Abstract
Objectives
This study was performed to assess skin–muscle–fascia specimen shrinkage and donor site changes, and to compare three techniques of specimen preparation for their effect on specimen shrinkage postexcision and after formalin fixation in feline cadaveric specimens.
Methods
Fifteen fresh feline cadavers were used for this study. Gelatin spheres were implanted in paired thoracic subcutaneous pockets and subsequently excised with 30 mm lateral margins and a fascial plane as the deep margin. Skin and fascia were either left unsutured, sutured together using four simple interrupted quadrant sutures (‘four-quadrant-sutured’) or sutured together in a continuous pattern (‘circumferentially sutured’). Specimens were measured for tumor-free margins on the excised and fixed specimens. The donor site defect was assessed for enlargement after specimen excision. Statistical analyses were performed to assess the donor site enlargement, and the influence of preparation technique on margin size, with significance set at P <0.05.
Results
The closest skin margins on the excised and fixed specimens were significantly smaller than the planned 30 mm margins; however, no significant difference was found between postexcision and postfixation specimens. No significant differences were found between the three techniques (P = 0.74) with regard to margins either after excision or fixation. The fascial and skin defects of the donor site were significantly larger than the planned excision.
Conclusions and relevance
Surgically obtained feline skin–muscle–fascia specimens will significantly decrease in size prior to formalin fixation, resulting in falsely decreased measurements from tumor to tissue margins. Affixing the skin to the fascia does not significantly influence this decrease in margin size in feline tissue specimens at this location.
Introduction
One of the important parameters in surgical oncology is the size of the surgically obtained deep and lateral margins. As a standard of care, the lateral and deep margins of excised specimens are inked and the correct orientation of the specimen is indicated. 1 The reliability of the histological assessment of surgically obtained lateral margins vs the true lateral margins is of vital importance to guide postoperative therapy.
It has been documented that various tissue specimens (liver, gastrointestinal tract, oral mucosa, skin) undergo a reduction in size (‘shrinkage’) after both excision and formalin fixation.2–13 Percentages as high as 34% and 40% between surgical removal and the final assessment of the fixed skin samples have been reported for humans and canines (M Riseelada et al, unpublished data).4,7,8,14 Tissue shrinkage from the initial surgical mapping to final sample measurements have been assessed in canines, 7 with a reported 21.1–32.0% decrease in tissue length, and in a canine cadaveric model with a reported 26–28% decrease in specimen size after fixation (M Riseelada et al, unpublished data). It has been suggested that the majority of shrinkage occurs prior to formalin fixation (M Riseelada et al, unpublished data). 4 A recent publication investigated shrinkage of skin strips in feline cadavers, and reported only a significant size decrease in hindlimb samples. 15 However, to our knowledge, no study exists reporting the degree of shrinkage in feline tissue specimens involving skin, muscle and fascia that mimics the clinical situation of tumor removal with wide margins prior to or after formalin fixation.
The aims of this study were first to compare skin–muscle–fascia specimens from feline cadavers excised from the mid-thorax with a 30 mm lateral surgical margin for margin changes postexcision and postfixation; second, to assess the effect of specimen preparation on lateral margin assessment; and, third, to assess the change in donor site defect after excision.
Our hypotheses were that feline skin would significantly decrease in size both postexcision and after formalin fixation, the donor bed would significantly increase in size and that specimen preparation technique would not influence the size of the lateral margins.
Materials and methods
Animals
Fifteen female intact young adult domestic shorthair cats were used, euthanized for reasons unrelated to the study. All specimens were harvested within 6 h of euthanasia, by a single surgeon and in a single session per animal. All processing and measurements were performed by the same surgeon. All cadavers were stored in a walk-in cooler specifically designed to store cadavers prior to use, with a temperature set at 7ºC. All specimens were processed immediately after harvest and then placed in 10% neutral-buffered formalin (specimen:formalin volume of 1:10) immediately after processing and allowed to fix for at least 72 h.
Procedures
All specimen preparation techniques were performed 10 times in a block design, allowing five direct comparisons of two of the three techniques. The thoracic areas were clipped and a 30 mm vertical skin incision and subcutaneous pocket was made, just large enough to introduce a premade gelatin sphere of 28 mm in diameter in the pocket and approximate the skin over the sphere using surgical staples or skin sutures (Figure 1). The pockets were centered over the mid-thorax, equidistant from the spine and sternum and equidistant between the caudal shoulder depression and last rib.
Close-up photograph of the 28 mm gelatin sphere
The spheres were produced in a spherical mold using gelatin (Knox Unflavored Gelatin; Kraft Foods Global) and methylene blue dye (1% methylene blue, 10 mg/ml; Akorn) (3.6 g gelatin in ¼ cup boiling water, followed by ¼ cup cold water and 2–3 drops of methylene blue dye) and allowed to gel for >12 h in a refrigerator prior to use.
A flexible ruler (Surgical Skin Marker, standard tip with ruler; Medline Industries) was used to mark 30 mm skin margins around the palpable subcutaneous sphere at eight equidistant points that were connected to form a circle similar to a surgical planning prior to excision (Figure 2a). The incision was performed over the preplanned line to the level of the underlying fascia and muscle plane (latissimus dorsi) (Figure 2b). The muscle–fascia complex was left intact and served as the deep tissue layer. The specimens were divided into three treatment groups including an ‘unsutured’ control group (n = 10), a ‘circumferentially sutured’ technique group (n = 10) or a ‘four-quadrant-sutured’ technique group (n = 10).
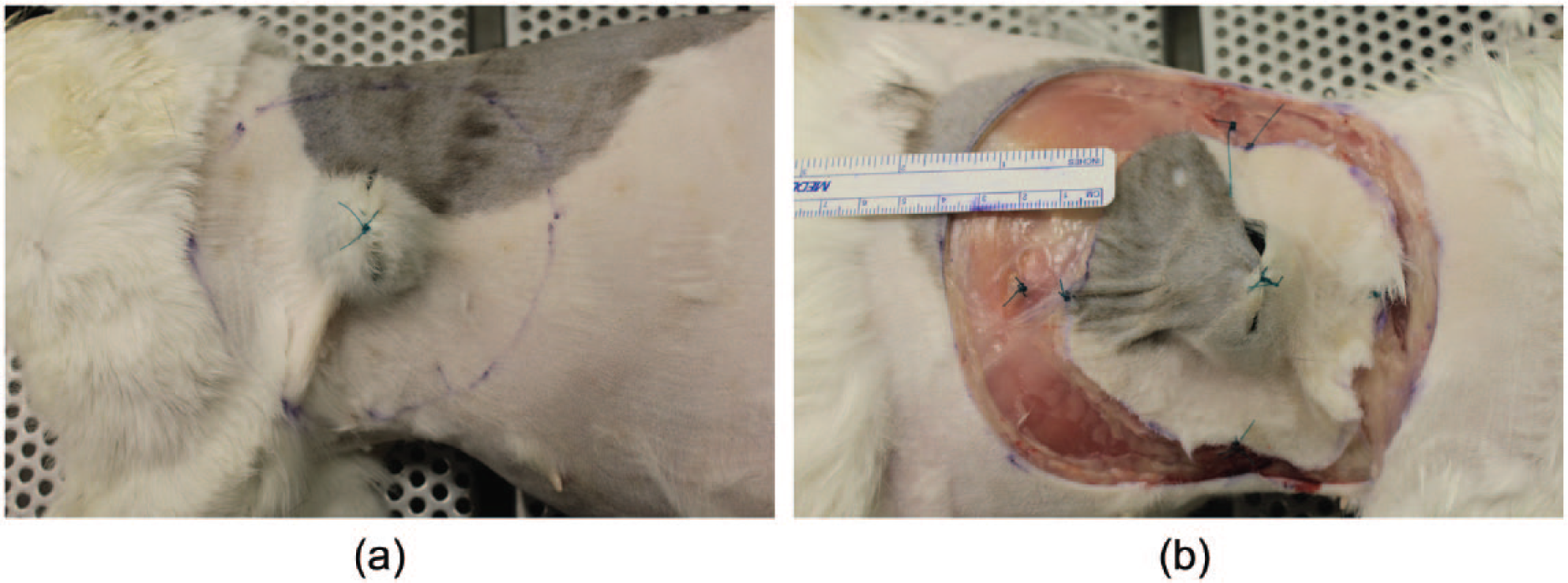
(a) The left side of a feline cadaver is shown, with the sphere implanted in the subcutaneous space peripheral to the latissimus dorsi muscle. Lateral margins of 30 mm were measured and indicated using a marker pen. A full circle, connecting the 30 mm lateral markers, was fully drawn. The resection was performed along the predrawn circle at the planned distance. (b) The right thorax of the same cat is shown with the contralateral specimen being excised. The caudal edges of the skin and muscle (deep) layer are sutured together using a simple continuous suture pattern, creating a ‘circumferentially sutured sandwich’. A ruler is shown in the image, indicating the gap between the specimen edge and the defect skin edge
The ‘circumferentially sutured’ specimens underwent suturing of the fascial layer to the skin layer in two separate simple continuous sutures prior to explantation (Figure 2b). The ‘four-quadrant-suture’ group had four simple interrupted (12, 3, 6 and 9 O’clock sites) sutures placed instead of the simple continuous suture pattern to fix the fascia to the skin, after which the specimens were fixed in 10% neutral-buffered formalin for at least 72 h.
Measurements
The skin and fascial defects were measured edge to edge in both a dorsoventral and a craniocaudal direction immediately after removal of the specimen.
The dorsal, ventral, cranial and caudal margins (palpable tumor edge to cut-skin edge) were measured immediately after specimen removal in an identical fashion to the surgical planning measurements with the specimen on a flat surface. Additionally, the fascial (deep) surface of the excised specimen was measured from cut edge to cut edge in a dorsoventral and craniocaudal direction.
The defect sizes (skin and fascia in both dorsoventral and craniocaudal direction) were compared in order to evaluate an influence on the tissue properties of the second site by the first defect made.
After fixation, the specimens were cut using the cross section method, 1 and the four lateral margins (cranial, caudal, dorsal and ventral) and the deep margins were measured (in mm) at the center of the sphere, as well as the cross-sectional width of the sphere (Figure 3).

(a) A fixed specimen sectioned in dorsoventral direction is shown, with the gelatin sphere embedded and fixed in the tissue. The cross-sectional diameter of the sphere was measured using this sectioned plane. (b) The final section has been completed on this specimen, allowing measurement of the cranial lateral margin
Statistical analysis
The width of the spheres was evaluated using a Wilcoxon sign-rank test. To test for differences in size from the measured 30 mm margins pre-excision the Wilcoxon sign-rank test was used. Pre-excision measurements were subtracted from both postexcision and postfixation measurements. The postfixation differences were separated into treatment groups and tested separately. Three-way ANOVA was used to compare the four margins across techniques and animals both postexcision and postfixation, with significance set at P <0.05. Individual margins were compared using Tukey’s adjustment for multiple comparisons. A paired t-test was used to compare the dorsoventral and craniocaudal defects between the grouped first made defects and the grouped results for the second defect. Commercial software (SAS, Version 9.3; SAS Institute) was used to conduct all analyses.
Results
Mean ± SD body weight of the feline cadavers was 3.05 ± 0.85 kg (range 1.60–3.63 kg); no significant differences in body weight were found between groups (P = 0.98).
The cross-sectional width of the gelatin sphere after fixation (27.2 ± 1.21 mm; median 28 mm; range 25–30 mm) was significantly smaller than the original diameter of 28 mm (P = 0.002).
The mean closest margin decreased by 42.4% after excision and by 58.7% after fixation compared with the originally planned margin.
The mean closest lateral margin was significantly smaller after excision than the originally measured sample (P <0.0001) for all three treatment groups, but did not differ between treatment groups (Kruskal–Wallis, P = 0.74). The closest lateral margin was also significantly smaller after formalin fixation compared with the originally measured sample (P = 0.002 for each of the three treatment groups) (Table 1). The difference in size between the postexcision and postfixation closest lateral margin was not significantly different for any of the three preparation techniques (‘unsutured’ group: P = 0.73; ‘four-quadrant-sutured’ group: P = 0.76, ‘circumferentially sutured’ group: P = 0.06).
The closest lateral skin margins of the excised and fixed specimens are presented as mean ± SD, as well as the median and range of the closest lateral margin of individual specimens, and as percentage decrease from the original margin in a separate column. The planned lateral margin was 30 mm in all specimens and in all directions

The P values for the difference between the postexcision and postfixation closest lateral margin are shown separately
The closest lateral margin in these postexcision specimens differed significantly from the planned lateral margin (P <0.0001)
The closest lateral margin of the processed postfixation specimen differed significantly from the planned margin (P = 0.002)
All four individual lateral skin margins were significantly smaller than the originally planned direction site-matched lateral margins post-excision (P = 0.01) and postfixation (P <0.0001) (Table 2). Postexcision, the dorsal margins were significantly greater than the caudal margins (P = 0.02). Postfixation, the dorsal and ventral margins were significantly greater than the cranial and caudal margins (P <0.02 for all differences).
Data for all four lateral margins of the three specimen preparation groups combined (n = 30) are presented as the mean ± SD, median and range of the individual specimens, and percentage decrease from the original planned margin. The planned lateral margin was 30 mm in all specimens and in all directions

The deep margin of the fixed samples was 2.45 ± 1.7 mm (range 1.0–6.0 mm).
The measured surgical skin defect was 11.69 ± 0.95 cm (median 12 cm, range 10.0–13.2 cm) in the dorsoventral and 12.777 ± 0.574 cm (median 12.6 cm, range 11.0–13.5 cm) in the craniocaudal direction. The measured surgical fascial defect was 9.244 ± 1.518 cm (median 10 cm, range 3.9–11.0 cm) in the dorsoventral and 10.472 ± 0.756 cm (median 10.5 cm, range 8.5–11.5 cm) in the craniocaudal direction. The defects were significantly smaller in the dorsoventral direction than in the craniocaudal direction, both for the fascial (P = 0.0008) and the skin defect (P <0.0001). The skin defect was significantly larger than the fascial defect, both in the dorsoventral and the craniocaudal direction (P <0.00001).
No significant differences were found between the first-site and second-site defects: skin dorsoventral (P = 0.66), skin craniocaudal (P = 0.20), fascia dorsoventral (P = 0.42), fascia craniocaudal (P = 0.66).
Discussion
The results of this study indicate that excised skin–muscle–fascia specimens taken from a thoracic location in feline cadavers significantly decreases in size, even more so than canine cadaver specimens using the same gelatin sphere tumor model (M Riseelada et al, unpublished data). The mean decrease in size of the closest lateral margins in this study (58.7%) is far greater than the reported decrease in size for canine cadaveric specimens (26.0–28.0%), as well as margins in a canine cadaveric tumor model identical to the current feline model (36.0%) (M Riseelada et al, unpublished data).
The decrease in lateral margins could not be attributed to an increase in sphere width as the mean cross-sectional diameter of the 28 mm spheres was significantly smaller after fixation (27.2 mm). We checked the accuracy of our margins using the same ruler after drawing the outline and letting the tissue relax, but did not use calipers to measure specifically the preplanned margin pre-excision. Tumor margins are important when deciding upon adjunctive therapies postsurgery, such as scar revision or radiation therapy.16–18 The results of this study suggest that tissue shrinkage postexcision and postfixation may result in an underestimation of the true lateral margins and subsequent overestimation of the number of feline patients requiring scar revision or radiation therapy. While this study used a solid model, without tumor infiltration into the surrounding tissues, we do feel that it provides useful information that can help guide the interpretation of the tumor-free margins reported histologically.
Using a model with 50 mm lateral margins in order to recreate a wide surgical excision for a feline injection-site sarcoma would have been ideal, 19 but bilateral excision of tissues with these dimensions was not possible. The current model allowed bilateral specimens to be removed without significant difference in defect size properties postremoval between the first and second site.
The majority of the specimen size and lateral margin changes occurred immediately after excision, prior to formalin fixation. Differences between the postexcision and postfixation specimens were not significantly different, which most likely reflects a type II error due to small sample size, as the differences between the specimens in the largest group (‘circumferentially sutured’ technique) approached significance. Previous canine studies have shown that significant changes occur during fixation (M Riseelada et al, unpublished data). 14 Interestingly, a recent paper investigating the size difference in feline cadaver skin specimens did not find a statistically significant difference in size after excision or after fixation of thoracic skin specimens. 15 This is counter to the findings in the current paper, as well as the clinical impression of specimen size difference after removal. This may be a result of stretching the skin samples after retrieving them from the formalin in the paper by Miller and Dark, 15 whereas the specimens were measured unaltered in the current study.
In the current study the size difference of the lateral margins of feline thoracic specimens was more pronounced than those in a similar canine study (M Riseelada et al, unpublished data). Differences in tissue composition (eg, collagen or elastin content) and thickness between feline and canine skin could account for differences in these findings. 20 The only difference histologically is that the feline epidermis does not have a continuous stratum granulosum and lucidum, as opposed to canine epidermis. 20 Potentially, this could allow the elastin in the feline skin to contract more freely.
Affixing the deep layer (latissimus dorsi muscle–fascia complex) to the skin did not significantly influence the size of the lateral margins compared with controls, nor did the technique used (four individual simple interrupted sutures or a simple continuous suture pattern) for affixing the two tissue layers. The difference in the measured closest lateral margins for the ‘circumferential suturing’ technique between postexcision and postfixation approached significance (P = 0.063). A post-hoc power analysis revealed that significance may be reached with a larger sample size (n = 11). The ‘unsutured’ and ‘four-quadrant-sutured’ technique groups would have required 391 and 315 specimens, respectively, to reach significance. The specimens treated with a continuous suture technique has less decrease in margin size after excision than either the control or the ‘four-quadrant-sutured’ specimens, but had a smaller closest lateral margin than the control specimens after formalin fixation. This difference immediately after excision could be due to differences in shrinkage between different tissue types, with the affixed latissimus dorsi muscle potentially influencing the amount of elastic contraction of the overlying skin. When assessing the wound bed, the skin defect increased significantly more in size than the underlying fascial–muscle layer, indicating a greater amount of elasticity in the skin, which could potentially explain the lesser decrease in size in samples with a more closely sutured specimen using a continuous suture. Interestingly, the mean closest lateral margin in the ‘circumferentially sutured’ group was smaller than the ‘unsutured’ specimen group, which potentially could be attributed to the different tissue properties and influence of formalin fixation, although that was not assessed separately.
While investigating the separation and translation of tissue layers was not a part of the current study, a previous study in canine cadavers has shown that affixing the different tissue layers will prevent translation and shifting of the deep plane relative to the skin (M Riseelada et al, unpublished data).
We used the closest lateral margin as the comparative margin across the samples, as well as evaluating all individual margins, as the closest lateral margin is the deciding factor for postsurgical recommendations in clinical cases. The individual margin assessment was to allow evaluation of directionality of the size decrease or increase of the lateral margins. We found that tissue shrinkage between pre- and postexcision and after fixation was greater at the cranial and caudal margins than in the dorsal and ventral margins. The width of the wound bed was also significantly greater in the craniocaudal direction, indicating a greater amount of elasticity, or tension, in this direction. Although, to date, no studies exist documenting skin tension lines in felines, the increased widening of the defect in the craniocaudal direction is in accord with the canine tension lines described by Irwin in 1966. 21 The difference in size of the lateral margins after excision (caudal significantly smaller than dorsal) and the fact that the dorsal and ventral margins were significantly greater after fixation than cranial and caudal margins could potentially be explained by the same directionality that influenced this increase in size of the wound bed.
Conclusions
The present study shows that the lateral margins of a fixed, processed specimen of feline skin – muscle and fascia – will underestimate the true, surgically obtained margins. Furthermore, both a simple interrupted pattern and a simple continuous pattern of affixing the tissue layers do not significantly alter the size of the lateral margins, and can therefore be performed to prevent tissue translation without compromising the assessment of lateral margins.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
