Abstract
Objectives
Despite comprehensive diagnostics, the aetiology of meningoencephalitis (ME) in cats often remains undetermined. As a result of recently published surveys, Encephalitozoon cuniculi has gained growing importance in cats not only with ocular disorders, but also with central nervous system disease. Therefore, it was hypothesised that E cuniculi may be an underestimated pathogen in the development of feline non-suppurative and/or granulomatous ME.
Methods
As a first step, histopathological sections of the brain of cats with encephalopathy were retrospectively reviewed to identify cases of granulomatous ME. In a second step, an immunohistochemical screening for detection of E cuniculi was performed in cases with ME of unknown origin.
Results
In 59/89 (66.3%) cats with ME, an aetiologically relevant pathogen was detected. Forty-three of 89 (48.3%) cats had a diagnosis of feline infectious peritonitis. In 14/89 (15.7%) cats, protozoan cysts were identified and infection with Toxoplasma gondii was confirmed by immunohistochemistry (IHC) in all cases. In 2/89 (2.3%) cats with granulomatous ME, fungal organisms were identified. Thirty of 89 (33.7%) cats with ME of unknown origin that underwent IHC for the detection of E cuniculi remained negative.
Conclusions and relevance
The results of this study suggest that E cuniculi is unlikely to be directly associated with (non-suppurative and/or granulomatous) ME in cats in Austria.
Introduction
Meningoencephalitis (ME) appears to be a frequent cause of neurological disorders in cats.1,2 According to histopathological surveys, inflammatory disorders have been identified in 30–45% of cats with central nervous system (CNS) disease.2,3 However, until now, only a few studies have been carried out regarding the aetiology of ME in cats.2–5 Granulomatous encephalitis still comprises a significant proportion of inflammatory diseases of the brain in cats. 4 Amongst them, feline infectious peritonitis (FIP), which typically causes pyogranulomatous ME, is the most frequently recognised infectious CNS disorder.3,4 Further causes for granulomatous ME in cats include protozoal, fungal, helmintic and some bacterial infections (eg, Mycobacterium species).1,3–6 Besides Toxoplasma gondii infections, Encephalitozoon cuniculi has been reported as an aetiological agent of granulomatous ME in some cats and therefore should be included in the list of differential diagnoses.7–9
Encephalitozoon cuniculi is known as an obligate intracellular microsporidian pathogen in mammals, including humans. Rabbits are the main host, in which infection usually runs a chronic latent course. 10 In rabbits, CNS, kidneys and eyes are primary target organs, where the pathogen commonly causes multifocal granulomatous ME, chronic interstitial nephritis and phacoclastic uveitis, respectively.11–14 Until now, E cuniculi infections in carnivores have primarily been reported in canids such as domestic dogs and farm-raised foxes, and mustelids, showing predilection sites and histopathological lesions that are (very) similar to those described in rabbits.15–17 Experimental E cuniculi infections in kittens have been shown to cause histopathological lesions (granulomatous ME and interstitial nephritis) compatible with those observed in other carnivores. 18 Meanwhile, it has become apparent that E cuniculi may have an underestimated aetiological role in cases of feline uveitis.14,19–20 Owing to a significant proportion of cats with granulomatous and/or non-suppurative ME, in which no causal agent can be identified, E cuniculi should be given particular attention as a possible causal pathogen of the condition.1,3,4,21 Therefore, it was hypothesised that E cuniculi might be an underestimated pathogenic agent in feline ME.
The aim of the present study was to evaluate retrospectively infectious causes of histopathologically diagnosed granulomatous and/or non-suppurative ME in cats. In addition, an immunohistochemical screening for detection of E cuniculi was performed in cases with ME of unknown origin.
Materials and methods
Histological brain sections of cats submitted for necropsy to the Institute of Pathology and Forensic Veterinary Medicine of the University of Veterinary Medicine, Vienna (Austria) between January 1999 and July 2012, where an encephalopathy was diagnosed histopathologically and immunohistochemically, were retrospectively reviewed to identify cases with inflammatory diseases. From 261 cats with encephalopathy, cases without inflammation (n = 171), neoplasia, degenerative diseases due to metabolic and/or circulatory disorders, malformation of the brain and traumatic injuries were excluded from further studies.
Ninety of 261 cats with an encephalopathy fulfilled the inclusion criteria of granulomatous and/or non-suppurative ME. One cat, in which a generalised E cuniculi infection had been diagnosed and published before, was excluded a priori from the present study. 9 Finally, 89 histopathological sections underwent statistical analysis. According to the pre-existing results, which were obtained as part of routine diagnostics using haematoxylin and eosin staining, in 59/89 a definitive diagnosis was evident owing to either pathognomonic histological morphology (FIP), special staining methods (Grocott’s methenamine silver [GMS] or periodic acid–Schiff (PAS) for predicted fungal infection), or immunohistochemistry (IHC; to confirm T gondii infections).
In the remaining 30 cases immunohistochemical examinations for the detection of rabies virus (performed in all cases with ME of unknown origin), Bornavirus species and Aujeszky’s disease virus, and feline herpesvirus type 1 (all of these viruses were only tested in selected cases) were conducted as part of diagnostic investigation before the survey and therefore the methods are not described in the present study.
According to our inclusion criteria, in all 30 cases which remained negative on testing for viral pathogens and did not match any known entity on the basis of typical morphology and/or immunohistochemical results, IHC was performed to assess for possible infection with E cuniculi.
Where protozoan cysts were present, which had not yet been further classified as the cause of the routine diagnostic investigation, IHC was performed in order to differentiate between cysts of T gondii and Neospora caninum.
IHC
For IHC, brain sections of areas with evident and characteristic histopathological lesions or areas with protozoan cysts were used. Immunohistochemical investigations using primary antibodies for the determination of infectious organisms (E cuniculi, T gondii, N caninum) were performed automatically on an autostainer (Lab Vision AS 360). Details concerning reagents and procedures are given in Table 1. Briefly, 3 µm paraffin-embedded sections of brain were placed on coated slides and dried to enhance tissue adherence. Antigen retrieval was performed on deparaffinised and rehydrated sections by heating in citrate buffer (pH 6) or pronase digestion (pH 8). Endogenous peroxidase activity was blocked by incubation in H2O2/methanol. After application of the primary antibody, a polymer detection system, consisting of a universal secondary antibody formulation conjugated to an enzyme-labelled polymer that recognises mouse and rabbit immunoglobulins, was used. The polymer complex was then visualised with an appropriate substrate/chromogen (diaminobenzidine [DAB]; Labvision/Thermo Fisher Scientific). Alternatively, biotinylated secondary antibodies were used, followed by incubation with streptavidin–peroxidase and visualisation with DAB. Subsequently, all sections were counterstained with haematoxylin, dehydrated and mounted.
Immunohistochemical reagents and procedures

HRP = horseradish peroxidase
For E cuniculi IHC, samples of a rabbit’s brain histopathologically characteristic and immunohistochemically positive for encephalitozoonosis, as well as brain samples of the kitten diagnosed with generalised encephalitozonosis, 9 served as positive controls. For immunohistochemical detection of T gondii, a section of cat brain with an immunohistochemically proven toxoplasmosis was used as a positive control. A section of kangaroo heart histopathologically characteristic and immunohistochemically positive for neosporosis served as positive control for N caninum IHC.
Statistical analysis
SPSS 14.0 (IBM) was used to perform a Student’s t-test in order to compare the mean age between cats with FIP and all other cats with non-suppurative and/or granulomatous ME. A χ 2 test was used to determine significant differences in breed between FIP cases and the rest of the cats.
Results
The 89 cats with ME had a mean age of 3.7 years and a median age of 1.2 years. Cats with FIP-induced ME were significantly younger (mean age 1.6 years; median age 0.7 years; P <0.001). The domestic shorthair breed represented 52/89 (58.5%) of affected cats. Regarding the breed distribution there was a significant difference between FIP cases and other cases (P <0.01). Maine Coon cats (n = 5) were only observed in cases with FIP. Both sexes were equally distributed. The cats in the present study comprised a mixed population of free-roaming and indoor-only animals.
In 59/89 (66.3%) cats, an aetiologically relevant pathogen was detected (Figure 1). The majority (43/89; 48.3%) revealed histopathological lesions typical for FIP (pyogranulomatous inflammation with typical involvement of plasma cells and neutrophils, localised around the lateral ventricles, in the meninges or the choroid plexus; Figure 2a). In 14/89 cases (15.7%) protozoan cysts, which were almost always accompanied by inflammatory and/or degenerative lesions, were diagnosed by histopathological examination. Fungal organisms associated with a granulomatous, inflammatory cell infiltrate were detected in 2/89 (2.3%) cats (Figures 1 and 2b).

Frequency of causative pathogens in 89 cats with non-suppurative and/or granulomatous meningoencephalitis.
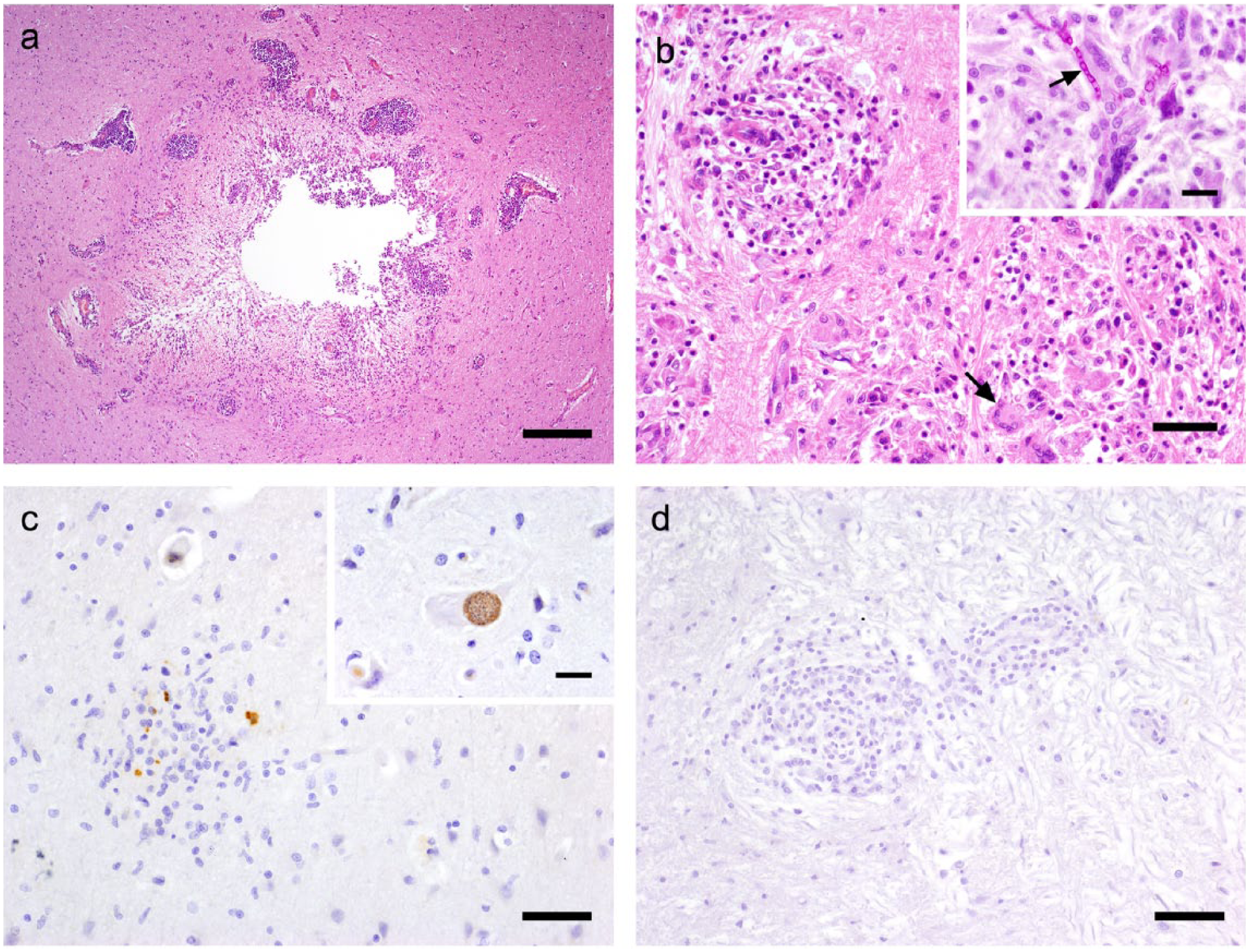
Granulomatous encephalitides in the cats. (a) Characteristic feline infectious peritonitis perivascular cuffing and pyogranulomatous ependymitis adjacent to the mesencephalic aqueduct (haematoxylin and eosin, bar = 400 µm). (b) Numerous fungal granulomas with giant cells (arrow; haematoxylin and eosin, bar = 150 µm); inset: fungal hyphae (arrow) in giant cells (periodic acid–Schiff; bar = 30 µm). (c) Immunolabelled tachyzoites of Toxoplasma gondii in an area of gliosis (bar = 80 µm); inset: specifically immunolabelled T gondii tissue cyst within a swollen axon (bar = 30 µm). (d) Aetiologically unclear granulomatous encephalitis of a cat remaining negative for Encephalitozoon cuniculi (bar = 80 µm)
Thirty of 89 (33.7%) cases with ME of unknown origin yielded immunohistochemically negative results for rabies virus in the course of routine diagnostic work-up. Furthermore, each of the selected cases with ME of unknown origin, were negative for Bornavirus species, Aujeszky’s disease virus and feline herpesvirus type 1 during routine diagnostic testing.
IHC, which was performed in the 14 cats with protozoan cysts, identified T gondii infection in all of these cases (Figure 2c), whereas IHC for N caninum and E cuniculi remained negative in all cases.
Furthermore, all 30 (33.7%) cats with non-suppurative and/or granulomatous ME of unknown origin or with an atypical histopathological appearance, yielded immunohistochemically negative results for E cuniculi (Figure 3).
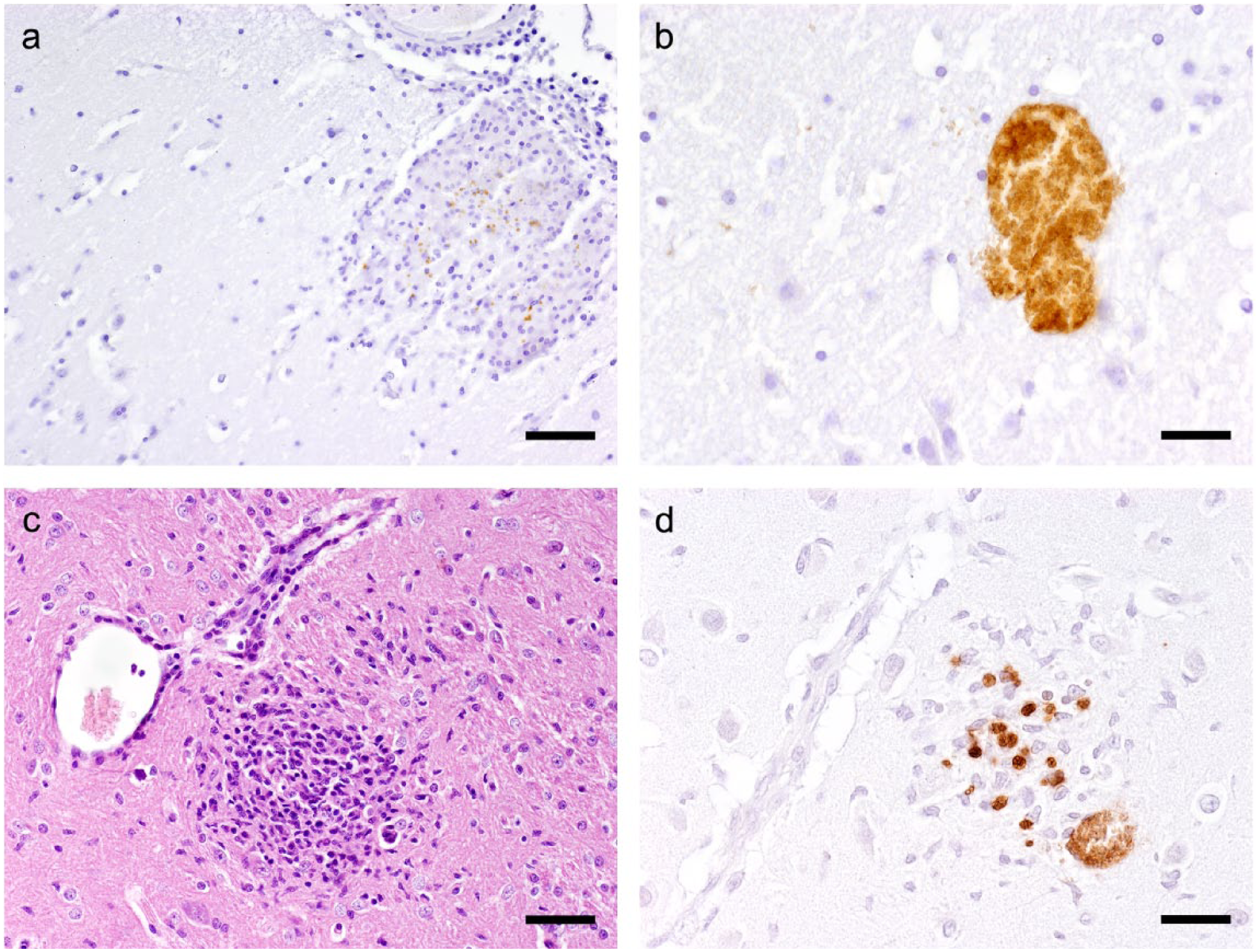
Brain sections from Encephalitozoon cuniculi-infected rabbit and kitten used for positive control for immunohistochemistry. (a) Submeningeal granuloma with specific brownish immunolabelling of E cuniculi spores in the rabbit brain (bar = 80 µm). (b) Specific immunostaining of a reactionless E cuniculi parasitophorous vacuole in the rabbit’s brain (bar = 40 µm). (c) Granulomatous encephalitis with reactive gliosis in the kitten (haematoxylin and eosin, bar = 80 µm). (d) Specifically immunolabelled E cuniculi spores and one parasitophorous vacuole in an area of gliosis in the brain of the kitten (bar = 40 µm)
Discussion
Based on histopathology and IHC results, ME was diagnosed in the cats of this study. A definitive cause for inflammatory brain lesions was detected in 66.3% of the cats with non-suppurative and/or granulomatous ME. The results of this study are similar to those of a previous study in which a causative agent was identified in 62.9% cats with ME. 4 Similar to previous studies, FIP was the most common cause for CNS disease in the cats of the present study.2–4 Moreover, in accordance with other reports, cats with ME due to FIP were significantly younger than the other animals with inflammatory CNS disorders.4,22 This is consistent with several surveys, in which cats <2 years of age with FIP (independent of organ involvement) were significantly overrepresented.23,24 Reasons for a greater risk of developing FIP in cats up to 2 years of age may be their immature immune systems, a higher viral load and stressful incidents that young cats are commonly confronted by.25,26 Although we observed a statistically significant difference between the percentage of certain breeds with FIP and cats with non-FIP encephalitis, owing to the low number of each different breed involved in the study it should not be overestimated.
Moreover, the percentage distribution of relevant pathogens responsible for (granulomatous) ME in cats of the present study (cases with FIP [48.3%], T gondii [15.7%] and fungal infection [2.3%]) revealed very similar results to the survey of Bradshaw et al (47 [51.1%] cases with FIP, eight [8.7%] with toxoplasmosis and one [1.1%] with a fungal infection). 4 However, in a previous study the number of cats with ME of unknown origin was even higher (approximately 50%) than in the cats of the present study (33.7%). 3 The reasons for the varying percentages in these studies may be different selection criteria, investigations procedures (eg, IHC was not performed in one study) and regional differences. 3 Only infections attributed to T gondii were relatively overrepresented in the present study than in other studies and may therefore be an underdiagnosed pathogen in cats with granulomatous ME.3,4 These differences may be due to regional differences in feeding habits (raw meat vs commercial diets) and husbandry conditions (outdoors vs indoors).27–30 However, ME due to an infection with T gondii may often run a subclinical course and it should be noted that diagnosis of ME in the present study was based on histopathological findings. 31 Similarly to other reports, fungal infections have been found to be rare in cats with ME.4,21 Nevertheless, special staining methods (GMS or PAS) should be applied in suspicious cases.
Several other pathogens (eg, feline leukaemia virus, feline panleukopenia virus, Bornavirus, rickettsiae) have been demonstrated to induce non-suppurative and/or granulomatous ME in cats.2,4,21,32,33 Owing to the pattern of inflammatory lesions (perivascular and parenchymal mononuclear infiltration, commonly associated with meningitis) viral agents or virus-triggered secondary immune-mediated mechanisms are believed to be the most probable cause for ME of unknown origin, although the proof is still lacking.1–4,34
In dogs, mainly puppies, E cuniculi has been detected as a cause of neurological and renal disease with histopathological lesions indicative of non-suppurative to granulomatous ME and nephritis.17,35 Encephalitozoon cuniculi has not been identified as a significant cause of chronic kidney disease in cats. 36 Nevertheless, it has been thought to cause neurological disorders in felids but has been sporadically documented.7,8 As a result of the diagnosis of generalised encephalitozoonosis in a kitten in Austria in 2011, the present study was conducted in order to evaluate whether E cuniculi may play a significant role in ME of unknown origin in cats. 9 However, in the present study, when this animal was excluded, all other cases in which an IHC for E cuniculi was performed, a negative result was determined.
Conclusions
Infections due to E cuniculi are increasing in cats. In the Austrian population of cats examined in the present study no evidence of E cuniculi as an aetiological pathogen for ME was found. Although a recognised cause of uveitis in cats, E cuniculi ME seems to be rare in cats.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
