Abstract
Objectives
The aim of the study was to evaluate the end-tidal concentration of isoflurane required to maintain heart and respiratory rate within ± 20% of basal measurement in cats undergoing ovariectomy that received buprenorphine, tramadol or a combination of both.
Methods
Thirty cats, divided into three groups, were enrolled in a simple operator-blinded, randomised study. Cats received acepromazine (0.03 mg/kg) and one of the following treatments: buprenorphine (0.02 mg/kg), tramadol (2 mg/kg) or a combination of both. Anaesthesia was induced with propofol and maintained with isoflurane titrated in order to maintain heart and respiratory rate within the target values recorded before premedication.
Results
Groups were similar for age, weight, dose of propofol administered, sedation and recovery scores. Cats receiving tramadol with buprenorphine were extubated earlier after isoflurane discontinuation. No statistical differences were detected in end-tidal fraction of isoflurane between buprenorphine alone or with tramadol. In cats that received tramadol or buprenorphine alone, ovarian pedicle traction caused a statistical increase in end-tidal isoflurane concentration compared with that measured during incision and suture of the skin. In cats that received the combination of tramadol plus buprenorphine no differences among surgical time points were observed.
Conclusions and relevance
Tramadol added to buprenorphine did not provide any advantage in decreasing the end-tidal fraction of isoflurane compared with buprenorphine alone, although it is speculated there may be an infra-additive interaction between tramadol and buprenorphine in cats.
Introduction
Perioperative multimodal analgesia is a technique that associates two or more analgesics with different mechanisms of action in order to improve pain control during surgery and the early postoperative period.1–4
Buprenorphine, a semisynthetic opioid with a long duration of action, is licensed as a postoperative analgesic in cats. This drug gained popularity for use in feline anaesthesia as it causes less nausea, vomiting, dysphoria, hyperthermia, cardiovascular and respiratory depression than other opioids.1,5 Buprenorphine is a partial agonist of mu (μ) opioid receptors (MOR) and has a high receptor affinity but lower intrinsic activity than morphine.4–6
Non-steroidal anti-inflammatory drugs are frequently associated with buprenorphine during ovariohysterectomy in cats.2–4 However, cats are more sensitive to the renal side effects of this class of drugs so they should be administered with caution in dehydrated, hypovolaemic or hypotensive patients. 1
Tramadol is a centrally acting analgesic, which provides intra- and postoperative pain relief for ovariohysterectomy.7,8 Tramadol decreases the minimum alveolar concentration (MAC) of volatile anaesthetics and produces dose-dependent analgesia with minimal cardiovascular and respiratory side effects.1,7,9 Its mechanism of action is only partially mediated by MOR; indeed, tramadol activates the monoaminergic descending inhibitory pathway to the spinal cord and reduces noradrenalin and serotonin reuptake into the neuronal terminal. 10 The main metabolite of tramadol, O-desmethyl-tramadol (M1), has higher affinity for the MOR than the parental compound. Compared with other species, M1 plasma concentration in cats is detected rapidly after tramadol administration.7,11 Tramadol with its peculiar mechanism of action could be added to buprenorphine for a multimodal approach to control acute intraoperative nociceptive stimulation in cats. In dogs the MAC of sevoflurane measured during an infusion of morphine was statistically decreased if tramadol was added to the infusion. 12
The aim of this study was to evaluate the effect of buprenorphine, tramadol or a combination of both on the end-tidal concentration of isoflurane in cats undergoing ovariectomy during selected surgical periods. The absence of significant differences in end-tidal concentration of isoflurane between cats treated with the analgesic combination compared with cats treated with each single agent was considered as a null hypothesis.
Materials and methods
Animals
The ethical committee of the University of Padua approved this study (Protocol 199912/2014) and the owner’s consent was obtained before anaesthesia. Thirty American Society of Anesthesiologists grade I and II cats admitted to the veterinary teaching hospital for elective ovariectomy were enrolled in a simple operator-blinded, randomised study. The animals were judged to be healthy, based on preanaesthetic examination. Cats younger than 5 months or older than 5 years, pregnant, weighing less than 2 kg, with a fractious temperament or treated with analgesics in the previous 2 months were excluded. Cats were fasted overnight and admitted to the hospital in the morning and surgeries were performed in the afternoon. The animals were housed in single cages with free access to water up to 1 h before premedication.
Treatments
Cats were divided into three groups using an online randomiser program (Research Randomizer Version 4.0; www.randomizer.org). All cats received acepromazine intramuscularly at 0.03 mg/kg (Prequillan; Fatro) combined in the same syringe as follows: group TR, tramadol at 2 mg/kg (Altadol; Formevet); group BU, buprenorphine at 0.02 mg/kg (Temgesic; Schering Plough); and group TR-BU, tramadol at 2 mg/kg and buprenorphine at 0.02 mg/kg. The mixture was prepared and injected by a clinician not involved in the study. Thirty minutes later, a 22 G intravenous (IV) catheter was inserted in the right cephalic vein and IV propofol was administered for the induction of anaesthesia. Lidocaine, 1.6 mg (Lidocaina 2%; Pfizer) diluted in sterile water for injection up to 0.2 ml, was sprayed on the larynx and then the trachea was intubated with a 3 or 3.5 ID endotracheal tube. Anaesthesia was maintained with isoflurane (Vetflurane; Virbac) delivered in 100% oxygen via an Ayre’s T-piece with a minimum fresh gas flow of 2 l/min.
A mixture of amoxicillin/clavulanic acid (0.1 ml/kg, Synulox; Fatro) was injected subcutaneously after induction. In the operating theatre, cats were placed in dorsal recumbency on an electric heating pad (Pet-Mat; Dale Ecotech). A multiparameter monitor (Datex S/5; GE Healthcare) continuously measured the respiratory rate (RR), end-tidal fraction of isoflurane (Fe’ISO), expired partial pressure of carbon dioxide (Pe’CO2), heart rate (HR) as displayed from an electrocardiogram, and oxygen saturation of haemoglobin (SpO2) and pulse oximetry plethysmogram as assessed using a clip probe placed on the tongue. An oesophageal temperature probe measured body temperature. Indirect arterial blood pressure (DoBP) was measured with an ultrasonic Doppler flow detector (Vettex; Huntleigh Diagnostics) placed on the clipped skin over the palmar digital artery. A paediatric cuff with a width of approximately 40% of the diameter of the leg was placed immediately below the elbow.
Two staff surgeons, along with a fifth-year veterinary student, performed a standard ventral midline approach ovariectomy. Lactated Ringer’s solution (Ringer lattato SALF; SALF Laboratorio Farmaceutico) was infused along with all the anaesthesia at 5 ml/kg/h. At the end of the surgery, but before isoflurane discontinuation, meloxicam at 0.2 mg/kg (Metacam; Boehringer Ingelheim) was injected subcutaneously.
Measurements
The basal HR and RR were measured before premedication; the dose of propofol required for anaesthesia induction was noted. A simple descriptive scale was used to assess sedation 30 mins after premedication and during recovery (Table 1).3,13 The time from isoflurane discontinuation to endotracheal tube removal and the time to regain sternal recumbency were also noted. Oesophageal temperature at the time of arrival in theatre and before extubation was recorded.
Simple descriptive scale of sedation and recovery scoring criteria

The Fe’ISO was adjusted to maintained HR and RR within ± 20% of basal values at the time of skin incision (Tincision), right ovarian pedicle traction (Tpdx), left ovarian pedicle traction (Tpsx) and during the suture of the skin (Tsuture). The vapouriser was initially set at 1% isoflurane. A 20% increase in either the HR and/or RR or spontaneous movement during surgery were taken as indicating inadequate anaesthesia. In such a situation, surgery was interrupted and Fe’ISO was increased by 0.2% and after 10 mins of equilibration, surgery was started again. This procedure was repeated until values returned to within the limits. If HR or RR were below the basal measurement Fe’ISO was decreased by 0.2% but, nevertheless, it was maintained >0.6%, in order to avoid any involuntary movement. A DoBP <80 mmHg for >10 mins was treated by increasing fluid to 15 ml/kg/h. If the blood pressure was still <80 mmHg after 10 mins a bolus of lactated Ringer’s at 5 ml/kg was administered and if the blood pressure remained <80 mmHg dopamine was infused at 7.5 µg/kg/min. A HR <100 beats/min was managed with atropine 0.02 mg/kg and the cat excluded from the study.
Statistical analysis
Sample size was calculated to detect a difference of 0.3% (SD of 0.1%) in Fe’ISO between group TR-BU and group TR or group BU, with a power of 0.85 and an alpha (α) of 0.05; the analysis returned a minimum sample size of eight cats per group. Normally distributed data were expressed as a mean ± SD, otherwise a median (interquartile range) was used. Differences between groups for age, weight, dose of propofol used, body temperature at arrival in the theatre and at the end of anaesthesia, sedation and recovery score, time from discontinuation of isoflurane to extubation and to sternal recumbency were analysed with an analysis of covariance linear model with protocol as fixed effects, and weight as a covariate, or with a Kruskal–Wallis non-parametric analysis of variance, as appropriate. A repeated linear mixed model was used to analyse Fe’ISO, HR, DoBP, RR and Pe’CO2 over the four surgical time periods. Treatment, time and their interaction were inserted as fixed effects and the animals as random effects; Bonferroni post-hoc pairwise comparison test between least squares means was used if statistical differences were present. Analyses were performed with a statistical package (SAS V.9.1; SAS Institute), and P <0.05 was considered statistically significant.
Results
Groups were similar for age and body weight (Table 2). Anaesthesia and the postoperative period were uneventful, and all cats completed the study. Thirty minutes after premedication, sedation was poor and the cats were calm but alert (Table 2). The dose of propofol for anaesthesia induction was not statistically different between groups and ranged between 4.2 and 6.5 mg/kg (Table 2). Recovery score was not influenced by the protocol used and was scored ‘good’ or ‘perfect’ in all of the cats. The anaesthesia time was not statistically different among treatment groups and ranged from 40–80 mins. Cats in group TR-BU were extubated earlier after isoflurane discontinuation compared with those in group TR and group BU (P = 0.013). No difference in time to regain spontaneously sternal recumbency after extubation was observed among groups. The oesophageal temperature measured at arrival in the theatre was higher in group TR-BU compared with the other two groups (P = 0.001), but the temperature at recovery was not statistically significantly different among groups (P = 0.075). None of the above parameters were influenced by the weight of the cats.
Results of analysis of covariance or Kruskal-Wallis non-parametric analysis of variance of the effects of tramadol (TR), buprenorphine (BU) and tramadol + buprenorphine (TR-BU) mixture
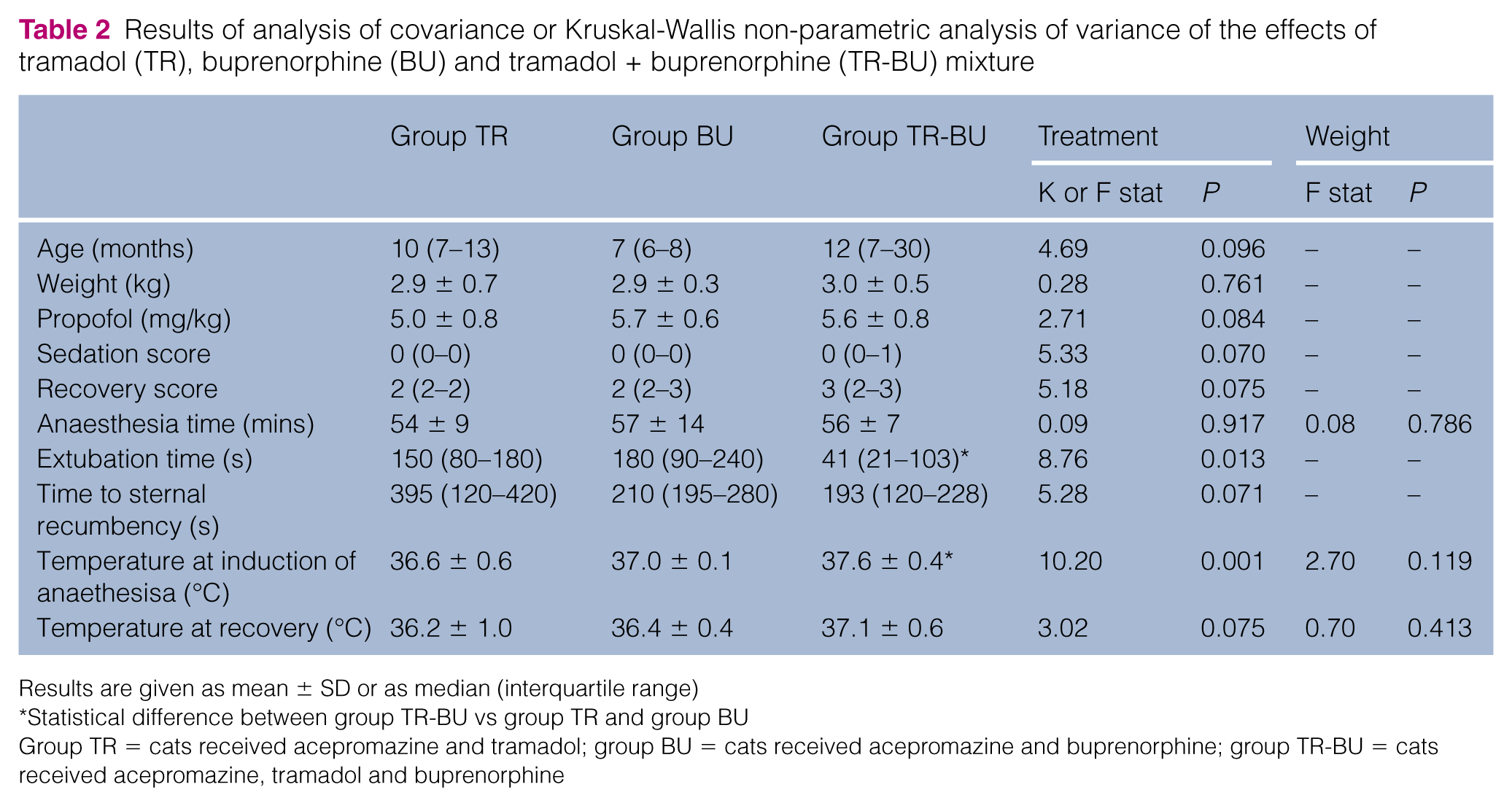
Results are given as mean ± SD or as median (interquartile range)
Statistical difference between group TR-BU vs group TR and group BU
Group TR = cats received acepromazine and tramadol; group BU = cats received acepromazine and buprenorphine; group TR-BU = cats received acepromazine, tramadol and buprenorphine
Treatment, time and their interaction affected the amount of isoflurane delivered during surgical time points. Among the groups, Fe’ISO at Tincision and Tsuture did not differ statistically (Figure 1), whereas cats in groups TR required a higher Fe’ISO at traction of both ovarian pedicles than those in group TR-BU (P <0.001). In group TR and group BU, Fe’ISO was statistically lower at Tincision than at the time of both ovarian pedicle traction or skin suture.
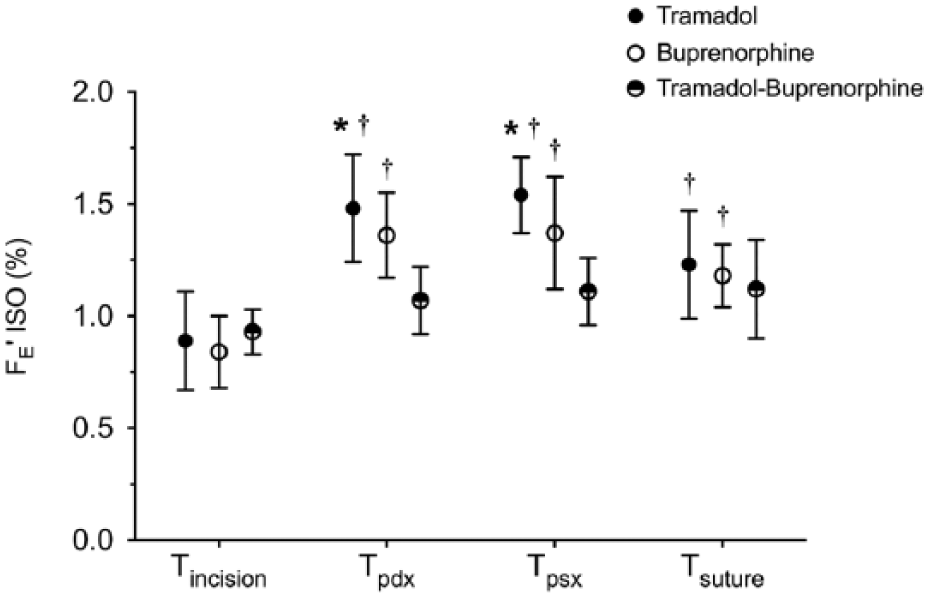
End-tidal fraction of isoflurane (Fe’ISO) at the time of skin incision (Tincision), traction of right and left ovarian pedicle (Tpdx and Tpsx, respectively) and suture of the skin (Tsuture) in cats premedicated with tramadol (TR), burprenorphine (BU) or tramadol + buprenorphine (TR-BU) undergoing elective ovariectomy. Data are expressed as mean ± SD. *Statistical difference between group TR vs group TR-BU at the same time point. †Statistical difference vs Tincision
Table 3 shows intraoperative variables over the surgical time points. Regardless of the treatment, HR measured at Tincision was statistically lower than that at Tpdx, Tpsx and Tsuture (P <0.001), but no differences among treatments were observed at each surgical point. At skin incision HR was <20% of basal recording despite decreasing Fe’ISO to 0.6% in 6/10, 5/10 and 6/10 cats in group TR, group BU and group TR-BU, respectively. There was a statistically significant effect of treatment (P <0.001), time (P = 0.005) and their interaction (P = 0.016) on Doppler arterial blood pressure. Arterial blood pressure measured at the skin incision in all groups was lower than at Tpdx and Tpsx, whereas at skin suture DoBP was statistically higher compared with that recorded at the skin incision only in group TR-BU. At both ovarian ligaments traction, a statistically lower arterial blood pressure was measured in group TR compared with the other groups. Three cats in group TR and three in group BU had a DoBP value <80 mmHg at the skin incision that increased within 10 mins and did not require treatment.
Intraoperative variables expressed as mean ± SD at skin incision (Tincision), traction of the right and left ovarian pedicle (Tpdx and Tpsx, respectively) and at the suture of the skin (Tsuture)
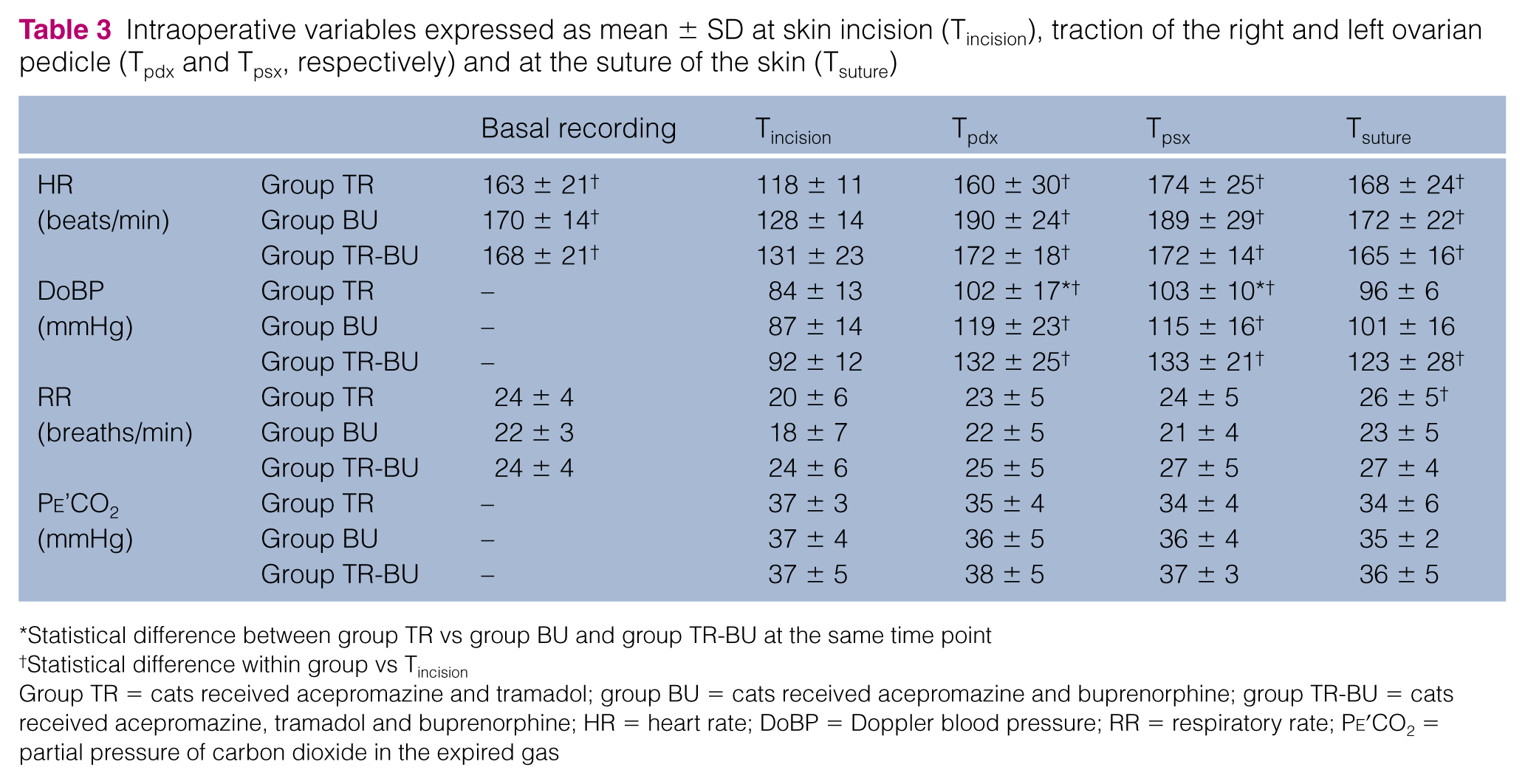
Statistical difference between group TR vs group BU and group TR-BU at the same time point
Statistical difference within group vs Tincision
Group TR = cats received acepromazine and tramadol; group BU = cats received acepromazine and buprenorphine; group TR-BU = cats received acepromazine, tramadol and buprenorphine; HR = heart rate; DoBP = Doppler blood pressure; RR = respiratory rate; Pe’CO2 = partial pressure of carbon dioxide in the expired gas
A statistical effect of time (P <0.001) was observed for RR, but the post-hoc test showed an increase in breaths per minute during skin suture compared with skin incision only in group TR. No other differences were observed, but RR at skin incision was lower than the 20% of basal recording in 5/10, 5/10 and 1/10 cats in group TR, group BU and group TR-BU, respectively. Treatment did not affect Pe’CO2 and there was no effect of time or interaction of time and treatment. Regardless the treatment scheduled, the SpO2 was >97% in all cats throughout anaesthesia.
Discussion
The results of this study showed that buprenorphine combined with tramadol was not clinically superior to buprenorphine alone in decreasing Fe’ISO in cats undergoing ovariectomy.
Acepromazine with tramadol or buprenorphine has been used in cats for premedication before inhalational anaesthesia for ovariectomy and ovariohysterectomy.3,7,8,13 In this study, the sedation scored at 30 mins after intramuscular injection was poor-to-mild, as described previously with acepromazine combined with buprenorphine.3,13 Moreover, tramadol and buprenorphine administered as single agents to cats not in pain produced mild or negligible signs of sedation.5,14 Those findings may account for the lack in statistically significant differences in sedation score among premedication protocols. Nevertheless, IV catheterisation was easily performed in all the cats, with minimal restraint.
Recovery was smooth, with similar scores among groups, as reported in previous studies in cats undergoing ovariohysterectomy with a comparable protocol.3,13 Acepromazine and both the analgesics used in the present study have a long duration of action, which may have contributed to an uneventful recovery with no detectable signs of excitement after extubation.5,11,14 Cats that received tramadol together with buprenorphine were extubated earlier than those receiving each single agent. This may represent a more rapid isoflurane washout as the concentration at the skin suture in this group did not differ statistically from that measured at the beginning of the surgery. Moreover, the higher body temperature at the end of the anaesthesia in this group may have contributed to shortened extubation time. On the contrary, no differences were observed in time to regain sternal recumbency spontaneously, and this was likely due to a similar sedative effect observed in all protocols.
In this study, tramadol administered with buprenorphine did not produce a statistically significant decrease in end-tidal fraction of isoflurane compared with buprenorphine alone. This result is similar to that of an experimental study in cats that showed no additional sevoflurane MAC reduction if hydromorphone was added to oral tramadol compared with the use of the opioid alone. 9 Moreover, buprenorphine 0.02 mg/kg IV or via the oral transmucosal route increases thermal threshold up to 51–53°C, while intramuscular tramadol 2 mg/kg causes a maximal threshold of about 44°C; this suggests that buprenorphine provides better analgesia than tramadol in cats.5,15 The dose of buprenorphine administered in this study might have been adequate to control the noxious stimulation due to ovarian pedicle traction, and the concurrent administration of tramadol would not have provided any further antinociception.
Buprenorphine or tramadol at the doses used in this study did not avoid the increase in isoflurane requirement during ovarian pedicle traction. This confirms previous results showing that such a surgical manoeuvre produces a more intense noxious stimulation as the stimulus intensity increases. 16 Buprenorphine has a low intrinsic activity compared with morphine and more opioid receptors have to be occupied to obtain a given effect as the intensity of noxious stimulation increases, shifting the dose–response curve on the right. 6 Buprenorphine may have decreased the pool of spare MOR available for tramadol and M1, which have a lower analgesic efficacy. This may further explain the absence of a statistical difference if tramadol is added to buprenorphine. Nevertheless, the isoflurane requirement during traction of both ovarian pedicles in cats receiving tramadol and buprenorphine was not different to that administered at skin incision and suture, indicating a possible beneficial, although suboptimal, effect of the combination. No clinical studies in veterinary medicine have investigated the interaction between tramadol and other opioids. In dogs, adding an infusion of tramadol to an infusion of morphine decreases the MAC of sevoflurane more than morphine alone, although the effect was less than additive. 12 In humans, tramadol showed an infra-additive pharmacological interaction with morphine on postoperative pain relief after different surgical procedures. 17 The results of that study suggested that increasing the dose of tramadol could have further decreased the requirement of morphine. In our study, higher doses of tramadol combined with buprenorphine could have produced a more significant decrease in end-tidal isoflurane. However, we decided to use the lowest doses of buprenorphine and tramadol that effectively provide analgesia by an intramuscular route.3,8,13,15
Regardless of the protocol used, most of the cats had a HR below the basal measurement at skin incision. This transitory decrease may represent the depressant effect of propofol administered for anaesthesia induction. 18 Probably decreasing the Fe’ISO below 0.6% would have allowed the HR to be maintained within the target frequency, although it was decided to maintain end-tidal isoflurane above 0.6% to avoid the risk of a sudden awakening.
Several cats in all groups had an arterial blood pressure <80 mmHg at skin incision that increased spontaneously within 10 mins. Acepromazine may lead to a decrease in arterial blood pressure; in cats an intramuscular dose of 0.1 mg/kg caused hypotension that resolved when surgical stimulation had started. 19 Also, propofol may have contributed to the low Doppler arterial blood pressure initially observed in all groups as observed in other studies.8,18 Moreover, cats treated with tramadol, compared with cats that received buprenorphine or buprenorphine and tramadol, required a higher concentration of isoflurane during ovarian pedicle traction. The difference in arterial blood pressure observed among groups may represent a dose-dependent vasodilation effect of volatile anaesthetics. 20
RR was lower than the basal reading at skin incision in most of the cats, regardless of the treatment. Those changes appear to be of little clinical relevance because mean respiratory rate, end-tidal CO2 and SpO2 were within clinically acceptable limits and comparable with those observed in other studies.7,8,13,18 Tramadol was reported to cause respiratory depression mediated by opioid receptors in cats, 21 although in our study all protocols preserved respiratory functions. Moreover respiratory depression due to volatile anaesthetics was avoided by isoflurane titration aimed at maintaining the target respiratory rate. 20
This study has some limitations: different surgeons, supervising veterinary students, performed the ovariectomy and this may represent a confounding factor as the noxious stimulation may have varied among operators, especially during the ovarian pedicle traction. In our study it was decided to target the end-tidal concentration of isoflurane based on the HR and RR measured before the administration of premedication. Those two parameters are easy to measure and are clinically used as signs of inadequate depth of anaesthesia or poor analgesia,8,18 although they may have been influenced by the stressful condition of an unfamiliar environment. This may have led us to underestimate the concentration of isoflurane needed to maintain a stable anaesthetic plane. Nevertheless, the values of end-tidal isoflurane, HR and RR measured intraoperatively were similar to those observed in cats undergoing ovariohysterectomy in other studies.2,3,7,8
Conclusions
The association of buprenorphine and tramadol was not superior to buprenorphine alone in decreasing the Fe’ISO required to maintain HR and RR within ± 20% of basal recording. Nevertheless, in cats premedicated with buprenorphine together with tramadol there were no differences in the end-tidal concentration of isoflurane at any surgical time points. This suggests tramadol provided a mildly superior antinociceptive effect when added to buprenorphine than that provided by buprenorphine alone.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
