Abstract
Series outline:
This is the second article in a two-part series on urinalysis in the cat. The specific focus is urine microscopic examination. Part 1, which appeared in the March 2016 issue, discussed urine macroscopic examination.
Practical relevance:
Urinalysis is an essential procedure in feline medicine but often little attention is paid to optimising the data yielded or minimising factors that can affect the results.
Clinical challenges:
For the best results, appropriately collected urine should be prepared promptly by specialist laboratory personnel for the relevant tests and assessed by a clinical pathologist. This is invariably impractical in clinical settings but careful attention can minimise artefacts and allow maximum useful information to be obtained from this seemingly simple process.
Audience:
Clinical pathologists would be familiar with the information provided in this article, but it is rarely available to general or specialist practitioners, and both groups can potentially benefit.
Equipment:
Most of the required equipment is routinely available to veterinarians. However, instructions have been provided to give practical alternatives for specialist procedures in some instances.
Evidence base:
The evidence base for feline microscopic urinalysis is quite poor and information has largely been extrapolated from the human literature. Information from feline studies has been included where available. In addition, practical clinicopathological and clinical observations are provided.
Introduction
Urine microscopic analysis is usually performed to aid in the diagnosis and monitoring of diseases of the urinary tract. Common clinical indications for this procedure in feline practice were outlined in Part 1. 1
Microscopic examination of urine involves the enumeration and identification of urine insoluble particles. The latter should be relatively straightforward for the experienced microscopist and can be performed on wet preparations, air-dried smears of urine or, ideally, a combination of the two. Enumeration of urine insoluble particles is more complex and, in veterinary medicine, this aspect of the urinalysis has often been problematic. Consequently results of urine sediment particle enumeration are poorly reproducible unless attention is directed towardsusing a standardised technique that enables a more critical assessment of the semiquantitative findings.2–5
In human medicine, urine insoluble particle enumeration has been simplified with the use of automated urine cell counters. Such machines are currently being validated for use in veterinary medicine as well.
Factors that may affect urine insoluble particle enumeration and microscopic identification are outlined in the box on page 374.
Urine Sample Collection and Preparation Techniques
Fresh urine (<60 mins post-collection) is the ideal sample for microscopic examination, as the constituents of urine are not stable. Casts and cells deteriorate rapidly, crystals may dissolve or form, and bacteria may die or proliferate. The method of urine collection will also influence the cellular composition of the sample: a voided sample may contain cells and bacteria from the genital tract; a cystocentesis sample may have increased numbers of red blood cells (RBCs); and catheterisation may result in increased numbers of urothelial cells.1,5


Traditionally, urine insoluble particle enumeration and identification in cats has been based on the microscopic examination of wet preparations comprising resuspended urine sediments following centrifugation of a consistent volume of urine (typically 3–5 ml, depending on the individual laboratory, although lesser volumes of urine may be adequate). With some modification, this technique may also be used to prepare air-dried smears for subsequent staining and cytological examination.
A standardised protocol for preparation of urine sediment samples for microscopic examination using conventional centrifugation (wet preparations or air-dried smears) is described below.3,5,6–9
Standardised protocol for preparing urine sediment samples
The microscopist’s level of expertise in examining urine wet preparations for the purposes of insoluble particle identification and enumeration will dictate whether staining will be used during this process (see box on page 375). Note, however, that semiquantitative results should always be determined using an unstained wet preparation, to avoid the dilutional effects of adding stain. Also, any crystals or microorganisms identified in a stained wet preparation should be confirmed on an unstained wet preparation. 9


Wet preparation of a urine sample. Two drops of unstained urine and a drop of urine to which a drop of new methylene blue stain has been added are placed side by side on the same glass microscope slide, with a coverslip applied to each
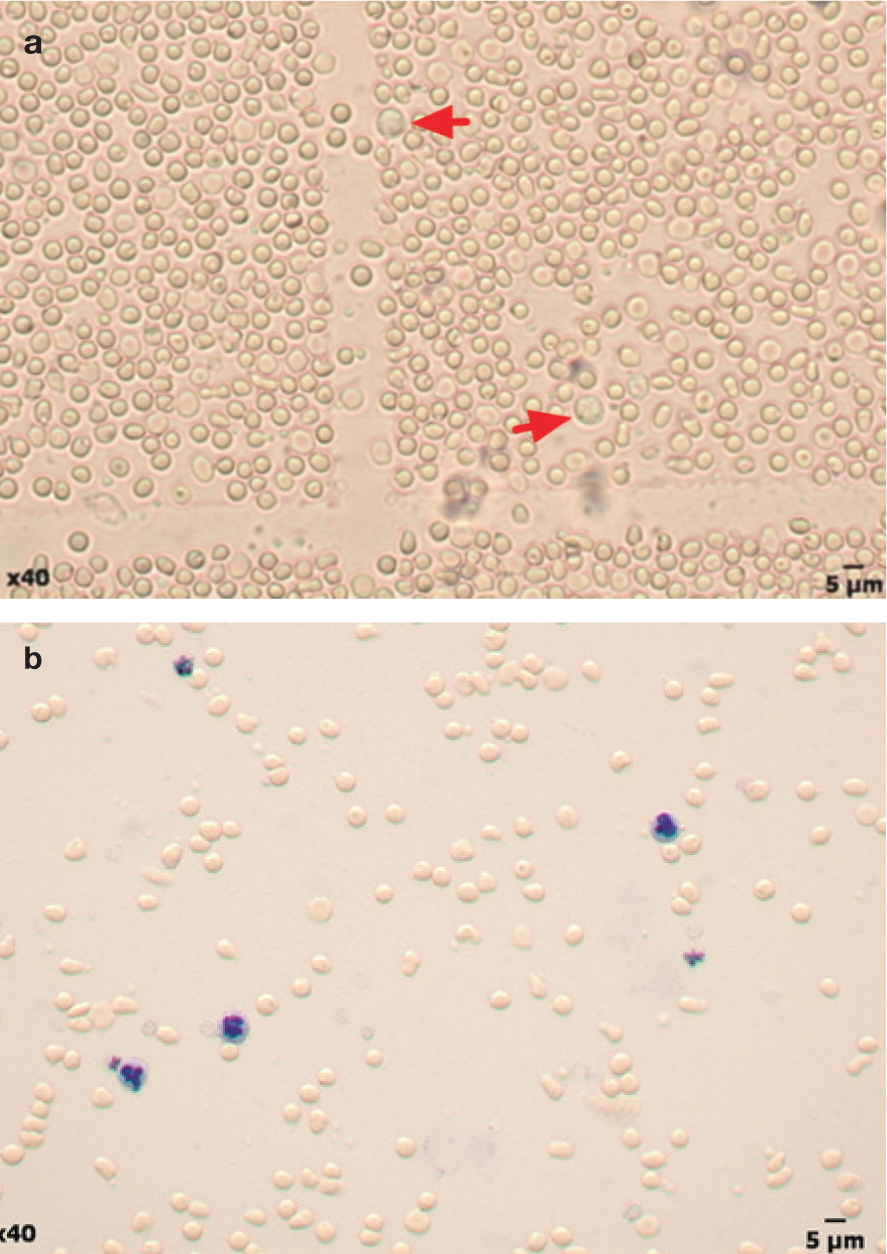
Wet-prep examination. (a) Unstained urine sample with numerous red blood cells and scant white blood cells (arrows). (b) Urine sample to which a drop of new methylene blue stain has been added, highlighting the neutrophils
Performing and Interpreting the Microscopic Analysis
Enumeration of urine insoluble particles
Insoluble particles in urine may be counted using conventional (semiquantitative) methods employed during routine wet-prep microscopic examination or using counting chamber methods.
Conventional method
The conventional method of counting urine insoluble particles in urine sediment preparations involves quantifying the number of structures seen per microscopic field (expressed as the number of insoluble particles per low power field [LPF] or high power field [HPF] when viewed under a × 10 or × 40 microscope objective, respectively).
This semiquantitative analysis may be problematic, especially as the width of the viewed microscopic ocular field varies between microscopes. Some microscopes use 25 mm ocular lenses, while older microscopes tend to have 20 mm ocular lenses. The area viewed through the former is approximately 50% larger than the area viewed through the latter. Consequently, if 15 WBCs/ HPF were seen with a 25 mm ocular lens, then only 10 WBCs/HPF would be seen through a 20 mm ocular lens on the same sample. 5
A coverslip is essential, particularly when examining a specimen under a × 40 microscope objective; not only to provide optimal microscopic resolution, but to avoid inadvertently placing the tip of the × 40 objective nosepiece in the specimen. The visibility of urine insoluble particles can be improved by lowering the microscope condenser or closing the substage iris diaphragm to increase the contrast. Focusing up and down on the slide, while systematically scanning it, enhances the examination of cellular detail. At low power magnification (× 10), the edges of the coverslip need to be checked for casts (and other heavier urine insoluble particles) that may have floated to the periphery of the field. At high power magnification (× 40), RBCs, WBCs and epithelial cells need to be counted, in addition to identifying the type of casts, small crystals, bacteria, sperm, lipid droplets, yeasts, parasitic ova and other insoluble particles.3,8
With the conventional method, the centrifugation step and removal of supernatant are important for concentrating urine insoluble particles in the specimen being examined, but are also a major source of error, especially when urine volumes available for testing vary. Consequently there is no precise reproducible information regarding the upper limit of normal formany insoluble particles found in urine sediment using conventional counting techniques. Typical semiquantitative results (based on centrifugation of 5 ml aliquots of urine, with a drop of resuspended urine sediment solution placed on a clean glass slide, and a coverslip applied) are listed in Table 1. 2
Microscopic urine sediment examination findings in cats utilising conventional measuring techniques
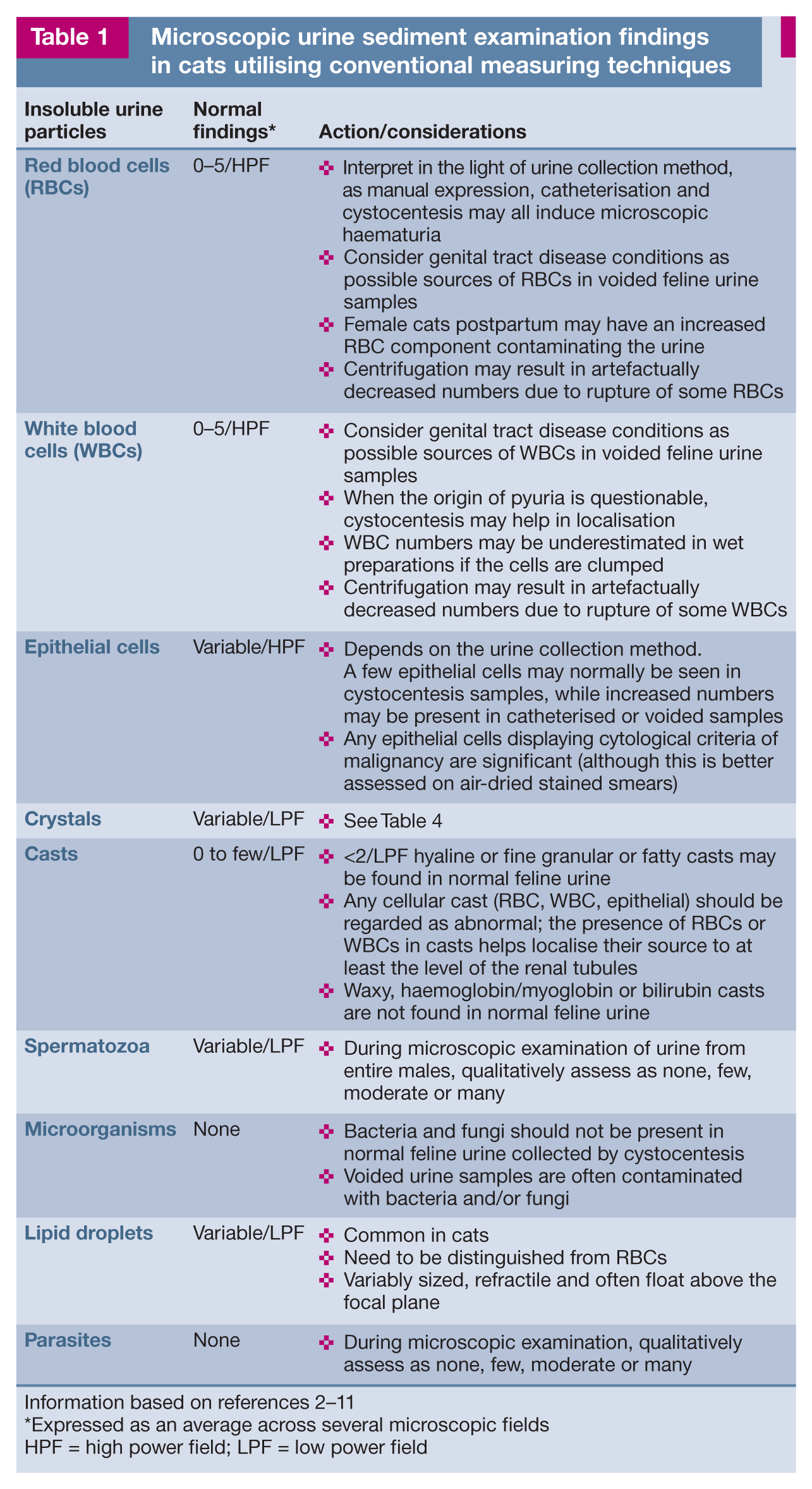
Information based on references 2–11
Expressed as an average across several microscopic fields
HPF = high power field; LPF = low power field
Counting chamber method
Enumeration of particles in urine is more accurately performed using a counting chamber method on an uncentrifuged urine sample. In human laboratories, this is also the reference measurement method used to monitor the performance of automated urine cell analysers. Quantitative estimations are based on direct counting and the results are expressed as particles per litre(rather than per low or high power field).12,13
Some veterinary laboratories have adopted the routine use of disposable plastic counting chambers with counting grids (Vetriplast or KOVA Glasstic Slide 10) to facilitate the enumeration of insoluble particles in uncentrifuged urine samples (Figures 3–5). 9 Counting techniques and calculations required for the use of such plastic counting chambers are included in the product insert literature and are beyond the scope of this article.

Disposable plastic counting chambers (KOVA Glasstic Slide 10) with counting grids to facilitate the enumeration of urine insoluble particles in uncentrifuged urine samples

(a) Vetriplast slide chambers and (b) appearance of the Vetriplast slide chamber grid under a x 2 objective lens

Vetriplast slide chamber grid lines viewed under the microscope using (a) x 20 and (b) x 40 objective lenses. Struvite crystals (circled) and white blood cells (arrows) are evident in (a). Struvite crystals (circled) and a transitional epithelial cell (arrow) can be seen in (b)
Identification of urine insoluble particles in wet preparations
Insoluble particles within feline urine include cells (epithelial cells, RBCs, WBCs and neoplastic cells), casts, microorganisms, crystals, lipid droplets, spermatozoa (gender specific), mucin and artefacts. Some insoluble particles are more difficult to identify in wet preparations of urine than in air-dried blood or cytology smears, as these particles may be subjected to varying periods of exposure to osmotic and pH changes as well as bacterial toxins, with resultant changes in their size, structure and transparency. 2
Cells
Epithelial cells
Epithelial cells can be found in low numbers in the urine sediment of healthy cats because they are constantly exfoliating into the urinary tract lumen as they are replaced by new cells. Accurate data regarding the number of epithelial cells normally present in the urine of cats is not available. 3
Three main types of epithelial cells may be found in urine, depending on their origin along the urinary tract – renal, transitional and squamous (see box). Some laboratories report these epithelial cell populations separately. However, this is often problematic on wet preparations, particularly as urine samples sent to the laboratory are often not fresh. Consequently, all epithelial cell types may be counted under the one general epithelial cell category. A high total epithelial cell count (usually enumerated as a range or mean of epithelial cells seen per HPF) on wet-prep examination should prompt closer cytological assessment of the epithelial cell component. For this purpose, a stained air-dried urine smear is often required to help differentiate between malignant, dysplastic and hyperplastic epithelial cells (the latter two cell types being common in urine from animals with cystitis and reactive hyperplasia).
Erythrocytes (RBCs)
Erythrocytes tend to be the smallest cellular constituents in urine when they are present (Figure 2). Their appearance may be affected by urine specific gravity (USG) and urine pH:2,3
If USG is 1.010–1.020, RBCs appear small (approximately half the size of WBCs), round, uniform in size, moderately refractile, and pale yellow to orange in colour.
If urine is concentrated, RBCs can become crenated and appear granular.
In hypotonic (especially if USG <1.006) or alkaline urine, RBCs swell, appear as balloons (with smooth edges and pale yellow cytoplasm) or colourless rings (ghost or shadow cells), or completely lyse. Osmotic lysis of erythrocytes can be complete within 2 h.

Leukocytes (WBCs)
Leukocytes in fresh urine generally appear as round cells with granular cytoplasm (Figure 2), intermediate in size between RBCs and transitional epithelial cells. However, their appearance may be affected by a number of factors:2,3
Nuclei may or may not be discernible using reduced illumination, bright-field microscopy but can be readily demonstrated in stained smears.
Crenation or swelling depends on USG and urine pH.
If kept at room temperature for an hour or longer, leukocytes may appear degenerate with foamy cytoplasm and mild karyorrhexis, pyknosis or karyolysis.
Up to 50% of WBCs may lyse within an hour at room temperature in alkaline dilute urine.
WBC numbers may be underestimated in smears if the WBCs are clumped.
Neoplastic cells
Neoplastic cells are rarely detectable on routine wet-prep urine sediment examination. Swelling and degeneration of cells can mimic malignancy, particularly when fresh urine samples have not been used. If exclusion of neoplasia in the urinary tract is required, a wet-prep sediment or, preferably, several air-dried smears must be prepared for examination from freshly formed urine (ie, not the first morning sample) immediately after centrifugation. Air-dried smears can be stained with a rapid Romanowsky stain and assessed more closely for the presence of cytological criteria of malignancy (see page 383).
Difficulties may be encountered when trying to differentiate hyperplastic, dysplastic and neoplastic changes within epithelial cells – particularly in the presence of significant inflammation and/or infection.2,3,5 In these instances, it would be prudent to refer the slides to an experienced veterinary cytopathologist for examination.
Casts
Casts are elongate, parallel-walled structures that form in the acidic and concentrated luminal environment of the ascending limb of the loop of Henle, distal renal tubules and collecting ducts. They are cylindrical moulds of the renal tubules formed on a matrix of Tamm–Horsfall mucoproteins secreted by the epithelial cells lining these tubules and ducts. RBCs, WBCs, epithelial cells, haemoglobin, myoglobin, lipid or bilirubin may be incorporated during their formation.2,3
Ideally, cast examination should be performed on unpreserved urine immediately after collection. If chemicals (eg, formalin) are used to preserve cast morphology, this should be noted on the laboratory submission form. Use of supravital stains may aid in the correct identification of casts, which can be difficult in unstained smears.2,3

Several types of casts may be identified on routine wet-prep urine sediment examination. These structures are classified according to their appearance, which reflects their content:2,3,5
Microorganisms
Bacteria
Detection of bacteria in the urine (bacteriuria) depends on the skill and experience of the observer and the numbers present. More than 104 bacterial rods/ml or 105 bacterial cocci/ml need to be present before they can be readily detected in the unstained sediment. 2 Bacteriuria is not a finding in healthy cats. 14 It was also thought to be rare in idiopathic feline lower urinary tract disease, although two studies have challenged this notion – with bacteriuria detected in 22% of catheterised samples in one 15 and 23% of cystocentesis samples in the other. 16 Bacterial urinary tract infections are more common in oldercats. Pyuria should not be a criterion for determining the presence or absence of bacteriuria. 10
If examining wet preparations of urine sediment, this should be performed under low light intensity to increase contrast. Bacteria usually refract light, and bob and quiver in the sediment. Single cocci are difficult to detect. Coliform rods may form chains, which need to be distinguished from fungal hyphae (air-dried smears allow differentiation). Stained wet preparations have been shown to have an unacceptably high false-positive rate for bacteria. 10
Examination of modified Wright-stained (or other rapid Romanowsky stained) air-dried smears of urinary sediment at high magnification (high dry or oil immersion, Figure 6) significantly improves the sensitivity, specificity and efficiency of microscopic detection and classification of bacteriuria compared with the wet-unstained method. 10
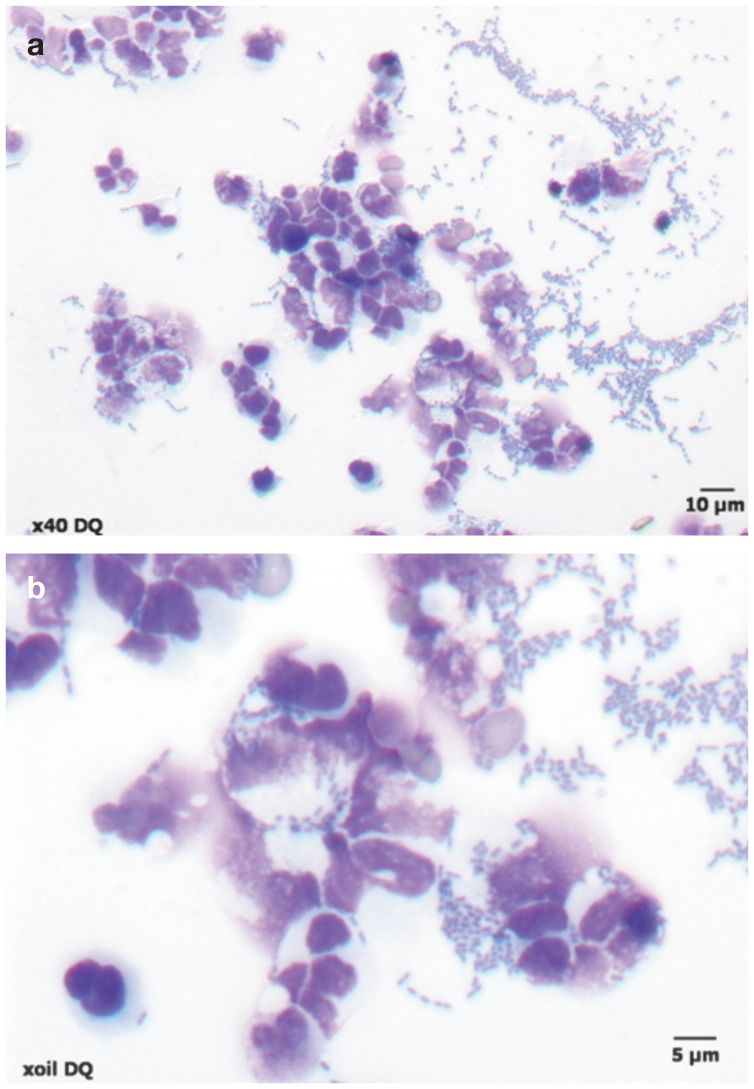
(a,b) Air-dried cytospin smear of urine from a cat with an Escherichia coli urinary tract infection. Note the short bacterial rods, located both intra- and extracellularly, accompanied by numerous degenerating neutrophils. Urine was obtained by cystocentesis and the smear was prepared using a cytocentrifuge and stained with Diff Quik
Factors that may affect the detection of bacteria during microscopic examination of urine sediment preparations are outlined in Table 2. Factors that may result in poor correlation between microscopy and urine culture results are listed in Table 3.
Factors that may affect detection of bacteria during microscopic examination of urine sediment preparations

Information based on references 2, 3 and 10
Factors that may affect culture of samples in which bacteriuria is correctly identified or ruled out on microscopy

Information based on references 2, 3 and 10
Yeasts/fungi
Fungi, including yeasts, are often contaminants in urine but in an appropriately handled cystocentesis sample should be regarded as significant and submitted for culture. Yeasts may be difficult to distinguish from RBCs or lipid droplets on microscopic examination of wet preparations. Budding forms and a double refractile wall may help identify yeast organisms more easily. Examination of air-dried preparations after cytocentrifugation of urine will also help determine the presence of a yeast/fungal infection.2,3
Fungal culture of appropriately collected urine samples is paramount if identification of a particular yeast/fungus is required.
Parasites
Eggs of Dioctophyma renale (giant kidney worm), Capillaria plica and Capillaria feliscati (bladder worms) may be present in urine.2,17
Crystals
Crystals are commonly found in cats on routine urinalysis (Table 4). Further investigation is required to assess the diagnostic and clinical significance of the crystalluria. The starting point should always be microscopic examination of a fresh (<1 h post-collection), non-refrigerated urine sample to avoid possible in vitro artefacts (eg, formation or dissolution of crystals in urine associated with prolonged storage or refrigeration).
Commonly encountered crystals and their significance in feline urine

Information based on references 2–4, 6, 9, 11 and 17–20

Struvite crystals in (a) an unstained wet preparation (arrows) and (b) an air-dried smear stained with Diff Quik

Ammonium biurate crystals in cat urine, showing the classic ‘thorny apple’ appearance. Image © Cornell University
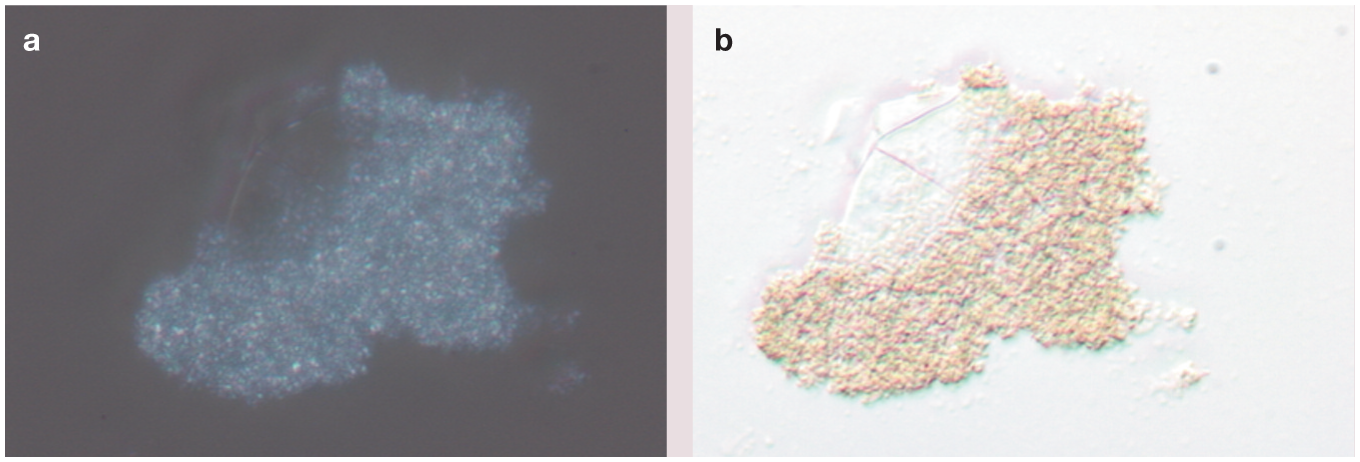
Amorphous urate crystals in unstained wet preparations; (a) with and (b) without polarisation

Calcium oxalate dihydrate crystals in a Diff Quik-stained smear viewed at (a) x 40 and (b) oil immersion x 100

Bilirubin crystals in a Diff Quik-stained smear

Preparation of air-dried smears for urine insoluble particle identification
When difficulty is encountered identifying insoluble particles, or increased numbers of epithelial cells or abnormal cells are seen on urine wet-prep examination, cytological examination of an air-dried smear should also be performed for further investigation. In veterinary practice, neoplastic cells in urinary sediment are best identified in air-dried smears.6,8,9 Liquid-based preparation techniques such as ThinPrep and SurePath, which have gained popularity for processing human urine specimens in the past few years, have yet to be investigated for use in screening cats and dogs with suspected urinary tract malignancies.
Preparing air-dried smears for cytological examination
Air-dried smears may be prepared for cytological examination using a variety of techniques. These attempt to preserve the cytomorphological features of the generally low numbers of nucleated cells, while simultaneously concentrating them onto a focal area of the glass slide for further assessment. The precise preparation technique will depend on the resources and preferences of the veterinary practice or veterinary laboratory.
Cytocentrifugation is routinely used in veterinary laboratories and some larger veterinary practices to prepare cytospin smears from urine samples. In general veterinary practice, preparing diagnostically useful smears from urine without the use of cytocentrifuge machines can be challenging, though several techniques can be used. Direct smears of the cell sediment can be prepared by conventional centrifugation (as described on page 375), although gravitational sedimentation techniques or a combination of conventional centrifugation and gravitational sedimentation techniques may be more useful in producing adequate cellular smears for further cytological examination.
Cytocentrifugation technique
Cytocentrifuge machines such as the Shandon Cytospin (Figure 12) or Cyto–Tek are used to prepare air-dried cytospin smears. An aliquot of 250–500 µl of well mixed, resuspended urine solution (prepared as described above) is placed in the cytocentrifuge’s cytospin chamber.

(a) A cytocentrifuge machine (Shandon Cytospin). (b) Components of the cytocentrifuge chamber
Cytocentrifugation produces a small central spot on the cytospin glass slide where cells are concentrated. The superior cell morphological preservation afforded by this technique makes the detection of cytological criteria of malignancy easier, particularly within epithelial cell populations (Figure 13). The ability to concentrate low numbers of urine insoluble particles into a central area on the slide also improves the likelihood of detecting fungal infections, as only scant fungal hyphae may be present in urine.

Cytospin smears of cat urine prepared with a cytocentrifuge and stained with Diff Quik. These show neoplastic urothelial cells accompanied by inflammatory cells and a mitotic figure (arrow in [a]) and display some criteria of malignancy (including prominent, irregularly shaped nucleoli, anisokaryosis and anisocytosis (arrows in [b])
Gravitational sedimentation technique
Smears from low cellular fluids, such as urine, can also be prepared using gravitational sedimentation techniques.
Specialised sedimentation chambers (Figure 14) with a rubber flange at one end (PrepStain Settling Chamber; Becton Dickinson), are available. When seated firmly on a glass slide using paper clips to create an impervious seal (Figure 14c), they may be filled with up to 4 ml of urine and left undisturbed for approximately 30 mins to facilitate gravitational cell sedimentation. The supernatant may be carefully aspirated using a pipette or, alternatively, tipped out by briskly inverting the chamber/glass slide unit contents before dismantling the chamber. The remaining contents of the sediment chamber adhering to the microscope slide can be rapidly dried with a hairdryer (set at medium to high for 15–30 s, or ceased sooner if the specimen on the slide appears dry), prior to unclipping the sediment chamber from the glass slide. The resultant smear is localised to a focal area on the slide (Figure 14c) that can then be stained and submitted for cytological examination.

(a) Apparatus required for the gravitational sedimentation technique includes a sediment chamber, paper clips and glass slide. (b) Close-up of a sediment chamber (PrepStain Settling Chamber; Becton Dickinson). (c) The fully assembled chamber (c) is filled and left undisturbed for 30 mins. The technique results in a cytospot which is air-dried and stained
Simpler gravitational sedimentation chambers may also be configured from syringe barrels or perspex tubing (Figure 15) that have been carefully cut, with one end dipped in melted paraffin wax and mounted perpendicularly on to a pre-warmed clean glass slide. The slide should be allowed to cool prior to filling with the resuspended urine sample. The rest of the procedure is similar to the gravitational sedimentation smearing technique described above.

Simpler gravitational sediment chamber apparatus for ‘in-house’ use with low cellular fluids such as urine
Direct line smear technique
Direct smears may be prepared from the resuspended urine sediment pellet (see page 374) by using the line smear technique described in Figure 16.

Line smear technique. Diagram 1 and glass slide (a) depict the position of the resuspended drop of urine (labelled S) close to one end of the frosted glass slide (which is labelled in pencil with the patient’s details) and the placement of the overlying spreader slide; this spreader slide is drawn backwards slowly to engage the resuspended drop of urine. Diagram 2 shows the varying angles at which the spreader slide can be held while making the smear (dependent on the desired thickness of the film to be produced). Diagram 3 illustrates the abrupt lifting of the spreader slide approximately 0.5 cm from the non-frosted edge of the glass slide; this action produces a dense ‘line’ at the end of the smear (b,c) in which most of the larger cells (including neoplastic epithelial cells) will usually be present. A better ‘monolayer effect’ may be achieved if, immediately after line smearing, the glass slide is tilted slightly with the ‘line’ uppermost, and rapidly dried with a hairdryer
Staining urine air-dried smears for cytological examination
Slides prepared using one of the above-discussed techniques can be left to air dry but should preferably be dried rapidly using a hairdryer (set at medium to high for 15–30 s, or ceased sooner if the specimen on the slide appears dry); this avoids slow-drying artefacts and helps ‘fix’ the cells to the glass slide, reducing the likelihood of cells ‘washing off’ the slide during staining. Other heat fixation methods are not encouraged as they are likely to alter cell morphology.
Both direct and cytospin air-dried smears submitted for microscopic examination can be stained using rapid Romanowsky stains. In practice, one of the easiest, quickest and most cost-effective Romanowsky-type stains available is Diff Quik.
As discussed earlier, air-dried smears stained with a rapid Romanowsky stain facilitate checking for bacteria (see Figure 6). Stained air-dried smears have been shown to have superior sensitivity (83%) and specificity (98%) for bacterial detection compared with the wet unstained method. 10 Air-dried smears can also be stained with a gram stain if bacteria are identified. Panfungal PCR testing (available at some specialist laboratories) may also be attempted on air-dried smears that reveal increased numbers of fungal hyphae. 21
Note that cytological stains must be replenished regularly to avoid artefactual bacterial presence in smears as a consequence of bacterial contamination and proliferation in unchanged stain media. Stains must also be renewed regularly and/or filtered to avoid deposition of stain precipitate, which may mimic bacteria in smears.
Key Points
Microscopic examination of urine, which includes urine sediment examination and urine cytological assessment, is more demanding and time consuming than urine macroscopic examination.
It involves greater skill and more equipment to perform competently.
Once the urine microscopic examination technique has been mastered and the limitations of the test have been considered, erroneous results and interpretations will be minimised.
Footnotes
Acknowledgements
Figure 8 (ammonium biurate crystals) is used with permission of Cornell University. The original image is located on the ‘eClinPath’ website at Cornell University. Cornell University retains full copyright.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
