Abstract
A 3-year-old male castrated domestic shorthair cat was presented with an acute history of lethargy and decreased appetite. Pertinent physical examination abnormalities included palpable irregularity of the right kidney and pain on palpation of the left kidney. Ultrasonographic imaging of the abdomen revealed gas present at the corticomedullary junction of the left kidney, consistent with emphysematous pyelonephritis, as well as emphysematous cystitis. While quantitative urine culture via pyelocentesis yielded a negative culture, a sample via cystocentesis was positive for Escherichia coli and emphysematous changes were presumed most likely secondary to an ascending infection. The purpose of this report is to describe the temporary management of ureteral obstruction secondary to emphysematous pyelonephritis using a ureteral stent in a cat.
Introduction
Emphysematous infections of the urinary tract are rare in veterinary medicine. Reported cases include emphysematous cystitis (EC), a condition characterized by the accumulation of gas within the bladder wall, lumen or both. This condition occurs most commonly in dogs with diabetes mellitus or primary renal glucosuria, and is rarely reported in non-diabetic patients. Glucose-fermenting bacteria within the urinary bladder, commonly Escherichia coli, produce CO2, which collects in the lumen or forms vesicles in the bladder wall.1–3 Even more rare are emphysematous infections of the upper urinary tract, namely the kidney. Emphysematous nephritis has been reported in the human literature and again most commonly occurs with diabetes mellitus.4–6 This article reports the temporary use of a ureteral stent in the management of a case of emphysematous pyelonephritis (EPN) suspected to have contributed to ureteral obstruction in a domestic shorthair cat.
Case description
A 3-year-old male castrated domestic shorthair cat was presented to the Animal Specialty and Emergency Center (ASEC) for evaluation and treatment of lethargy and a decreased appetite. The clinical signs developed over a course of 5 days while the owners were away, and initial diagnostics performed by the primary care provider included a complete blood count (CBC), serum biochemical profile and urinalysis (UA) obtained by cystocentesis. The CBC revealed a leukocytosis (19,600/µl, reference interval [RI] 3500–16,000/µl) with a neutrophilia (15,800/µl, RI 2500–8500/µl). Abnormalities noted on serum biochemical profile included elevations in blood urea nitrogen (BUN; 224 mg/dl, RI 14–36 mg/dl), creatinine (Cr; 3.5 mg/dl, RI 0.6–2.4 mg/dl), BUN/Cr ratio (64, RI 4–33), magnesium (6.4 mEq/l, RI 1.5–2.5 mEq/l), phosphorus (10.1 mg/dl, RI 2.4–8.2 mg/dl), potassium (9.2 mEq/l, RI 3.4–5.6 mEq/l), cholesterol (261 mg/dl, RI 75–220 mg/dl), triglycerides (593 mg/dl, RI 25–160 mg/dl), amylase (1491 IU/l, RI 100–1200 IU/l) and creatinine kinase (1604 IU/l, RI 56–529 IU/l), as well as decreases in sodium (126 mEq/l, RI 145–158 mEq/l), chloride (78 mEq/l, RI 104–128 mEq/l) and Na/K ratio (14, RI 32–41). UA revealed a specific gravity of 1.018, a pH of 6.5, 3+ protein, 3+ blood, 4–10 white blood cells per high-power field (HPF), 0–1 squamous epithelial cells/HPF, 2–3 red blood cells/HPF and >100 cocci and rods/HPF. A quantitative urine culture was submitted and performed via broth dilution (minimum inhibitory concentration). The culture identified 50,000–100,000 colony-forming units per ml of E coli, which later returned as susceptible to both amoxicillin/clavulanic acid and enrofloxacin. Plain abdominal radiographs showed multifocal gas lucencies in the region of the left kidney, and the patient was administered subcutaneous ampicillin (Polyflex; Boehringer Ingelheim), intramuscular enrofloxacin (Baytril; Pfizer) and began on oral clindamycin prior to referral. Doses of all drugs from the primary veterinary were unavailable for review. The cat was seen for an initial consultation at ASEC within 24 h of its visit with the primary care veterinarian.
At presentation, the patient weighed 6.7 kg and was given a body condition score of 5/9. Temperature, respiration and heart rate were all within normal limits. Physical examination abnormalities included mild periodontal disease, 5% dehydration, generalized flaking of the hair coat, slight irregularity to the right kidney and pain on palpation of the left kidney. Thoracic radiographs, abdominal radiographs and abdominal ultrasound were performed. Thoracic radiographs were unremarkable and abdominal radiographs revealed changes consistent with initial radiographs. The right kidney was unaffected and continued evidence of EPN existed owing to multifocal gas lucencies visualized in the left kidney (Figure 1). Abdominal ultrasound revealed irregularity to the left kidney, as well as severe hydronephrosis (renal pelvis measuring 2.77 cm) and a moderate amount of gas present at the corticomedullary junction. The left proximal ureter displayed no evidence of obstruction, measuring 0.19 cm in width. Additionally, there was a moderate amount of echogenic, gravity-dependent sediment within the renal pelvis (Figure 2). Although the emphysematous changes were presumably secondary to the same E coli in the bladder (confirmed via urine culture), a urine sample was taken from the left renal pelvis via ultrasound-guided pyelocentesis in order to document the same infection. This culture was negative, but the patient had been receiving oral enrofloxacin for approximately 36 h prior to sample submission. Although anterograde pyelography would have been the preferred diagnostic to confirm a diagnosis of ureteral obstruction, and this option was recommended to owners, this was not performed owing to financial constraints.

Initial abdominal radiographs showing free gas within the left kidney and urinary bladder

Preoperative left renal ultrasound showing severe left hydronephrosis with gas shadowing within parenchyma
The cat was treated for the moderate azotemia with a buffered crystalloid solution (Plasma-Lyte A Injection pH 7.4 Multiple Electrolytes Injection, Type 1, USP; Abbott Laboratories) with no additives (at a maintenance rate as calculated for 70* body weight [BW]3/4), buprenorphine (0.1 mg/kg IV q6h Buprenex; Reckitt-Benckiser), famotidine (Famotidine Injection, USP; 0.5 mg/kd IV q12h; West-Ward Pharmaceuticals), enrofloxacin (5 mg/kg IV q24h Baytril; Bayer) and ampicillin Na/sulbactam Na (30 mg/kg IV q8h Unasyn; Pfizer). As the left kidney exhibited continued severe hydronephrosis after 3 days of hospitalization, an ultrasound-guided decompressive pyelocentesis was performed under heavy sedation with midazolam 0.3 mg/kg and a propofol constant rate infusion (total 10 mg/kg IV) to effect throughout the procedure. Turbid urine (35 ml) was removed from the left renal pelvis, with bacteria and degenerate neutrophils visible on in-house cytologic examination. Serial biochemistry and electrolyte analyses were performed to follow resolution of the azotemia, with normalization of serum BUN (29 mg/dl) and creatinine (0.7 mg/dl) after 3 days. The patient remained normotensive throughout hospitalization. It was discussed with the owners that given the degree of persistent hydronephrosis, emphysematous changes visualized radiographically and a positive quantitative urine culture, severe inflammation from EPN causing left-sided ureteral obstruction was inferred. It is important to note that although a diagnosis of ureteral obstruction was never confirmed prior to surgical intervention and placement of a ureteral stent, it was strongly suspected based on the cat’s clinicopathologic and radiographic abnormalities. The decision to place a stent was made based on strong indications of an obstructive process.
Following resolution of azotemia, an exploratory celiotomy and distal cystotomy/proximal urethrotomy were performed to allow placement of a ureteral stent. Other than the severely enlarged left kidney, the remainder of the abdominal viscera was unremarkable. The left kidney and the bladder were isolated with laparotomy sponges. The fluoroscopy unit was positioned to allow visualization of both the left kidney and the bladder in the field of view. A ventral distal cystotomy combined with a ventral proximal urethrotomy was performed to visualize the ureterovesicular opening of the left ureter. A 2.5 Fr 14 cm ureteral stent was placed under fluoroscopic guidance with the use of an 0.018 guide wire-advanced retrograde into the left renal pelvis via the left ureter. No evidence of an extra- or intraluminal mechanical obstruction (such as a mass or calculi) was identified in the region of the left proximal ureter during surgery. Prior to closing the cystotomy incision a urinary catheter was placed aseptically in order to ensure adequate postoperative urine output (1–2 ml/kg/h). Postoperative radiographs were performed that confirmed appropriate placement of the ureteral stent and urinary catheter (Figure 3).
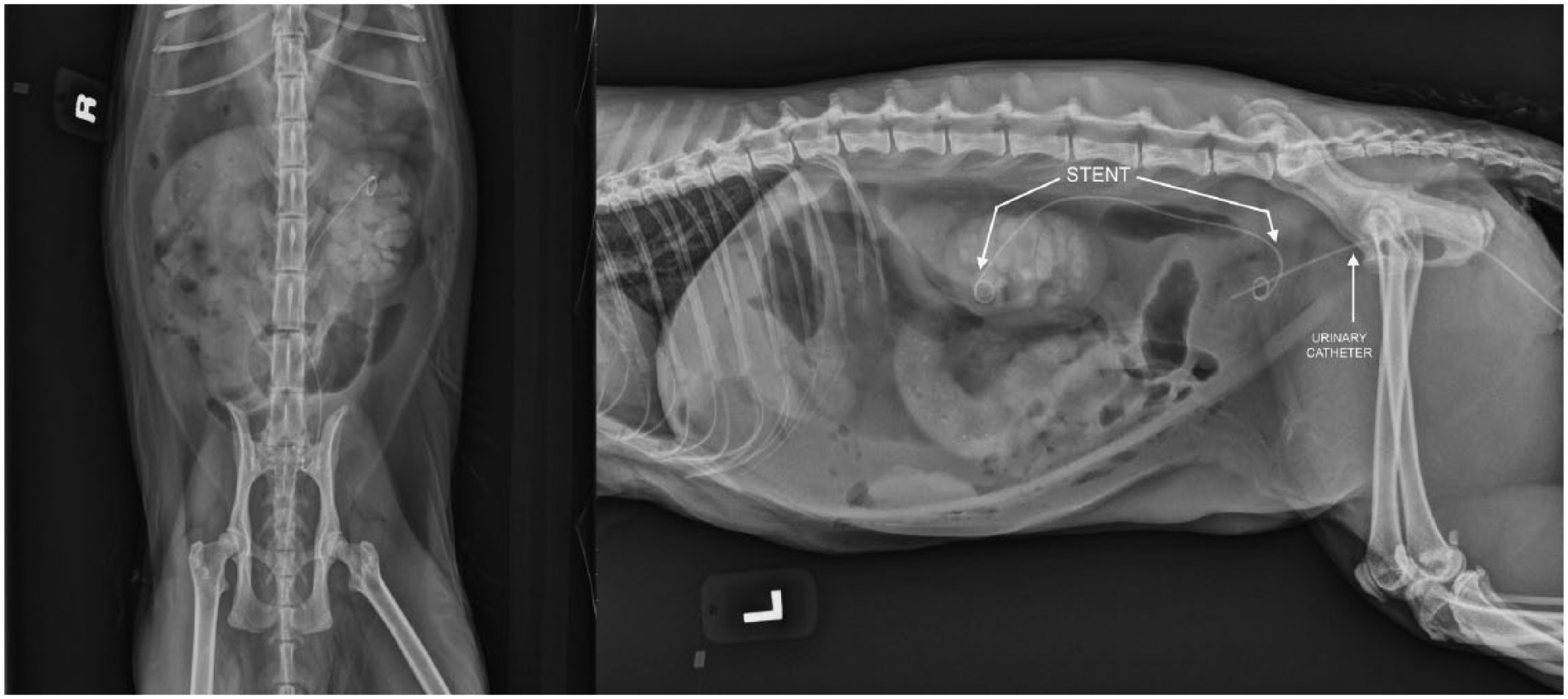
Immediate postoperative abdominal radiographs showing appropriate placement of left ureteral stent and urinary catheter with contrast within the left renal pelvis
Following surgery, urine output was monitored and the fluid rate was adjusted to compensate for postobstructive diuresis. Urine production was noted to be appropriate (~2 ml/kg/h), and the hematuria began to resolve within 24 h of surgery. It was at this time that the urinary catheter was removed. The patient was discharged to the owner 3 days postoperatively with enrofloxacin (5.6 mg/kg PO q24h), buprenorphine (0.01 mg/kg PO q12h) and lactated Ringer’s solution (100 ml SC q24h).
One week after discharge, the owner reported that the cat had resumed normal energy and appetite levels, with no evidence of hematuria, stranguria or pollakiuria. A follow-up abdominal ultrasound revealed persistent but improved distension of the renal pelvis, measuring 1.40 cm in height and 0.95 cm in width but improved in comparison with prior to stent placement (1.58 cm in height and 2.77 cm in width) (Figure 4). Two weeks following initiation of antibiotics, a quantitative urine culture obtained by cystocentesis was negative for bacterial growth. Recommendations included UA and culture every 3 months for the next year or if clinical signs recurred. Three months postoperatively, no gross hematuria, stranguria or pollakiuria were observed.

One week postoperative left renal ultrasound showing improvement of left hydronephrosis with absence of gas
Nine months following initial stent placement, recurrent marked hydronephrosis of the left kidney was found, with the renal pelvis measuring up to 3.0 cm in width and 3.0–3.5 cm in length. A repeat chemistry panel showed normal serum BUN (26 mg/dl, RI 15–34 mg/dl) and creatinine (1.5 mg/dl, RI 1.0–2.2 mg/dl), which was attributed to presumptive partial function of the right kidney. No UA was performed here given that serial UA (every 3 months) while the patient was clinically normal showed decreased urine concentrating ability (eg, urine specific gravity ranging from 1.020–1.026). Although renal scintigraphy would have been ideal to document remaining renal function, this was not financially feasible for the owners. Based upon the severity and chronicity of the hydronephrosis, the left kidney was presumed to contain little-to-no functioning tissue. The owners were presented with several treatment options, which included nephrectomy, stent removal and pyelocentesis. The owners elected to perform nephrectomy based on financial considerations while also seeking short- and long-term resolution of the patient’s peruria and hematuria. A culture of renal pelvis was performed at the time of the nephrectomy to decide the use of postoperative antibiotics, which was not indicated owing to the negative culture. The result indicated that the pyelectasia was likely due to stent obstruction/irritation, which is a known complication with ureteral stents. 7 It is once again important to note function of the right kidney was unknown prior to left-sided nephrectomy.
A left nephrectomy was performed with concurrent removal of the previously placed ureteral stent. No other abnormalities were identified during the laparotomy. The abdomen was lavaged with sterile saline prior to closure. The patient was maintained on a buffered crystalloid solution (Plasma-Lyte A Injection pH 7.4 Multiple Electrolytes Injection, Type 1, USP; Abbott Laboratories) with no additives (at a maintenance rate as calculated for 70*BW3/4), fentanyl continuous rate infusion (2 μg/kg/h IV), ampicillin (22 mg/kg IV q8h), enrofloxacin (5 mg/kg IV q24h), famotidine (0.5 mg/kg IV q12h) and dolasetron (0.6 mg/kg IV q24h).
Discussion
To our knowledge, this is the first reported case in the veterinary literature of attempted management of EPN with a ureteral stent. EPN is described in the human literature as infection that results in gas formation in the renal parenchyma, collecting systems or perinephric tissues. 4 Gas production is secondary to glucose-fermenting bacteria, with E coli, Klebsiella species and a few Proteus species documented as the most common bacterial isolates for this condition.4,8–10 However, other reported cases in the human literature suggest that certain bacterial strains are capable of producing high levels of nitrogen, CO2 and hydrogen. This may explain how emphysematous urinary tract infections develop in the absence of hyperglycemia with subsequent glucosuria.4,11 In humans, periodontal disease and immunosuppression have also been reported as predisposing factors.6,12 Traditional surgery including ureteronephrectomy was the recommended therapeutic intervention in human medicine until recently, with the increasing utilization of interventional techniques such as percutaneous catheters. A shift occurred in the 1980s with the standard of care being minimally invasive techniques in combination with medical management for EPN and other causes of ureteral obstruction.4–6,8,11 Percutaneous drainage (eg, drainage via catheters or stents placed with computed tomographic guidance) is recommended for patients with localized areas of gas who still possess functioning renal tissue. 6 Complication rates and failure following minimally invasive techniques alone does increase with increasing severity of EPN.Two types of EPN have been identified in humans based on ultrasound, with type I indicating parenchymal destruction and/or fluid accumulation, and type II denoted by the presence of fluid in the renal or perirenal spaces and gas in the collecting system. 13 Type I EPN has been associated with a worse prognosis than type II. One study reported that all patients classified as class I or II EPN were successfully managed with a ureteral stent (and/or percutaneous drainage catheters) and antibiotic therapy, whereas patients with more severe classes (classes III and IV) had failure rates as high as 92%. 14 A review of 10 retrospective studies found significantly less mortality (13.5%) in eligible patients managed with percutaneous drainage and antibiotics compared with patients who underwent emergency nephrectomies. 8 Although it is likely that patients requiring emergency nephrectomies were of advanced stages with systemic involvement, these studies underscore the value of minimally invasive procedures in patients with lower-grade EPN.
Contrary to EPN, numerous reports of EC are documented in the veterinary literature, and are also most commonly reported in diabetic dogs, with no sex predilection.9,10 The most common bacteria isolated have been E coli, along with Clostridium species and Aerobacter aerogenes. In non-diabetic dogs, development of EC has been secondary to a primary condition such as bladder trigone diverticulum, long-standing steroid administration or chronic urinary tract infections.1,3,10,15 Two recent cases were documented in a publication by Moon et al, 16 which reviews diagnosis and medical management for both a dog and cat with concurrent EC and EPN. Although other previous reports of both EC and EPN exist,1,3,12,15,17 it is important to note that this is only the second documented case of EPN in a non-diabetic feline, and the first to employ a ureteral stent. Davies and Williams reports EC (in the absence of concurrent EPN) caused by an E coli species in a non-diabetic cat. 12 Another report details a diabetic dog with emphysematous EC, peritonitis and endocarditis secondary to a gas-producing E coli. 2 Previous cases utilize medical management but do not attempt minimally invasive therapeutic options documented in the human literature for patients with EPN. Similar to other studies, this case details diagnosis and treatment strategies, but no conclusions regarding prognosis may be drawn.
Diagnostic imaging tools have been reported for both EPN and EC in the human and veterinary literature, respectively. Ultrasound has shown to be superior to radiographs for detection of EC in veterinary patients, 10 so it is reasonable to extrapolate that it may also prove more sensitive and specific than radiographs alone for EPN. Reports in the human literature have used CT scanning as a diagnostic tool for detection of EPN, with a reported sensitivity and specificity as high as 100%.4,8 CT has also been used to grade the severity of disease prior to institution of therapy, with a new classification scheme of subcategories of type I and II developed in the early 2000s. 14 CT is the recommended imaging modality of choice for diagnosis, monitoring and prognosis of EPN in humans.4,5,8
Several studies have also shown that minimally invasive techniques such as placement of J stents and pigtail catheters have favorable outcomes in patients without poor preoperative prognostic factors.4,8 Conservative management such as intravenous fluids, antibiotics and glycemic control have been successful in patients with early disease or mild involvement (gas only in the pelvicalyceal space, but not the peri- or paranephric space or kidneys themselves).6,11 A comprehensive review of the current human literature has shown that, overall, mortality rates from medical management alone are 50%; medical management combined with an emergency nephrectomy 25%; and medical management with percutaneous drainage only 13.5%. 8 It is also now the standard of care that medical management in combination with minimally invasive techniques should be attempted first, but, if indicated, nephrectomy should not be delayed.5,8,18
It is important to note that in this patient, a bacterial urinary tract infection was identified but there was no evidence of concurrent glucosuria. This demonstrates that an ascending urinary tract infection with subsequent EPN should not be ruled out based on the absence of glucosuria. As far as we are aware, this is the first reported case of a ureteral stent used in a veterinary patient with EPN. Therefore, we cannot comment on the long-term efficacy of stents for this condition in veterinary patients. It is reasonable to presume that the stent may need to be removed in the future as it can act as a nidus for infection, but it is unknown if re-stenting the patient prior to nephrectomy would have resulted in a favorable outcome. A recent study by Berent et al showed that ureteral stents have a high overall success rate if placed by a qualified specialist, 7 regardless of etiology of obstruction, and that 27% of cats in this study were re-stented successfully with either a larger diameter or softer stent. This suggests that re-stenting may have been a reasonable option for this patient prior to nephrectomy. The stent may be removed with a minimally invasive procedure such as a percutaneous cystotomy or laparoscopic-assisted cystotomy. Another recent case series documents the use of ureteral stenting and pelvic lavage as renal-sparing treatment for 13 dogs suffering pyonephrosis secondarily to either ureterolithiasis or ureteral stricture. 19 Seven of these 13 dogs had positive cultures from either the bladder or renal pelvis, and eight were azotemic prior to stenting. Only two dogs experienced intraoperative complications (eg, stent shearing and ureteral wire penetration at the site of the ureterolith), and post-procedural complications were either short term or not clinically significant (eg, stent encrustation, migration and tissue proliferation).
Another therapeutic option would have been placement of a subcutaneous ureteral bypass (SUB). The decision against placing a SUB was made because of concerns that, as with a ureteral stent replacement, nephrectomy would still eventually be required. In an attempt to minimize the number of surgical procedures performed, the owner elected a nephrectomy. Although a nephrectomy was ultimately performed, it is important to note that the stent allowed temporary relief of the condition. It is hard to say how well this cat may have done if its azotemia and progression of hydronephrosis at initial presentation had not been so severe.
Conclusions
Despite major limitations in the management of this case, given that neither anterograde pyelography nor renal scintigraphy were performed prior to nephrectomy, this report demonstrates temporary resolution of hydronephrosis secondary to EPN following placement of a ureteral stent. Although recent studies have documented emphysematous infections of the upper and lower urinary tract, 16 this case investigates the first-time use of ureteral stents for EPN in veterinary medicine. It is unknown whether the ureteral stent or appropriate antibiotic therapy contributed more to temporary resolution of a suspect obstructive process, but we feel this case demonstrates that minimally invasive techniques should be considered as a reasonable option for patients with EPN, and further studies are warranted.
Footnotes
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this case report.
