Abstract
Objectives
The aim of the study was to evaluate the efficacy of a 4.7 mg deslorelin implant in tom cats.
Methods
Nine mature male cats were included in the deslorelin group and five cats in the control group. Before the study started, all cats were confirmed to have distinct sexually dimorphic behaviour. Blood samples were taken on the implantation day, at day 7 and at day 15, then monthly, in order to measure serum dihydrotestosterone (DHT) and 17beta(β)-oestradiol concentrations. The deslorelin group (n = 9) was divided into two subgroups: five cats (cats 1–5) were neutered in the postimplantation period during suppression of sexually dimorphic behaviour, and four cats (cats 6–9) were neutered after re-expression of sexually dimorphic behaviour. The control group cats (n = 5) were castrated without administration of the implant.
Results
Sexually dimorphic behaviours ceased within a mean ± SD of 13–58 days (23.30 ± 14.17) after implantation. DHT concentration decreased within 30 days. The mean duration of suppression was 26.5 ± 7.42 months and reactivation coincided with increased DHT values reaching preimplantation concentrations within 1 month. 17β-oestradiol concentrations significantly correlated with DHT concentrations (P <0.01). For cats castrated during suppression of sexual behaviour, the length of the long axes of the nuclei of Leydig cells, the diameter of seminiferous tubules and the height of the epithelium of the seminiferous tubules did not change until 3–6 months after implantation, whereas at 12 and 32 months the measured values were even lower than in the control group. For cats castrasted after reactivation, the length of long axes of the nuclei of Leydig cells and the diameter of seminiferous tubules approached the values of the control group between 4 and 6 months after reactivation.
Conclusions and relevance
A deslorelin implant (4.7 mg) suppresses sexually dimorphic behaviour in tom cats without any side effects and with full reversibility; however, duration of suppression is highly individual.
Introduction
The number of companion animals is increasing gradually; in parallel, the need for effective contraceptives for pets and for animals living in shelters is also increased. Progestins like medroxyprogesterone acetate, megestrol acetate and proligestone have been used for the control of fertility in domestic male and female felids for suppression of gonadotropin secretion from the anterior pituitary lobe. However, cystic endometrial hyperplasia, pyometra, mammary neoplasms, fibroepithelial hyperplasia of the mammary gland and diabetes mellitus have been described after treatment with progestines, especially after the prolonged use of medroxyprogesterone.1–8 The search for alternative methods is therefore ongoing. Slow-release gonadotropin-releasing hormone (GnRH) agonist implants were approved for long-term fertility control in recent years and have been shown even to postpone puberty in domestic female cats.9,10 In adult queens, oestrous behaviour and oestradiol (E2) secretion were successfully suppressed for 18.5 months. 11 Another study revealed that administration of a deslorelin implant in tom cats resulted in a gradual decrease in testosterone (T) concentrations, testis size and total sperm count within 4 months. 12 Spermatogenic suppression was found to be highly individual at different time points after deslorelin application. 12 After removal of the implant sexual activity started again within 4 months. 12 The sexually dimorphic behaviour of tom cats, such as urine marking, roaming, mounting other animals and aggression towards other cats or the owner, may be controlled effectively by castration. 13 However, castration might include some complications such as scrotal swelling, haemorrhage and infection. 14 Therefore, alternatives to surgical castration should be investigated, particularly for toms intended for future breeding and toms that have risk factors for anaesthesia and surgery.
The behavioural neuroendocrinology shows a close relationship between the classically male hormone, T, and the female hormone, E2. T can be converted to E2 in a number of tissues. Further, T can also be converted to a more potent androgen, dihydrotestosterone (DHT), by the 5alpha(α) reductase enzyme. The metabolism of T to E2 is often critical for its behavioural actions in males. The ability of oestrogens and androgens together to regulate male behaviour was confirmed by investigators who demonstrated that co-treatment of castrated male animals with E2 and DHT restored all parameters of male sexual behaviour comparable to that of animals treated with testosterone alone. 15
The aim of the present study was to provide more data concerning endocrinology and testicular function in tom cats after administration of the 4.7 mg deslorelin implant Suprelorin (Virbac Animal Health); for this purpose, samples were taken at different time points after application and after re-activation of sexual activity. We measured the individual DHT concentrations instead of T, as DHT is the biologically more active form of T. Moreover, the E2 concentrations were measured in the present study. Oestrogens have an inhibitory effect on testis tissue and therefore on testosterone biosynthesis.16,17 In addition, certain studies suggested that oestrogens play a role in the regulation of spermatogenesis.18,19 The question was whether, in cats, individually increased oestrogen concentrations might be responsible for individual reactions to the GnRH implant. As the duration of the suppression of libido, testicular function and fertility proved to be highly individual, already available data will be supplemented by the present study.
Materials and methods
Ethical approval was obtained from the animal ethics committee, University of Ondokuz Mayis, Samsun, Turkey (approval number 2010/56).
Animals
Fourteen tom cats (mean ± SD weight 4.17 ± 0.62 kg) were assigned to this study. Nine cats (seven domestic shorthair cats, two Van cats and one Siamese) aged between 1 and 6 years were included in the deslorelin group, and five cats aged between 1 and 3 years were included in the control group (all domestic shorthairs). All cats were privately owned, healthy at presentation and showed marked sexually dimorphic behaviour. They were housed indoors and allowed to go out each day for a few hours.
Study design
Treatment
The study population comprised cats brought to the clinic with the request of neutering by their owners. The owners were informed about the study and volunteered for their cats to take part in the study. Cats were randomly assigned to the deslorelin and control groups, and neutered at different times after implantation. The testicles were investigated histologically.
In the deslorelin group (n = 9) 4.7 mg deslorelin implants (Suprelorin; Virbac Animal Health) were implanted subcutaneously between the shoulder blades, according to the manufacturer’s instructions. The control group did not receive any treatment.
Andrological examinations and blood sampling
Anamneses included the frequency and places of urine spraying, severity of aggression towards the owners, intensity of urine smell and contact with female cats, as well as sexual behaviour such as mating, mounting or masturbation. Clinical examinations and blood sampling for DHT and 17beta(β)-oestradiol (E2) measurements were performed on implantation day, at day 7 and day 15, and then once monthly following deslorelin implantation.
In the control group, blood samples were taken on the day of castration, at day 7 and day 15, and then once monthly for 2 months.
Observation of sexually dimorphic behaviour
The cats were visited in their homes and observed for signs of sexual behaviour: alterations in the odour of urine, urine marking, presence of a female cat, mating, mounting or masturbation, aggression (eg, biting, scratching) towards the owners or other cats. The owners observed their cats and filled in questionnaires every week during the month after implantation and then monthly thereafter. All recorded data were controlled by the same researcher in the cats’ households once weekly during the month after implantation and then monthly.
Deslorelin and control groups
Table 1 presents the castration time of the cats in the deslorelin group after implantation (cats 1–5) and after re-expression of sexual behaviour (cats 6–9). The control group (n = 5) included healthy untreated tom cats that were neutered while showing sexually dimorphic behaviour.
Castration time, time until and duration of suppression of sexual behaviour in the deslorelin group after implantation and after reactivation

Castrated in the postimplantation period before reactivation of sexually dimorphic behaviour
Hormone assays
Serum was obtained after centrifugation at 3000 g for 10 mins and stored at −20°C until assayed. DHT was measured instead of T as this is the biologically more active hormone. For this purpose, a human ELISA kit was adapted and validated for cat serum (EIA-4132; DRG International; J Thuroczy, A Wölfling and L Balogh, unpublished data). Sensitivity was 6 pg/ml; and for the highest detection limit readings of >2500 pg/ml have to be diluted 1:8 at the maximum. Intra- and inter-assay coefficients of variation (CV) were 3.9% and 5.9%; rates of recovery were 97.9%.
For E2 measurement, the DRG Estradiol Sensitive ELISA (EIA-4399; DRG International) was used and validated for cat serum (J Thuroczy, A Wölfling and L Balogh, unpublished data). Sensitivity was 1.39 pg/ml; intra- and inter-assay CVs were 5.52% and 6.78%, respectively; and rates of recovery were 95.7%. Measured values are given in pg/ml for both DHT and E2.
Histopathological evaluation of the testes
After castration, testes were fixed in Bouin’s solution for 18 h. Thereafter, testes were cut longitudinally and submerged into Bouin’s solution for postfixation. The Bouin-fixed samples were then washed several times in 70% ethanol. Each specimen was processed routinely. Tissue samples were embedded in paraffin wax, sectioned at 5 μm and stained with haematoxylin and eosin for histopathological assessment under a light microscope (Nikon Eclipse E600) by a pathologist (groups blinded).
For histopathology, testicular parenchyma of each sample was evaluated. In testicular parenchyma the length of the long axes of 10 nuclei of Leydig cells was measured at × 40 magnification on 10 different areas of each slide. Furthermore, the diameters and heights of 10 seminiferous tubules from 10 different areas were measured at × 20 magnification.
Statistical analyses
The commercial software PASW statistics 18.0 software for Windows (SPSS; IBM) was used for all statistical analyses. The normality and homogeneity of groups were determined by Shapiro–Wilks test and Barlett box tests. Alterations in the long axes of the nuclei of Leydig cells, and the diameter and height of seminiferous tubules in the normally distributed data were compared by the repeated measures define factor. For non-normally distributed data, the Mann–Whitney U-test was used for comparison between two groups. The Kolmogorov–Smirnov Z-test was used for the comparison between more than two groups. Owing to the small number of data and non-normal distribution, the Friedman test was performed to test the differences between the groups over the time period. After the Friedman test, the results were statistically tested for significant differences between the two groups using the Wilcoxon test. Differences were considered significant at P <0.05.
Results
During the course of the study the cats’ health status remained good and no reaction at implantation site or other side effects were observed.
Sexually dimorphic behaviour
Before the study started, all cats of both groups showed strong sexually dimorphic behaviour, including daily urine spraying indoors and the intense urine smell peculiar to intact male cats. Half of the cats were showing aggression towards their owners, including scratching and biting them. Five cats in the deslorelin group had contact with female cats.
In the deslorelin group, all signs of sexually dimorphic behaviour ceased gradually within a mean ± SD of 13–58 days (23.30 ± 14.17) after implantation (Table 1). Six of nine cats showed a temporary increase in sexual behaviour for 1 week after the implantation, then the clinical signs gradually ceased. The sexual behaviour in the control group ceased within a mean ± SD of 1–4 (1.8 ± 0.4) days after castration (P <0.001).
The mean ± SD duration of suppression of sexually dimorphic behaviours in cats 6–9 was 16–30 (22.7 ± 5.8) months (Table 1). The increase in sexual behaviour coincided with a marked increase in DHT. Although the order of re-expression of sexually dimorphic behaviour was different in individual cats, a gradual increase in urine marking and the typical urine smell of intact tom cats were the first signs of re-activation of sexual behaviours in cats 6–9 of the deslorelin group.
Hormone concentrations
DHT concentrations
The mean ± SD DHT concentrations of the deslorelin group (n = 8) before deslorelin implantation was 47.63–1992.04 (726.00 ± 644.44) pg/ml and decreased to 7.15–77.52 (39.66 ± 29.51) pg/ml after 1 month of implantation. The mean ± SD DHT concentration of the control group (n = 5) was 298.97–704.73 (501.85 ± 286.91) pg/ml and decreased to 33.99–109.90 (49.45 ± 42.08) pg/ml after 1 month of castration.
The DHT concentrations of cats 1–5, neutered in the postimplantation period before reactivation of sexually dimorphic behaviour, are given in Table 2. The highly individual concentrations at each time point are striking.
Preimplantation, precastration and postcastration dihydrotestosterone (DHT) concentrations of cats 1–5, castrated before reactivation of sexually dimorphic behaviour in the deslorelin group (reference intervals 30–700 pg/ml) 20

0 d = day of implantation
The DHT concentrations of cats 6, 7 and 9, neutered at different times after reactivation of sexually dimorphic behaviour, are shown individually as the individual variability was high (Figure 1). Mostly, DHT concentrations increased dramatically 1 week after deslorelin implantation and then significantly decreased within the first month thereafter. The concentrations then varied individually until a significant increase up to 2500 pg/ml coincided with reactivation of sexually dimorphic behaviour (15–21 months).

Individual concentrations of dihydrotestosterone (DHT, pg/ml) and 17β-oestradiol (E2, pg/ml) from deslorelin implantation onwards and until reactivation and postcastration in cats 6,7 and 9. 0 d = day of implantation; Mo∞ = time of reactivation of sexually dimorphic behaviour; MoAkt* = follow-up period after reactivation
The initial DHT concentration of cat 6 was 2500 pg/ml, which remained stable until day 7 and then decreased continuously until month 11, when the DHT concentration was still 500 pg/ml. Despite this, the signs of sexually dimorphic behaviour ceased at day 16. In the last 4 months before sexual reactivation, DHT concentration again reached 2500 pg/ml. This cat also had an exceptionally high E2 concentration, and the DHT and E2 concentrations were not included in the calculation of mean concentrations of the study group. In the other cats, DHT concentration reached the preimplantation value 3 or 4 months after reactivation (Figure 1).
Cat 8 was castrated 4 months after reactivation of sexually dimorphic behaviour; however, as the owner moved to another city, the blood samples of that cat could not be collected 8 months after implantation as planned. These data are excluded from Figure 1.
E2 concentrations
The mean ± SD E2 concentrations of the deslorelin group (n = 8) before deslorelin implantation and 1 month after implantation were 2.17–22.39 (12.12 ± 7.80) pg/ml and 3.17–21.18 (11.38 ± 7.64) pg/ml, respectively. The mean ± SD E2 concentrations of the control group (n = 5) were 6.49–31.83 (22.04 ± 10.91) pg/ml and decreased to 0.46–30.44 (10.04 ± 13.77) pg/ml 1 month after castration.
The initial E2 concentration of cat 6, neutered after reactivation of sexually dimorphic behaviour, was very high (321.61 pg/ml) and not consistent with the reference interval of E2 concentrations in intact adult male cats (12.1–16.1 pg/ml). 20 This value was therefore not included in the calculation of the mean E2 concentration of the deslorelin cats above. The mean E2 concentrations of eight deslorelin cats and the control group were within the reference interval.
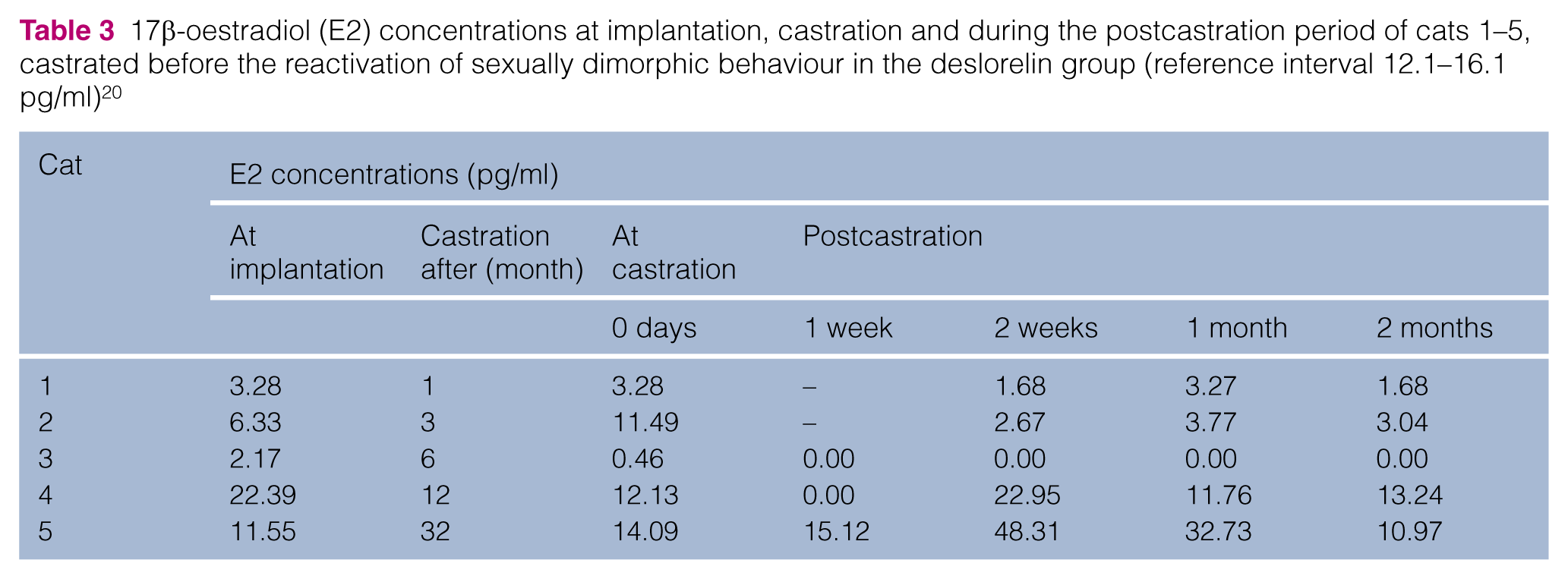
The serum E2 concentrations of cats 1–5, neutered in the postimplantation period before reactivation of sexually dimorphic behaviour, are given in Table 3. At the time of castration 1 and 32 months after the implantation, concentrations were decreased in 3/5 cats only. In the following 2 months, concentrations were highly variable, and basal in only one cat.
17β-oestradiol (E2) concentrations at implantation, castration and during the postcastration period of cats 1–5, castrated before the reactivation of sexually dimorphic behaviour in the deslorelin group (reference interval 12.1–16.1 pg/ml) 20
The serum E2 concentrations of cats 6–9, neutered at different times after reactivation of sexually dimorphic behaviour, are shown in Figure 1.
Histopathological findings
The results of the histopathological evaluation of all cats are presented in Tables 4 and 5. No significant difference was found between the length of the long axes of the nuclei of Leydig cells when cats were castrated 1, 3 or 6 months after implantation. Similarly, the difference was not significant between 12 and 32 months after implantation. However, between cats castrated at 1 and 6 months, and those castrated 12 and 32 months after implantation, a significant difference in the length of long axes was assessed (P <0.001); similarly, there was a significant difference between the cats castrated 12–32 months after implantation and the control group (P <0.001). When the diameters of seminiferous tubules were compared, a significant difference was observed between those castrated 1 and 12 months after implantation (Figure 2). With regard to the height of the epithelium of the seminiferous tubules, significantly higher values were measured after castration at 3 months after implantation than at 32 months (P <0.01; Table 4).
Comparison of long axes of the nuclei of Leydig cells, diameter of seminiferous tubules and height of the epithelium of the seminiferous tubules between cats 1–5 in the deslorelin group and the controls

All values are mean ± SD. Different letters indicate differences between groups (in column); P <0.001 and P <0.01 was considered significant
Number of measurements for each cat
Comparison of long axes of the nuclei of Leydig cells, diameter of seminiferous tubules and height of the epithelium of seminiferous tubules among cats 6–9 in the deslorelin group and the controls

All values are mean ± SD. Different letters indicate differences between groups (in column); P <0.001 and P <0.01 were considered significant
Number of measurements for each cat
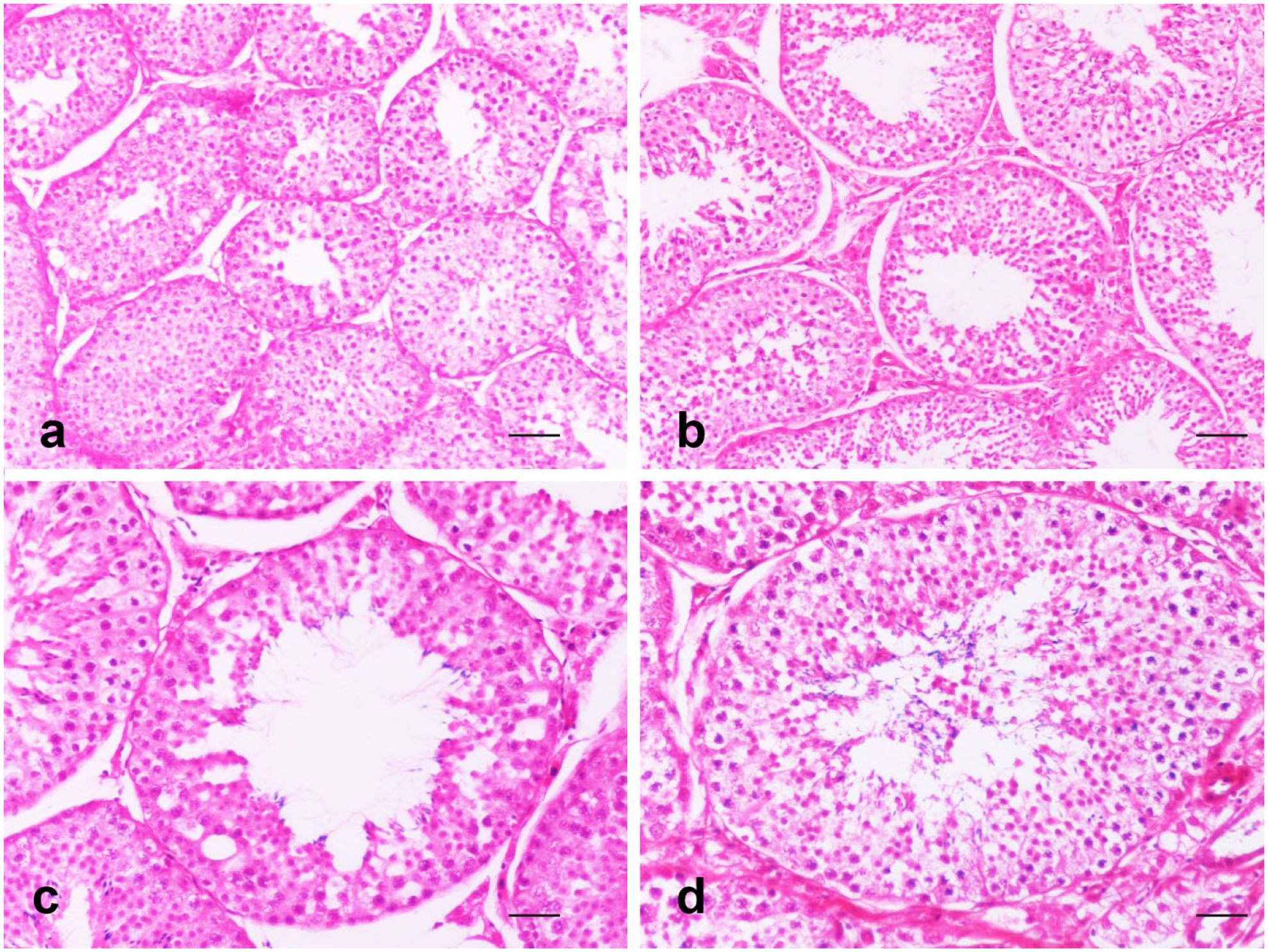
Photomicrographs of the diameters and heights of the epithelium of the seminiferous tubules in sections of the cats’ testes. The diameter of the seminiferous tubules was significantly decreased at 12 months (a; haematoxylin and eosin [HE], bar = 95 μm) compared with 1 month (b; HE, bar = 95 μm) after implantation. The seminiferous tubules with the wide lumen were lined by lower germinal epithelium at 32 months (c; HE, bar = 18 μm) compared with at 3 months (d; HE, bar = 18 μm) after implantation
In cats 6–9, castrated after the reappearance of sexual behaviour, the length of the long axes of the nuclei of Leydig cells was similar 2, 4 and 6 months after reactivation. However, in cats castrated 2 months after reactivation, values were signicantly higher (P <0.05) than in the control group. Four months after reactivation, the difference compared with the control group was no longer measurable. The diameter of seminiferous tubuli was significantly higher (P <0.001) at 2 months after reactivation than at 6 months and in the control group. Similarly, the height of the epithelium of the seminiferous tubule at 6 months was even lower than in the control group (Table 5).
Discussion
Sexually dimorphic behaviour
Manifold studies have proved that the application of long-term release formulations or the continuous administration of GnRH agonists result in reversible suppression of reproductive function in male and female cats.8–12,21–25 The duration of suppression of sexually dimorphic behaviour in our study was 22.7 ± 5.8 months, which is similar to the 88.7 weeks stated by Goericke-Pesch et al. 21 However, this might only reflect the significant individual times of duration, as animal numbers in both studies were relatively low.
DHT concentrations
In this study, DHT concentrations significantly decreased within 1 month of administration of a 4.7 mg deslorelin implant. This is comparable with results from other studies,12,24–26 even though we measured DHT concentrations instead of T. As enough data are available concerning the course of the peripheral T concentration during suppression with deslorelin, we aimed to observe the course of the biologically more active DHT in peripheral blood. This has been carried out previously by Tsutsui et al, 27 who compared peripheral DHT and T concentrations in six tom cats during the breeding season. They observed highly individual variations in DHT concentrations (range 0.03–0.7 ng/ml; mean ± SE: 0.35 ± 0.1 ng/ml) and no correlation with T values (range 0.72–1.60; mean ± SE: 1.03 ± 0.11 ng/ml). This high individual variability was also seen in the present study.
In one cat, sexually dimorphic behaviour ceased at day 16 postimplantation, despite high DHT concentrations (>500 pg/ml) after 6 months. Additionally, E2 concentrations were extremely high and followed the course of DHT. In this cat, deslorelin resorption may be delayed owing to DHT metabolism or other mechanisms; this remains to be investigated. The elevated serum E2 concentrations could be caused by aromatisation and storage in fat tissue or increased secretion from the adrenal gland.
In the present study, the course of DHT after administration of 4.7 mg deslorelin resembled the course of T observed by others.21,28 T and its metabolite, DHT, are responsible for normal breeding behaviour. 13 However, there are no comparable data in the relevant literature concerning the course of DHT after application of deslorelin; more investigations are therefore needed before any conclusion can be drawn.
E2 concentrations
Kaya et al stated that the serum E2 concentrations of bitches increased at 37–49 weeks after 9.4 mg deslorelin implantation in the prepubertal period. 29 This finding shows that deslorelin might affect serum E2 concentrations in the dog. However, there are no previous data about E2 concentrations after deslorelin implantation in tom cats. Therefore, the serum E2 concentrations before and after implant insertion were measured as we hypothesised that this might have an impact on individual effectiveness of the implant. Despite the individual variability between the cats, serum E2 concentrations in the postimplantation period decreased significantly and increased again approaching the reactivation time of sexual behaviours. The results of the study revealed that both DHT and E2 concentrations were low from months 5–6 of deslorelin implantation. Marino et al stated that E2 concentration in male dogs declined markedly despite individual variability (8–138 pg/ml). 30 This finding is similar to the E2 concentrations of tom cats in our study. Possible reasons for this variability of serum E2 concentrations could be individual differences in drug absorption or metabolism. However, in the majority of cases, the effect of deslorelin, as measured by the decrease in sexually dimorphic behaviour and DHT concentrations, was normal.
Histopathological findings
Histologically, in this study, the decrease in the length of the long axes of nuclei of Leydig cells was more marked in cats castrated 12 and 32 months after implantation than in those castrated 1–6 months after implantation, indicating that length might decrease in individual cases until 32 months after implantation. In another study, the nuclei of the Leydig cells were determined to be clearly atrophied in dogs treated with deslorelin for 41 and 101 days when suppression was maximal. 31 Thus, we could show that testicular atrophy in tom cats after deslorelin application takes longer than in dogs.
In our study we furthermore observed that at 2 months after the reactivation of sexual behaviour, the length of nuclei of Leydig cells and the diameter of seminiferous tubules increased again and reached values comparable with those of the control cats and the cats castrated at 4 and 6 months after reactivation, even after suppression of 16–32 months. Another study reports preliminary data concerning the recurrence of testicular function after long-term suppression with deslorelin in seven male cats; 21 the authors found normal spermatogenesis between 2 weeks and 5 months after the end of efficacy of deslorelin. Others observed fertile matings in tom cats 7–42 weeks after the last measurement of basal testosterone concentrations.12,21 Unfortunately, it was not possible to evaluate the fertility of tom cats by matings in this study.
Conclusions
Our findings and those from others show that, in tom cats, sexually dimorphic behaviour may reappear 16–30 months after the implantation of 4.7 mg deslorelin, dependent on the increase in DHT. However, it must be emphasised that achievement of fertility may precede normal sexually dimorphic behaviour and complete testicular regeneration. Additionally, it should be kept in mind that high individual variability in DHT and E2 concentrations, and the onset and duration of efficacy of the deslorelin implant will be observed.
Footnotes
Acknowledgements
We would like to thank Virbac Animal Health, Carros, France, for providing the Suprelorin implants.
Conflict of interest
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
