Abstract
Objectives
Congestive heart failure secondary to cardiomyopathy is a common manifestation of cardiac disease in cats, carrying a variable prognosis. The objective of this retrospective study was to evaluate the relationship between red blood cell distribution width (RDW) and survival time in feline patients with acquired heart disease with and without congestive heart failure (CHF).
Methods
Three hundred and forty-nine client-owned cats with echocardiograms and complete blood count, including RDW measurement, performed between March 2006 and December 2011, were included in the study. Patient characteristics, including signalment, hematocrit, RDW, echocardiographic parameters and survival, were recorded. Comparisons between RDW in cats with asymptomatic acquired heart disease and those with CHF were made. Survival was documented and compared at 30 days and 6 months.
Results
CHF was present in 80 cats and absent in 269 cats. Cats with CHF had an increase in mortality compared with cats without CHF at 30 days and 6 months (P = 0.007 and P = 0.04, respectively). RDW was not significantly associated with survival in cats with or without CHF at 30 days or 6 months. A significant difference was found between median RDW values in cats with CHF vs cats without CHF (16.3% vs 15.8%; P = 0.02). The median RDW value was significantly higher in cats with unclassified cardiomyopathy compared with cats with other types of cardiomyopathy (16.3% vs 15.8%; P = 0.03).
Conclusions and relevance
Single RDW values did not predict mortality in cats with acquired heart disease but may be useful in determining if cats have decompensated heart disease and CHF. Human studies indicate that incremental increases in serial RDW measurements are associated with decreased survival; serial RDW measurements in cats may be an area of future study.
Introduction
The most common form of cardiovascular disease in cats is hypertrophic cardiomyopathy (HCM), a condition characterized by concentric left ventricular hypertrophy, impaired myocardial relaxation and diastolic dysfunction. Other acquired cardiomyopathies reported in cats include restrictive cardiomyopathy (RCM), unclassified cardiomyopathy (UCM), dilated cardiomyopathy (DCM), and arrhythmogenic right ventricular cardiomyopathy (ARVC). All have the potential to decompensate to congestive heart failure (CHF).1,2 Decompensation may involve activation of the renin–angiotensin–aldosterone system, activation of the inflammatory cascade and decline in cardiac function with cardiac remodeling. 3
An owner’s decision of whether or not to pursue treatment for CHF is the result of multiple factors, with prognosis and survival time likely playing an important role in the decision-making process. Reported survival times for cats with acquired cardiomyopathy range dramatically from 2–4418 days.4,5 Survival times in cats from the onset of CHF can range from days to a few years; however, typically, median survival time is 6–12 months.4–7 Established parameters that correlate with survival in CHF cats include weight, age, left atrial size and function, extreme left ventricular (LV) hypertrophy, LV systolic function, presence of systolic anterior motion and body temperature at the time of hospital admission.4–6,8 Recently, cardiac troponin I has been shown to be correlated with time to cardiac death;9,10 however, reference intervals (RIs) and validation can vary greatly between analyzers. Despite these findings, veterinary medicine has yet to discover an accurate prognostic indicator for cats with acquired cardiovascular disease and CHF. While N-terminal prohormone brain natriuretic peptide levels have been evaluated to assess for cause of dyspnea, they have not been used as prognostic indicators in cats with CHF. Determining an inexpensive and easy method to predict survival for cats presenting with CHF would be beneficial, and may assist an owner’s decision to go with medical management vs euthanasia.
Prediction of prognosis and survival times in human CHF is largely based on clinical risk scores, clinicopathologic abnormalities and biomarkers.11–19 Common parameters used in prediction models include age, systolic blood pressure, ejection fraction, presence of renal insufficiency, anemia, plasma sodium concentration, brain natriuretic peptide, atrial natriuretic peptide and troponin levels.12,13,15,18,19 While these models are helpful for stratification of patient populations, the calculations can be cumbersome, difficult to perform owing to the large number of data points required, and do not directly predict individual outcome. 17 Recently, red blood cell (RBC) distribution width (RDW) has shown promise as a prognostic indicator in people with both acute and chronic heart failure, with an increasing RDW value correlating to a decreasing survival time.20–25 RDW is appealing as a prognostic indicator because it is readily available on an automated complete blood count (CBC) analyzer and is easy to obtain.
To our knowledge, RDW has not been investigated in cats as a prognostic indicator in any disease process. The aim of this study was to determine whether a correlation between RDW (independent of hematocrit [Hct]) and length of survival exists in cats with CHF. We hypothesized that an increasing RDW would correlate with a decreased survival time in cats presenting with CHF secondary to acquired cardiac disease.
Materials and methods
Case selection
The electronic medical records system at a private specialty hospital was searched for all cats that had echocardiograms (ECHOs) performed between March 2006 and December 2011. Included in the study were cats diagnosed with acquired cardiac disease that had CBCs performed within 3 days of their ECHOs (CBCs were run in-house by the Clinical Pathology Department on an Advia 120 hematology machine [Siemens]). Cats were excluded from the study if they were diagnosed with congenital heart disease, were anemic (Hct <30%) or if CBCs were not completed within the allotted time frame.
Data collected from the medical records included signalment, medical history, physical examination results, thoracic radiographic results (when available), Hct (RI 30–48.5%), RDW (RI 13.8–17.9%), total survival time in months following initial ECHO and whether the cat was alive at 30 days and 6 months following initial ECHO. Thirty day and 6 month survival times were selected based on the recheck schedules established by the Cardiology Department for cats with CHF and asymptomatic heart disease, respectively. If cats did not return for a follow-up visit then survival data were obtained from clinical records, or a telephone call to the owner or referring veterinarian within a 3 month period of time.
Echocardiography
ECHOs were performed at the discretion of the primary clinician, based on physical examination findings and patient history. ECHOs were completed or supervised by two board-certified cardiologists, and were performed with chemical restraint as needed based on cat temperament. Standard transthoracic ECHO was performed using a sonograph with a 12.0 MHz transducer, as previously described elsewhere. 26 Data collected included left atrial diameter, aortic root diameter, left atrium to aortic root ratio (LA:Ao) and ECHO diagnoses. Cats with no abnormal ECHO findings were diagnosed as normal. Cats were otherwise diagnosed as HCM, hypertrophic obstructive cardiomyopathy (HOCM), UCM, four-chamber dilation, DCM, RCM, ARVC and endomyocardial disease, according to previously published guidelines.1,2,27–33
Cats were divided into two groups based on the absence or presence of CHF. CHF was diagnosed by identification of pulmonary edema and/or pleural effusion on thoracic radiographs, by identification of pleural, abdominal or pericardial effusion on ECHO, or by a combination of both. Cardiogenic edema and effusions were confirmed based on cat medical history and concurrent diagnostics. Cats with diagnosis of CHF served as the study group, while cats without CHF were used as the control group.
Statistics
Differences in RDW between various groupings, including those with and without CHF, and type of structural heart disease were tested using the Wilcoxon rank-sum test. The χ2 test was used to compare 30 day and 6 month mortality in cats with and without CHF. Logistic regression was used to evaluate the association between RDW tertiles and LA:Ao tertiles with survival at 30 days and 6 months. As the assumption of linearity between RDW and LA:Ao and mortality was not met, both RDW and LA:Ao were modeled as tertiles. A multivariable regression analysis was performed to examine the association of RDW and CHF, both defined a priori, as well as potential confounders that were significantly associated with mortality in univariate regression: LA:Ao and LV systolic function with 30 day and 6 month mortality. These models were checked for multicollinearity. A statistical analysis software program was used for all analyses (SAS version 9.2). P ⩽0.05 was considered statistically significant.
Results
Seven hundred and twenty-six cats had ECHOs performed during the time period evaluated. Three hundred and forty-nine cats met the inclusion criteria for the study, with 80 of these cats having a diagnosis of CHF. Figure 1 shows a flow chart of the study design and cat population.

Flow chart depicting the study design. Study population of 80 cats with echocardiograms, red blood cell distribution width (RDW) data available within the 3 day time frame, and presence of congestive heart failure (CHF) during the study period
Domestic shorthair cats were the most common breed overall, including both the study and control populations (207/349). Population characteristics of the study and control population are presented in Table 1.
Study and control population characteristics

ECHO = echocardiography; HCM = hypertrophic cardiomyopathy; UCM = unclassified cardiomyopathy; RCM = restrictive cardiomyopathy; HOCM = hypertrophic obstructive cardiomyopathy; DCM = dilated cardiomyopathy; ARVC = arrhythmogenic right ventricular cardiomyopathy
RDW tertiles were defined as low (13.3–15.4), medium (15.5–16.5) and high (16.6–23.4). The median RDW was 16.3% (range 13.6%–19.6%) in the study group and 15.8% (range 13.3%–23.4%) in the control group. A statistically significant difference was found between the two groups (P = 0.02). No statistically significant association was found between RDW tertiles and 30 day or 6 month mortality in the entire cohort or in the study or control groups (Table 2 and Figures 2 and 3). The percentage of deceased cats at 30 days (Figure 2) and 6 months (Figure 3) for the study and control populations were evaluated for the low, medium and high RDW tertile, and no statistically significant correlation was appreciated between RDW and survival at 30 days or 6 months.
Comparison of red blood cell distribution width (RDW) values (expressed in low, medium or high tertiles) with 30 day and 6 month mortality in cats with and without a diagnosis of congestive heart failure (CHF)
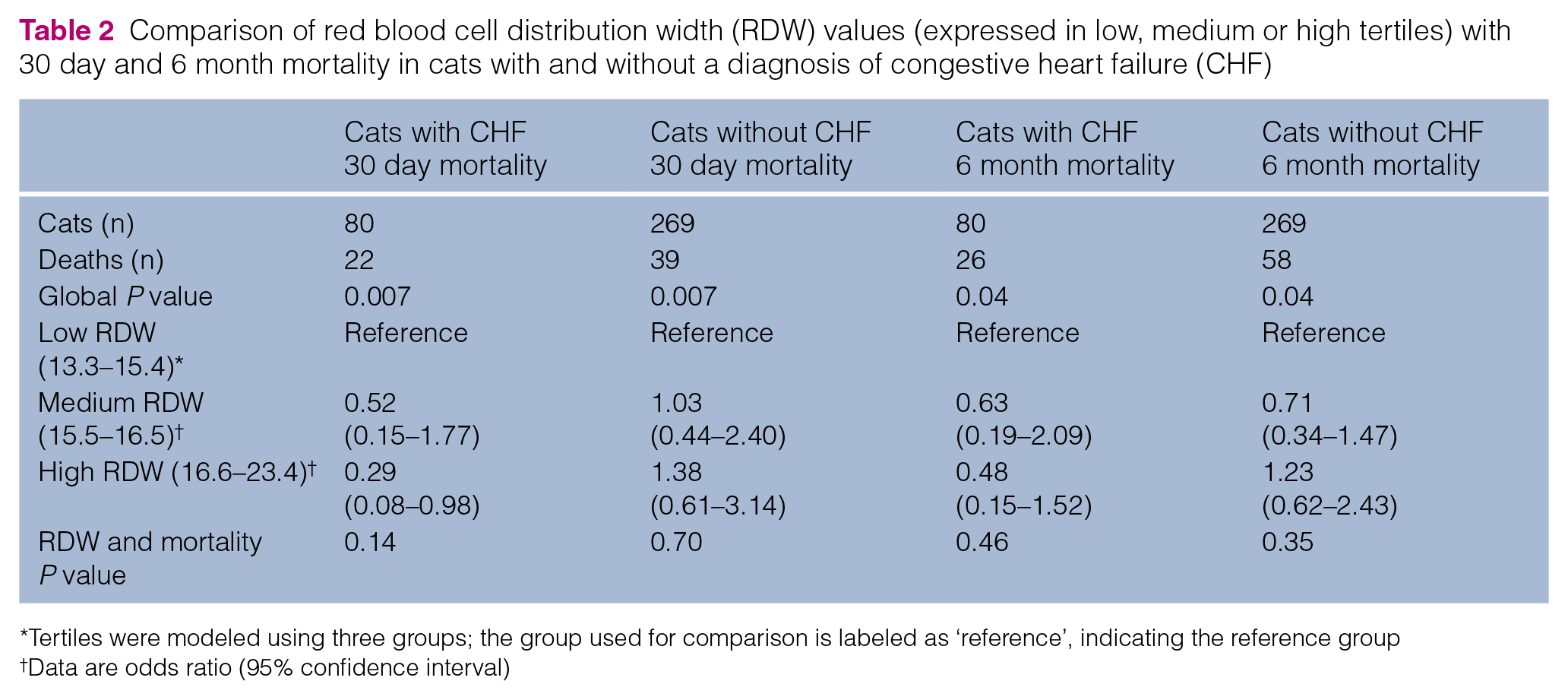
Tertiles were modeled using three groups; the group used for comparison is labeled as ‘reference’, indicating the reference group
Data are odds ratio (95% confidence interval)

Comparison of red blood cell distribution width (RDW) tertiles and mortality at 30 days

Comparison of red blood cell distribution width (RDW) tertiles and mortality at 6 months
Cats with a diagnosis of CHF had an increase in mortality at 30 days and 6 months in comparison with cats without a diagnosis of CHF (P = 0.007 and P = 0.04, respectively). The percentage of deceased cats with CHF vs absence of CHF was 27.5% vs 14.50% at 30 days. The percentage of deceased cats with and without CHF at 6 months was 32.5% and 21.6%, respectively. From 30 days to 6 months, there were only an additional four deaths. The correlation between RDW value and presence of CHF for cats that were alive or dead at 30 days and 6 months is given in Table 3.
Comparison of red blood cell distribution width (RDW) values, presence of congestive heart failure (CHF), and mortality at 30 days and 6 months

The cardiac diagnosis or type of cardiomyopathy (HCM, HOCM, UCM, DCM, RCM, ARVC, four-chamber dilation, endomyocardial disease) was associated with survival at 30 days and 6 months (P = 0.03 and P = 0.004, respectively) as shown in Table 4. This was irrespective of CHF diagnosis. Cats with HCM, HOCM and without structural heart disease had a greater percentage of survival at 6 months vs cats with DCM and UCM.
Comparison of red blood cell distribution width (RDW) values (%) with echocardiographic (ECHO) diagnoses vs cats without a particular ECHO diagnosis
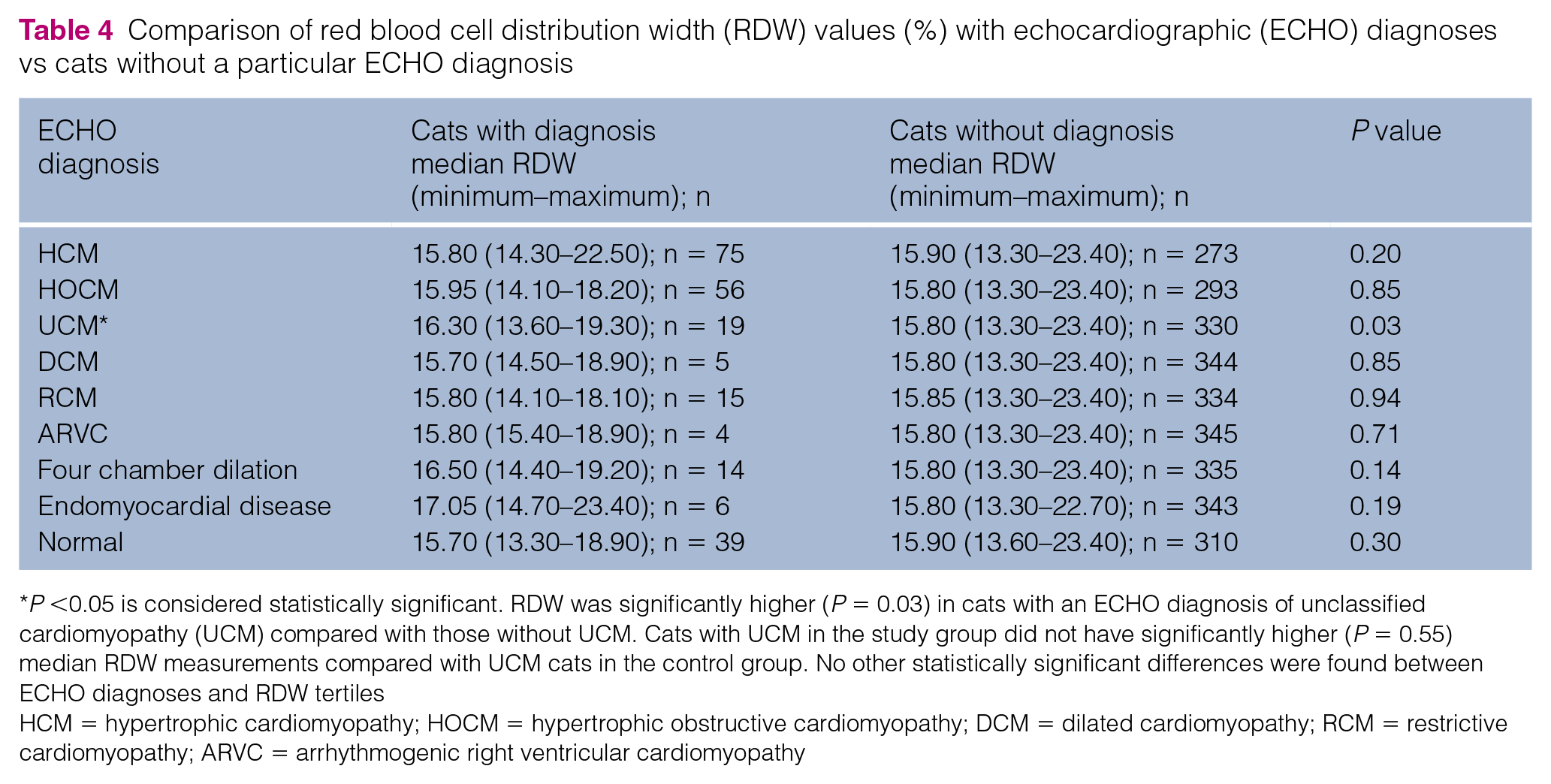
P <0.05 is considered statistically significant. RDW was significantly higher (P = 0.03) in cats with an ECHO diagnosis of unclassified cardiomyopathy (UCM) compared with those without UCM. Cats with UCM in the study group did not have significantly higher (P = 0.55) median RDW measurements compared with UCM cats in the control group. No other statistically significant differences were found between ECHO diagnoses and RDW tertiles
HCM = hypertrophic cardiomyopathy; HOCM = hypertrophic obstructive cardiomyopathy; DCM = dilated cardiomyopathy; RCM = restrictive cardiomyopathy; ARVC = arrhythmogenic right ventricular cardiomyopathy
Nineteen cats were diagnosed with UCM, 10 in the study group and 9 in the control group. When RDW was compared between cats with various ECHO diagnoses, median RDW was significantly higher (P = 0.03) in cats with an ECHO diagnosis of UCM compared with those without UCM (16.3% vs 15.8%). Cats with UCM in the study group did not have significantly higher (P = 0.55) median RDW measurements (16.7%; range 13.6%–19.3%) compared with UCM cats in the control group (16.3%; range 15.1%–18.1%). No other statistically significant differences were found between ECHO diagnoses and RDW (Table 4).
LA:Ao was correlated with survival time. LA:Ao was divided into tertiles, group 0: 0.53–1.20; group 1: 1.21–1.46; group 2: 1.47–2.84. Group 2, with the largest LA:Ao, was associated with an increase in mortality at 30 days (P = 0.04). Age, systolic anterior motion and extreme LV hypertrophy were not found, in univariate analysis, to be significantly associated with either 30 day or 6 month mortality. LA:Ao and low LV systolic function FS <30% were found to be significantly associated with mortality in univariate regression. RDW, LV systolic function, CHF and LA:Ao were entered into multivariable logistic regression models, separately for 30 day and 6 month mortality. Only LV systolic function FS <30% was associated with increased mortality at 30 days and 6 months (P <0.05, data not shown).
Discussion
RDW has traditionally been used to assist clinicians in determining causes of anemia by providing a quantitative measurement of the variability in size of circulating erythrocytes, with larger RDW values indicating greater diversity in RBC size.34–36 Classic etiologies in dogs, cats and people for increased RDW measurements include conditions associated with ineffective erythropoiesis such as iron deficiency, anemia of chronic inflammatory disease, vitamin B12 or folate deficiency, and hemoglobinopathies.24,34–36 More recently, RDW has been used to predict the outcome in a variety of disease states in people, and has shown promising results as a prognostic indicator in people with congestive and chronic heart failure, acute myocardial infarction and coronary artery disease.11,13,18–25,37–39 Arbel et al reported that the occurrence of a major cardiovascular event increased by 10–15% for every 1% increase in RDW values. 37
The underlying mechanism and pathologic basis of increased RDW values identified in people with cardiovascular disease is unknown. A popular theory suggests that RDW is increased secondarily to systemic inflammation.23,35,38–41 It is hypothesized that inflammatory mediators such as interleukin (IL)-1, IL-6, tumor necrosis factor-alpha and interferon-gamma released in certain disease states cause suppression of erythropoietin-induced erythrocyte maturation, resulting in an increased release of reticulocytes into the circulation.23,35,38–41 Persistent inflammation is a principal pathophysiologic finding and poor prognostic factor for people with heart failure.14–23,35,37,41–43
In people with cardiovascular disease, increased RDW has been linked to decreased survival time.20–25,37–39 We hypothesized that this relationship would hold true in cats with CHF secondary to acquired cardiomyopathy. However, the results of our study did not support this finding and no association was found between 30 day or 6 month survival with RDW value. One reason RDW may not be a suitable prognostic indicator in cats is owing to feline-specific differences in RBC physiology compared with human RBC physiology. Feline RBCs have a shorter life span (70–80 days vs 120 days), are smaller in diameter (5–6 μm), contain a greater number of hemoglobin sulfhydryl groups (8–10 vs 2–4) and have marked rouleaux formation, with or without inflammation.34–36,44–48 The greater number of sulfhydryl groups and limited glutathione stores make feline RBCs more prone to oxidative stress and Heinz body formation, and may lead to greater variability in normal RDW values in cats vs humans.34,36,44,45,49,50
An additional difference between feline RBC physiology and people is that cats have two distinct subpopulations of reticulocytes: aggregate and punctate (mature) reticulocytes.51–55 Healthy cats also have a high number of circulating reticulocytes (2–17%),34,51,54–56 in comparison to normal human adults whose reticulocytes are 0.5–1.5% of RBCs and therefore may have a more uniform distribution of RDW.46–48
Despite the differences in feline RBC physiology compared with people, we did find an association between RDW value and the presence of CHF in cats. Cats with CHF had a significantly increased median RDW value compared with cats in the control population. This is an interesting finding and suggests that cats with CHF may have increased levels of inflammatory mediators. Additionally, it suggests that while RDW is not valuable as a prognostic indicator in cats with CHF, it may be useful when used in conjunction with other tests to support a diagnosis of CHF. However, RDW would be impractical used alone for making a diagnosis of CHF in an acutely dyspneic patient, as it has not been evaluated in cats with other causes of respiratory distress. This finding is also in contrast to a recent study performed in dogs comparing RDW values in healthy dogs, dogs with compensated chronic degenerative valvular disease and dogs with CHF. In this study, the authors found that RDW values were not associated with the presence of valvular disease or CHF. 57
Not surprisingly, cats with CHF vs cats without CHF had an increase in mortality at 30 days and 6 months. In addition, the larger LA:Ao was correlated with an increase in mortality; this makes sense as cats with larger left atriums are more likely to have CHF, which decreases survival time.
Although not the specific goal of our study, we did find an association between RDW and the presence of UCM. Cats with UCM, regardless of concurrent CHF, had increased RDW compared with cats with other ECHO findings. Cats in our study were diagnosed with UCM based on a diagnosis of exclusion. Cats that had abnormal ECHO findings that did not fit with the characteristics of one of the recognized disease conditions such as HCM, DCM, RCM or ARVC were diagnosed with UCM.1,2,31,32 Therefore, cats with UCM demonstrated a variety of ECHO abnormalities that were not consistent across the population of cats. As the mechanistic link for elevation in RDW with cardiovascular disease in people is largely theorized, and the pathogenesis of UCM in cats is unclear, we can only speculate as to the cause of the association.30–32 Perhaps there is a larger cascade of inflammatory mediators in cats with UCM vs other feline cardiomyopathies, or UCM is associated with a more chronic inflammatory disease state. Further studies may be warranted in order to determine if there is any clinical relevance between UCM, inflammation and increased RDW measurements in cats.
In a prospective multicenter cohort study of people with CHF, serial RDW values were found to predict cardiac disease progression. The authors reported that as serial RDW measurements increased, underlying cardiac disease progressed. 38 Therefore, based on these studies, serial RDW measurements throughout the course of disease may provide a more clinically relevant prognostic indicator than single RDW measurements.22,38,39 Although the single RDW measurements at 30 days or 6 months did not correlate with survival in cats with CHF, routine serial measurements of RDW may prove to be a more reliable prognostic indicator in cats.
Limitations of our study include the relatively small sample size of cats with CHF, as only 80 cats were diagnosed with CHF by a cardiologist and had available RDW data obtained within 3 days of ECHO. Cats were not considered to be in CHF unless there was a written diagnosis in the medical record, which may have erroneously excluded some cats with CHF from the study group. Variability between the ECHO skills and interpretation of those images may provide another limitation as cardiology residents or specialists may have slightly different interpretations or criteria for classification of feline cardiomyopathies. Given the retrospective nature of the study, the criteria used for diagnosis of the various cardiomyopathies are unknown. A prospective study would offer more control over the time at which the RDW measurements were taken (ie, prior to or after therapy for CHF), as well as allow for comparison of serial RDW values. It is unknown whether there is day-to-day variability in a patient’s RDW measurement, as this has not been explored in either veterinary or human medicine.
Similarly to studies conducted in people, other causes of increased RDW value, such as iron deficiency, vitamin B12 or folate deficiency, and hemoglobinopathies, were not examined in this cat population; anemia was included in the exclusion criteria. Only RDW measurements were evaluated in this study and these measurements were not interpreted in relation to the complete CBC or other laboratory data such as biochemical analysis. It is uncertain if other CBC indices such as the percentages of aggregate and punctate reticulocytes or RBC morphology would have been able to provide additional insight.
Conclusions
A quick, easy and inexpensive prognostic indicator would prove valuable for cats presenting with CHF. Unfortunately, no significant association between RDW and 30 day or 6 month survival in cats with CHF was found. A significant association between increasing RDW and the presence of CHF in cats was identified, suggesting that RDW may provide additional evidence for a diagnosis of CHF in cats with clinical signs. Based on these findings and results in people, additional studies evaluating serial RDWs in cats from time of ECHO diagnosis of acquired heart disease to CHF and death may prove beneficial.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The study was funded by a private donation from a client of the Angell Animal Medical Center in Boston, MA, USA, for resident research projects, and the Barkley Fund at Cummings School of Veterinary Medicine at Tufts University.
