Abstract
Objectives
The goal of the present study was to compare the antifungal efficacy of an essential oil (EO) shampoo proven to be effective against Microsporum canis with miconazole/chlorhexidine for topical haircoat disinfection in cats treated concurrently with oral itraconazole.
Methods
Cats received treatment with oral itraconazole (Itrafungol) at a dose of 5 mg/kg/day pulse administration for 1 week, every 2 weeks for at least 6 weeks and were washed twice a week with a neutral shampoo with added EOs of Thymus serpyllum (2%), Origanum vulgare and Rosmarinus officinalis (5% each) for the period of systemic treatment. This protocol was compared with a conventional treatment (oral itraconazole + 2% miconazole/2% chlorhexidine shampoo).
Results
The treatment was well tolerated and adverse effects were not recorded. All cats were clinically negative at week 11. With respect to animals with extensive lesions, the speed of resolution was higher in cats with focal lesions. The animals showing diffuse lesions required more than a course of treatment to achieve a mycological cure. There was no significant difference between the number of weeks to obtain mycological cure for cats treated with EOs and animals treated conventionally.
Conclusions and relevance
The treatment appeared to be effective and well appreciated by the owners. The use of shampoo with the added EOs of T serpyllum, O vulgare and R officinalis would seem an interesting, natural alternative to conventional topical treatment.
Introduction
Microsporum canis, a zoophilic and zoonotic dermatophyte that is highly infectious and has a broad host range, is the main responsible agent for dermatophytosis in cats worldwide. The infection, even if not life threatening, is highly contagious and spontaneous healing can require several months. 1
Therapeutic measures of feline microsporiasis should include the combination of systemic and topical treatment. 2 The main goal of local drug administration is to minimise the spreading of infective arthrospores, which are the source of both reinfection and new infections. Topical therapy is needed to disinfect the hairs, as systemic therapy combined with the host immune response eradicates the infection from the coat. Repeated disinfection is needed as the haircoat is reseeded with infective arthrospores until this occurs.
The most recent systemic treatment protocol licensed for use in cats in Europe is based on oral itraconazole 5 mg/kg/day pulse administration for 1 week, every 2 weeks, 3 with a total treatment period of 6 weeks, while most commonly recommended topical options include rinses and shampoos. Many commercial rinse formulations containing enilconazole, lime sulfur, accelerated hydrogen peroxide and miconazole/chlorhexidine are available. These products have been tested and reviewed both in vitro and in vivo, with excellent results,4–10 and as no rinsing is required, their administration is advisable in multiple cat situations. Some (enilconazole, lime sulfur) are indicated as first-choice options. 1 However, shampoo combines the antimycotic effect and the physical act of shampooing, 8 helping to remove fungal propagules, and is recommended in animals kept as pets. A combined 2% miconazole/chlorhexidine shampoo is largely available on the market and has been proven to be effective.8–10
In recent years, the interest in selecting sustainable products from landscape plants has increased and some data are available for M canis, indicating that a number of chemically defined essential oils (EOs) from several plants can yield antifungal activity both in vitro and in vivo.11–13 In particular, EOs derived from Thymus serpyllum, Origanum vulgare and Litsea cubeba have shown strong efficacy against several dermatophyte species.12,13 Such plant-derived compounds are of interest as they have not yet been manipulated by pharmaceutical industry. Herbal remedia are usually well accepted by pets and owners, and many owners are interested in alternative treatments.
The goal of the present study was to compare the antifungal efficacy of an EO-based shampoo with miconazole/chlorhexidine for topical haircoat disinfection in cats treated with oral itraconazole.
Materials and methods
Cats
Fourteen symptomatic cats affected by spontaneous dermatophytosis caused by M canis were included in the study after obtaining informed consent from the owners. The animals were of both sexes, of different breeds (11 domestic shorthairs and three Persians), with ages ranging from 3 months to 8 years.
Fungal infection was confirmed by direct hair examination, Wood lamp examination when possible and culture. Mycotic load was evaluated by counting colony-forming units (CFUs) as previously described, indicating each infection as heavy (⩾50 CFUs/plate), mild (49–5 CFUs) and low (<5 CFUs). 14
Inclusion/exclusion criteria
Inclusion criteria were the presence of local or generalised lesions due to M canis, associated with positive culture (heavy or mild) of hair drawn with the brush technique, achieved on Sabouraud CAF agar + actidione (Liofilchem). Animals treated from less than 6 months before the inclusion day and/or with concomitant dermatoses were not admitted to the study.
Scoring and monitoring
Skin lesions were evaluated at day 0 and scored. 9 In detail were considered ease of epilation (1 = within normal limits; 2 = mild but excessive; 3 = moderate; 4 = severe and extensive), degree of seborrhoea (1 = none; 2 = mild; 3 = moderate; 4 = severe) and extent of the primary lesions (1 = none; 2 = single, small area; 3 = more than one small area; 4 = extensive lesions). The three scores were then added to give a total lesion score. The number of skin lesions ranged from four to 12 for the two groups. The occurrence of human infection (n = 7) was also recorded. The owners of Persian cats were advised to clip their cats. Detailed data are reported in Tables 1 and 2.
Anamnestic and clinical data in treated cats, and lasting outcome of therapies
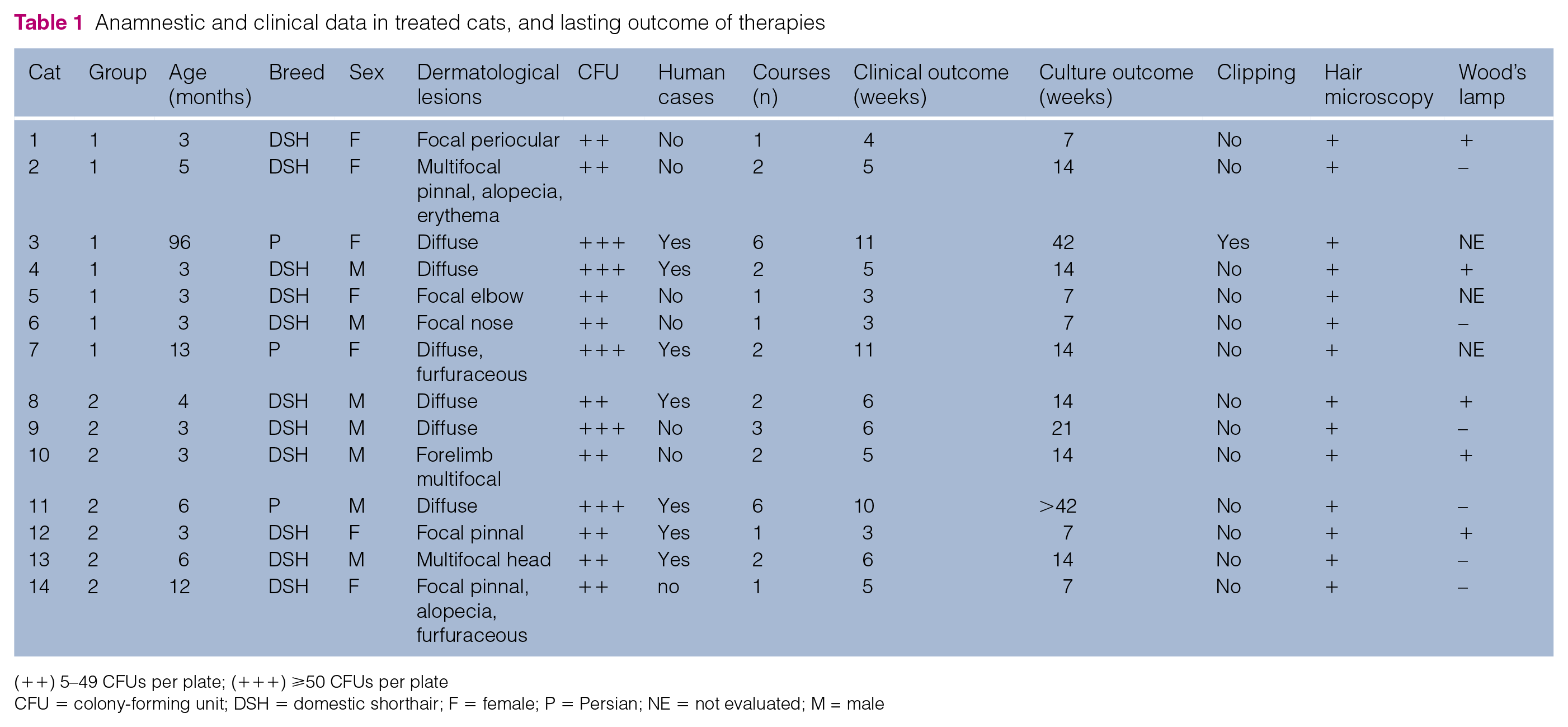
(++) 5–49 CFUs per plate; (+++) ⩾50 CFUs per plate
CFU = colony-forming unit; DSH = domestic shorthair; F = female; P = Persian; NE = not evaluated; M = male
Clinical score data before and after treatment in cats from groups 1 and 2

Study design
Diagnosis was achieved in different private clinics. Once diagnosed the cats enrolled in the present open study were assigned to two different groups at one time. The treatment took place at home. Cats in group 1 received oral itraconazole (Itrafungol; Eli Lilly Italia) at a dose of 5 mg/kg/day pulse administration for 1 week, every 2 weeks, with a total treatment of at least 6 weeks and they were washed twice a week with about 5 ml of a neutral shampoo with the added EOs of T serpyllum (2%), O vulgare and R officinalis (5% each). The EOs were provided by Flora; their chemical composition and proven antimycotic activities have been reported elsewhere. 11 The shampoo was immediately removed by rinsing after the application.
Cats in group 2 received oral itraconazole at the same dose plus 2% miconazole/2% chlorhexidine shampoo (Malaseb; Eli Lilly Italia) twice a week for the period of systemic treatment. The shampoo was left on the haircoat for 10 mins before rinsing with warm water. 8 All treatments were administered by one of the authors (AGC). Cats were examined weekly to evaluate an improvement of their clinical status and a fungal culture was achieved by brush technique. The animals were treated until they had two negative consecutive weekly cultures, 7 and the protocol treatment was stopped at week 44.
Statistical analysis between the groups was performed by means of Wilcoxon, and Mann–Whitney tests to evaluate significant differences in weeks needed to obtain mycological cure. Statistical significance was defined as P <0.01. When not all study subjects reach mycological cure, the adjusted Kaplan–Meier method was applied to evaluate the probability of healing.
To avoid passive contamination of haircoat, environmental mycotic pollution was monitored by the use of both an air sampler (Sas super-100 Air Sampler; PBI) and contact plates, as previously reported. 15 The owners were advised to clean the environment thoroughly by vacuuming followed by a deep clean with disinfectants available commercially in Italy (accelerated hydrogen peroxide, bleach, ammonium quaternary compounds and enilconazole). Cultural controls were repeated weekly.
Results
All enrolled cats had never been treated with antimycotic drugs, except for cat 3 (group 1), who had been treated with griseofulvin and then with ketoconazole, without any improvement of clinical and mycological features. Antimycotic treatments had been stopped about 1 year before the beginning of the present study.
Both treatments were well tolerated and adverse effects were not recorded. In group 1, two animals were clinically healthy at week 3 post-treatment, while all other cats were clinically healthy at week 11. All cats were culturally negative at the end of the trial. In group 1 cats, mean time to clinical and mycological cure was 6 weeks (median 4 weeks, range 3–11 weeks) and 15 weeks (median 14 weeks, range 7–42 weeks), respectively.
One cat in group 2 was dermatologically normal by week 3, while all cats were clinically cured at week 10. By the end of the study 6/7 animals were negative on fungal culture. For cats in group 2, the mean time to clinical and mycological cure was 5.9 weeks (median 6 weeks, range 3–10 weeks) and 12.8 weeks (median 6 weeks, range 7–21 weeks), respectively.
With respect to cats with extensive lesions, as expected, the speed of resolution was higher in cats with focal lesions, ranging from 3 to 4 weeks in group 1 cats and from 3 to 5 weeks in group 2 cats. Cats with diffuse lesions required more than one course of treatment (requiring 2–6 treatments for both groups) to achieve a mycological cure. A reduction in CFUs was observed by week 3 post-treatment in all examined cats. All cats with focal lesions had healed culturally at week 7. After the start of treatment no cases of new human infection or reinfection were reported. Two Persian cats out of three were randomly assigned to group 1; only one was clipped (the other Persian cat from group 2 was clipped as recommended).
With regard to aetiological cure, a significant difference between treatments was not observed (z score −0.1429; U = 19.5), even if the probability of healing at week 42 was 46% more for cats in group 1.
Detailed data on treatment outcome are reported in Tables 1 and 2.
The correct cleaning instructions given to owners led to environmental clearance from fungal propagules from week 1 post-treatment in all cases.
Discussion
Treatment with shampoo with added EOs yielded results comparable with conventional therapy. Owing to the lack of contact time required, it was particularly appreciated by the owners; the miconazole/chlorhexidine shampoo had to be left on the cats’ coats for 10 mins, and some cats exhibited nervous behaviour during this time.
Studies on the use of antifungal rinses to controldermatophytosis have been conducted on cats, bothnaturally and experimentally infected, living in catteries and other communities.4–10 To the best of our knowledge, our trial is the first study carried out on owned cats. We are aware of our small sample size, but this was due to the difficulty in the simultaneous recruitment of pet cats that met the inclusion criteria. Although the epidemiology and clinical situation in shelters is more controlled and homogeneous, such studies can not be fully applicable to indoor cats, living in very close contact with people, including children, in an environment with furniture, curtains, cushions and other household items. The best treatment protocol is difficult to identify, depending on the number of cats involved, the owner’s resources and global health of cats, 1 so the use of shampooing in such animals is advisable, while in catteries the rinse is more useful.
In the present study the determination of CFUs was applied to evaluate the efficacy of local treatment and the capacity of active compounds to limit the spreading of arthrospores. Both local treatments were able to decrease heavy and mild mycotic loads until elimination of arthrospores on the haircoat. An effective topical treatment together with correct management of environmental disinfection are of primary importance to cure dermatophytoses and to avoid reinfection and/or new infections. Therefore, in our study no relapses or new cases of human infections were recorded during the observation period.
Considering their potential toxicity, EOs should be carefully administered in animals, especially in cats. Oils from Thymus species are toxic when administered orally, 16 and carvacrol and thymol, the main components of both Thymus and Origanum oils are skin sensitisers and antigens, 17 so the use of these oils undiluted should be avoided. Rosemary oil is considered safe for mammals, although chronic exposure to rosemary oil at high concentrations has rarely been reported to cause contact dermatitis; acute toxicity of rosemary oil has not been reported. 18 In general, toxicity testing is concerned with pure single oils rather than mixtures. 19 In the present study, EOs as a mixture were administered to optimise their efficacy and to minimise toxic effects.
Nevertheless, even if EOs, properly diluted, are generally safe, attention must be paid to use chemically defined compounds, 20 under the supervision of a skilled phytotherapist.
Conclusions
On the basis of our observations, the use of shampoo with the added EOs of T serpyllum, O vulgare and R officinalis would seem a natural and interesting alternative to conventional topical treatment.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
