Abstract
Objectives
The aim of the study was to describe the normal abdominal echoanatomy of the tigrina and to compare it with the abdominal echoanatomy of the domestic cat. Reference intervals for the normal abdominal ultrasonographic anatomy of individual species are important for accurate diagnoses and interpretation of routine health examinations. The hypothesis was that the echoanatomy of the tigrina was similar to that of the domestic cat.
Methods
Eighteen clinically healthy tigrina were selected for abdominal ultrasound examination, in order to obtain normal parameters of the bladder, spleen, adrenal gland, kidney, gastrointestinal tract, liver and gall bladder, and Doppler parameters of liver and kidney vessels.
Results
The splenic parenchyma was consistently hyperechoic to the kidneys and liver. The liver, kidneys and spleen had similar echotexture, shape and dimensions when compared with the domestic cat. The gall bladder was lobulated and surrounded by a clearly visualized thin, smooth, regular echogenic wall. The adrenal glands had a bilobulated shape. The urinary bladder had a thin echogenic wall. The Doppler parameters of the portal vein and renal artery were similar to the domestic cat.
Conclusions and relevance
The results support the hypothesis that the ultrasonographic parameters of the abdominal viscera of the southern tigrina are similar to those of the domestic cat.
Introduction
The tigrina (Leopardus guttulus) is an endangered species in Brazil,1–3 and efforts should be made by the scientific community to reverse the current situation. The tigrina is extremely difficult to observe in its natural habitat and is considered one of the least known Felidae; therefore, scientific references are scarce. 4 They are free-roaming wild animals in Brazil and South America, and are frequently involved in road accidents and are sometimes presented to small animal or wildlife veterinarians with the resultant injuries. Captive tigrinas may present similar infectious and non-infectious diseases to domestic cats, so ultrasonographic examination may be used for diagnostic purposes.
The use of ultrasound in wildlife medicine is growing, mainly because it is a tool that aids in geriatric assessments, quarantine and preimport situations; 5 however, diagnostic imaging in wildlife medicine is still a challenge owing to the lack of normal parameters that can guide the veterinarian. Ultrasound research has been conducted in order to obtain normal reference intervals (RIs) in non-domestic cats; for example, the cheetah (Acinonyx jubatus – Schreber, 1775), caracal (Caracal caracal – Schreber, 1776) and even in a study describing the renal anatomy of the tigrina.5–7
Ultrasound has the advantage of being non-invasive, quick, affordable and mobile, and can provide relevant information on abdominal organs.
The Doppler technique is a tool that monitors vascular disorders in, for example, the kidney and liver, and may indicate early changes in organ disease. Kidney disease is common in cats, especially in older cats, and Doppler ultrasound (DUS) has been used in domestic cats for kidney evaluation, 8 and in dogs for hepatic evaluation. 9
Therefore, the aim of this study is to describe the abdominal ultrasonographic anatomy of the liver, gall bladder, gastrointestinal tract, kidneys, adrenal glands, spleen and urinary bladder, and DUS of the portal vein and renal artery, in captive tigrina and to provide RIs for this species. It was hypothesized that abdominal ultrasonographic anatomy of the tigrina is similar to that of the domestic cat.
Materials and methods
Population sample
Eighteen tigrinas underwent ultrasonographic examination under anesthesia. The tigrinas were all originally free-living and were rescued from urban areas near São Paulo and Paraná (Southern and Southeastern territory of Brazil). This study was approved by the ethics committee of the São Paulo State University, the Sorocaba Zoo (Brazil) committee and the Brazilian Institute of Environment and Renewable Natural Resources. The healthy status of the tigrinas was assumed by history, clinical and laboratory examinations. The examinations were performed during routine annual examinations.
All the tigrinas were adults and were kept in captivity in the Sorocaba Zoo. The approximate age of the tigrinas was estimated by dental arcade and history of the individual. The age of the animals ranged from 17–48 months. The mean weight of the animals was 2.4 kg (range 1.8–3.5 kg).
Eleven tigrinas (four females, seven males) were examined at the Diagnostic Imaging Department of the São Paulo State University in Botucatu (group 1). Seven other tigrinas underwent ultrasonographic examination at Sorocaba Zoo (group 2). Animals were considered healthy based on history, and physical and clinical examination. Complete blood count, and kidney and liver function tests were performed and considered to be the RIs for this species, as RIs for biochemistry and hematology in tigrina have not been reported. The RIs were also within the normal limits for healthy domestic cats. All animals were fasted overnight, but water was given ad libitum until shortly before the examination. Blood samples were obtained by femoral venepuncture and about 3–4 ml of blood was collected from each cat. Urinalysis was not performed as the animals urinated during physical restraint.
Anesthetic protocols
Two anesthetic protocols were used. All the tigrinas were first physically contained by netting. Tigrinas in group 1 underwent anesthetic induction with ketamine hydrochloride (Ketamine 10%, 7 mg/kg IM; Agener) associated with dexmedetomidine (Dexdomitor, mean dose 15–30 mg/kg IM; Pfizer). Group 2 underwent general anesthesia with xylazine 2% (Calmium, mean dose 1–2 mg/kg IM; Agener) and 10% ketamine hydrochloride (Ketamine, 10 mg/kg IM; Agener), 10 and maintenance of anesthesia under isoflurane (Vetflurano; Virbac).
Ultrasonography
Group 1 underwent ultrasonographic examinations using a 7.5–10 MHz multi-frequency linear array transducer (Logic Model 3; GE). Tigrinas in group 2 underwent an examination with a 8–13 MHz multi-frequency linear array transducer (MyLab Alpha; Esaote). Each tigrina was positioned in dorsal and lateral recumbency; the ventral abdominal area was clipped of hair and ultrasound coupling gel was applied.
Ultrasound examination was performed systematically starting with the urinary bladder, left kidney, left adrenal gland, spleen, stomach, pylorus, duodenum, jejunum, colon, liver, gall bladder, right kidney and right adrenal gland. When a structure was not initially viewed another attempt was made before the end of the examination.
Abdominal images of anatomic structures were evaluated for size, shape, position, echotexture and echogenicity. The structures were measured during the examination by manual positioning of electronic markers on frozen images.
The bladder was evaluated for filling, content and wall thickness. The kidneys were evaluated for shape, contour, dimensions, corticomedullary distinction, echogenicity and echotexture.
Luminal bladder volume was assessed subjectively when little or moderate anechoic fluid was identified. The bladder wall was measured at the ventral wall. The length of the kidneys was measured in the sagittal plane and the width was acquired in the transverse plane. Renal volume was calculated according to the formula for ellipsoids previously reported for measurement of renal volume in tigrinas. 7 The resistivity index, pulsatility index (PI), mean velocity, peak systolic velocity and end diastolic velocity were measured in the renal artery at the renal hilum in all animals, with special attention paid to the angle of insonation.8,11
The length and height of the adrenal glands were measured in the sagittal plane and the width was acquired in the transverse plane. The wall thickness of the gastric fundus, pylorus and gall bladder were measured at the ventral wall. The gastric wall was measured in regions where gastric folds were not present and at least two measurements were performed in order to achieve the average wall thickness. The duodenum, jejunum and colon had mural thickness measurements made from the echogenic mucosal surface to the outer hyperechoic serosal surface in the transverse plane. No quantification of intestinal motility or peristalsis in any portion of the gastrointestinal tract was made. The splenic thickness was measured at the level of the splenic hilum. 12
Portal vein diameter was obtained in the transverse plane in the right hilar region. Portal and hepatic veins were identified by mapping with color Doppler with the sample volume positioned in the hepatic parenchyma. Caudal vena cava and Doppler measurements of portal vein flow were taken in the transverse plane at the level of the 10th and 11th right intercostal spaces. 11 The average flow velocity of the portal vein was measured by uniform insonation. 13 The pancreas, ileum, lymph nodes and other large abdominal vessels were not evaluated in this study.
Data analysis
The mean, range and SD were calculated for each variable. Paired t-tests were used to compare dimensions of the left vs right kidneys; mural thickness of layer thickness of the stomach; and the left vs right adrenal gland dimensions. Statistical significance was accepted at P ⩽0.05. Data are expressed as mean ± SD.
Results
There are no current RIs for standard hematology and biochemistry of the tigrina. However, the results of the 18 animals studied herein were within normal limits when compared with domestic cats, and are therefore assumed to be normal. Techniques for the evaluation and imaging of abdominal organs are grouped and also presented individually below. Table 1 summarizes the ultrasonographic findings of the spleen, gall bladder, gastrointestinal tract and adrenal glands and their respective measurements. Table 2 shows the ultrasonographic findings of the kidneys.
Ultrasonographic measurements of the portal vein, caudal vena cava, spleen, gall bladder, stomach, adrenal glands and urinary bladder of healthy captive tigrinas

Ultrasonographic measurements of the kidneys and urinary bladder of healthy captive tigrinas (n = 18)

Data are mean (range) ± SD
Liver and gall bladder
The liver was visualized cranially to the stomach in all tigrinas. The right portion of the liver was more pronounced, extending caudally to the costal arch in all individuals. Smooth and sharp lobar edges were identified. The echogenicity of the liver compared with the spleen ranged from hypoechoic (13/18) to isoechoic (4/18) and only one individual hepatic parenchyma was hyperechoic.
The cross section on the right side, between the 11th and 12th intercostal spaces was considered the best region for access to the portal vein, vena cava, hepatic hilum, gall bladder and aorta. The wall of the gall bladder was hyperechoic in all animals and it was filled with anechoic luminal content; however, hyperechoic dots were also seen, detected in 55% (10/18) of the cats. The gall bladder was located cranioventrally among hepatic lobes in the medial region of the abdomen and it was lobulated in 83% (15/18) of animals. The common bile duct was identified in 15/18 animals and had a slightly tortuous appearance, hyperechoic walls and anechoic content (Figure 1). The average diameter of the common bile duct was 0.3 cm. The duodenal papilla was not identified.

Ultrasound image of a sagittal section of the liver and gall bladder (GB) of a male, 6-year-old tigrina. The arrows indicate the dorsal and ventral wall of the common bile duct
The extrahepatic portal vein was identified in 15 animals and the presence of echogenic walls also facilitated the identification of the hepatic hilum. The hepatic parenchyma portal vessels showed a hyperechoic wall when compared with hepatic veins, and the main portal vein branched left and right. The portal vein was identified in the hilar region – porta hepatis – through the transverse plane between the 10th and 11th intercostal spaces in 15/18 tigrinas and was identified with hepatopetal flow. The spectral wave form was parabolic and relatively constant with some variations according to respiratory movements. The average velocity of the vein is described in Table 1. The caudal vena cava was measured in the hilar region in eight tigrinas (Table 1).
Spleen
The spleen was identified caudolaterally to the left kidney and craniodorsally to the gastric fundus in all tigrinas. The shape of the spleen ranged from triangular to elongated, and showed a regular and echogenic capsule. The splenic parenchyma presented and a homogeneous echotexture (Figure 2). The average thickness of the spleen in the transverse plane was 0.87 cm. Branches of the splenic vein were identified as anechoic structures within the splenic parenchyma and exiting the spleen at the hilum.
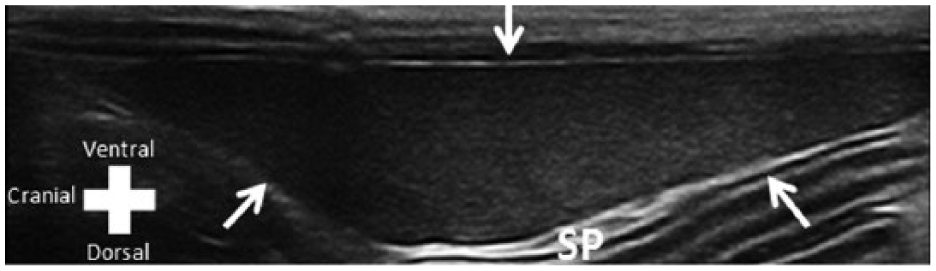
Cross-sectional ultrasound image of the spleen (SP) of an approximately 5-year-old male tigrina. Arrows delimit the SP
Adrenal glands
The right and left adrenal glands were identified in all of the cats and were located craniomedially to the respective kidneys. Corticomedullary differentiation was detected on the left adrenal gland in 67% (12/18) of tigrinas and in the right adrenal gland in 61% (11/18), and the cortex was consistently hypoechoic relative to the medullary region. The left adrenal was bilobulated and scanned parallel to the aorta (Figure 3a). The right adrenal gland was generally more elongated than the left, but retained the bilobulated shape, and, owing to the subcostal approach, was more often positioned at oblique angles to the caudal vena cava (Figure 3b). The left and right phrenicoabdominal veins were identified passing over the midbody of the respective adrenal gland. The average length of the left adrenal gland was 1.17 cm (range 0.97–1.32) cm and the mean height was 0.39 cm (range 0.29–0.53 cm). The average length of the right adrenal gland was also 1.17 cm (range 0.92–1.37 cm) and the mean height was 0.35 cm (range 0.29–0.50 cm).

Longitudinal ultrasonographic image of the (a) left and (b) right adrenal gland in an adult male tigrina. Cursors indicate glands limits. (a) The left adrenal gland is located between the left kidney (LK) and the aorta (arrow). Color Doppler was used to identify the flow of the aorta and the LK. (b) The right adrenal gland is located adjacent to the right renal artery (arrow) and next to the aorta (arrowhead). The phrenicoabdominal vessel crosses the mid-body of the gland. ADRENAL DIR = right adrenal gland
Gastrointestinal tract
The stomach was visualized in all animals and was positioned caudal to the left hepatic lobes and craniomedially to the left kidney. Four layers of the gastric wall were observed in all individuals. The gastric mucosa and muscle layer were hypoechoic, whereas the submucosa and serosa were hyperechoic (Figure 4). The luminal content of the stomach was predominantly gaseous. The thickness of the gastric wall and pylorus was a mean of 0.24 cm (range 0.20–0.35). The duodenum and jejunum had a luminal mucous pattern and had a similar anatomic position to that reported in domestic cats. 14 The wall thickness of the duodenum was significantly greater than that of the jejunum (P = 0.01) and colon (P <0.01). In all tigrinas the descending colon had a luminal gas pattern. The colonic wall was significantly thinner compared with the wall thickness of the stomach (P = 0.02), duodenum (P <0.01) and jejunum (P <0.01). Duodenal, jejunum and colon wall thickness measurements were as follows: duodenum (mean 0.23 cm; range 0.20–0.25 cm), jejunum (mean 0.22 cm; 0.21–0.25 cm) and colon (mean 0.16 cm; range 0.14–0.18 cm).

Transverse ultrasound image of the collapsed gastric fundus of an adult female tigrina
Urinary system
The bladder was identified cranial to the pubis and was visualized in all tigrinas. In 83% (15/18) it presented little luminal anechoic content; the other three tigrinas presented a moderately filled bladder with anechoic content (urine). Anechoic urine was observed in all tigrinas, and floating point echoes, which did not form acoustic shadowing, were present in five cats.
The bladder wall had a flabby appearance in all cats, and the descending colon – positioned dorsally – indented the ventral bladder wall. It was possible to identify three layers in the bladder wall, and they alternated in nature (hyperechoic, hypoechoic and hyperechoic) (Figure 5). The average thickness of the bladder wall was 0.20 cm (range 0.35–0.11 cm). The urethra extended caudally from the bladder into the pelvic canal and had an echogenic wall.

Longitudinal plane of the urinary bladder of an adult male tigrina. The bladder shows the stratification of three layers and scant luminal content. The animal urinated upon restraint. Cursors delimit the thickness of the bladder wall
Kidneys
In all tigrinas, the left kidney was visualized caudally to the gastric fundus (Figure 6), and the right kidney was visible adjacent to renal fossa of the liver. The echogenicity of the left renal cortex was compared with the splenic parenchyma and appeared as hypoechoic in 83% (15/18) of the tigrinas; one presented isoechogenicity and two showed increased echogenicity. The right kidney cortex was compared with the liver and was found to be hypoechoic (9/18), isoechoic (3/18) and hyperechogenic (6 /18). A clear distinction between cortex and medulla was identified in 83% (15/18) in the left kidney and 67% (12/18) in the right kidney. The ratio between renal cortex and medulla was 1:1.

(a) Dorsal and (b) transverse planes of the left kidney of an adult male tigrina. Arrows delimit peripheral cortex/renal capsular surface
One tigrina showed the corticomedullary rim sign. Cortical renal cysts were present in two males. The cysts had an anechoic content and were circular in shape with thin capsules measuring approximately 0.5 cm in diameter. One cyst was located in the right kidney of a male tigrina and another individual had a kidney cyst in the left kidney.
After B-mode measurements, color Doppler mapping of the renal vasculature and pulsed spectral Doppler study of the renal artery at the hilum were performed with a sample area of 2 mm (Figure 7).

Triplex Doppler ultrasonography of the renal artery of the right kidney in an adult male tigrina. Peak systolic velocity = 1; end diastolic velocity = 2
Student’s t-test revealed no significant difference between the measurements from the left and right kidney measurements.
Discussion
Tigrinas in captivity exhibit cautious behavior, 15 which hinders the observation of clinical signs of disease; therefore, abdominal ultrasonography may be a valuable diagnostic tool in these animals. In this study, abdominal ultrasonography, with transducers of 10 and 13 MHz, provided images of abdominal organs in the tigrina. Owing to time constraints on examination and anesthesia, other abdominal structures were not identified in this study.
The location of the hepatic parenchyma is similar to the domestic cat. 16 The spectral trace of the portal vein was similar to that found in non-sedated cats and cheetahs (A jubatus) under anesthesia.6,16 The average velocity of the portal vein is within the RI for domestic cats (10–18 cm/s), as are the diameters of the portal vein and caudal vena cava. 16
The relative echogenicity of the liver of tigrinas compared with the spleen and kidney had the same relation as other species such as dogs and cat, 16 and big cats like the cheetah. 6 Considering that none of the tigrinas showed clinical signs of disease, hepatic echogenicity is probably a poor parameter for determining normality. Ultrasonographic appearance may be insufficient to discriminate among canine and feline diffuse infiltrative liver diseases, 17 and this may also apply to other species such as the tigrina. The location of the gall bladder in the tigrina is typical of domestic cats. The lobulated gall bladder is common in cats and has been reported in domestic cats. 18
The relative status of a ‘full’ gall bladder can be attributed to preanesthetic fasting. Ten tigrinas (55%) had hyperechoic debris in the lumen of the gall bladder; this finding may represent a high incidence of biliary sludge in wild cats in captivity, as reported in normal domestic dogs but not in the cheetah.6,19 Gall bladder sludge is an unusual ultrasonographic finding in cats that is predictive of increased liver enzymes and total bilirubin. 20 More studies are needed to establish potential links between gall bladder sludge and hepatobiliary disease in the tigrina. The tortuous bile duct is compatible with other species of cat, such as the domestic cat and the cheetah.6,21
The thickness of the tigrina gall bladder wall was at the upper limit of the RI for domestic cats (0.06–0.09 mm) 14 and larger cats – with an average weight of 10 kg – such as the caracal (C caracal) (0.6–0.9 mm). 5 The thickening of the gall bladder wall is an important finding as gall bladder disease in domestic cats. 14
The location of the spleen in tigrinas is similar to the domestic cat and its ultrasonographic identification was easily accomplished. In some tigrinas, a dorsolateral approach was needed for better identification. Often, an oblique angle may falsely increase the thickness of the organ. The RI found for the thickness of the spleen was similar to that described in domestic cats. 12
An asymmetrical shape of the adrenal gland is also reported in domestic dogs and cats.16,22 The average length of the adrenal gland in the tigrinas was higher (1.17 cm) than the average reported length in domestic cats (1.07 cm), but the average height (0.41 cm) was similar to that of domestic cats (0.43 cm). 22 The adrenal shape of the tigrina differs from reports in the domestic cat. Variation in the shape of the adrenal glands is also seen in domestic cats and dogs. 16 Further investigation is warranted on the adrenal shape of the tigrina and its relation with captive vs non-captive animals and cortisol production.
The stomach, pylorus, duodenum, jejunum and colon showed no difference to domestic cats regarding, anatomic position, stratification and wall thickness. 23
The distinction between cortical and medullary regions of the kidney was clearly observed in most animals (15/18), as in normal domestic dogs and cats. Poor corticomedullary differentiation in dogs and domestic cats can be attributed to the presence of diseases such as congenital renal dysplasia, chronic inflammatory disease and chronic kidney disease. 24 The renal cortex echogenicity was hypoechoic compared with the hepatic parenchyma, and this correlation is similar to that previously reported in domestic dogs and cats,16,25 and also agrees with findings in large cats such as cheetahs. 6
Renal dimensions were similar to those found in a previous study of renal ultrasound evaluation of healthy tigrinas. 7 The results obtained in this study showed no difference in kidney size between males and females, or between the left and right kidneys, confirming the previous data. 7 Eight of the 36 kidneys evaluated were hyperechoic compared with adjacent organs, which can be considered abnormal for domestic cats and dogs. 16 This finding may be incidental in the tigrina and can be explained – similar to the domestic cat – by the presence of fat vacuoles in renal cortical tubular epithelium.26,27 However, renal biopsy would be required to confirm the diagnosis.
The medullary rim sign may be an incidental finding in cats and dogs or can represent nephritis, hypercalcemic nephropathy or leptospirosis.28–30 The medullary rim sign appears to be a non-specific ultrasonographic sign; however, the possibility cannot be excluded that it may be a sentinel sign of subclinical renal disease. 31 Although no renal biopsy was performed, the medullary rim sign was assumed to be an incidental finding in this study as the animals were healthy and presented no clinical signs of renal disease. Renal cysts found in two males did not alter renal dimensions previously reported in the tigrina. 7
The renal artery arises laterally from the aorta and the renal hilum protrudes, branching within the kidney in the tigrina. The resistivity index can be altered in dogs anesthetized with ketamine, 32 and this may have influenced the values obtained in the tigrina. However, the values obtained are within the normal range when compared with domestic cats (>0.70) and large cats such as cheetahs (>0.54).6,13,33 The PI of the renal artery in the tigrina (PI 1.85) showed results comparable within the normal range for domestic cats anesthetized with isoflurane (PI 2.8). 33
The wave morphology of the renal artery presented a parabolic flow, as reported in unsedated domestic cats. 8 The peak systolic velocity and end-diastolic velocity showed higher values than in non-anesthetized domestic cats; 8 however, there are no reports that discuss the influence of anesthesia for these parameters in wild and domestic cats. The influence of anesthetics on these values should be studied as the diagnosis of renal diseases in wild animals requires anesthesia in order to perform diagnostic imaging examinations. Kidney failure has a high morbidity among domestic cats and a decrease in blood supply to the kidneys may be the first sign of kidney dysfunction.8,34 The prevalence of kidney disease in the tigrina is currently unknown. Other renal arteries such as the interlobar arteries and arcuate were not evaluated owing to limited examination time.
The bladder was often found empty, as the tigrinas urinated upon physical restraint, and this may have influenced the values of the wall thickness. Bladder location and ultrasonographic appearance were similar to the dog and domestic cat, 16 and larger cats – with an average weight of 10 kg – such as the caracal (C caracal) 5 . The average thickness of the bladder wall in the present study (0.20 cm) was slightly higher than that reported in domestic cats (range 0.13–0.17 cm) 35 and within the normal range for dogs (0.14–0.23 cm). 36 However, the thickness of the bladder can vary according to the degree of distension, 36 and this may slightly increase the average thickness of the bladder wall.
Five (5/18) tigrinas had echogenic debris floating in the bladder. The thickness of the bladder wall was similar to those animals that had no debris in the bladder lumen, and echogenic points in the lumen of the bladder may be a normal finding in normal domestic cats. 16 Ultrasonographic echoes within the urinary bladder of clinically normal cats may be due to urine lipid. 37
Ultrasound is an imaging modality that provides information about the shape, position, architecture and vasculature of the abdominal organs of the tigrina. Differences in ultrasonographic anatomy and DUS parameters occur in different species. Thus, knowledge of the abdominal ultrasonographic anatomy and parameters may have a positive impact in the preservation of species.
Conclusions
This study documents the normal ultrasonographic features of the abdominal organs of clinically healthy southern tigrinas kept in captivity. The ultrasonographic parameters of the structures studied in the southern tigrina are similar to those found in domestic cats. These data may be useful in the diagnosis of diseases that affect the tigrina.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funded by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP).
