Abstract
Objectives
The aims of the study were to describe the radiographic and computed tomographic features in cats naturally infected with Aelurostrongylus abstrusus, and to identify signs of pulmonary hypertension with echocardiography.
Methods
Fourteen cats positive on Baermann test for A abstrusus were included in the study. All cats underwent thoracic radiography, CT and echocardiography.
Results
The most common clinical signs were coughing (10/14) and dyspnoea (5/14). Radiographic findings included a generalised unstructured interstitial pulmonary pattern (8/14), mixed bronchointerstitioalveolar pattern (3/14) and bronchointerstitial pattern with bronchial wall thickening (3/14). Sternal lymphadenopathy was detected on thoracic radiographs in six cats. On CT, features were mixed bronchointerstitioalveolar pattern with ground-glass appearance in six cats, interstitioalveolar with multiple pulmonary nodules in five, interstitial ground-glass infiltrates in three, regional lymph node enlargement in 11 (10 sternal, three cranial mediastinal and three tracheobronchial lymph nodes) and subpleural thickening in four. None of the thoracic radiographs revealed subpleural thickening. In all cases, pulmonary vessels were normal in terms of size, shape and attenuation on both radiography and CT. Pulmonary hypertension and cardiac abnormalities were not observed in any cat during echocardiography.
Conclusions and relevance
CT provided a more thorough characterisation of pulmonary and mediastinal lesions compared with thoracic radiographs in cats naturally infected with A abstrusus. Although feline aelurostrongylosis has been previously associated with histopathological lesions in lung arteries, in this cohort clinical evidence of pulmonary hypertension was not documented.
Introduction
Feline aelurostrongylosis has a global distribution and, although previously considered a sporadic disease, is now recognised with increasing frequency. Indeed, the parasitic infection has been recently identified in up to 22−25% of free-roaming and shelter cats across Europe and in up to 5% of animals in the USA.1–3 The infection is either subclinical or characterised by varying respiratory clinical signs, including mild-to-intense longstanding cough, sneezing or wheezing, mucopurulent nasal discharge, tachypnoea, tachycardia and dyspnoea with open-mouth or abdominal breathing. 4 Vague non-respiratory but severe clinical signs such as lethargy, depression and loss of body weight may also accompany the disease. Death due to respiratory failure can occur if aelurostrongylosis is left untreated. 4 Clinical signs develop as a consequence of a strong inflammatory response due to egg production and migration of first-stage larvae (L1) in lung parenchyma.5,6 Adult parasites live in the terminal bronchioles, alveolar ducts and alveoli of cats. The females lay eggs, which go through embryonic maturation within the parenchyma. Secondary endothelial arterial damage is induced by the inflammatory response due to presence of the parasite. After hatching, L1 pass up the respiratory escalator to the pharynx, where they are swallowed and released via faeces into the environment. L1 continue their life cycle into mollusc intermediate hosts, in which they reach the third, infective, larval stage (L3). The cat, which is the definitive host, is infected by ingesting the intermediate or paratenic hosts. 4 All parasitic stages are able to damage the lung parenchyma.
Radiographic changes associated with Aelurostrongylus abstrusus in experimentally infected cats include generalised, structured nodular interstitial mixed with bronchial lung pattern of varying severity with mild changes in location and distribution.7,8 Although CT is less readily available than conventional radiology, its use is growing considerably because it allows better characterisation of pulmonary diseases in both dogs and cats. To our knowledge, only two articles have described the CT findings in cats, either naturally or experimentally infected with A abstrusus.8,9 In case of experimental infections, the severity of lesions documented with CT positively correlated with parasitic burden and the most frequent lesions were multiple nodules of variable size distributed throughout the lungs. Other CT findings included severe multifocal thickening of the bronchi and interstitial changes blurring the margins of the outer serosal surface of bronchi and vessels. 8 In naturally infected cats, CT findings include multiple small hyperattenuating areas widely distributed throughout the lung fields. Vascular inflammation and thrombosis of the lungs might be important pathogenic mechanisms leading to secondary pulmonary hypertension. 10 However, it is unclear whether natural infection with A abstrusus causes pulmonary hypertension in cats. Increased arterial pressure of the lungs has been uncommonly described in cats, in particular with reverse patent ductus arteriosus, pulmonary thromboembolism, heartworm and upper airway obstruction; pulmonary hypertension has been documented using echocardiography in only one cat with naturally acquired aelurostrongylosis. 11
The purposes of this study were to describe the radiographic, CT and echocardiographic findings in a cohort of cats naturally infected with A abstrusus and to investigate if a relationship exists between the parasitic lung infection and the presence of pulmonary hypertension.
Materials and methods
Client-owned cats with positive results on the Baermann test for A abstrusus identified between December 2012 and June 2013 at the Veterinary Institute of Novara were included in the study if treatment naive. Radiography, CT of the thorax and echocardiography were performed in all cats under general anaesthesia and within the same day if clinical conditions were stable. Radiographic and CT studies were evaluated by two radiologists (EA, GL) and echocardiographic studies were performed by two cardiologists (OD, FM).
Dorsoventral and right-to-left lateral radiographs of the thorax were obtained (GE Medical Systems). Radiographs were evaluated for an abnormal pulmonary pattern, lymphadenopathy and cardiovascular changes. Abnormal pulmonary patterns were classified as: (1) generalised unstructured interstitial, (2) mixed bronchointerstitioalveolar and (3) bronchointerstitial with bronchial wall thickening. The distribution of the pulmonary patterns was classified as diffuse (multifocal) or focal.
Pulmonary CT images were taken in sternal recumbency with a four-slice scanner (GE Medical Systems), with a detector width of 1.25 mm. Scanning parameters were 180 mA, 120 kVp, tube rotation time 1 s, collimator pitch 0.75 and standard reconstruction algorithm. The field of view was optimised on the thorax, and all scans were acquired in a craniocaudal direction. A total of three scans (precontrast, pulmonary angiography and delayed phase) were acquired before and after intravenous (IV) injection of iodinated contrast medium (Omnipaque 350 mgI/ml; GE Healthcare) in the cephalic vein at a dose of 2 ml/kg with flow rate of 2 ml/s using a power injector (Envision CT; Medrad Italia). A dedicated medical image viewing station (Advantage Workstation 4.2; GE Medical Systems) was used for CT image analysis. CT studies were evaluated for pulmonary pattern abnormalities adopting the above criteria used for radiographic classification, including thoracic lymphadenopathy, subpleural thickening and presence of pulmonary thromboembolism, as shown by intravascular filling defects of the pulmonary arteries during the angiographic phase.
Echocardiographic examinations were performed with a commercial instrument (GE Medical Systems). All studies were performed immediately after CT examination under general anaesthesia. Each cat was positioned in left and right recumbency to obtain the two-dimensional and motion mode (B-mode and M-mode, respectively) acquisition and to obtain Doppler parameters. Pulmonary hypertension was identified if the main pulmonary artery, right atrium or right ventricle were dilated; if concentric and eccentric right ventricular hypertrophy were observed; if a high-velocity diastolic pulmonic valve insufficiency (>2.2 m/s) or high-systolic tricuspid regurgitation velocity (>2.8 m/s) were documented; or if an acceleration time to ejection time ratio <0.3 was calculated in the pulmonary outflow profile.
To perform the examination, all cats were sedated with intramuscular dexmedetomidine (Dexdomitor; Elanco Animal Health) at a dose of 7 µg/kg, ketamine (Imalgene 1000; Merial Italia) at a dose of 2 mg/kg and butorphanol (Dolorex; Intervet Italia) at a dose of 0.2 mg/kg. General anaesthesia was induced with IV propofol (Proposure; Merial Italia) to effect to achieve the orotracheal intubation and maintained with isoflurane (Isoflurane Vet; Merial Italia) at 1.3 EtIso in 100% oxygen through an endotracheal tube. To allow optimal CT scans, IV atracurium besylate (Tracrium; GlaxoSmithKline) was administered as a single bolus (0.15 mg/kg) to induce apnoea and intermittent positive pressure ventilation (11 ± 2 cm H2O) was imposed to have normocapnia (between 30 and 35 mmHg EtCO2) and until resolution of the neuromuscular block. Only during the CT scans was apnoea induced by switching off the mechanical ventilation.
Results
Fourteen cats met the inclusion criteria. The median age of the cats was 24 months (range 8−120 months) with a median body weight of 3.1 kg (range 2.0−3.5 kg). There were seven neutered males and seven neutered females; none was intact. All cats were domestic shorthairs. Radiographic and CT findings are listed in Table 1. The most common presenting clinical signs were cough and dyspnoea, reported in 10 (71.4%) and five (35.7%) cats, respectively.
Radiographic and CT findings in the 14 cats naturally infected with Aelurostrongylus abstrusus

F = female; DSH = domestic shorthair; ST = sternal lymph nodes; CM = cranial mediastinal lymph nodes; TB = tracheobronchial lymph nodes; M = male
Based on thorax radiography, a generalised unstructured interstitial pulmonary pattern was seen in eight (57.1%) cats, a mixed bronchointerstitioalveolar infiltrate in three (2.1.4%) (Figure 1a,b) and a bronchointerstitial pattern with thickening of the bronchial walls in three (21.4%) (Figure 2). The distribution of the described pulmonary infiltrates was multifocal in all cases. Sternal lymphadenopathy was suspected in six (42.9%) cats. Cardiovascular structures, when evaluable, were within normal limits.

(a) Right-to-left lateral and (b) dorsoventral thoracic radiographs of a cat. LL = latero-lateral; SX = left; DV = dorsoventral
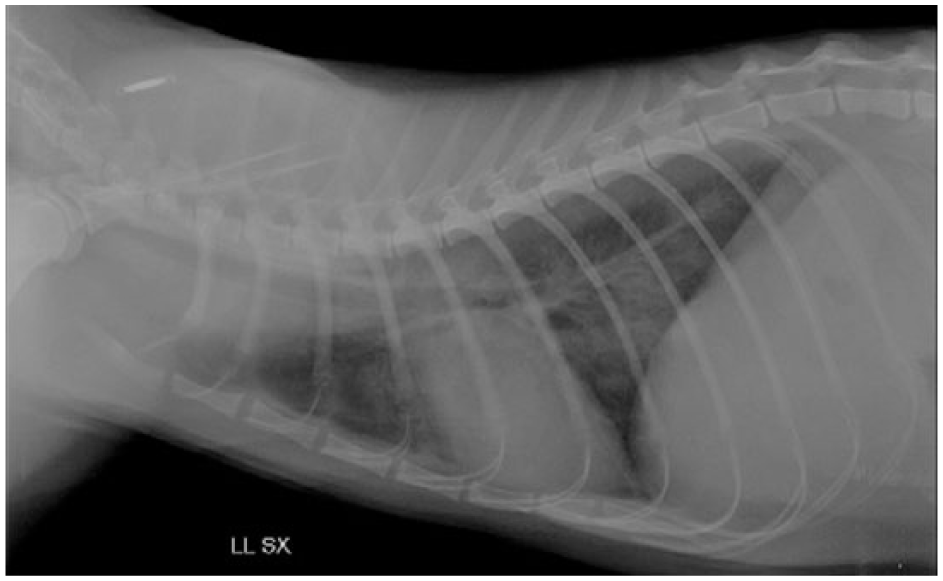
Right-to-left lateral thoracic radiograph of a cat. Bronchointerstitial pattern with thickening of the bronchial walls LL = latero-lateral; SX = left
On CT imaging, bronchointerstitioalveolar infiltrates with ground glass appearance were seen in six (42.9%) cats (Figure 3), interstitioalveolar with multiple pulmonary nodules in five (35.7%) (Figure 4) and interstitial ground glass appearance in three (21.4%). Thoracic lymph node enlargement was seen in 11 (78.6%) cats. Subpleural thickening was observed in four (28.6%) cats. No evidence of intravascular filling defects in the pulmonary arteries compatible with pulmonary thromboembolism was documented in any case during the CT angiographic phase. All cats with lymph node enlargement documented with thoracic radiographs had lymphadenopathy that was also detected by CT; in contrast, four cats documented by the latter were not identified with radiographs.
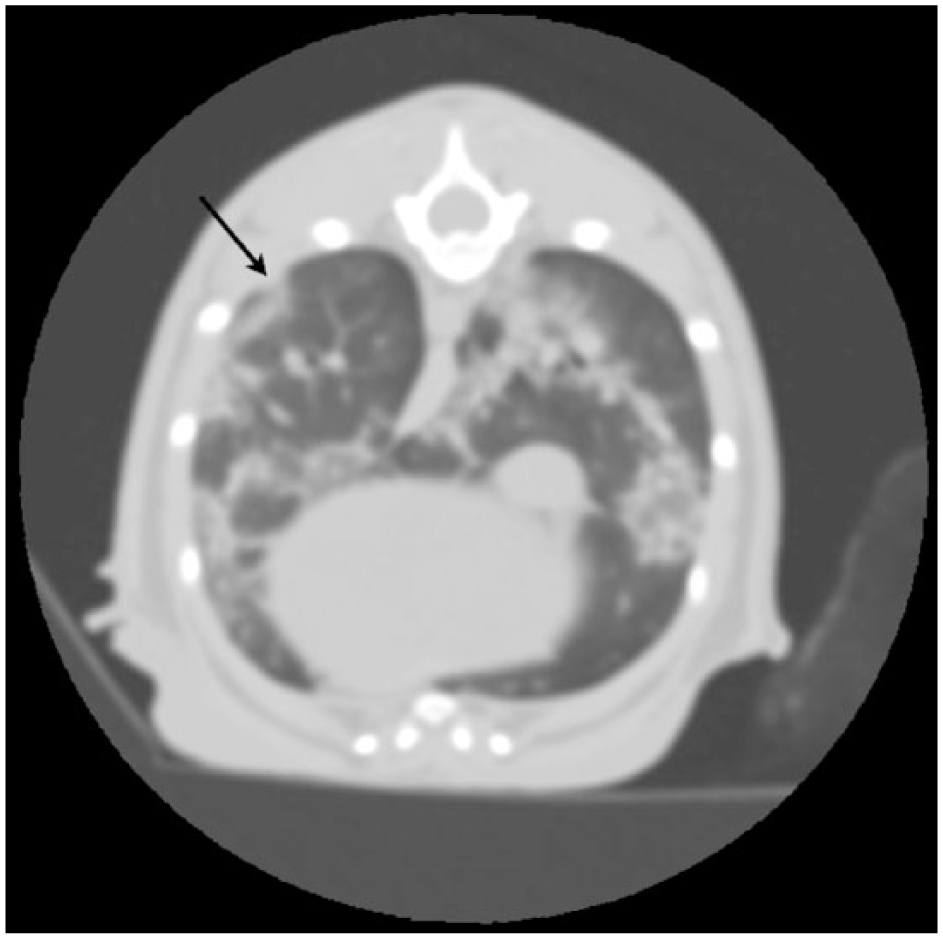
CT image of the same cat shown in Figure 2. Bronchointerstitioalveolar infiltrates with ground glass appearance. Subpleural thickening is also noted (black arrow)
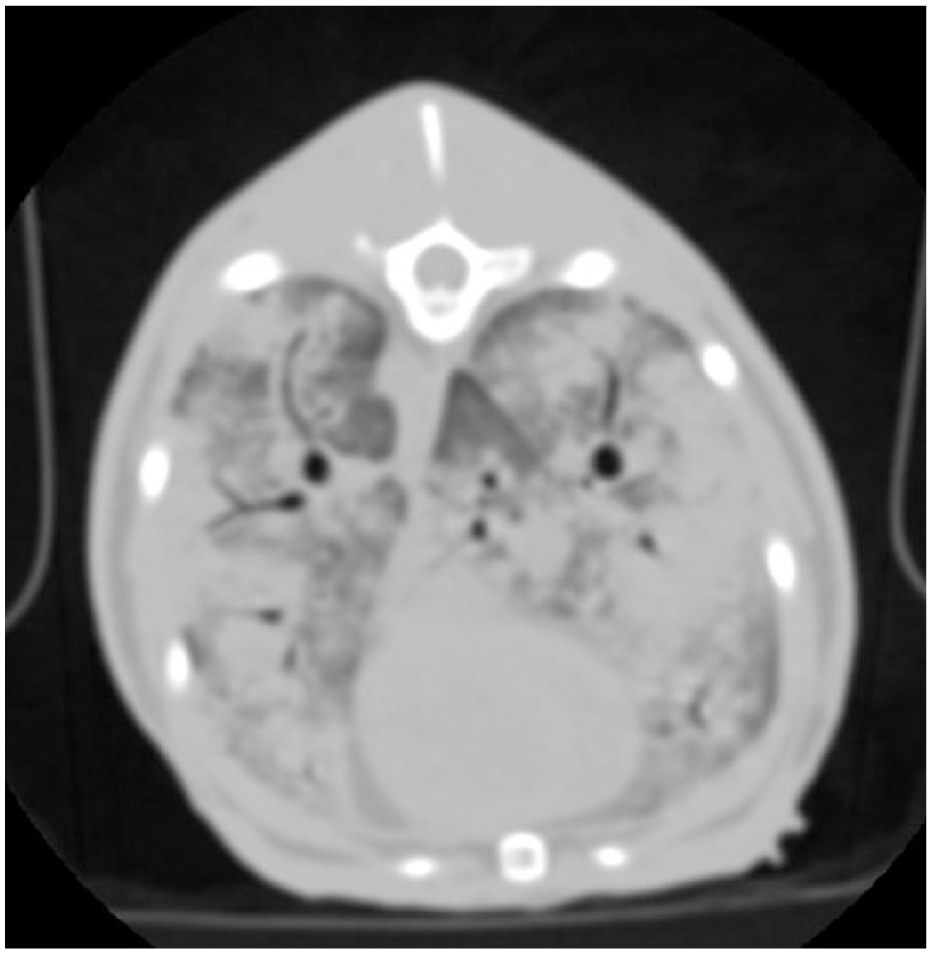
CT image of the same cat shown in Figure 1. Interstitioalveolar infiltrate with multiple ill-defined pulmonary nodules
In three cases where radiography showed an unstructured interstitial pulmonary pattern, CT revealed a multifocal nodular infiltrate. In three other cases, CT allowed identification of an interstitial ground glass opacity; this feature was not clearly recognisable in radiography. In seven cats CT showed a generalised bronchial wall thickening, which was less clear on radiographs. Subpleural thickening was also detected in four cats by CT and not visible with conventional radiology.
Furthermore, lymphadenopathy was also better evaluated by CT as conventional radiology detected sternal lymph nodes enlargement in only six cases compared with 11 cases with the former. In addition, CT was able to identify cranial mediastinal and tracheobronchial lymphadenomegaly in three cases.
Echocardiographic findings are shown in Table 2. According to the echocardiographic findings, there was no evidence of pulmonary hypertension or cardiac disease in any cat. A mild jet of tricuspid insufficiency was identified in three (21.4%) cats. The median peak velocity was 1.3 m/s (range 1.2–1.4 m/s), which, by using the modified Bernoulli equation, yielded an estimated pressure gradient between the right atrium and ventricle of 6.8 mmHg (range 5.8−7.4 mmHg). Only one cat had pulmonary insufficiency, with a peak velocity of 1.7 m/s that was suggestive of a diastolic pressure gradient between the right ventricle and main pulmonary artery of 11.6 mmHg.
Echocardiographic measurements of the 14 cats naturally infected with Aelurostrongylus abstrusus

Data are median (range)
LAD = left atrium dimension; IVSd = interventricular septum; LVIDd = left ventricle internal diameter; LVPWd = left ventricle posterior wall at diastole; LA/Ao = left atrium-to-aorta ratio; Ao ann = aortic artery annulus dimension; AP ann = pulmonary artery annulus dimension; RVDd = right ventricle internal diameter at diastole; LVPWd = left ventricle posterior wall at systole; E wave = early velocity of mitral inflow; E dec T = deceleration time of E wave; A wave = late velocity of mitral inflow; P VTI = pulmonic velocity–time integral; P Vmax = peak blood velocity of pulmonary inflow; Ao Vmax = peak blood velocity of aortic inflow; RV AT = right ventricle ejection time; RV ET = acceleration time of the pulmonary inflow; RV AT/ET = acceleration-to-ejection time ratio; Tric reg = peak velocity of tricuspidal regurgitation; IPd = peak velocity of diastolic jet of pulmonic insufficiency; PW Doppler = pulsed-wave Doppler; CW Doppler = continuous-wave Doppler
Discussion
The findings in this study suggest that cats naturally infected with A abstrusus present with a broad spectrum of radiographic and CT changes but do not have pulmonary hypertension based on echocardiography. Radiographic features of aelurostrongylosis in cats have been previously described in experimental infections, and severity was associated with the infective dose and, possibly, chronicity. 8 Among radiographic findings, bronchial and peribronchial infiltrates, multiple pulmonary nodules and hilar lymphadenopathy were frequent. In this study, we confirm that similar radiographic findings are also present in cats with naturally occurring aelurostrongylosis, with a diffuse unstructured interstitial pulmonary pattern being the most represented. Furthermore, we noted that the severity of clinical signs was associated with imaging findings, with the most severely dyspnoeic cats having multifocal alveolar pattern and lack of lung aeration (data not shown). Because the listed radiographic signs are non-specific, in addition to lung parasites other differential diagnoses should be considered, including pulmonary fibrosis, pulmonary oedema, fungal disease, asthma and neoplasms. 12 Pulmonary fibrosis has been recently described in cats and the most common radiographic features were bronchointerstitial or alveolar pattern, pulmonary masses or bullae, and pleural effusion. 13 Two of nine cats with lung fibrosis presented right-sided heart enlargement due to pulmonary hypertension. 13 Pulmonary oedema is characterised by an increased pulmonary opacity associated with a range of patterns and variable distribution of infiltrates. If the cardiac silhouette is recognised, cardiomegaly is often present. 14 However, cardiomegaly was not documented in any of our cats. Recent reports described the CT findings of cats infested by Dirofilaria immitis and Toxocara cati.15,16 Overlapping imaging features are present between those parasites and A abstrusus infection, and should therefore be added in the differential diagnosis list of feline lung disease. In our case series, no evidence of congestion of pulmonary arteries was detected as is often present with other pulmonary parasites.15,16
Primary pulmonary tumours typically present as a solitary nodule or mass and may be accompanied by additional lesions representing metastatic disease, although they can also manifest as multifocal parenchymal disease with peribronchial infiltrates or patchy alveolar patterns. Lobar consolidation occurs if a lung lobe is gradually infiltrated. Occasionally, concurrent cranial mediastinal or hylar lymphadenomegaly and/or pleural effusion are radiographically visible. 17 In our study, if pulmonary nodules were documented, they were not isolated but had a multifocal-to-generalised distribution.
With regard to pulmonary mycosis, Histoplasma capsulatum can cause granulomatous disease with lung involvement. Radiographic patterns in cats include fine, diffuse miliary, nodular, or bronchointerstitial infiltrates.18–20 Histoplasmosis is also frequently accompanied by bone lesions consistent with osteomyelitis. 21 At our latitude, histoplasmosis has never been described in cats and bone lesions were not identified in our series. Although feline asthma may have unremarkable radiographs, common findings are peribronchial cuffing, alveolar infiltrates, consolidation, air trapping and hyperlucent lungs. 17 None of the cats with A abstrusus had evidence of pulmonary hyperinflation associated with peribronchial or alveolar infiltrates.
CT has been previously compared with conventional radiology in the evaluation of non-cardiac thoracic disease in dogs and cat. 22 This advanced imaging technique has several advantages over survey radiography, including elimination of superimposed anatomy and superior contrast resolution. These properties allow clarification of intrathoracic lesions when radiographic findings are negative or non-specific. CT has been shown to have a higher accuracy for characterisation of lower airway disease than radiography (89 vs 50%).23,24 In our study, CT allowed a very detailed characterisation of lung infiltrates compared with conventional radiology. This difference might be explained by the superimposition of bone and soft tissues structures and inferior contrast resolution of radiography, as previously demonstrated. 22 Furthermore, lymphadenopathy was also better evaluated by CT as conventional radiology detected sternal lymph node enlargement in only six cases compared with 10 cases with the former. In addition, CT was able to identify cranial mediastinal and tracheobronchial lymphadenomegaly in three cases.
Differential diagnoses for focal or diffuse lymphadenopathy include inflammatory granulomatous or neoplastic metastatic disease. Distinction between neoplastic and granulomatous lymphadenopathy cannot be achieved by diagnostic imaging and lymph node histology is required.
In a canine study on CT assessment of lymph nodes, a cut-off was established for tracheobronchial lymph node enlargement; in non-inflammatory diseases, a size of >12 mm has been considered 85.7% sensitive and 95.2% specific for metastases. 25 In addition, rim contrast enhancement in metastatic lymph nodes was considered a very common finding and useful in identifying metastatic lymph nodes. To our knowledge, no data have been reported on differentiation between normal and pathological lymph nodes of the feline thorax based on CT. Because our cats had A abstrusus infection, we assume that thorax lymphadenopathy was due to the associated inflammatory condition, even if histology was not performed.
Although thoracic radiographs and CT examination suggested that the lungs were severely injured, none of the cats had echocardiographic evidence of pulmonary hypertension. Pulmonary hypertension results from an imbalance of arterial vasodilators and vasoconstrictors, from vascular smooth muscle cell proliferation and thrombosis. 26 It can be classified as pre- or postcapillary and can be distinguished based on the underlying disease process. 27 Our results are in agreement with a previous study of cats experimentally infected with A abstrusus and evaluated with pulmonary artery catheterisation and arteriograms; pulmonary hypertension was not detected in any cat. 28 However, in a recent report a cat with natural occurring aelurostrongylosis and reversible pulmonary hypertension was described. Furthermore, the same cat also displayed right-sided heart enlargement, based on echocardiography. 11
Histological changes that occur in the pulmonary arteries due to A abstrusus infection have been well characterised in cats.10,27 The most common features are hyperplasia and hypertrophy of smooth muscle cells of the media and intimal proliferation. These changes developed approximately 12 weeks after infection. Because our cats were naturally infected, and the exact timing of infection was unknown, it cannot be excluded that A abstrusus was present for less than 12 weeks in all cases, although we deem this unlikely. Thus, we hypothesise that pulmonary hypertension is an uncommon finding in cats naturally infected with the parasite. It may be argued that there are still limited data regarding echocardiographic findings of cats with pulmonary hypertension. However, it has been assumed that the same echocardiographic criteria adopted for dogs may also be applied to study cats. 29
There are some limitations to our study that need to be addressed. In particular, all cats had thoracic radiographs, CT and echocardiography performed at first admission, but none were re-evaluated during the weeks following lungworm treatment. It would have been interesting to verify whether the documented lung abnormalities were partially or fully recovered. Furthermore, none of the cats included in the study received a proper lung work-up to rule out underlying pathology. Another limitation is the absence of objective criteria to assess bronchial wall thickening based on CT examination, as recently proposed in small animals. 17 However, because cats are smaller than many dog breeds, performing accurate measurements is hampered. Finally, invasive pulmonary arterial pressure was not measured in the present series. Pulmonary capillary wedge pressure would have yielded a more objective quantification of lung hypertension than echocardiography.
Conclusions
Aelurostrongylosis should be included in the list of radiographic differential diagnoses in all cats presenting with interstitial to bronchoalveolar or nodular pattern and with suspected lymphadenopathy. Computed tomographic documentation of ground glass appearance associated with interstitioalveolar infiltrate, multifocal nodular structures, generalised lymphadenopathy, and bronchial wall and subpleural thickening should be considered suspicious for A abstrusus infection in cats. Furthermore, based on the present series, pulmonary hypertension in cats with naturally occurring aelurostrongylosis does not seem to develop, as assessed by echocardiography.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
