Abstract
Objectives
This study aimed to describe the ultrasonographic, endoscopic and histological characteristics of the caecum and ileocaecocolic junction in cats suffering from chronic clinical signs compatible with caecocolic disease.
Methods
Cats presenting with clinical signs suggestive of a caecocolic disease were prospectively recruited. All cats underwent an ultrasonographic examination of the caecum, ileum, colon, ileocolic lymph nodes and local mesenteric fat, in addition to comprehensive abdominal ultrasonography. This was followed by a colonoscopy with a macroscopic assessment of the caecocolic mucosa; caecocolic tissue samples were systematically collected for histologic analysis.
Results
Eighteen cats were included. Eleven of 18 cats had ultrasonographic abnormalities adjacent to the ileocaecocolic junction (lymphadenopathy, local steatitis) and 13/18 cats had abnormalities directly related to the junction (wall thickening, loss of wall layering). Seventeen of 18 cats had at least one ultrasonographic abnormality. Endoscopically, hyperaemia, oedema, discoloration and/or erosions were found in all cats. Each cat was classified as having mild or moderate-to-severe lesions according to endoscopic results; no classification could be established statistically for ultrasonographic results. The accentuation of the dimpled pattern tended to be inversely related to the severity of endoscopic lesion scoring. Histologically, a large proportion of cats showed typhlitis (13/16), one had lymphoma and two were normal. All cats with typhlitis also had colitis. There was only slight agreement between endoscopic and histological caecal results regarding the severity of lesions. Loss of caecal wall layering on ultrasound was found in 7/18 cats and, surprisingly, did not appear as a reliable predictor of the severity of inflammation or of malignancy; neither did local steatitis nor lymph node size.
Conclusions and relevance
Ultrasonography and endoscopy should not be used as the sole methods to investigate the ileocaecocolic region in cats with clinical signs suggestive of caecocolic disease. The presence of chronic clinical signs should routinely prompt histological biopsy.
Introduction
Cats suffering from chronic ileocaecocolic (ICC) disease, including typhlitis, although rare in this species, usually present with diarrhoea, dyschezia, tenesmus, haematochezia and/or constipation, evolving in a continuous or intermittent way, with a duration in excess of 21 days. 1 The infectious causes of chronic ICC disease include, but are not limited to, feline infectious peritonitis, parasites such as Trichuris species, Ancylostoma species, Tritrichomonas foetus, Cryptosporidium parvum, Giardia duodenalis, Isospora species, Toxoplasma gondii and, much less frequently, bacterial enterocolitis (Campylobacter species, Escherichia coli, Salmonella species, Yersinia species, Clostridium species). Other differentials for chronic ICC diseases include inflammatory diseases, inversion of the caecum into the colon, obstruction, stenosis, neoplasia (lymphoma, adenocarcinoma, mastocytoma) and fecaliths.1–7 After exclusion of parasitic and food-responsive inflammatory bowel diseases, diagnostic investigations usually lead to an ultrasonographic examination, often followed by colonoscopic assessment and collection of biopsies for histological evaluation. Few descriptions of pathological ultrasonographic and endoscopic appearances of the feline ICC junction are available.1,2,4,8,9 Ultrasonographic abnormalities at the level of the ICC junction have previously been reported and are clinically significant.2,8,9 While guidelines exist for histological evaluation of colonic biopsies, criteria for caecal evaluation are not specifically described in cats. 10
The association of the ultrasonographic, endoscopic and histological appearances of the caecum in healthy cats was reported in 2015 by the present authors. 11 Ultrasonographically, a caecal follicular layer was described as a combination of mucosal and submucosal layers, considered as a unique ultrasonographic layer because of the lymphoid follicles overlapping them both. 11 An ultrason-ographic cut-off value for this follicular layer in cats without clinical signs ascribable to ICC pathology has been established to differentiate non-invasively cats with and without mild histological inflammation. 11 On endoscopy, the normal caecal mucosa showed a ‘dimpled aspect’ in all cats due to the presence of small surface depressions. 11 This dimpled aspect could be of variable intensity, usually mild to moderate in healthy cats, although a severe dimpled aspect could occasionally be found. 11
The aim of this study was to determine and describe the association between the ultrasonographic changes, the macroscopic appearance during colonoscopy, and the histological findings in cats presented with clinical signs suggestive of caecocolic disease.
Materials and methods
Study population
This prospective study was conducted, with informed owner consent, on cats presented to the small animal gastroenterology service of Alfort Veterinary School Hospital, between February 2014 and July 2015. The following clinical signs were considered inclusion criteria: mucoid diarrhoea, haematochezia, dyschezia, tenesmus and, less frequently, constipation. Cats were included if they presented with one or more of these clinical signs for more than 3 weeks, and underwent all of the following diagnostic tests, performed within less than 1 week of each other: an abdominal ultrasonography, a colonoscopy and caecocolic biopsies. Cats receiving a treatment such as antibiotics or corticosteroids (>0.2 mg/kg q24h) were not included. For each cat, a physical examination was performed, and data regarding age, breed, sex and medical history were collected by one study author (VF). Standard investigations were performed before ultrasonography and colonoscopy (ie, biochemical and haematological analyses, urinalysis, coproscopy, feline immunodeficiency virus and feline leukaemia virus point-of-care tests and serum total thyroxine [T4] concentrations, when necessary).
Ultrasonographic evaluation
The abdomen was clipped and each cat was placed in dorsal recumbency for the ultrasonographic examination, performed with a linear 18 mHz transducer (MyLab 30; Esaote), by the same observer (HH), under the supervision of a board-certified radiologist (PP). For each cat, after completion of a full abdominal ultrasound, the ileum, caecum, ascending and descending colon in longitudinal planes, the ileocolic lymph nodes (LNs) and the adipose tissue surrounding the ICC junction were examined.
The following parameters were recorded: the full thickness of the ileal, colonic and caecal walls on longitudinal planes of the wall in the near field; the appearance of wall layering; the thickness of the follicular layer in the proximal and distal caecum (combination of the mucosal and submucosal layers, with the proximal part being the closest to the colon and the distal part closest to the cul-de-sac) (Figure 1); the content (gas, solid, liquid) of the caecum and the colon; the thickness, number, shape, echogenicity and homogeneity of the ileocolic LNs; the presence of peritoneal effusion; and the echogenicity of the mesenteric surrounding fat. 11 The last was qualified as follows, according to a grading scheme defined by the authors and used in a previous publication: grade 0 = normoechoic fat compared with adjacent mesenteric fat and well-defined contours of the LNs; grade 1 = hyperechoic fat and well-defined contours of the LNs; and grade 2 = hyperechoic fat and blurred contours of the LNs. 11

Ultrasonographic image on longitudinal plane of a feline caecum (Ca). The thickness of the proximal (closest to the colon [Co]) and distal (closest to the cul-de-sac) parts of the caecal wall was measured, from the interface of the ultrasound with the luminal content to the external border of the serosa. The intermediate calliper depicts the outer border of the combined mucosal plus submucosal layers, the so-called follicular layer
Normal ileal and colonic upper limit wall thicknesses used were 3.2 mm and 2.5 mm, respectively.8,12,13 For upper caecal wall thicknesses, a previous paper on healthy cats, performed by the present authors, revealed maximal thicknesses for cats with no caecal inflammation (as determined by histology and after exclusion of cats with asymptomatic mild inflammation) of 3.8 mm and 4.1 mm (proximal and distal parts, respectively). 11 These values appeared higher than those previously published (2.6 mm and 3.6 mm, respectively) and so were selected as upper limit caecal wall thicknesses. 12 In the same paper, the caecal follicular layer had a maximum thickness of 2.6 mm and 3.2 mm in the proximal and distal parts, respectively. 11 The normal range for ileocolic LNs is between 2.9 mm and 4.1 mm; they appear ovoid in shape and homogeneous; two, three or more can often be seen.11,12,14 The ultrasonographic parameters from the cases enrolled were compared with those from a feline population without clinical signs, established in a previous paper by the same authors. 11
Endoscopic evaluation
All cats underwent a colonoscopy (Video Endoscope GIF 180 [Olympus]; 8.8 mm diameter insertion tube [VF and AB]) in left lateral recumbency. 15 Colonoscopies were performed by the two endoscopists together, both of whom were unaware of the ultrasonographic results. The procedures were performed under general anaesthesia: diazepam (Valium, 0.25 mg/kg IV; Roche) and propofol (Propovet, 5 mg/kg IV; Axience) for induction; the cats were then intubated and isoflurane gas was used for maintenance anaesthesia (1.5 l/min) (Vetflurane; Virbac).
The mucosal appearance of the caecum and colon was described. For each cat, macroscopic evaluation of the distal ileal mucosa was performed when possible, requiring repositioning of some cats in order to facilitate the access to the ICC junction. Criteria from the World Small Animal Veterinary Association (WSAVA) Gastrointestinal Standardization Group were recorded using a standardised form. 16 Each criterion was scored according to the following scale: 0 = normal; 1 = mild; 2 = moderate; 3 = severe. As no standard is suggested in terms of number of modified parameters to evaluate the severity of lesions, the authors decided to classify a cat as having moderate-to-severe lesions if at least two WSAVA criteria were modified (including at least one with a score of 2/3 or more) or a single WSAVA criterion was modified with a score of 3/3. The others were classified as having mild lesions (or no lesion if all criteria were normal). The operators also focused on the dimpled aspect and it was scored subjectively as absent, mild, moderate or severe. The presence of stenosis, intussusception, mass, foreign body or parasites was also recorded. Furthermore, caecal and colonic wall rigidity was also subjectively assessed with the biopsy forceps.
At least five mucosal biopsy samples were systematically collected with the same endoscopic forceps (smooth and fenestrated jaws of 2.3 mm external diameter [Endojaw; Olympus Medical Systems]): two from the caecum, one from the ascending and two from the descending colon, or more when possible. At least one ileal biopsy was also collected when possible, either by direct visualisation or while blindly catheterising the ileocolic junction with the endoscopic forceps. All samples but one from the descending colon were sent for histological evaluation. This other biopsy was sent for coronavirus detection by the reverse-transcriptase polymerase chain reaction (RT-PCR) method.
Histological evaluation
The biopsies were routinely processed, sectioned at 3 µm and then stained with haematoxylin–eosin–saffron. The slides were evaluated by two pathologists (EL and JC), who were unaware of the ultrasonographic and endoscopic results. A scoring system developed by the WSAVA Gastrointestinal Standardization Group was used. 16 Ileal samples were evaluated as if they were duodenal samples; caecal samples were evaluated as if they were colonic biopsies. A sample was considered as inflammatory if the intensity of the leukocytic infiltrate exceeded normal values, as defined by the WSAVA Gastrointestinal Standardization Group. 16 In all biopsies, the final grade of inflammation severity corresponded to the higher grade of leukocytic inflammation (lymphoplasmacytic, eosinophilic or neutrophilic) seen on biopsies. For colonic and caecal samples, animals with a final diagnosis of inflammation were allocated into two groups: one group corresponding to mild inflammation, the other group corresponding to moderate-to-severe inflammation. Histological analysis was considered as the gold standard when evaluating the inflammatory or tumoral status of cats.
Detection of feline coronavirus
Pathology consistent with feline coronavirus was screened for by systematically examining the caecal biopsies. Briefly, RNA was extracted from biopsies by means of RNeasy Mini Kit (Qiagen), following the manufacturer’s protocol, and the RNA templates were stored at −70°C until their use. Quantitative RT-PCR assays were performed using a previously described protocol. 17
Statistical analysis
Medians and interquartile ranges (IQRs; first and third quartiles) were provided for all quantitative variables, as some of them were not normally distributed. The distribution of the proximal caecal wall was compared with that of the distal wall within the feline population, by using the Wilcoxon signed rank sum test for paired data. Receiver operating characteristic (ROC) curves were used to assess the accuracy of each measured ultrasonographic parameter in classifying cats according to the severity of the lesion (mild vs moderate-to-severe), using histology as the gold standard. 18 A higher area under the ROC curve (AUROC) indicated a better accuracy of the parameter. Younden’s index was used to determine the optimal cut-off values for each measured ultrasonographic parameter. 19 The cut-off values provided were then applied to calculate sensitivities and specificities for each measured parameter. The ultrasonographic parameter with the highest AUROC would then be used to classify cats with moderate-to-severe lesions vs no or mild lesions, for the caecum. Kappa (κ) coefficients were calculated to quantify the agreement on severity of lesions between the results of histology, ultrasonography, endoscopy and virology, both in the colon and the caecum. The strength of agreement was considered poor, slight, fair, moderate, substantial and almost perfect for κ values of <0.00, 0.00–0.20, 0.21–0.40, 0.41–0.60, 0.61–0.80 and 0.81–1.00, respectively. 20 Ultrasonographic parameter medians for this feline population and the previous healthy population from another paper by the same authors were compared using a Mann–Whitney test. 11 A P value <0.05 was considered significant.
Results
Eighteen cats (seven females, 11 males) were included in the study. Sixteen were neutered (nine males, seven females). Represented breeds were domestic shorthair (11/18), Maine Coon (2/18), Birman (2/18), Oriental Shorthair (1/18), Chartreux (1/18) and Angora (1/18). The age of the cats ranged from 6 months to 18 years, with a median (IQR) of 6.5 (3.2–12) years. The median (IQR) weight was 4.7 (4.1–5.7) kg. Nine cats were regularly dewormed; the others had a negative coproscopy on inclusion. Cats were mainly presented for haematochezia (15/18) and diarrhoea (7/18). Associated clinical signs were vomiting (5/18), constipation (1/18), dysorexia (2/18), weight loss (1/18) and hyperthermia (1/18). Only one cat had a previous history of inflammatory bowel disease with lymphoplasmacytic infiltration of the jejunal and ileal mucosa; its medical treatment at presentation was prednisolone 0.16 mg/kg q24h, which was interrupted 1 week before the study examinations. Median (IQR) duration time of clinical signs was 7.5 (2–12) months.
Ultrasonographic findings
The caecum was visualised in all cats. Eleven of 18 cats (61%) had abnormalities adjacent to the ICC junction (abnormal LNs or steatitis). Two LNs (11/18) or more (7/18) were visible for each cat. Three of 18 had at least one enlarged LN (⩾4.1 mm). Median LN size was 3.1 mm (range 2–8 mm; IQR 2.4–3.9). All LNs were homogeneous; 3/18 (17%) were rounded and hypoechoic (only one of these was considered as enlarged) (Figure 2). The adipose tissue surrounding the ICC junction displayed grade 0 (8/18), 1 (7/18) or 2 (3/18) steatitis (Figure 2). No peritoneal effusion was found in any cat.
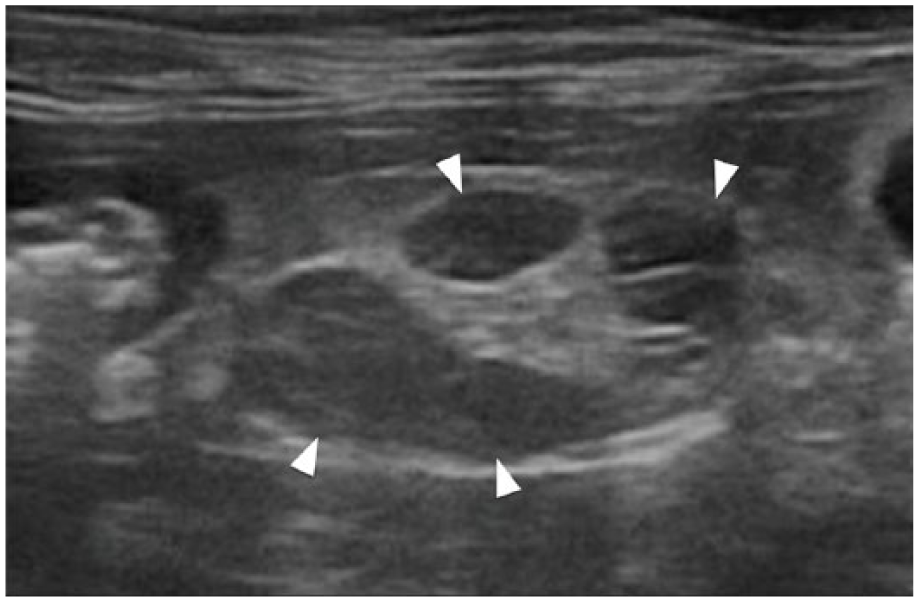
Ultrasonographic image of ileocolic lymph nodes (arrowheads). Note their hypoechogenicity, their number (three in this image), the rounded shape of the two lymph nodes in the top field and the mildly hyperechoic surrounding mesenteric fat (grade 1)
Thirteen of 18 (72%) cats presented with abnormalities directly related to the ICC junction. The caecum wall was thickened in its proximal part for 2/18 cats; it was considered as normal for all in the distal part (18/18). The caecal layers previously described could not be identified in 7/18 cats (39%) in either proximal or distal parts; the wall then had a diffuse hypoechoic aspect (Figure 3). The follicular layer was thus measured in 11/18 cats and appeared thickened for 3/11 in the proximal part and for none in the distal part. Colonic and ileal layers were preserved in all cats but walls were thickened in 1/18 and 8/18 cats, respectively. Median (IQR) thicknesses are detailed in Table 1. The median for the proximal caecal wall (3.3 mm) appeared slightly higher than for the distal wall (3.0 mm), but they were not significantly different from each other (P = 0.10). Indeed, 12/18 cats had a proximal caecal wall thicker than the distal one. The caecum was empty (with or without a small amount of gas) in all cats. When all caecal, ileal, colic, mesenteric fat and LN parameters were combined, 1/18 cats had no abnormality, 5/18 had one abnormality, 3/18 had two, 4/18 had three, 3/18 had four, 1/18 had five and 1/18 had seven.
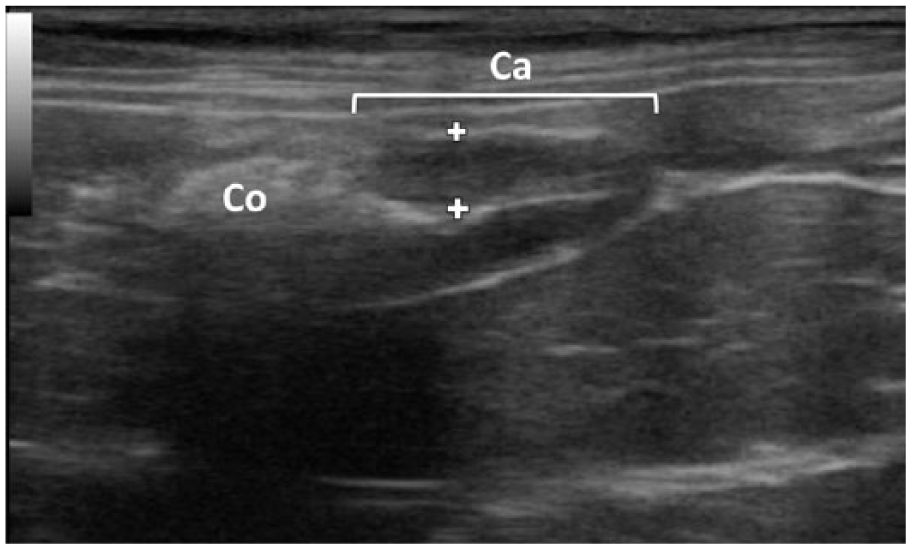
Ultrasonographic image on longitudinal plane of a feline caecum (Ca) with loss of wall layering. The full thickness of the ventral wall is measured between the callipers. The colon is visualised on the left of the image (Co)
Median thicknesses and interquartile ranges (IQRs) of the different ultrasonographic parameters

ROC curves were calculated for the following five caecal parameters, in order to classify cats according to the severity of caecal lesions (as determined by histology): proximal and distal caecal wall and follicular layer thicknesses (by attributing to the cats with no measurements [loss of layering] the highest value obtained for cats with a measurable follicular layer) and size of the most enlarged LN. All AUROC curve values were lower than 0.81. Given this low value, cut-off values, specificities and sensitivities were not calculated. No classification of the feline population according to the severity of caecal lesions could be established for any caecal ultrasonographic parameter.
Wall thickness medians for the previous healthy population from another paper by the same authors (2.4 mm) and the present feline population (3.3 mm) were significantly different in the proximal caecum (P = 0.005), but were not significantly different in the distal part (3.0 mm for both populations) (P = 0.39) (Figure 4). 11 Follicular layer medians could not be compared as they were not measurable in all cats in this study. Medians were not significantly different between the two populations with respect to thicknesses of ileocolic LNs (2.9 mm and 3.1 mm, respectively) (P = 0.90).

Caecal (proximal and distal) wall thicknesses in the present population (with clinical signs) and the previously described population (without clinical signs11). The horizontal black lines represent the medians of each parameter
Endoscopic findings
Adequate visualisation of the caecocolic mucosa was effectively confirmed after colonic lavage. The caecum was thus assessed for all cats and appeared hyperaemic (12/18; 10 mild, two moderate), oedematous (16/18; eight mild, three moderate, five severe), discoloured (3/18; two mild, one moderate) and/or eroded (5/18, mild), according to the WSAVA classification. One cat presented a modified caecal wall texture (mild). Cats showed a modification of one criterion (6/18), two criteria (6/18), three criteria (4/18), four criteria (1/18) or five criteria (1/18). None of the cats were considered to have a normal caecum. One cat had only one modified criterion with a score of 3/3 (oedema) (Figure 5). Taking into account all WSAVA criteria, each cat was classified according to the severity of caecal lesions on endoscopy: 7/18 (39%) cats showed mild lesions and 11/18 (61%) showed moderate-to-severe lesions (Table 2). Considering the seven cats with a mild classification, 3/7 had no visible dimpled aspect and 4/7 had a mild dimpled aspect. Among the 11 cats with a moderate-to-severe classification, a dimpled aspect was either not visible (7/11), mild (3/11) or moderate (1/11) (Figure 6). Caecal wall rigidity was assessed with the biopsy forceps and was more intense than in the colon, as described previously. 11
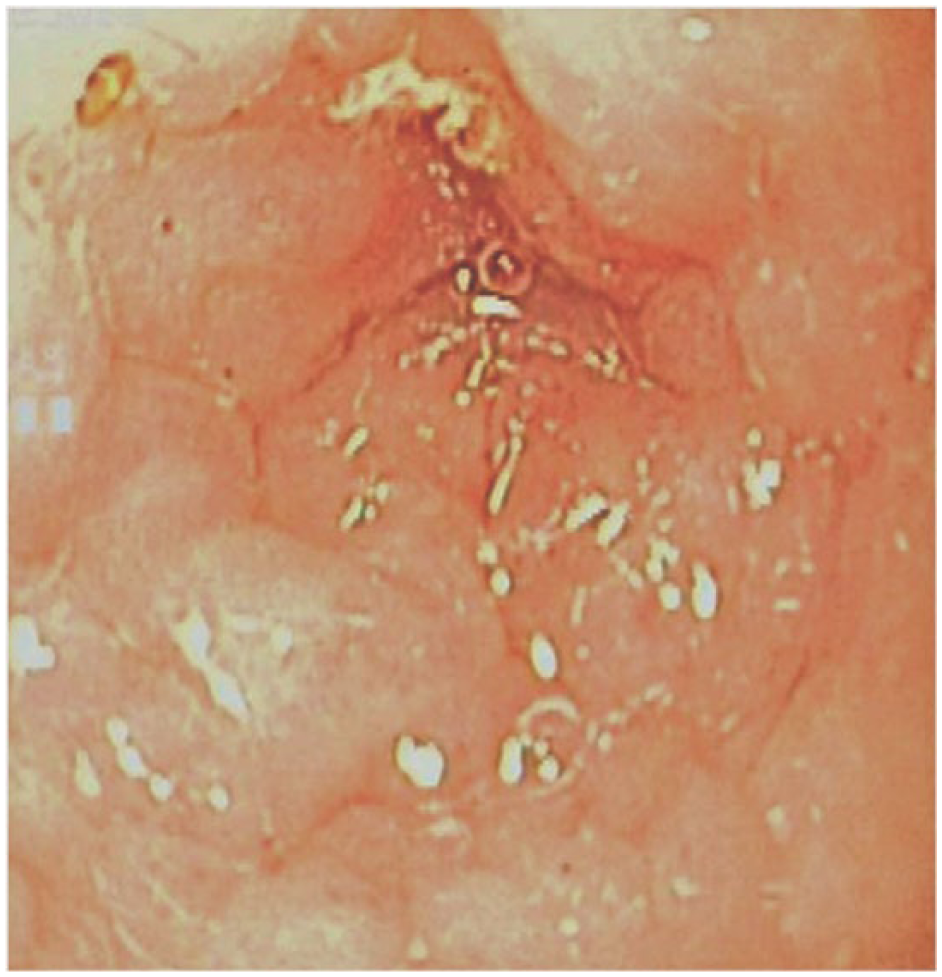
Endoscopic image of the caecum of a cat with severe oedema and a mild dimpled aspect. Histologically, it was considered as having mild typhlitis
Endoscopic and histological classifications of severity of caecal and colic lesions
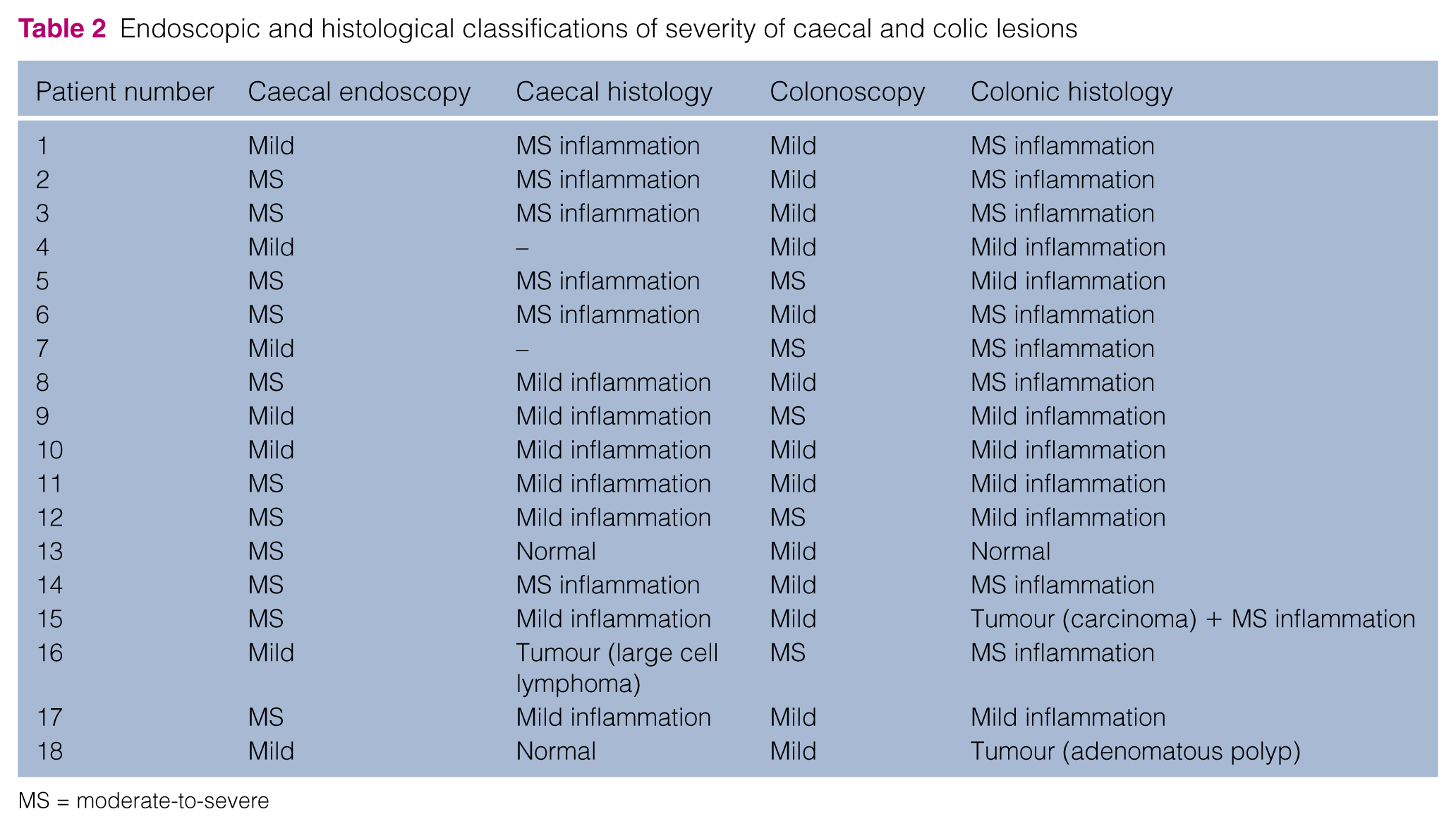
MS = moderate-to-severe

Endoscopic image of the caecum of a cat with a moderate dimpled aspect. Histologically, it was considered as normal
With respect to macroscopic colonic evaluation, 13/18 (72%) cats showed mild lesions and 5/18 (28%) showed moderate-to-severe lesions (Table 2). No foreign body, no invagination and no parasites were found in the caecum or the colon. Two cats presented with a distal colonic mass; one of them had an associated stenosis. The ilealmucosa was directly assessed in 2/18 cats and was considered normal; biopsies were obtained. In 12/18 cats, biopsies were taken through blinded catheterisation of the ileocolic junction with the endoscopic forceps (Figure 7). In the other four cats, the angulation between the ascending and transverse colon was too marked to allow ileocolic junction catheterisation; no ileal sample could then be obtained, despite repositioning of the cat during the procedure.

Catheterisation of the ileocolic junction with the endoscopic forceps in order to obtain a distal ileal biopsy
Feline coronavirus analysis
RT-PCR for feline coronavirus was performed in 14 colonic biopsies (biopsies were not available for analysis in four cats). Feline coronavirus RNA was isolated in 5/14 cats.
Histological evaluation
For each cat, a set of biopsies (between two and five, except two cases with only one biopsy) was obtained from each location (caecum, colon, and when possible, ileum). For 2/18 cats, caecal biopsies were performed but could not be processed owing to technical problems and only ileal and colonic biopsies were analysed. For 4/14 cats, ileal biopsies could not be processed owing to technical problems or inadequate quality. All the other biopsies were of adequate quality for evaluation (ie, at least one biopsy per set of biopsies, for a given location and cat, was adequate).
Caecal biopsies were evaluated in 16/18 cats. Most cats presented with an inflammatory caecum; ie, typhlitis (13/16). Among them 11/13 had lymphoplasmacytic typhlitis (often with neutrophilic and/or eosinophilic components; Figure 8) and 2/13 had neutrophilic typhlitis (Figure 9); 7/13 had mild typhlitis and 6/13 had moderate-to-severe typhlitis. One cat had a caecal lymphoma. The two other cats (2/16) presented with normal caecal biopsies (Table 2).

Photomicrograph of caecal mucosa with moderate lymphoplasmacytic inflammation, at high magnification. Lymphocytes (arrowheads) and plasma cells (arrows) fill the lamina propria between the glands. Haematoxylin–eosin–saffron (scale bar = 20 µm)

Photomicrograph of caecal mucosa with mild neutrophilic inflammation, at high magnification. Note the mild increase in neutrophils (arrows) in the lamina propria. Lymphocytes and plasma cells do not exceed normal numbers. Haematoxylin–eosin–saffron (scale bar = 20 µm)
Concerning colonic biopsies (available for all 18 cats), one cat had both a colonic carcinoma and lymphoplasmacytic colitis. The other cats had lymphoplasmacytic colitis (14/18, often with a neutrophilic component), neutrophilic colitis (1/18), an adenomatous colonic polyp (1/18) or a normal colon (1/18). In animals with colitis (including the one with colonic carcinoma), seven animals showed mild colitis and nine showed moderate-to-severe colitis (Table 2). All cats with typhlitis also had colitis (13 cats); the severity of inflammation (normal, mild, or moderate-to-severe) was identical in colonic and caecal samples in the majority of them (11/13). The cat with caecal lymphoma had colitis. The two cats with a normal caecum had either a normal colon (1/2) or a colonic polyp (1/2). The cat with both a colonic carcinoma and lymphoplasmacytic colitis presented typhlitis.
Ileal biopsy specimens were evaluated in 10/18 cats: seven were classified as normal, two as inflammatory (lymphoplasmacytic inflammation; mild in one cat and moderate in the other) and one showed a high-grade large cell lymphoma (as in the caecal biopsies).
Agreement between endoscopic, coronavirus and histological results
Regarding the caecum, κ coefficients did not exceed 0.25 (Table 3). In particular, there was a slight agreement between caecal histology and endoscopy. Considering the colonic results, the agreements between histological, endoscopic results and coronavirus state were also unsatisfactory (Table 3).
Kappa (κ) values (95% confidence intervals [CIs]) between endoscopic, histological and virological results in the caecum and colon

Discussion
In this study we prospectively compared ultrasonographic, endoscopic, virological and histological findings in 18 cats presented with clinical signs of caecocolic disease. All cats but one had at least one abnormal ICC ultrasonographic parameter. Loss of caecal wall layering on ultrasonography did not allow discrimination of histological lesion severity but seems, nonetheless, to be a marker of mild-to-severe histological caecal inflammation. On endoscopy, all cats presented with a macroscopically abnormal caecum. The accentuation of the dimpled aspect tended to be inversely related to the severity of macroscopic endoscopic lesions; evaluation of the caecal dimpled aspect could be used by internists as an extra caecal scoring parameter. On histology, cats with adequate samples had mild-to-severe typhlitis and one had a large cell lymphoma; however, two were normal regarding the caecum. The correlation between endoscopy and histology was unsatisfactory regarding the severity of caecal and colic lesions; thus, endoscopy does not seem reliable as the sole means of assessing the severity of caecocolic lesions with the criteria set in this study. However, an abnormal caecum on ultrasonography or endoscopy in the context of caecocolic clinical signs should encourage histological biopsies to be performed. Clinical signs were predictive of histological abnormal findings on ICC biopsies.
ICC disorders can affect all age categories of cats, as was the case in our study where cats, presented with ICC disease with confirmed inflammation on histopathology after exclusion of parasites, ranged in age from a few months to senior age group.3,5 The domestic shorthair breed was dominant in our feline population, probably as it is the most common breed presented for consultation in our facility, and thus may not represent a predisposition but rather breed popularity. The disease was chronic in all cats; haematochezia was the most frequent clinical sign in our population, compatible both with a caecal or colonic origin. One cat received low doses of prednisolone in the weeks before presentation. This was interrupted before the study examinations were performed. Furthermore, to our knowledge, this low dose of corticosteroids should not have modified ultrasonographic or macroscopic aspects, nor histological samples.
Ultrasonographically, caecal wall thicknesses were rarely increased in our population of cats (2/18 for the proximal part, none for the distal part). The normal upper caecal wall thicknesses considered in this paper may have been too high; however, they corresponded to the highest values in the healthy population with no histological inflammation from a previous study by the same authors, which seemed statistically appropriate. 11 Thus, there seems to be considerable overlap in caecal wall thicknesses between healthy cats and cats with typhlitis (Figure 4). Ultrasonographic results were compared with those of the above-mentioned healthy population. 11 In terms of caecal wall thicknesses, medians between the two populations were significantly different for the proximal caecal wall but were not significantly different in the distal part. Indeed, 67% cats in the actual population had a proximal wall thicker than the distal one; this was never found in the previous feline population. This finding is quite surprising, as lymphocenters are most numerous in the distal caecum, as previously established.11,21,22 Thus, we expected the distal caecal region to be the most thickened in cases of inflammation. The reason for this actual finding is unknown but may be related to the difficulty in performing reliable and repeatable measurements in the distal part, owing in part to the variable degree of caecal filling. However, this finding suggests that the ultrasonographic follow-up of the proximal caecal wall thickness is the most relevant in a cat; for example, if a patient has been scanned previously before the onset of caecocolic clinical signs, the follow-up of determining the proximal caecal wall thickness seems most appropriate. Thus, the proximal caecal wall thickness should always be measured on routine feline abdominal ultrasonography.
As 39% of cats had no visible caecal wall layering, comparison of follicular layer thicknesses was not possible between the two populations. Loss of wall layering on ultrasonography is most often associated with neoplasia in the digestive tract.23,24 In our feline population, of the seven cats with loss of caecal wall layering, three were classified by histology as having moderate-to- severe inflammation; the other two had mild inflammation (two had no histology available). None had neoplasia. Surprisingly, in the cat with caecal lymphoma, layering was preserved. Thus, it seems that loss of wall layering in the feline caecum may not be a reliable predictor of severity of inflammation or malignancy. Nonetheless, this finding seems an important feature of mild-to-severe inflammation, as no cat from the previous feline population without clinical signs showed loss of layering on ultrasonography.
Medians were not significantly different either between the two populations regarding thicknesses of ileocolic LNs. Only 3/18 cats had ileocolic LNs above the highest normal value defined in this paper (1/3 with severe ICC inflammation, 1/3 with caecal lymphoma and severe colic inflammation, and 1/3 with mild ICC inflammation and ileal thickening but no biopsy): it thus appears that LN size is not a reliable indicator of caecal disease and that normal ileocolic LNs should not be the basis of exclusion of severe typhlitis/colitis or even neoplasia (the cat with colic carcinoma had normal ileocolic LNs). Both cats with caecal or colic tumours (lymphoma and carcinoma) had round and hypoechoic ileocolic LNs, which agrees with previous descriptions of metastatic LNs.25,26 One other cat with severe colitis and typhlitis also had rounded and hypoechoic LNs. This finding thus seems non-specific. The cat with lymphoma, however, had the largest LNs of all cats (8 mm).
Regarding pericaecal mesenteric fat inflammation, only 56% of the present population had some degree of hyperechogenicity: among the cats with grade 2 steatitis, 1/3 had moderate-to-severe inflammation, 1/3 had mild inflammation and 1/3 had no histology. Among cats with grade 1 steatitis, 3/7 had severe caecal inflammation, 3/7 had mild inflammation and 1/7 had normal histology. Furthermore, in the previous paper, 7/20 cats without clinical signs had grade 1 steatitis and the statistical correlation between the presence of steatitis and histology was very poor. 11 It thus seems that pericaecal mesenteric fat hyperechogenicity (ie, steatitis) is not a good indicator of caecal inflammation or its severity.
Overall, between the present population and the healthy cats of the previous publication, raw data were not very different for the different ultrasonographic ileocaecal parameters, except for the proximal caecal wall. 11 It thus seems that ultrasonographic evaluation of the ICC region may be of limited value when used solely to diagnose inflammation (or neoplasia), and that a normal caecum and ICC region on ultrasonography in a cat with colic signs should not keep it from having endoscopy and biopsies performed. Nonetheless, ultrasonography remains an essential preliminary examination before endoscopy in order to evaluate the rest of the abdomen and to explore potential concomitant diseases that cannot be evaluated by endoscopy, including the size and aspect of loco-regional LNs. Moreover, fine-needle aspirates of the latter can be performed if necessary, and a diagnosis may be achieved without needing general anaesthesia and endoscopy.
No classification in relation to the severity of caecal lesions could be established for any caecal ultrasonographic parameter. The AUROC curve values were unsatisfactory for all ultrasonographic parameters. Indeed, the ‘best’ cut-off, ie, the caecal wall thickness in the distal part, had an AUROC curve value of 0.81; the others were inferior to this value. The unsatisfactory AUROC curve values obtained for the different ultrasonographic parameters have three possible explanations: (1) the parameters were not appropriately measured; (2) they simply do not allow discrimination regarding severity of lesions; or (3) the small sample size led to non-accurate AUROC curve values. As all measures were performed by a board-certified radiologist in a standardised way, the first proposition is quite unlikely.
On endoscopy, all cats showed modification of at least one WSAVA criterion during the macroscopic caecal evaluation. The main modifications were oedema (89%) and hyperaemia (66%). Severe alterations of the caecal mucosa, as erosions, were only present in 5/18 (28%) cats; histology revealed mild (2/5) or moderate-to-severe (3/5) inflammation in these cats. The dimpled aspect was absent (10/18), mild (7/18) or moderate (1/18) in the present population, whereas it was present in all and more marked in the previous population of healthy cats. 11 Moreover, the accentuation of the dimpled aspect tended to be inversely related to the severity of macroscopic endoscopic lesions. We hypothesise that oedema, observed in many cases of typhlitis, could have attenuated the small depressions of the caecal mucosa. Thus, an absence of caecal dimpled aspect on endoscopy could be a sign of typhlitis and should lead the endoscopist to look thoroughly for other signs of inflammation and to perform caecal biopsies. We suggest that evaluation of the caecal dimpled aspect should be used by internists as an extra caecal scoring parameter. Despite modifications on endoscopy, two cats showed normal caecal biopsies. We hypothesise that an inflammatory site could have been missed while taking mucosal biopsies during endoscopic examination (although one had an adenomatous colic polyp, possibly explaining the clinical signs). Also, the discrepancy between endoscopic and histological evaluations may have been due to our endoscopic criteria, determined by the WSAVA. 16 Endoscopic evaluation includes assessment of hyperaemia and oedema, while these two criteria are not taken into account for standard histological evaluation of endoscopic biopsies. 10 Thus, in cases mainly affected by oedema and hyperaemia, the severity of lesions may have been overestimated by endoscopy, in comparison with histology. It appears that endoscopy should not be used as the only mean of assessing the severity of caecocolic lesion. Endoscopic evaluation of the ileal mucosa can be challenging in cats as the distal ileum may be difficult or impossible to reach, even by blind catheterisation with endoscopic forceps by experienced operators. The ileum is a frequent digestive disease location in cats; thus, not being able to perform ileal biopsies in all cats in this study is an important limitation.
Histologically, the majority of caecal and colonic samples displayed lymphoplasmacytic inflammation, consistent with a chronic disease. In a few cases (three cats), the inflammation was only neutrophilic, suggesting a more acute process, despite all cats showing chronic signs; however, in each case, if the caecum showed a neutrophilic inflammation, the colon conversely showed a lymphoplasmacytic inflammation indicating in all cases a chronic disease. These findings suggest that the caecum and the colon may experience asynchronous inflammation. We could speculate that the chronic inflammation may spread progressively from one large intestine segment to the other, with neutrophilic infiltrate being replaced (or enriched) by lymphocytes and plasma cells. Except for one cat previously mentioned with clinical signs but normal ICC biopsies, cats with clinical signs of caecocolic disease always presented histological lesions on caecal or colonic biopsies. Clinical signs were thus predictive of histologically abnormal findings.
As feline coronavirus may occasionally induce ICC lesions, the presence of the viral genome was systematically searched for on colonic biopsies by RT-PCR. 27 Five of 14 cats had positive results; four of them displayed severe histological inflammation. However, histological findings did not support a causal link with inflammation as no pyogranulomas were found; thus, infectious peritonitis in those cats was unlikely. Moreover, only a fair statistical agreement was observed between coronavirus status and histological lesions.
This study has some limitations. Histological results on endoscopic biopsies were considered as the gold standard in this study. However, endoscopic biopsies are not fully representative of the bowel and have been judged as inadequate for differentiating lymphoma from inflammatory bowel disease in cats. 28 Focal lesions may have been missed, as these biopsies do not sample the whole surface of the intestine. Moreover, deep lesions could easily have been missed because endoscopic biopsies are limited to the mucosa. For example, three cats displayed ileal wall thickening on ultrasonography involving mainly the muscular layer, but no histological lesion on ileal biopsies: this discrepancy could result from the absence of the muscular layer on the biopsies. However, endoscopic sampling was elected by the authors as laparotomy is a more invasive and expensive procedure; endoscopy requires shorter anaesthesia time, permits macroscopic exploration of the caecocolic mucosa, leads to faster recovery and, moreover, there is no alternative sampling technique for the colon. 27 Another limitation to this study is the relatively low number of animals recruited, which should lead to a cautious use and application of the conclusions in this study. Only a small number of cats with caecocolic neoplasia were included. This is an infrequent disease in cats. We included only patients undergoing both ultrasonography and endoscopy. This may have biased the population sample as patients presenting with a caecal mass or lesions with severe ileocolic lymphadenopathy on ultrasonography were not always sent to endoscopy; instead they underwent ultrasound-guided fine-needle aspiration for cytological analysis, to avoid anaesthesia and reach a faster diagnosis. Further studies that include more neoplastic cases need to be performed, in order to better define the discriminating power of ultrasonography in the diagnosis of caecal disease.
Conclusions
Clinical signs of caecocolic disease are predictive of abnormal findings on endoscopy and inflammatory or tumoral lesions on histology of ICC biopsies. Ultraso-nographic and endoscopic evaluations did not permit accurate prediction of the severity of lesions, when considering histology on endoscopic biopsies as the gold standard. Thus, the severity of typhlitis cannot be appropriately judged macroscopically and histological biopsies need to be performed. Unexpectedly, loss of wall layering on ultrasonography did not necessarily imply neoplasia but did seem to imply some degree of typhlitis. Local steatitis did not reflect caecal inflammation severity or the presence of neoplasia. However, loss or severe attenuation of the macroscopic caecal dimpled aspect may suggest inflammation or tumoral lesions. These findings in 18 animals suggest that histological biopsies remain mandatory to evaluate ICC lesions in cats.
Footnotes
Conflicts of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
