Abstract
Objectives
The aim of this study was to investigate the role of thyroid transcription factor-1 (TTF-1) in the diagnosis of feline lung–digit syndrome (FLDS) and to investigate the associations between the morphological features of FLDS and TTF-1 expression. We also compared the reliability of TTF-1 and transmission electron microscopy (TEM) in establishing the diagnosis of FLDS.
Methods
Histology records of feline digit tumours were retrieved, including patients from 2008–2015. If formalin-fixed, paraffin-embedded tissues were available for review, patients were included in the study. As a control group we included 12 feline primary tumours of the digits. All the histological slides of the study group were blindly reviewed by the same veterinary pathologist. Representative sections of the lesions were selected for immunohistochemistry (IHC) analysis. To confirm the respiratory origin of the neoplastic tissue, TEM was used as a gold standard in all cases.
Results
Five cases of FLDS were included. TTF-1 was weakly to moderately positive in 60% of the cases, showing no correlation with the microscopic presence of ciliated epithelium. When IHC results were combined with the presence of cilia, 80% of the cases from the study group could be identified as FLDS. TEM confirmed the presence of ciliated epithelium in all five cases, confirming the respiratory origin of the neoplastic tissue and therefore the diagnosis of FLDS.
Conclusions and relevance
TTF-1 expression is maintained in FLDS. While the combination of TTF-1 and identification of cilia confirms FLDS, TEM should be considered in those cases where diagnosis is uncertain and FLDS is suspected.
Introduction
Feline primary lung tumours are uncommon and aggressive neoplastic diseases, which are mainly diagnosed in geriatric patients. Persian cats may be overrepresented; however, sex or neutering status predilection has not been reported. 1 Pulmonary adenocarcinoma presents more often (64%), whereas bronchioloalveolar carcinoma and adenosquamous carcinomas are less common (20.5% and 15.4%, respectively).1,2
In a recent study, approximately 80% of feline pulmonary carcinomas metastasised within the thorax (38.5%), to regional lymph nodes (33.3%) and distant visceral organs (10.2%). 1 Within the patients exhibiting distant metastatic disease, approximately 18% develop metastasis to the digits and this represents a peculiarity of feline primary lung tumours, known as ‘feline lung–digit syndrome’ (FLDS). FLDS is seen only with primary lung tumours and particularly with bronchioloalveolar carcinomas.1,3–5 Tumour metastases are found at the distal phalanges, where the weightbearing digits are most frequently affected; multiple digit and multiple limb involvement is common. 3 Histopathological examination of digital tumours may suggest or even confirm a respiratory epithelial origin if goblet cells or ciliated cells are observed, respectively; however, the differentiation between FLDS and primary digit tumours becomes a challenge for pathologists when there is lack of these features.3,6 Even in the case of radiological evidence of a lung mass, a lesion of a distal phalange should not be assumed to be FLDS when its respiratory origin cannot be confirmed. Although it seems clear that valuable diagnostic tools are needed in order to diagnose FLDS, to our knowledge no studies have yet covered this topic.
Thyroid transcription factor-1 (TTF-1) is a 38 kDa nuclear protein member of the Nkx2 homeodomain transcription factor family, expressed by human and canine type II alveolar pneumocytes, bronchiolar and thyroid epithelial cells.7,8 In the feline species, positivity to TTF-1 is mainly restricted to bronchiolar and thyroid cells; type II alveolar pneumocytes are rarely positive. 1 TTF-1 is known to play a crucial role in the differentiation of lung epithelial cells and remains expressed in neoplastic tissue, becoming a useful tool in confirming the primary pulmonary origin of lung tumours; non-pulmonary and non-thyroid neoplastic tissues do not express TTF-1.1,7,9 Regardless of the high sensitivity and specificity observed in dogs, sensitivity in cats is a matter of controversy.1,7,9 Recent research data suggested that TTF-1 expression is conserved in well-differentiated feline pulmonary neoplastic tissue, whereas less differentiated tumours have low or negative expression of TTF-1, questioning its value as diagnostic tool. 9
The objective of this study was to explore the usefulness of TTF-1 in confirming the diagnosis of FLDS and to associate the morphological features of cases of FLDS with TTF-1 expression. We also compared the reliability of TTF-1 with transmission electron microscopy (TEM), which was used as the gold standard to confirm the respiratory origin of metastatic feline digital tumours.
Materials and methods
Population
Histology records of feline digit tumours submitted to the Section of Veterinary Pathology, School of Veterinary Science, University of Liverpool (UK) and from the archive of the Laboratorio Veterinario Bresciano (Italy) were retrieved through database searches, including cats from 2008–2015. If formalin-fixed, paraffin-embedded (FFPE) tissues were available for review, patients were included in the study. Signalment, patient history, clinical signs and imaging findings were collected from the cat’s file if this was referred for investigations; in the case of external cats, the veterinarian submitting the samples was asked via a telephone call.
Original diagnoses made at the time of sample submission were based on haematoxylin and eosin staining.
As a control group (primary digit tumours), we included 12 feline primary tumours of the digits: four samples each of squamous cell carcinoma, apocrine carcinoma and basal cell carcinoma. The selected control tumours had been previously diagnosed as primary of the digit based on histology features and absence of pulmonary lesions, as a result of radiology findings.
Histopathology and immunohistochemistry
All the histological slides of the study group were blindly reviewed by the same veterinary pathologist (LR), who was not aware of the cats’ clinical data. The slides had been prepared by FFPE tissues fixed in 10% neutral buffered formalin that were then routinely stained with haematoxylin and eosin and observed under a bright-field upright microscope.
The pattern of cellular arrangement (tubular or solid) was classified according to the most represented within the section. Cilia were identified as elongated slender eosinophilic apical structures present on the luminal side of the neoplastic cells and semi-quantitatively graded as follows: no structures compatible with typical cilia present (–), typical apical cilia present in <10% of the neoplastic cells (+), typical apical cilia present in 10–50% of the neoplastic cells (++) and typical apical cilia present in >50% of the neoplastic cells (+++). The mitotic index was calculated as the total number of mitotic figures in 10 microscopic 400 × high-power fields (HPFs). Necrosis was assessed semi-quantitatively as absent (–), <5% of the section area (+), between 5% and 20% of the whole section area (++), >20% of the whole section area (+++).
Representative sections of the lesions were selected for immunohistochemistry (IHC) analysis. As primary antibodies we used anti-pan-cytokeratin (AE1/AE3/PCK26 MoAb, ready to use; Ventana Medical Systems) and anti-TTF1 (8G7G3/1 MoAb, ready-to-use, Ventana Medical Systems). All tissue sections were placed in an automated staining system (BenchMark XT; Ventana Medical Systems) within which samples were deparaffined, rehydrated and processed for blocking endogenous peroxidase and epitope retrieval using Cell Conditioning 1 (Ventana Medical Systems) according to the manufacturer’s recommendations. Primary antibodies were incubated according to the protocol suggested by Ventana at 37°C for 16 mins. As detection system, we used the biotin-free ultraView Universal DAB Kit (detection of mouse IgG, mouse IgM and rabbit primary antibodies, Ventana Medical Systems). Upon completion of the immunostaining, sections were counterstained with Mayer’s haematoxylin. Positive controls comprised feline normal skin (pan-cytokeratin), normal thyroid gland and normal lung (TTF-1).
FLDS was considered TTF-1-positive if any number of cells were positive. The intensity of the TTF-1 stain was semi-quantitatively assessed as mild (+, barely perceptible brown nuclear stain), moderate (++, positive stain, weaker if compared with normal bronchial epithelium control) or marked (+++, positive stain comparable with the stain of normal bronchial epithelium control). The number of positive cells was calculated among the total number of neoplastic cells in five randomly selected, tumour representative HPFs and expressed as percentage of positive cells.
TEM
To confirm the respiratory origin of the neoplastic tissue, TEM was used as a gold standard in all cases.
From FFPEs, the area of interest was identified and the excess of wax was trimmed out. FFPE sections (0.3 × 0.3 cm) were dewaxed in warm xylene, subsequently washed in a graded series of ethanol (100%, 100%, 95%, 90%) and then rehydrated in distilled water. Rehydrated tissue samples were then fixed in 2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer for a maximum of 5 h at 4ºC. Fixed tissue samples were then washed in sodium cacodylate buffer for 10 mins and submitted to a secondary fixation/stain step with osmium tetroxide (1% aq) for 60 mins at room temperature. Tissue samples were then washed briefly with distilled water and stained with uranyl acetate (2% uranyl acetate in 0.69% maleic acid) for 60 mins and dehydrated with ascending concentrations of ethanol for 10 mins each step, except for 15 mins at 100% ethanol and then 3 × 5 mins at 100% acetone. Tissues were embedded in TAAB epoxy resin using an increasing series of resin:acetone solutions (30:70, 70:30, 100:0, 100:0) for 60 mins each step. Samples were finally embedded in fresh resin in polyethylene moulds and polymerised at 60ºC overnight. Toluidine blue-stained, 0.5 m-thick semi-thin sections were used for selecting relevant areas for 75 nm ultrathin sections contrasted with lead citrate and uranyl acetate. Sections were examined under a Phillips EM208S (FEI UK) at 80 kV equipped with a charge-coupled device camera (Gatan, ES500W).
Respiratory epithelial ultrastructural differentiation was assessed upon presence of cilia structures, characterised by electron-dense basal bodies and elongated cytoplasmic apical projections containing groups of microtubules.
Results
Study population
Five FFPE samples of feline digit tumours were included in the study group and there was a FFPE sample for each patient. All the cats were domestic shorthairs. There were three neutered males and two neutered females; median age was 14 years (range 13–15 years). The presenting complaint of the five patients was lameness. In three cases pain during palpation of the digits was reported; in two cases nail deviation was described. Three of the five cats had radiographs of the thorax performed and these revealed the presence of a pulmonary mass; however, sampling of the lung mass was not performed in any of the cases. Radiography of the affected limbs revealed presence of osteolytic lesions and soft tissue swelling, affecting the phalanges of the second and/or third fore digits in all the study cases. The characteristics of the cats are summarised in Table 1.
Signalment and clinical features of cats with feline lung–digit syndrome

DSH = domestic shorthair; MN = male neutered; FN = female neutered; NR = not reported
Histopathology and IHC
Four out of the five samples were originally diagnosed as FLDS and one as undifferentiated carcinoma, although highly suspicious of FLDS based on the radiological report (presence of a pulmonary mass) attached to the submission form (cat 4, Table 1).
Histology of the lesions revealed in all cases infiltrative, not well delimited, unencapsulated monomorphic neoplastic cell proliferations characterised by large aggregates of cohesive cells supported by scant fibrous stroma and extending to the biopsy cut borders (Figures 1 and 2). There was a tubular pattern in 4/5 cats (Figure 1), while cat 4 was predominantly solid (Figure 2; Table 2). Cells exhibited a variable range of anisokaryosis and anisocytosis from mild (cat 2) to marked (cat 4). Ciliated epithelial cells were observed in 40% of cases (cats 2 and 5; Table 2); although they were abundant in cat 5, these were rare in cat 2. Goblet cells were not noted. The mitotic index ranged from 14–56 per 10 HPFs (Table 2). There was evidence of necrosis in all the samples; this was moderate to severe in 80% of cases (n = 4/5; Table 2). Based on morphology and blind review of the samples, FLDS could be confirmed in 40% of cases (cats 2 and 5) owing to the presence of cilia, or suspected in another 40% (cats 1 and 3) owing to the presence of a tubular pattern; however, this could not be confirmed based on the sole histological examination in cat 4 given the presence of a solid pattern, very rare lacunar areas with doubtful cellular apical structures and the absence of other pathognomonic features (Figure 2). In the control group no cilia or goblet cells were observed with haematoxylin and eosin staining.

Feline lung–digit syndrome in case 5. Neoplastic cells forming tubular structures infiltrating the dermis. Elongated eosinophilic apical structures consistent with cilia are identified (arrow). Electron microscopy (inset) identified those structures as well-differentiated electron-dense basal bodies (arrow) and cilia containing microtubular structures (arrowhead). Haematoxylin and eosin, scale bar = 35 μm. Inset: transmission electron microscopy. Scale bar = 0.4 μm

Feline lung–digit syndrome in case 4. Neoplastic cells are arranged in solid structures infiltrating the dermis. Occasional lacunar areas are recognised, with rare cells exhibiting pale amorphous apical rim of doubtful interpretation (black arrow). Cellular atypia (white arrow) and atypical mitoses (arrowhead) are frequent. Electron microscopy (inset) identified moderately electron-dense structures consistent with abortive basal bodies (arrow) and broad projections with intra-cytoplasmic sparse microtubules consistent with abortive cilia (arrowhead). Haematoxylin and eosin, scale bar = 35 μm. Inset: transmission electron microscopy. Scale bar = 0.4 μm
Immunohistochemical, morphological and ultrastructural features of patients with feline lung–digit syndrome

TTF-1 = thyroid transcription factor-1; IHC = immunohistochemistry; TEM = transmission electron microscopy; MI = mitotic index; NA = not assessable
As expected, IHC for anti-pan-cytokeratin was positive for all cases, confirming the epithelial origin of neoplastic cells. Anti-TTF-1 was mild-to-markedly positive in 60% of cases (n = 3/5; Figures 3 and 4), showing a poor correlation with the presence of ciliated epithelium: 2/3 positive cases had no evidence of cilia (cats 3 and 4) and cat 5, which had frequent ciliated cells, was negative for TTF-1 (Table 1).

Feline lung–digit syndrome in case 2. Neoplastic cells are arranged in tubular structures infiltrating the dermis. The majority of the cells exhibit predominant moderate nuclear thyroid transcription factor-1-positive stain (arrow), while the minority of neoplastic cells are negative (arrowhead). Indirect immune peroxidise; scale bar = 35 μm
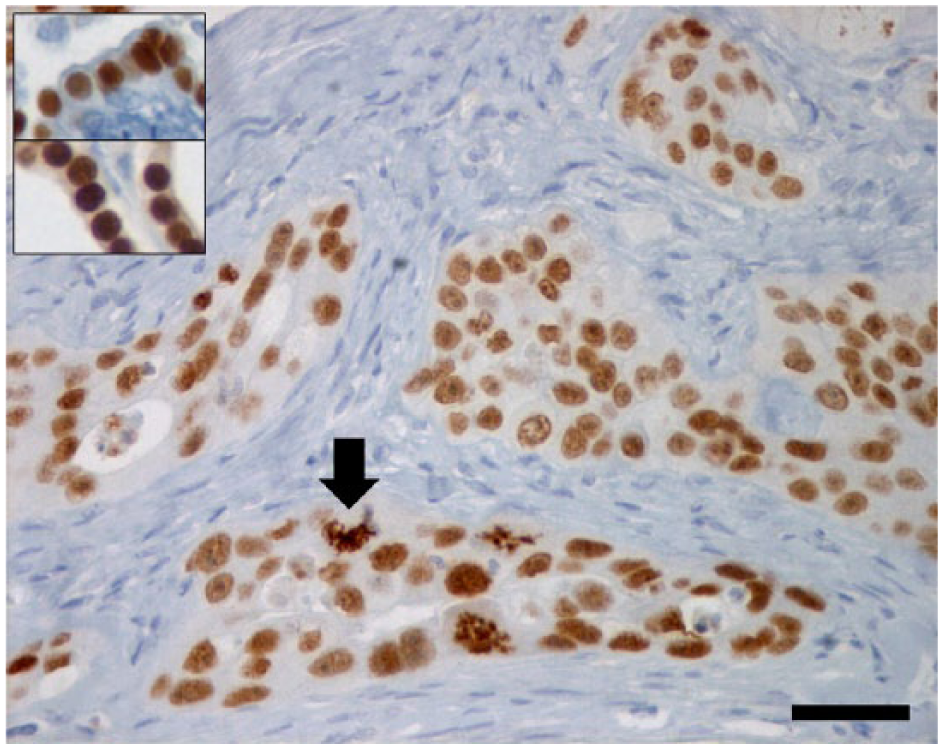
Feline lung–digit syndrome in case 4. Neoplastic cells are arranged in solid structures infiltrating the dermis exhibiting strong and diffuse nuclear thyroid transcription factor-1 (TTF-1)-positive stain that is preserved in mitotic cells (arrow). Inset: TTF-1-positive stain in normal bronchus (upper quadrant) and normal thyroid (lower quadrant) used as control. Indirect immune peroxidise; scale bar = 35 μm
In the control group, IHC for anti-pan-cytokeratin was positive in 100% of the cases, confirming the epithelial origin of neoplastic cells; anti-TTF-1 was negative in 100% of the samples.
In summary, 60% (n = 3/5) of the study group samples had positive nuclear labelling for TTF-1 and when this result was combined with presence of cilia, 80% of the cases could be identified as FLDS.
TEM
Ultra-sections obtained from FFPEs were of average-to-good quality owing to the method of fixation (formalin fixation) but suitable for ultrastructural investigation. In the two cases where cilia were evident histologically (cats 2 and 5), ultrastructurally well-developed cilia and basal bodies were present (Figure 1, inset). In the remaining samples, in which cilia were not evident histologically (cats 1, 3 and 4), short broad structures containing evidence of microtubules (aborptive cilia) often associated with electron-dense intra-cytoplasmatic basal bodies were detected (Figure 2, inset). TEM confirmed the presence of ciliated epithelium in all the five cases confirming the respiratory origin of the neoplastic tissue and therefore the diagnosis of FLDS.
TEM performed on the control group revealed no presence of cilia in any of the cases, and this result confirmed the haematoxylin and eosin and IHC findings. Therefore, none of the control group cases was reclassified as FLDS.
Discussion
The present study aimed to investigate if TTF-1 was a suitable marker in the diagnosis of FLDS, as this condition remains a diagnostic conundrum for the pathologist if clear features of respiratory epithelium cannot be identified with haematoxylin and eosin staining. Previous reports have suggested the use of histochemical and IHC stains, such as periodic acid–Schiff and anti-CAM5.2 antibody to identify digital tumours as metastatic; however, none of these would confirm the respiratory origin of the metastasis.4,6 IHC for anti-TTF-1 has been applied in feline lung tumours as a tool to confirm their pulmonary origin, with high specificity.1,9
Our results confirm that TTF-1 maintains a very high specificity for respiratory epithelium, as suggested by previous studies,1,7,8 even in the event of metastasis to the digit in FLDS. Its sensitivity is low, as just 60% of the cases (n = 3/5) stained positive in our dataset. These data are not surprising as they are in agreement with the study of D’Costa et al, 1 where only 66.7% of pulmonary carcinomas had a positive staining result. However, owing to the low sensitivity, Kujawa et al recently questioned the usefulness of TTF-1 as a diagnostic tool in cats. 9 The results of their study revealed that >50% of the bronchioloalveolar carcinomas stained negative for TTF-1 and that its expression was significantly lower in malignant tumours compared with benign neoplasia. 9 Our results confirm that TTF-1 expression is maintained in distant metastatic sites and that this marker can be useful in confirming the pulmonary origin of digit metastases. Although we have no information on histological type, grade or expression of TTF-1 by the primary pulmonary neoplasias, the presence of FLDS confirms an aggressive biological behaviour of the primary tumours. In contrast to the study by Kujawa et al, 9 in our cases TTF-1 IHC intensity was moderate to high, which was surprising; we could therefore hypothesise that, although biologically aggressive primary pulmonary tumours may lack or have low TTF-1 expression, this could be re-expressed at a higher intensity in distant metastatic foci. This phenomenon requires further investigation in order to be confirmed and to understand its biological significance.
In our population TTF-1 expression was not associated with the presence of ciliated epithelium and therefore morphological degree of differentiation (2/3 positive cases had no cilia seen with haematoxylin and eosin staining), and the combined result of TTF-1 positivity and presence of cilia identified FLDS in 80% of the cases (n = 4/5) and was superior to haematoxylin and eosin morphology or IHC alone, suggesting that this combination may be useful in a diagnostic setting.
Goblet cells have been reported to be present in FLDS, but according to our case series no goblet cells could be detected; this is likely due to the specific characteristic of our population compared with a previous study that found this to be a common feature of FLDS. 6 The lack of goblet cells highlights the fact that this feature may not be as common as hypothesised by previous authors.3,6
FLDS has so far appeared to be more common in the elderly cat, with an average age at presentation of 12 years, and no apparent sex or breed predisposition; these data are in line with our populations.4,6,10 More recent epidemiological data have shown that primary pulmonary carcinomas are four times more common in Persian cats, therefore suggesting a possible genetic predisposition in this breed. 1 Surprisingly, none of our cats was Persian and this could be owing to the small population of cats included in the study or simply to a different genetic variability of Persian cats in our regions and subsequent lack of predisposition towards pulmonary carcinomas.
Analysis of radiographic findings, treatments and outcome data was beyond the purpose of this study and was therefore just partially reported.
The results of this study also provide important evidence on the use of TEM in the diagnosis of FLDS, which, to our knowledge, has not been previously investigated. As we hypothesised, TEM was 100% sensitive and 100% specific in identifying the respiratory origin of neoplastic cells, confirming itself as a gold standard diagnostic tool for FLDS.
The prognosis for cats with FLDS is grave;11,12 one study revealed a median survival time (MST) of 67 days after presentation and the majority of cats were euthanased, owing to persistent lameness, lethargy or anorexia. 6 However, cats with primary epithelial neoplasia of the digit have a MST of 206 days. 6 A final diagnosis is therefore important in ambiguous cases as cats with a metastatic pulmonary tumour would be expected to have a poorer prognosis than those with two different primary neoplasias. However, we would like to emphasise the importance of performing thoracic radiology when a digital tumour is diagnosed in a cat.
The small number of cases enrolled represents the main limitation of this study, which is most likely to due to the rarity of FLDS. 3 Although this has to be taken into account during results interpretation, we consider the data provided useful both for pathologist and clinicians submitting biopsies.
A further limitation is the lack of biopsies from the primary lung neoplasia, which, unfortunately, made it impossible to correlate the expression of TTF-1 between the primary and metastatic site. Therefore, any assumption based on the two recent studies from D’Costa et al or Kujawa et al would be speculative.1,9
Conclusions
The results of this study confirm that TTF-1 expression is maintained when feline pulmonary tumours metastasise to the digits and that this marker can be useful in confirming the pulmonary origin of digit metastases. A combination of TTF-1 and the morphological identification of cilia confirms FLDS; however, TEM should be considered in those cases where diagnosis is uncertain and FLDS is suspected.
Footnotes
Acknowledgements
We thank M Pope for the excellent electron microscopy technical assistance.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
