazolin is somewhat more active against gram-negative bacteria than cephalothin, 2 providing more benefit for prophylaxis of surgical wound infections.
Cefazolin pharmacokinetics have been extensively studied in dogs.8–13 They have also been studied in other animal species, such as goats, 14 horses, 15 calves 16 and laboratory animals. 8 Cefazolin is rapidly distributed throughout most body fluids, penetrating poorly across biological membranes and physiological barriers. Reported volumes of distribution are in the range of 0.2–0.3 l/kg. Cefazolin protein binding has been proved to be very variable among species (from 80% in people to 19% in dogs);6,9 however, it has not been determined in cats. In dogs, the low protein binding percentage is related to antibiotic diffusion to tissues, which parallels plasma concentrations,9,11 favoring a rapid distribution from plasma to interstitial fluid. Cefazolin is rapidly eliminated in urine by glomerular filtration and tubular secretion; the reported elimination half-life (t½) in dogs is around 1 h.8,9,11,13
The main indication of cefazolin in human and veterinary medicine is for perioperative prophylaxis in surgeries classified as type 2 (clean-contaminated wounds). 17 For this purpose, it is administered intravenously around 30 mins prior to surgical incision (to allow achievement of therapeutic concentrations at tissue level when incision is performed). Administration can be repeated 90 mins later in lengthy surgeries or when significant blood loss occurs. According to Rosin et al, 11 one intravenous administration is enough in dogs; however, if necessary, an additional subcutaneous administration can be performed.
To the best of our knowledge, cefazolin pharmacokinetics have not yet been described in domestic cats. Therefore, the aim of this study was to characterize the plasma pharmacokinetics and renal excretion of cefazolin, as well as to determine its concentrations in some selected tissues in cats under surgical conditions.
Materials and methods
Experimental animals
Nine experimental mixed-breed cats (three females, six males) aged 1–1.5 years, with mean ± SD weight of 4.02 ± 0.90 kg, were included. All cats were healthy, as determined by clinical examination, complete blood and serum biochemical analysis, and urinalysis. Animals were housed in the facilities of the Facultad de Ciencias Veterinarias, Universidad de Buenos Aires, and allowed to acclimatize for 2 months before the experiment. Access to a high-quality commercial dry food (Purina ProPlan; Nestle) and water was ad libitum.
All animal procedures were approved by the Institutional Animal Care and Use Committee, Facultad de Ciencias Veterinarias, Universidad de Buenos Aires, Argentina.
Dosage form
An aqueous 10% (cefazolin equivalent) w/v solution of cefazolin sodium salt (Cefazolina Richet; Richet) was administered intravenously at a dose rate of 20 mg/kg. The dose was diluted in 2.5 ml saline solution (NaCl 0.9%) before administration and injected through the cephalic vein over a 2 min period.
Experimental design
Prior to starting the study, two catheters (Jelco; Smiths Medical International) were placed into the cephalic veins. One (24 G) was for antibiotic and fluid (NaCl 0.9%, 10 ml/kg/h) administration and the other (22 G) for blood sample withdrawal.
The urethra was also catheterized to collect all urine produced during the complete duration of the study (6 h).
Surgical procedures (ovariectomy or orchiectomy) were performed under general parenteral anesthesia, according to conventional standard techniques (midazolam 0.2 mg/kg [Midazolam; Richmond Vet Pharma]; ketamine 10 mg/kg [Ketonal 100; Richmond Vet Pharma]; xylazine 0.5 mg/kg [Rompun; Bayer]). Cefazolin was administered intravenously, at a dose rate of 20 mg/kg, 30 mins prior first surgical incision.
Blood sampling
Blood samples (0.7 ml) were collected into heparinized syringes at 0.08; 0.16; 0.33; 0.50; 0.75; 1; 1.50; 2; 3; 4; and 6 h after cefazolin administration.
Samples were centrifuged at 1500 g for 15 mins and the harvested plasma stored at −20°C until analysis.
Urine sampling
Starting with an emptied urinary bladder, all urine produced after cefazolin administration was collected at 1 h intervals. The volume of each sample was recorded and an aliquot was stored at −20°C.
Tissue sampling
Tissue samples (0.02–0.1 g) were taken at 0.5–1 h postadministration. Selected tissues/organs were skin, subcutaneous, muscle, ovary and uterus in female cats, and skin, testicle and epididymis in males. Samples were rinsed briefly with saline solution and dried through gentle pressure with sterile gauze for removing blood contamination. Subsequently, they were weighed and stored at −20°C.
Cefazolin was eluted from tissue samples following a technique described by Bamberg et al. 18 Briefly, samples were cut into small pieces and diluted with phosphate buffer pH 6 (in a ratio 1:2 w/v) and incubated for 24 h at 4°C, applying agitation for the first 40 mins of incubation. Finally, they were centrifuged (1500 g for 15 mins) and the supernatant harvested and stored until assay.
All collected samples (plasma, urine and tissue) were assayed within 2 weeks of collection.
Cefazolin determination
Cefazolin plasma, tissue and urine concentrations were determined by microbiological assay, 19 using Bacillus subtilis ATCC 6633 as test microorganism. Standard curves were prepared on normal cat plasma, phosphate buffer pH 6 or normal cat urine, depending on the sample matrix to be quantified.
Each sample was seeded in triplicate and each standard dilution in quintuplicate.
Limit of detection and quantification of the method for the three assayed matrices was 0.78 µg/ml and 1.56 µg/ml.
The method was linear between 0.78 and 100 µg/ml (r = 0.9982). Inter- and intra-assay coefficients of variation were <10%.
Pharmacokinetic analysis
Individual cefazolin plasma concentration vs time curves were analyzed by non-linear least square regression analysis using computer software (Phoenix WinNonlin 6.3, 2005–2012; Certara).
Initial estimates were determined using the residual method, 20 and refitted by non-linear regression.
The number of exponents needed for cefazolin intravenous administration data were determined by applying the Schwartz and Akaike criterions,21,22 and the residual distribution around the estimated concentrations.
Most pharmacokinetic parameters were calculated using classic equations associated with compartmental analysis. 20
Results
Adverse effects were not observed during or following intravenous administration of cefazolin in any of the cats.
Cefazolin plasma disposition curves in all the animals were best fitted to a biexponential model (Figure 1) according to an open bicompartmental model:
Where C(t) (µg/ml) represents cefazolin plasma concentration at t time; Y1 and Y2 (µg/ml) are the concentrations extrapolated to 0 time of the distribution and elimination phase, respectively; and λ1 and λ2 (per h) are the slopes for the distribution and elimination phase, respectively.

Simulated cefazolin plasma concentration–time profile after intravenous administration at a dose of 20 mg/kg (−) and mean ± SD (▲) (n = 9). Minimum inhibitory concentration (MIC) values correspond to breakpoint for cefazolin (MIC = 2 µg/ml)
The average equation for the model was:
The main pharmacokinetic parameters are reported in Table 1.
Pharmacokinetic parameters of cefazolin after intravenous administration to cats at a dose rate of 20 mg/kg (n = 9)
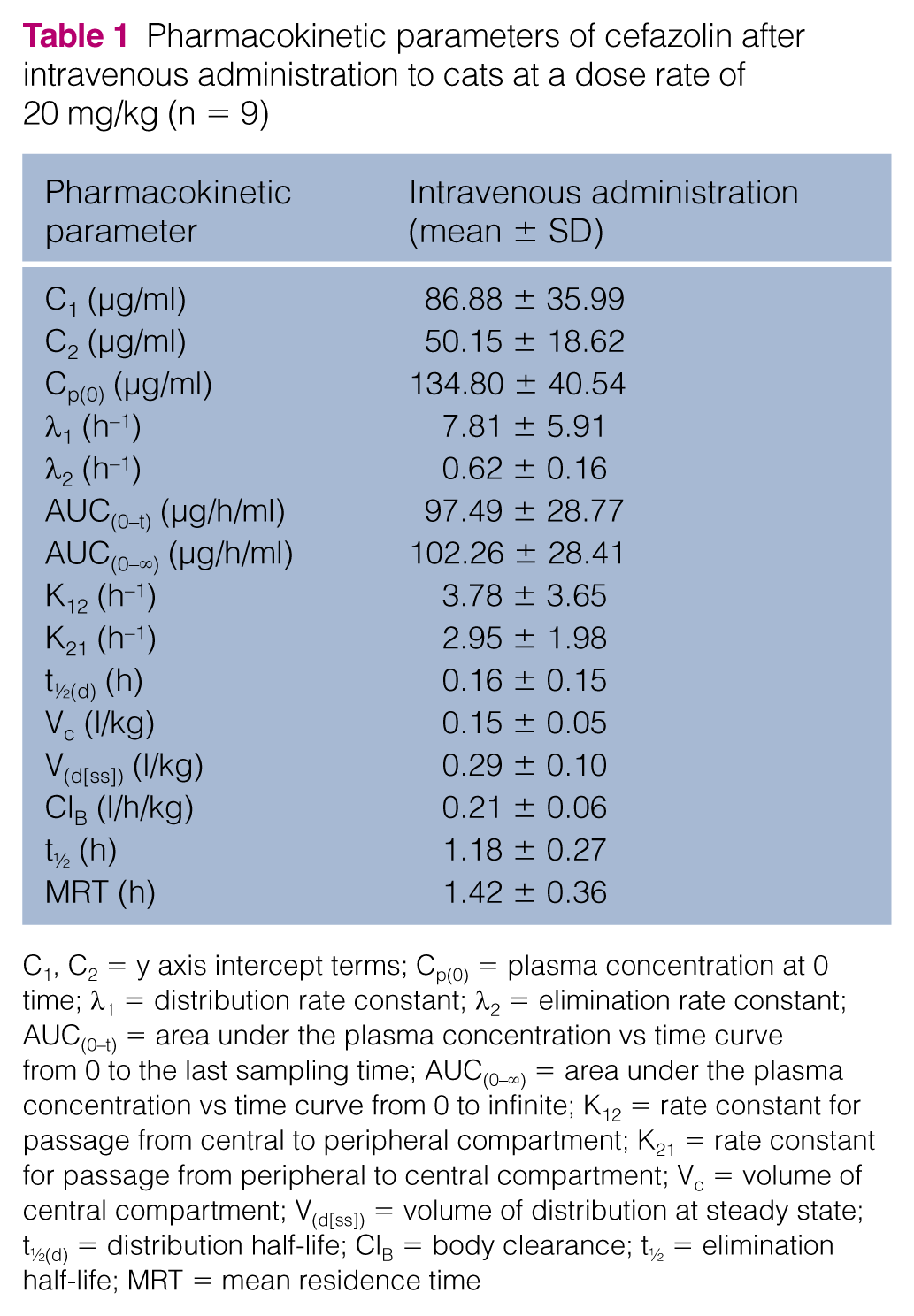
C1, C2 = y axis intercept terms; Cp(0) = plasma concentration at 0 time; λ1 = distribution rate constant; λ2 = elimination rate constant; AUC(0–t) = area under the plasma concentration vs time curve from 0 to the last sampling time; AUC(0–∞) = area under the plasma concentration vs time curve from 0 to infinite; K12 = rate constant for passage from central to peripheral compartment; K21 = rate constant for passage from peripheral to central compartment; Vc = volume of central compartment; V(d[ss]) = volume of distribution at steady state; t½(d) = distribution half-life; ClB = body clearance; t½ = elimination half-life; MRT = mean residence time
Cefazolin showed a fast distribution, reflected by the rate constant of the process (λ1 7.81 ± 5.91 h–1) and its short half-life (t½(d) 0.16 ± 0.15 h). The extent of distribution was moderate (V(d[ss]) 0.29 ± 0.10 l/kg).
Cefazolin was rapidly eliminated from the body with a total body clearance (ClB) of 0.21 ± 0.06 l/h/kg, a t½ of 1.18 ± 0.27 h and a mean residence time (MRT) of 1.42 ± 0.36 h.
Cefazolin tissue concentrations and tissue/plasma concentrations ratio are shown in Table 2. Tissue concentrations were always lower than the corresponding plasma concentrations. The highest concentrations were found in ovaries, uterus and epididymis, while the lowest were found subcutaneously and in muscle and testicles.
Cefazolin tissue concentrations (mean ± SD) and tissue/plasma concentration ratios after intravenous administration (20 mg/kg) (n = 9)

Cefazolin was rapidly and almost totally eliminated in urine (84.22% of the total administered dose in a 6 h period). The cumulative amount excreted in urine throughout the study is shown in Figure 2. Cefazolin urine concentrations were very high at all sampling times, with a peak value of 1378.13 ± 832.02 µg/ml during the first hour and, a minimum of 332.41 ± 237.01 µg/ml during the sixth hour.
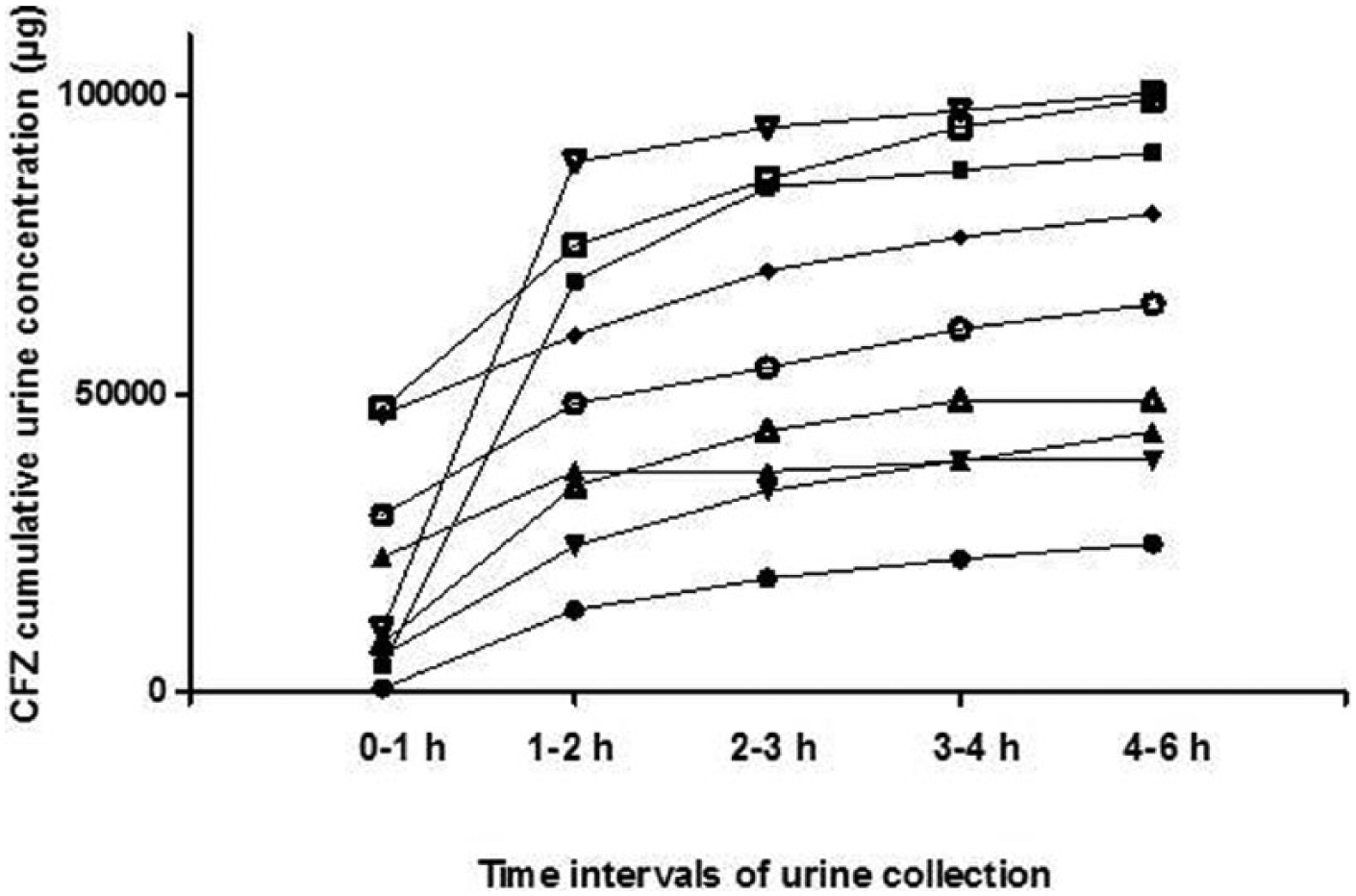
Cumulative cefazolin (CFZ) urinary elimination after its intravenous administration to nine cats at a dose rate of 20 mg/kg
Discussion
Perioperative antimicrobial prophylaxis is recommended for various surgical procedures to prevent surgical site infections. Optimal antimicrobial agents for prophylaxis should be bactericidal, non-toxic and achieve active concentrations against the typical pathogens that can cause surgical site infection postoperatively. Owing to its characteristics, cefazolin is the usual choice for prophylaxis in the perioperative period in both human and veterinary medicine.
It is important to highlight that to achieve a proper prophylactic effect it is necessary to know the antibiotic plasma disposition, as well as its permanence at the site of action.
Pharmacokinetic studies of antimicrobial agents are most often performed in awake animals. Because the physiology of the anesthetized surgical patient is different from that of the awake patient, in the present study the pharmacokinetics of cefazolin were examined in cats under general anesthesia.
A weakness of the present study is that samples taken from tissues correspond to a unique time point after cefazolin administration. However, the sampling time was chosen taking into consideration surgical timing when the highest antibiotic concentrations in tissues are required.
Although the temporal pattern of cefazolin tissue concentrations cannot be predicted from a unique data point, it has been reported that tissue concentration declines in parallel to the plasma disposition curve.9,11
Cefazolin concentrations in all of the studied matrices were determined through the microbiological assay. 19 This method is criticized by some authors; however, for cefazolin, it was selected because its sensibility, simplicity and high correlation with high-performance liquid chromatography determination.23,24 Moreover, as cefazolin is almost completely eliminated through the kidneys as the active compound in all studied species (rats, dogs, sheep, goats and humans),8,25 the microbiological assay would be appropriate to quantify its concentrations in biological specimens. Another issue with this method is its low sensitivity (limit of quantification [LOQ] 1.56 µg/ml). However, considering that the LOQ of the method is below the cumulative cafazolin breakpoint concentration (2 µg/ml) it can be applied for determination of efficacious cefazolin concentrations in biological matrices.
Plasma cefazolin disposition curves after intravenous administration were best fitted by an open bicompartmental model in all the cats. This is in agreement with studies performed in dogs, 13 calves 16 and goats. 14
As shown in Figure 1, immediately after cefazolin intravenous administration, plasma concentrations were very high (Cp(0) 134.80 µg/ml), and declined sharply (as is reflected by the distribution and elimination rate constants). Concentrations remained above the MIC breakpoint (2 µg/ml) for >4 h and were above the limit of detection (>1 µg/ml) up to the last sampling time in 8/9 animals.
The observed plasma pharmacokinetic profile of cefazolin in cats is as expected for a β-lactam antibiotic; similar results have been reported in other species.8–16 Moreover, cefazolin pharmacokinetic parameters in cats are very similar to those reported for cephalothin in this species. 26
The mean cefazolin tissue concentrations found in this study were relatively high and estimated tissue/plasma ratio for all tissues were always less than unity. These results confirm that distribution of cefazolin is restricted to the vascular space and the extracellular fluid. However, it is known that antibiotic concentration measured from tissue homogenates should be carefully considered and not used to draw direct conclusions on drug activity or to predict antibiotic efficacy in tissue infections. 27
As with many β-lactams, cefazolin is eliminated in the urine by glomerular filtration and tubular secretion. The high urine concentrations observed in the present study are in good agreement with values found in human studies (≈4000 µg/ml). 6
The rapid urinary excretion of cefazolin (84.2% of the administered dose in a 6 h period – equivalent to five half-lives) found in this study is similar to that reported for dogs (80% in 8 h). 8
According to its bacterial-killing kinetics, cefazolin is classified as a time-dependent antimicrobial agent, and the ideal dosing regimen would maximize the duration of drug exposure. The length of time that its concentration remains above the MIC, that is the T >MIC, is the parameter that best correlates with efficacy. Therefore, to predict the efficacy of cefazolin for surgical infection prophylaxis, the MIC values that should be used are those reported for pathogenic bacteria that most commonly colonize wounds. In this context, Staphylococcus species is one of the most commonly reported bacteria. Applying the Clinical and Laboratory Standards Institute MIC breakpoint (⩽2µg/ml), 3 cefazolin plasma and tissue concentrations will remain well above this value for more than 4 h.
Conclusions
According to its pharmacokinetic profile and tissue concentrations, a unique cefazolin intravenous administration of 20 mg/kg to cats provides adequate plasma and tissue concentrations to guarantee antimicrobial coverage for most common pathogens that colonize surgical sites.
Footnotes
Acknowledgements
The authors are indebted to Purina ProPlan, Argentina, for the kind provision of the animal food.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by a grant (Research Project UBACyT 20020130100400, 2014-2017) of Secretaría de Ciencia y Técnica, Universidad de Buenos Aires, Argentina.


