Abstract
Objectives
The aim of the study was to describe the ultrasonographic and endoscopic appearance and characteristics of the caecum in asymptomatic cats, and to correlate these findings with histology.
Methods
Ex vivo ultrasonographic and histologic evaluations of a fresh caecum were initially performed. Then, 20 asymptomatic cats, privately owned or originating from a reproductive colony, were recruited. All cats had an ultrasonographic examination of the ileocaecocolic junction, where the thickness of the caecal wall, ileocolic lymph nodes and the echogenicity of the local fat were assessed. They all underwent a colonoscopy with a macroscopic assessment of the mucosa and biopsies for histology.
Results
An ultrasonographic hypoechoic nodular inner layer, which corresponded to the coalescence of multiple lymphoid follicles originating from the submucosa and protruding in the mucosa on histology, was visible in all parts of the caecum. The combined mucosa and submucosa was measured ultrasonographically and defined as the follicular layer. Although all cats were asymptomatic, 3/19 cats showed mild caecal inflammation on histology. The most discriminatory ultrasonographic parameter in assessing this subclinical inflammation was the thickness of the follicular layer at the entrance of the caecum, with a cut-off value of 2.0 mm. All cats (20/20) showed some degree of macroscopic ‘dimpling’ of the caecal mucosa on endoscopy.
Conclusions and relevance
Lymphoid follicles in the caecal mucosa and submucosa constitute a unique follicular layer on ultrasound. In asymptomatic cats, a subtle, non-clinically relevant inflammation may exist and this is correlated with an increased thickness of the follicular layer on ultrasound. On endoscopy, a ‘dimpled aspect’ to the caecal mucosa is a normal finding in the asymptomatic cat.
Introduction
The caecum is a blind diverticulum of the colon, at the transition between the colon and the ileum, located in the right hemiabdomen.1–4
Ileocaecocolic pathology is a relatively frequent disorder in feline gastroenterology. An ultrasonographic (US) examination on a cat presenting with dysorexia, diarrhoea, haematochezia, weight loss or vomiting calls for a proper scan of the ileocaecocolic junction and the caecum.
The normal US appearance of the feline caecum and ileocolic junction has been previously described by Besso et al. 5 Normal caecal wall thicknesses commonly used in cats are 1.0 mm at the inlet of the caecum and 2.5 mm in the cul-de-sac; the colonic wall upper limit is 2.5 mm.5–7 Normal caecal lymph nodes measure between 3.0 and 4.1 mm.5,8 Yet the exact location of the caecal measurements was not clearly specified in the previous study and the authors hypothesised that advances in US technology with the use of a higher-frequency probe might enable more information on this specific organ to be obtained. US changes of the ileocolic junction in cats are clinically relevant and have occasionally been reported in asymptomatic cats. 9 Despite the absence of digestive signs, the authors therefore hypothesised that a mild histological caecal inflammation could be found and that the caecal wall thickness would be related to this inflammation.
Moreover, the anatomy of the feline caecum is well described but there is little information on the endoscopic appearance of the normal feline caecum, whereas several textbooks and scientific papers discuss the colon.1,3,4,10
The first aim of this study was to describe the US appearance of the caecum in the asymptomatic adult cat. The second aim was to describe the macroscopic appearance of this organ during a colonoscopy and to obtain histological samples. Additionally, we aimed to correlate US, endoscopic and histological findings.
Materials and methods
The first step of this prospective study was to evaluate ex vivo a fresh caecum of an adult cat in order to depict precisely the location of the US measurements for the rest of the study.
Immediately after the death of a recently dewormed cat with no digestive signs, euthanased for chronic heart failure, the ileocaecocolic junction and ascending colon were withdrawn by a pathologist (JC) and placed in a bath of water for a US examination. Longitudinal images of the caecum were achieved sonographically with a linear 18 mHz transducer (MyLab 30) (HH and PP). The thickness of the proximal (the closest part to the colon) and distal (the closest to the cul-de-sac) parts of the caecal wall were both measured on a longitudinal plane, in the ventral wall, from the interface of the ultrasound with the luminal content to the external border of the serosa.
The caecum was fixed in formalin, then routinely processed and sectioned at 3 µm. It was stained with haematoxylin–eosin–saffron (HES) and evaluated by light microscopy by a board-certified pathologist (EL).
Study population
All procedures were approved by the Ethical Committee of the National Veterinary School of Alfort (agreement 29-08-2014). Healthy cats without clinical evidence of gastrointestinal disease (no diarrhoea, haematochezia, weight loss, vomiting or dysorexia during the last 3 months) were recruited from February to May 2014. Study cats were provided by employees or students of the veterinary school (7/20) or originated from a colony of reproductive laboratory cats (13/20). For each cat, a physical examination was performed and data regarding age, breed, diet and deworming were collected (HH and AB).
Anaesthesia
Cats were fasted for at least 12 h. The US and endoscopic evaluations were achieved under general anaesthesia: diazepam (Valium, 5 mg/kg IV; Roche) and propofol (Propovet, 0.25 mg/kg IV; Axience) were used for induction; the cats were then intubated and isoflurane gas anaesthesia (Vetflurane; Virbac) was used (2 l/min).
Ultrasonographic evaluation
The abdomen was clipped and each cat was placed in dorsal recumbency for the US examination, performed by the same observer (HH), under the supervision of a board-certified radiologist (PP). For each cat, the caecum, ascending, transverse and descending colon in longitudinal planes, the ileocolic lymph nodes and the adipose tissue surrounding the ileocaecocolic junction were examined.
The following parameters were recorded: the full thickness of the colonic and caecal walls on longitudinal planes, the thickness of the combined mucosa plus submucosa in the proximal and distal caecum, the content (gas, solid, liquid) of the caecum and the colon, the thickness of the ileocolic lymph nodes and the echogenicity of the mesenteric surrounding fat. The latter was qualified as a grade 0 – normoechoic fat compared with adjacent mesenteric fat and well-defined contours of the lymph nodes; grade 1 – hyperechoic fat and well-defined contours of the lymph nodes; and grade 2 – hyperechoic fat and blurred contours of the lymph nodes.
Endoscopic evaluation
Immediately after the US evaluation, cats were placed in left lateral recumbency and received an enema with warm water. 10 Smooth transabdominal palpation was performed in order to help faeces evacuation. All cats underwent a colonoscopy (8.6 mm diameter insertion tube [Video endoscope GIF 160; Olympus]) (VF and AB). The internal appearance of the caecum was described. Criteria from the World Small Animal Veterinary Association (WSAVA) Gastrointestinal Standardization Group were recorded. 11 As no standard was suggested in terms of number of modified parameters to be considered as inflammatory, we decided to classify a cat as such if at least two WSAVA criteria were modified. The operators also focused on the number of small caecal surface depressions (<10, 10–30, >30). Five mucosal biopsy specimens were collected for all cats with the same endoscopic forceps (smooth and fenestrated jaws of 2.3 mm external diameter [EndoJaw; Olympus Medical Systems]), two from the caecum, and one from the ascending and two from the descending colon. All specimens but one descending colonic biopsy were sent for histological evaluation. The remaining biopsy was sent for coronavirus detection by PCR.
Histological evaluation
The biopsies were routinely processed, sectioned at 3 µm and then stained with HES. Two authors (JC and EL) blindly evaluated the slides. A scoring system developed by the WSAVA Gastrointestinal Standardization Group was used. 12 Caecal samples were evaluated as if they were colonic biopsies and were classified as normal or inflammatory. A sample was considered as inflammatory if the intensity of the leukocytic infiltrate exceeded normal values. 12 Histological analysis was considered as the gold standard when evaluating the inflammatory status of cats.
Morphometric evaluation of lymphoid follicles on biopsies
In large intestinal samples, lymphoid follicles size was assessed by computer-assisted morphometric analysis (EL and JLS). A caecal and a colonic slide containing at least one lymphoid follicle were selected for each cat. Serial sections were stained with HES and the one with the largest lymphoid nodule was selected for evaluation. Digital pictures of the biopsies containing lymphoid follicles were acquired with a fully motorised research microscope (AxioCam HRc, logiciel AxioVision 4.6.3 SP1; Carl Zeiss). The total area of each biopsy and the area occupied by lymphoid follicles were calculated on digital picture using a dedicated software (Image J; US National Institutes of Health). 13 For each case, the ratio of the lymphoid follicles area to the area of the biopsy (lymphoid area ratio) was calculated for the colon and the caecum.
Detection of feline coronavirus
Feline coronavirus was systematically searched on caecal biopsy specimens. Quantitative reverse transcriptase PCR (RT-PCR) assays were performed. 14
Statistical analysis
An epidemiologist (LD) was involved in the design of the study and performed statistical analyses (SAS version 9.1; SAS Institute). Medians and interquartile ranges (IQR; first and third quartiles) were provided for all quantitative variables, as some of them were not normally distributed. In order to be able to compare our results with those in the literature, means were also provided when necessary. Receiver operating characteristic (ROC) curves were used to assess the accuracy of each measured US parameter and the morphometric ratio in classifying cats with inflammation vs without inflammation of the caecum, according to histological diagnosis that was considered as the gold standard. 15 A higher area under the ROC curve (AUROC) indicated a better accuracy of the parameter. Youden’s index was used to determine the optimal cut-off values for each measured US parameter. 16 The cut-off values provided were then applied to calculate sensitivities and specificities for each measured parameter. The US parameter with the highest AUROC was used to classify the cats as inflammatory vs normal for the caecum.
κ coefficients were calculated to quantify the agreement on the inflammatory status between histological, US and endoscopic results, both in the colon and the caecum. The strength of agreement was considered poor, slight, fair, moderate, substantial and almost perfect for κ values of <0.00, 0.00–0.20, 0.21–0.40, 0.41–0.60, 0.61–0.80 and 0.81–1.00, respectively. 17
Results
During the ex vivo evaluation of the fresh caecum, both proximal and distal parts of the caecum appeared to have clear layering. From the innermost part to the most external part of the caecum, the following layers were observed: the interface with the luminal content (hyperechoic line), a thick layer of multiple hypoechoic coalescent nodular areas, a thin hyperechoic band with scalloped borders, a thin hypoechoic and a thin hyperechoic even layer (Figure 1). On histological samples, the caecum had a typical structure of a tubular digestive organ with abundant gut-associated lymphoid tissue, as previously described.1,2 In the submucosa, solitary-to-confluent follicular lymphatic nodules were present; they were most numerous in the distal part (Figure 2).2,4 Almost all of these lymphatic follicles extended into the mucosa by disrupting the lamina muscularis. The US hypoechoic nodular inner layer thus corresponded to the coalescence of multiple lymphoid follicles originating from the submucosa and protruding in the mucosa on histology. Consequently, it was not appropriate to refer to the inner hypoechoic layer as the ‘mucosa’. The most accurate measurement for this layer of follicles was to evaluate the combined mucosal and submucosal layer (Figure 1). Henceforth, it is referred to as the ‘follicular layer’ (FL).
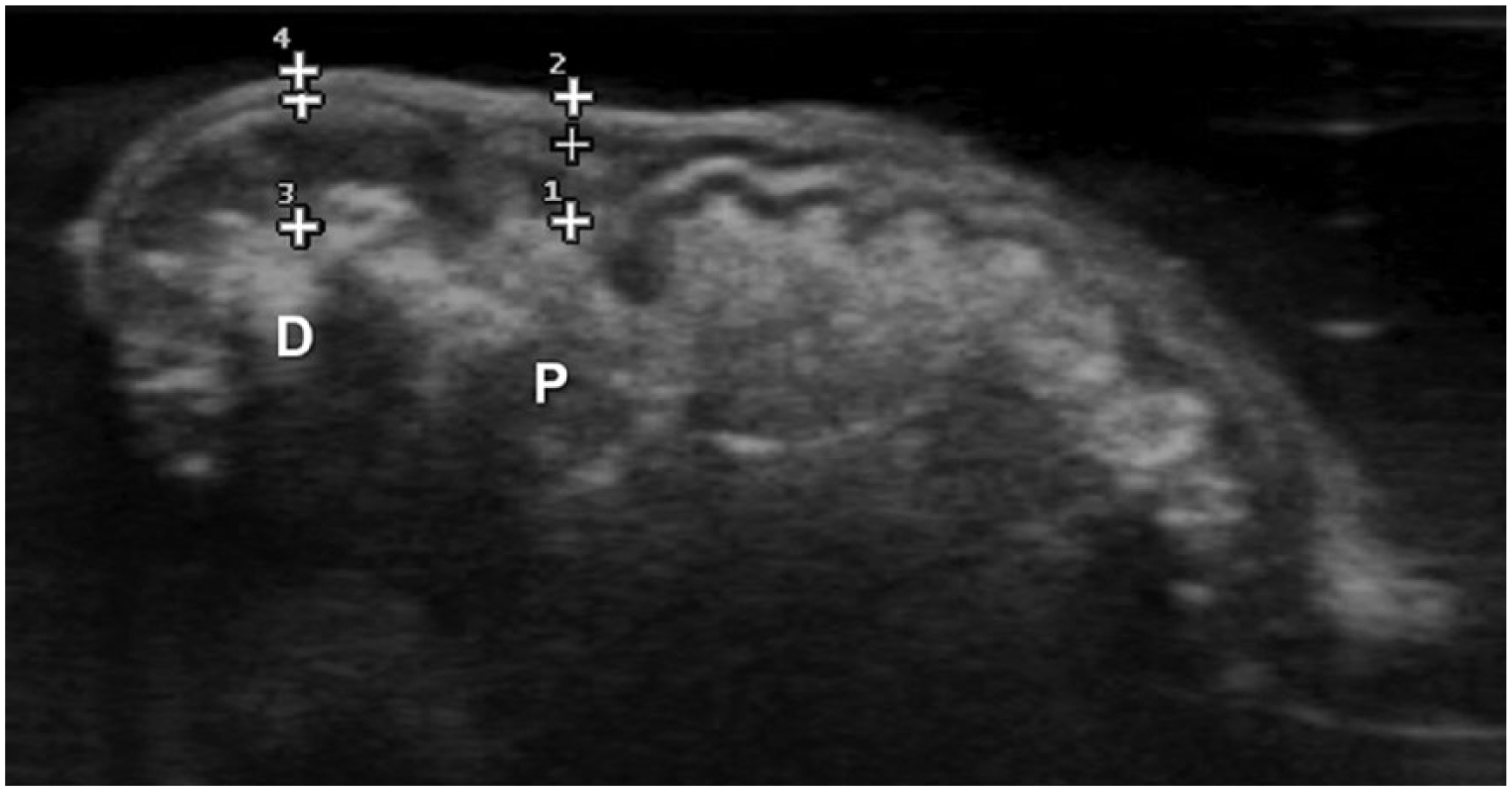
Ultrasonographic image on longitudinal plane of an ex vivo caecum placed in a water bath. The thickness of the proximal (closest to the colon; letter P) and distal (closest to the cul-de-sac; letter D) parts of the caecal wall were measured in the ventral wall, from the interface with the luminal content to the external border of the serosa (from callipers 1 to 2 and callipers 3 to 4, respectively). The intermediate calliper depicts the outer border of the combined mucosal plus submucosal layers, the so-called ‘follicular layer’

Photomicrograph of a normal feline caecum (sagittal section). The wall is made of four layers: (1) mucosa, (2) submucosa, (3) tunica muscularis and (4) serosa. Follicular lymphatic nodules (squares) originate from the submucosa and protrude into the mucosa. They are more numerous in the distal part of the caecum (D) than in the proximal part (P). The serosa contains ileocolic lymph nodes (arrowheads). Haematoxylin–eosin–saffron, scale bar = 2000 µm
Twenty cats (four females, 16 males) were included in this study. Seven were neutered (six males, one female). Represented breeds were mixed Siamese (10/20), domestic shorthair (9/20) and Birman (1/20). The age of the cats ranged from 1–8 years, with a median (IQR) of 3 (1–7) years. The median (IQR) weight was 4.3 (3.8–4.6) kg. Eighteen cats had recently been dewormed; the two others had not been dewormed in the previous 6 months. All cats lived indoors.
Ultrasonographic findings
The caecum was identified in all cats, but its position varied between individuals: it was found in the right central ventral abdomen (12/20). In eight cats, the caecum was in a more cranial position, close to or under the last rib. Once the ileocaecocolic junction was located, the caecum was found in most cases with a slight clockwise rotation (14/20); in the six other cats, it was found with an anticlockwise rotation, to reach a longitudinal view.
The layers previously described were visible in all parts of the ventral wall on all cats (Figure 3). The caecum was empty with or without a small amount of gas (15/20); a solid or liquid content was also observed (5/20).
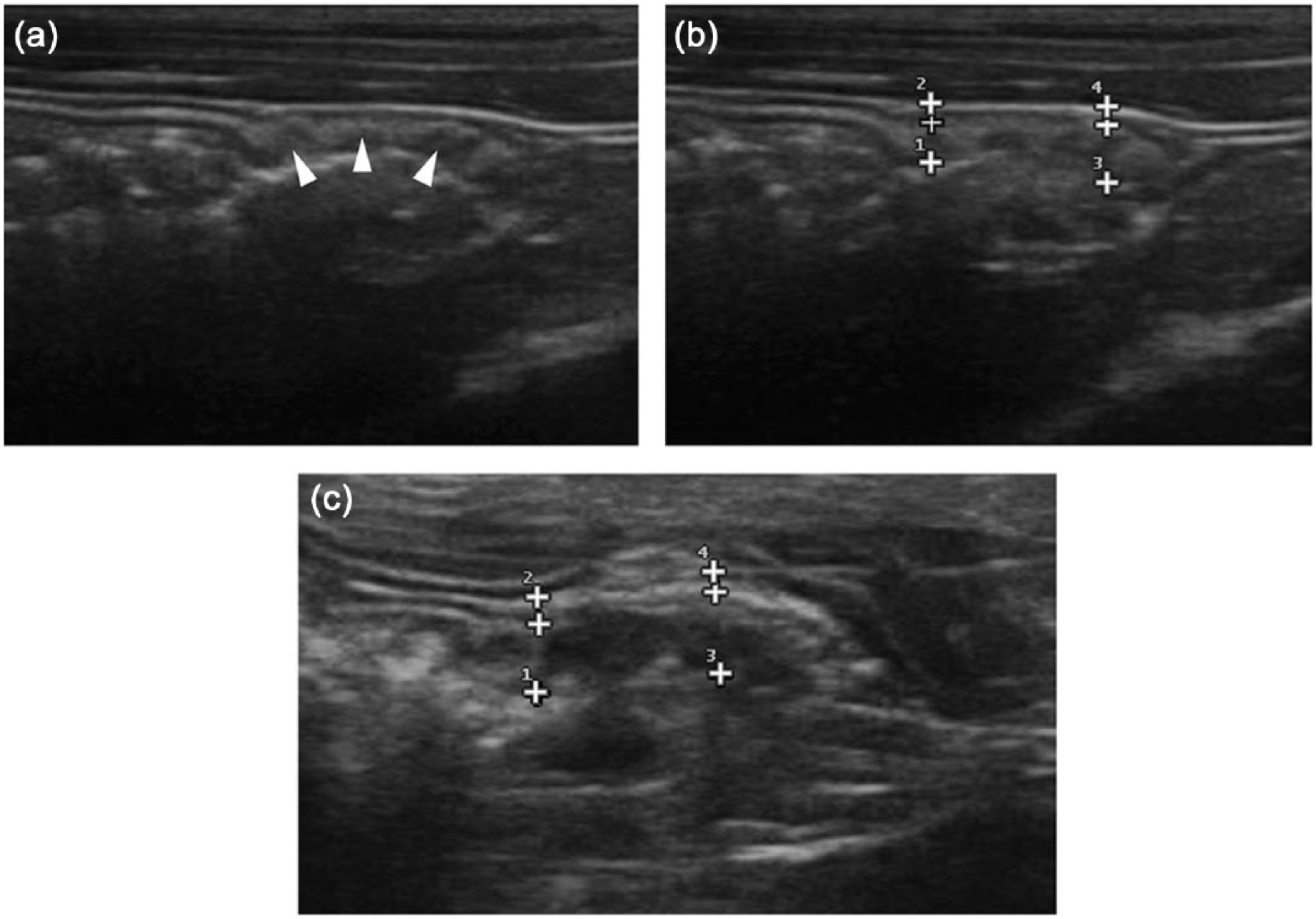
Ultrasonographic caecal longitudinal images of two different asymptomatic cats. The caecum of the cat represented in (a) and (b) was classified as normal on histopathology and the caecum of the cat depicted in (c) as mildly inflammatory. Note the multiple hypoechoic coalescent nodular areas (arrowheads) (a) surrounded by a thin hyperechoic band with scalloped borders. The follicular layer is measured between callipers 1 or 3 and the intermediate calliper (b,c). Note that the follicular layer occupies a larger proportion of the full thickness of the caecum in (c) compared with (b)
The medians (IQRs) of the full-thickness caecal walls and of the FL are presented in Table 1. Means (minimum–maximum) of the proximal and distal full-thickness caecal walls were, respectively, 2.5 (1.7–3.6) mm and 3.1 (2.4–4.1) mm. The caecal FL had a mean (minimum–maximum) thickness of 1.8 (1.3–2.9) mm and 2.4 (1.5–3.2) mm in the proximal and distal parts, respectively. Median thicknesses (IQRs) of the colon and ileocolic lymph nodes are presented in Table 1.
Median and interquartile range of the different parameters measured ultrasonographically

FL = follicular layer; IQR = interquartile range
The adipose tissue surrounding the ileocaecocolic junction displayed a grade 0 (13/20) or 1 of steatitis (7/20) and no grade 2 (0/20).
Among caecal US parameters, the two follicular layer parameters (proximal and distal FL) had the highest AUROC: 0.94 and 0.83, respectively. A proximal FL >2.0 mm resulted in a sensitivity of 87% and a specificity of 100% in detecting mild caecal inflammation. A distal FL >2.5 mm resulted in a sensitivity of 75% and a specificity of 100%. The other US parameters and their AUROC values are presented in Table 2.
Area under the receiver–operating characteristic (AUROC) curves to quantify the accuracy of the quantitative caecal ultrasonographic parameters in detecting mild caecal inflammation

CI = confidence interval; FL = follicular layer; VLN = ventral lymph node; DLN = dorsal lymph node
Concerning the ROC analysis of the colonic US parameters, the size of the dorsal lymph node (DLN) had an AUROC of 0.76 and the size of the ventral lymph node had an AUROC of 0.71. The thickness of the colonic ascending wall had an AUROC of 0.66. The cut-off value was estimated at 3.0 mm for the DLN with a sensitivity of 71% and a specificity of 80%.
All cats were then classified as normal vs inflammatory for both the caecum and the colon using the parameter with the highest AUROC and its cut-off value.
With an inflammatory caecum defined by a proximal FL >2.0 mm, cats were classified as inflammatory (5/20) or normal (15/20). With an inflammatory colon defined by a DLN >3.0 mm, cats were classified as inflammatory (9/20) or normal (11/20).
Endoscopic findings
The normal colonic mucosa was smooth, pale orange–pink and glistening. Submucosal blood vessels were often seen throughout the colon.
The caecum appeared in all cats as a small blinded cavity in direct continuity with the ascending colon. The mucosa displayed a ‘dimpled aspect’ due to the presence of small surface depressions. This dimpled aspect had a variable intensity and was noted in all cats: it was either mild (9/20), moderate (7/20) or severe (4/20) (Figure 4). Caecal wall rigidity was evaluated during biopsies and was more intense than in the colon, in all cats.
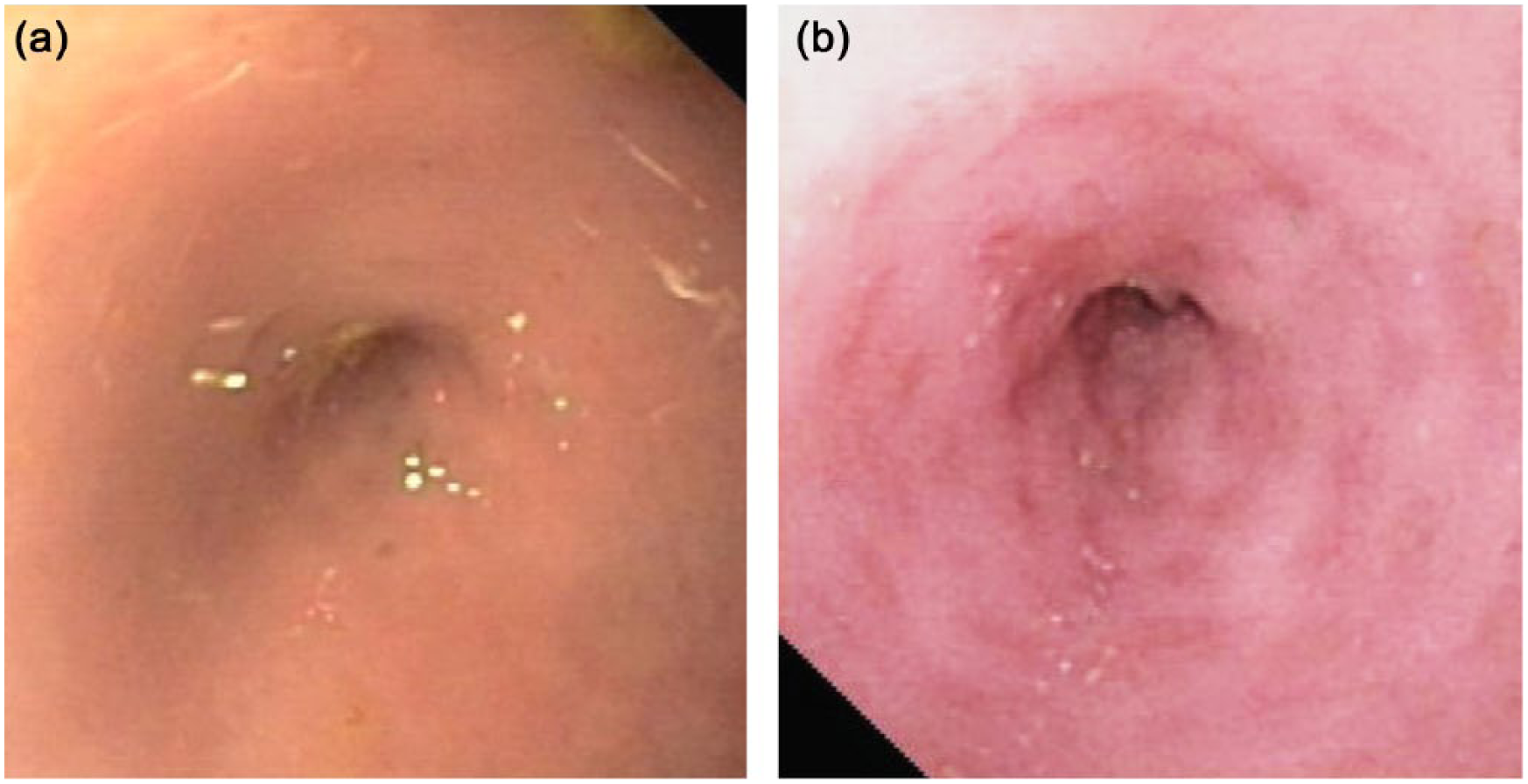
Endoscopic images of the caecum in two asymptomatic cats. Note two different degrees of the normal mucosal ‘dimpling’; (a) mild; (b) moderate
According to WSAVA classification, the caecum of the studied cats showed a modification of one criterion (7/20), two criteria (3/20) or three criteria (1/20). Cats were then classified as normal (16/20) or inflammatory (4/20).
Macroscopic colonic evaluation was considered as normal (15/20) or inflammatory (5/20). Only one cat showed inflammatory changes both in the caecum and the colon.
Feline coronavirus analysis
Feline coronavirus was searched in 17 cats (biopsies were not available for RT-PCR analysis for three cats). Results were slightly positive (1; 4/17), highly positive (2; 4/17) or negative (0; 9/17).
Histological evaluation
Caecal biopsies were evaluated (19/20; one cat had an inadequate biopsy for histological analysis) and classified as normal (16/19) or inflammatory (3/19). The inflammatory caecal biopsies only displayed mild neutrophilic inflammation, with 5–10 neutrophils in the lamina propria per × 40 field (Figure 5).

Photomicrographs of a normal caecal mucosa. (a) Glands in cross section and lamina propria with low cellularity without any neutrophils; and (b) caecal mucosa with mild neutrophilic inflammation (neutrophils are indicated with arrows). Five to 10 neutrophils per × 40 field were found in the lamina propria in this biopsy. Haematoxylin–eosin–saffron, scale bar = 20 µm
Colonic biopsies were evaluated (19/20; one cat had inadequate material for histological analysis) and classified as normal (14/19) or inflammatory (5/19). The inflammatory colonic biopsies only displayed mild neutrophilic inflammation, but one cat for which moderate neutrophilic inflammation (between 20 and 30 neutrophils per × 40 field) was associated with an overlying ulcer.
A histologically normal colon corresponded to a histologically normal caecum in most cats (13/18). An inflammatory colon corresponded to an inflammatory caecum (2/18). Two cats had an inflammatory colon and a normal caecum; one cat was the other way round.
There was no correlation between inflammation on histology and a non-dewormed status.
Morphometric evaluation of lymphoid follicles on biopsies
A biopsy containing one or more lymphoid follicles could be obtained from sets of caecal (16/19) and colonic biopsies (8/19). The values of calculated areas are summarised in Table 3. The AUROC value for lymphoid area ratio in the caecum was 0.56; given this low value, cut-off value, specificity and sensitivity were considered as not relevant.
Average values obtained by morphometric evaluation of caecal and colonic biopsies

Data are median (interquartile range)
Agreement between US, endoscopic and histological results
Concerning the caecum, the agreement between histological and US inflammatory classification was substantial (κ = 0.69); there was a slight agreement between histological and endoscopic results (κ = 0.13; Table 4).
κ values (95% confidence intervals [CIs]) between ultrasonographic, endoscopic and histological results in the caecum

Regarding the colon, the agreement between histological and US results was moderate (κ = 0.48); the agreement between histological and endoscopic results was almost perfect (κ = 0.85).
κ values assessing the agreement between caecal and colonic inflammation among a single evaluation technique were all moderate or less satisfying (κ <0.50).
The κ scores between the caecal histological inflammatory state and the presence of a surrounding steatitis on ultrasonography, between the caecal histological inflammatory state and the presence of a third ileocolic lymph node, and between the caecal histological inflammatory state and the intensity of the dimpled aspect were all considered as very poor (κ <0). Agreement between the caecal histological inflammatory state and the coronavirus state was fair (κ = 0.31).
Discussion
In this study, we prospectively compared US, endoscopic and histological caecocolic findings in a population of 20 asymptomatic cats. This study demonstrated that a subclinical mild caecal inflammation could be found in asymptomatic cats. Among all measured US parameters, the most accurate one in detecting this subclinical state was the thickness of the caecal FL and more specifically its thickness at the entrance of the caecum. Ultrasonography had a higher agreement with histology than with endoscopy in the evaluation of a mildly inflamed caecum, when an ultrasonographically ‘normal’ caecum was defined as a proximal FL ⩽2.0 mm.
The caecal wall measurements in this study revealed themselves to be higher than those provided by Besso et al: 5 the study population had a mean caecal wall of 2.5 mm in the proximal part and 3.1 mm in the distal part. We hypothesised that this was due to the higher frequency probe used in this study, which allowed more precise measurements, or to a different measurement location and/or orientation. Hypoechoic follicles have already been described ultrasonographically in the submucosa of the colon of dogs and cats, and characterised as a reactive intraparietal lymphoid follicles. 18 The authors hypothesised them to be an indicator of colonic inflammatory disease. In this study, we revealed that these follicles are rather overlapping the caecal mucosa and submucosa and that they can be seen ultrasonographically in a normal caecum, especially in the distal part; however, their increased number and/or size, especially in the proximal part of the caecum, revealed itself as a US marker of mild caecal inflammation, with a cut-off thickness value of 2.0 mm for the proximal FL. Given the imperfect sensitivity, a full assessment of the caecum seems nonetheless necessary in order to minimise false-negative normal caecum. In a previous paper on a population of cats with US abnormalities of the caecum, ileocolic junction, ileocolic or colic lymph nodes, and where almost all cats had digestive clinical signs, the most common US findings were lymphadenopathy, increased caecal wall thickness and hyperechoic fat. 9 Surprisingly, there was a very poor agreement in the present study between the caecal histological inflammatory state and the presence of steatitis, which is a US parameter commonly used to assess inflammation. We hypothesised that some cats may have had a scar steatitis, resulting from a previous local inflammation.
Despite the absence of clinical signs, 16% of the study population showed histological signs of mild caecal inflammation and 26% of mild colonic inflammation. Thus, the absence of digestive signs does not rule out colonic or caecal mild inflammation. In symptomatic cats, an increased caecal wall thickness can be due to other lesions than inflammation, including neoplasia. Cut-off values, sensitivities and specificities of the different US parameters to detect other lesions (such as neoplasia) remain to be evaluated in symptomatic cats.
Feline coronavirus was systematically searched on caecal biopsies as a large number of cats in this study lived collectively. 19 Almost 50% of the population was coronavirus-positive but all cats were asymptomatic as this was an inclusion criteria. A fair agreement was observed between coronavirus status and inflammation on histology. However, histological findings do not support a causal link as the inflammation was mild and not specific. No pyogranulomas were found; thus, feline infectious peritonitis was unlikely. There was also no correlation between inflammation on histology and a non-dewormed status.
Based on the WSAVA endoscopic criteria, only a slight agreement was found between endoscopic and histological evaluation in the inflammatory state of the caecum, whereas this agreement was almost perfect regarding the colonic inflammatory state. It thus seems that the WSAVA endoscopic criteria may not be relevant in detecting mild forms of asymptomatic caecal inflammation; another possibility is that the decision made by us to define an abnormal caecum as a caecum with at least two WSAVA modified criteria was not suitable; however, it was statistically the most appropriate combination. A variable intensity of the caecal mucosal dimpled aspect was found in all cats. Nevertheless, no concordance was found between this particular aspect and histological inflammation: it thus reveals itself as a normal caecal finding in asymptomatic cats.
The agreement between caecal and colonic inflammation among a single evaluation technique was unsatisfactory for all three techniques. Thus, evaluating the colon is not sufficient to assess the caecal inflammatory state and likewise.
Concerning morphometric evaluation, the follicle area on the whole biopsy, the number of follicles and their diameter were parameters considered for evaluation. The last two were impossible to evaluate owing to a frequent confluence of lymphoid follicles. Measuring the relative area occupied by lymphoid follicles on the biopsy was then elected. This criterion had already been described for evaluation of lymphoid follicles in the stomach of dogs and cats. 12 In this study, the obtained lymphoid area ratios were highly variable for caecal, as well as for colonic biopsies. This could be explained, on the one hand, by variability in sampling location, even though all biopsies were taken from the distal part of the caecum, and variability in depth of biopsies. On the other hand, for a given biopsy, the orientation of the biopsy in the block and its cutting plane were inconstant. Therefore, for a given lymphoid follicle, the maximal lymphoid area could not always be reached, except with systematic serial cutting, which is, obviously, not possible in routine diagnostic. Crush artefacts could also have distorted area measurement. For the caecum, statistical analysis indicated a poor discriminatory ability of this ratio for caecal inflammatory and normal status.
There were some limitations to this study. The sample of cats was relatively small and there was a majority of Siamese intact male cats, which is not representative of the feline population. But, to our knowledge, there was no direct incidence of the breed or sex of the cats on the parameters measured ultrasonographically. The low number of cats may have led to imprecise estimations of the κ values between histological, endoscopic and US results.
Conclusions
Lymphoid follicles are numerous in the caecum of asymptomatic cats and constitute a unique follicular layer on ultrasonography. A subtle, non-clinically relevant inflammation may exist in the caecum of asymptomatic cats and the US thickness of this follicular layer at the entrance of the caecum is the best parameter for its evaluation. A dimpled endoscopic aspect to the caecum can be found in asymptomatic cats. Ultrasonography, endoscopy and histopathology are complementary to evaluate the caecum fully.
Footnotes
Acknowledgements
The authors thank, in particular, Dr Alain Fontbonne and his team at the Reproduction Department, Ecole Nationale Vétérinaire d’Alfort, for help with the practical realisation of this study. The study was performed at the National Veterinary School of Alfort, Maisons-Alfort, France.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
