Abstract
Objectives
The aim of the study was to evaluate ultrasonographic changes in the small intestine of cats with clinical signs of gastrointestinal disease and low or low–normal serum cobalamin concentrations.
Methods
Records for client-owned cats presenting to the small animal hospital with signs of gastrointestinal disease and in which serum cobalamin concentrations were measured from 2000–2013 were reviewed. Inclusion criteria were cobalamin concentrations <500 ng/l, abdominal ultrasound within 1 month of cobalamin testing and definitive diagnosis.
Results
Of 751 serum cobalamin measurements, hypocobalaminemia or low–normal cobalamin was identified in 270 cats, abdominal ultrasound was performed in 207 of those cats and a diagnosis was available for 75 of them. Small intestinal ultrasound changes were detected in 49/75 (65%) cats. Abnormalities included thickening, loss of wall layer definition, echogenicity alterations and discrete masses. Serum cobalamin concentrations <500 ng/l were observed with diagnoses of inflammatory disease, neoplasia, infectious disease and normal histopathology. Cobalamin concentration was significantly lower in cats with lymphoma or inflammatory bowel disease compared with other gastrointestinal neoplasia (P = 0.031). No difference was found between cobalamin concentration and the presence of ultrasound abnormalities, specific ultrasound changes or albumin concentration.
Conclusions and relevance
One-third of symptomatic cats with hypocobalaminemia or low–normal cobalamin concentrations may have an ultrasonographically normal small intestine. For the majority of cats in this study, histopathologic abnormalities were observed in the small intestine, regardless of ultrasound changes. These findings suggest gastrointestinal disease should not be excluded based on low–normal cobalamin concentrations, even with a concurrent normal ultrasound examination. Additional studies are needed in cats with low–normal serum cobalamin concentrations, as a definitive diagnosis was not pursued consistently in those cats. However, data from this study suggest that careful monitoring, histopathologic evaluation and future cobalamin supplementation may be warranted.
Introduction
Serum cobalamin and abdominal ultrasound aid in diagnosis of intestinal disease in veterinary patients. Cobalamin (vitamin B12) serves as a marker of gastrointestinal, specifically ileal, function. Cobalamin absorption requires gastric pepsinogens, R-protein, pancreatic proteases, intrinsic factor and cobalamin–intrinsic factor receptors in the ileum. Cobalamin concentrations and half-life depend on intestinal absorption and enterohepatic circulation, leading to a shortened half-life from 13 to 5 days with gastrointestinal disease. 1 Studies have documented the prevalence of hypocobalaminemia in cats with gastrointestinal disease, with varying results.1–5 While most research regarding cobalamin in cats focuses on subnormal concentrations, recent work highlights the association of elevated cobalamin concentrations with hepatic disease and neoplasia. 6
Abdominal ultrasound is also used to document intestinal disease. Changes cited as significant include thickening of the intestinal wall or relative thickening among layers.7–9 Complete ultrasonographic evaluation of the small intestine includes echogenicity, differentiation among wall layers, identification of masses, motility and intraluminal contents.10,11
The objective of this study was to determine the relationship between low or low–normal serum cobalamin concentrations and small intestinal ultrasonographic changes in cats with signs of gastrointestinal disease.
Materials and methods
Study population
The medical record database from Texas A&M University Veterinary Medical Teaching Hospital was searched from 1 January 2000 to 31 December 2013 in order to identify all cats on which serum cobalamin measurements were performed. Inclusion criteria were serum cobalamin <500 ng/l, abdominal ultrasound examination within 1 month of cobalamin measurement, definitive diagnosis and clinical signs of gastrointestinal disease.
Information regarding breed, clinical signs, duration of clinical signs and date of cobalamin measurement were obtained from the medical records. Serum albumin concentration and concurrent non-gastrointestinal disease were reviewed when available.
Data collection
Cobalamin measurements were performed through the Texas A&M University Gastrointestinal Laboratory using a previously described immunoassay. 12 The lower limit of reported cobalamin concentration changed during the study period but the assay method remained constant. Ultrasound findings were based on final reports in the medical record. Ultrasound examinations were performed by the board-certified radiologist or radiology resident on clinical duty; resident examinations were reviewed by a board-certified radiologist. In cats with multiple ultrasound examinations, the first ultrasound associated with hypocobalaminemia or low–normal cobalamin was included. Cats with ultrasound changes in only the stomach or colon were included in the number of cats with normal small intestine. No distinction was made among ultrasound changes noted in the ileum vs general small intestine because distinction was not made in all reports. Ultrasound reports were not reviewed for accuracy.
Definitive diagnoses were based on small intestinal histopathologic or cytologic examinations, as documented in pathologists’ reports obtained from fine-needle aspiration, endoscopy, surgery or necropsy. Diagnosis of lymphoma was included if obtained from fine-needle aspiration of an intestinal lymph node, liver or spleen.
Data analysis
Cats were assigned to groups based on cobalamin concentration as follows: concentrations <300 and 300–499 ng/l. Hypocobalaminemia (<300 ng/l) was based on the current laboratory reference interval (RI 290–1500 ng/l), as well as standard cut-off for normal in recent studies.12–14 The borderline group (300–499 ng/l) was based on the observation of cats with consistent clinical signs of gastrointestinal disease, that fell within accepted normal cobalamin parameters. Cobalamin concentrations below the reported limit at the time of measurement were recorded as 99 ng/l (RI 100–1500 ng/l; 2000–2006) and 149 ng/l (RI 150–1500 ng/l; 2007–2013) depending on year. Different machines for ultrasonography and cobalamin measurements were used as equipment was replaced; there were routinely two ultrasound machines in use at all time points. To assess the impact of time, groups were evaluated in 4 year increments.
Statistical analysis was performed using IBM SPSS Statistics 22.0 and MedCalc 14.12.0. Significance was set at P <0.05. Data were assessed for normality using the Shapiro–Wilk test and were not normally distributed (P <0.0001). Cobalamin concentration was compared with the presence of ultrasound abnormalities, hypoalbuminemia, diagnoses of inflammatory bowel disease (IBD) and lymphoma, and purebred vs mixed-breed cats using the Mann–Whitney U-test. Cobalamin concentration was compared among ultrasound groups, and definitive diagnosis using the Kruskal–Wallis test. Post-hoc analysis (Conover test) was performed on factors reaching statistical significance. A receiver–operator curve (ROC) was applied to examine the presence of ultrasound abnormalities based on cobalamin concentration. The χ2 test and trend was used to assess the impact of year on the frequency of ultrasound abnormalities. Fisher’s exact test was performed to assess cobalamin group based on diagnosis.
Results
Population
Seven hundred and fifty-one records of cats with serum cobalamin measurements were reviewed. Seventy-five cats met the inclusion criteria, including 53 (71%) cats with hypocobalaminemia and 22 (29%) with low–normal cobalamin. Reasons for exclusion included cobalamin ⩾500 ng/l (n = 481/751; 64%), lack of abdominal ultrasound (n = 63/270; 23%), and lack of definitive diagnosis (n = 132/270; 49%) (Figure 1). The median cobalamin concentration for the study population was 208 ng/l, with a mean concentration of 242 ng/l (mean 95% confidence interval [CI] 217–266; range <99–484 ng/l). The most common breed across both groups was domestic shorthair; six breeds were represented, including domestic shorthair, domestic longhair and four pure breeds (Table 1). There was no difference between mixed breed and purebred cats and cobalamin concentration (P = 0.207).

Study population
Summary of breed distribution, presenting clinical signs and non-gastrointestinal illnesses of cats with low or low–normal serum cobalamin concentrations and clinical signs of gastrointestinal disease
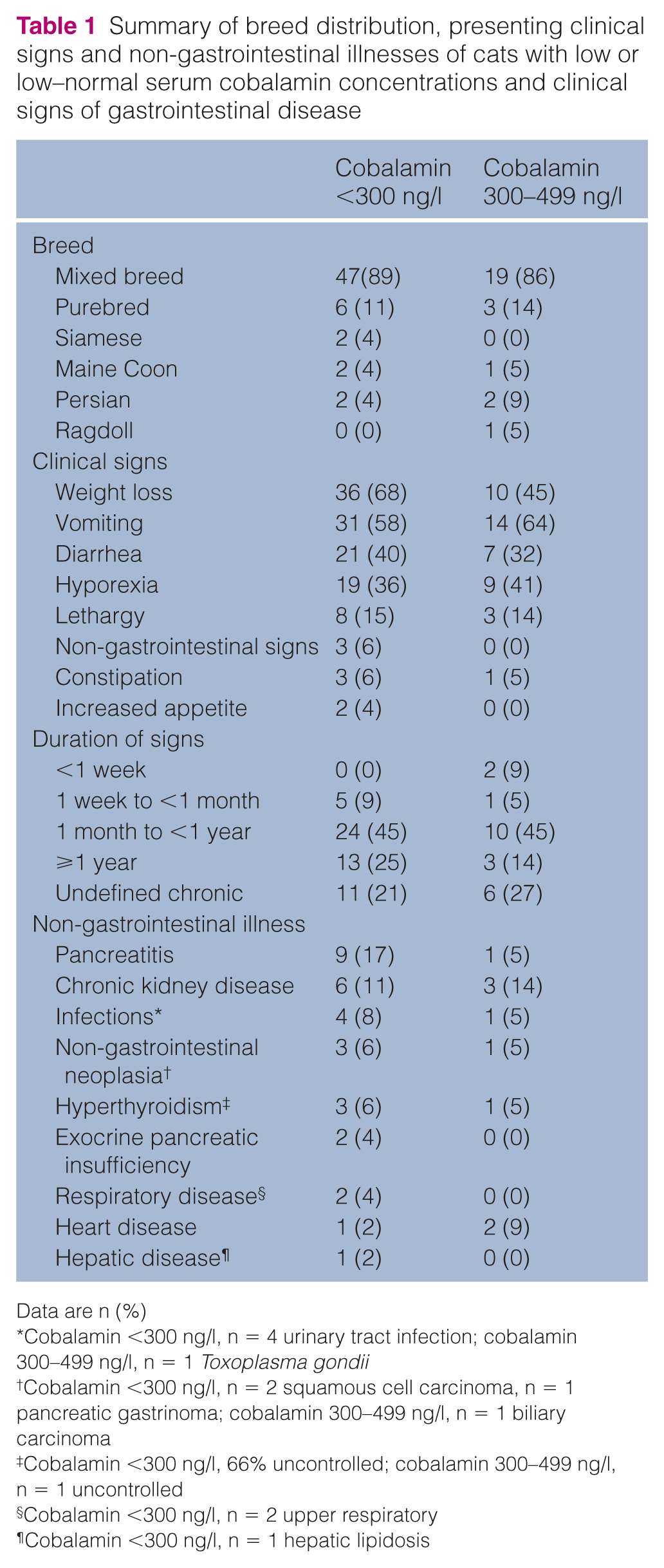
Data are n (%)
Cobalamin <300 ng/l, n = 4 urinary tract infection; cobalamin 300–499 ng/l, n = 1 Toxoplasma gondii
Cobalamin <300 ng/l, n = 2 squamous cell carcinoma, n = 1 pancreatic gastrinoma; cobalamin 300–499 ng/l, n = 1 biliary carcinoma
Cobalamin <300 ng/l, 66% uncontrolled; cobalamin 300–499 ng/l, n = 1 uncontrolled
Cobalamin <300 ng/l, n = 2 upper respiratory
Cobalamin <300 ng/l, n = 1 hepatic lipidosis
Clinical signs and concurrent illness
Most cats presented with clinical signs of at least 1 month’s duration (<2 weeks to 4 years cobalamin <300 ng/l; 3 days to 6 years cobalamin 300–499 ng/l). In some cats, the exact duration of signs was unknown but was recorded as ‘chronic’ in the medical record, with notation of multiple weeks or months. The most common presenting clinical signs in both groups were vomiting and weight loss. The most common non-gastrointestinal illnesses were chronic kidney disease and pancreatitis in cats with cobalamin <300 and chronic kidney disease and heart disease in cats with cobalamin 300–499 ng/l (Table 1).
Ultrasound and cobalamin concentration
Ultrasound findings included increased wall thickness, loss of wall layer definition, changes in echogenicity, focal mass lesions and an ultrasonographically normal small intestine. Sixteen (16/53; 30%) and 10/22 (45%) cats with cobalamin <300 and 300–499 ng/l, respectively, had an ultrasonographically normal small intestine. The most common ultrasound change noted across all groups, accounting for 44% (n = 33/75) of ultrasound findings, was increased wall thickness. (Figure 2). There was an increased frequency of abnormal ultrasound findings over the time course of the study (χ2 trend [2, n = 75] = 4.5 [P = 0.032]) (Figure 3). There was no difference between cobalamin concentration (P = 0.239; area under the ROC curve (AUC) = 0.582, SE = 0.074, 95 % CI 0.462–0.695) and documentation of an abnormal ultrasound. No specific ultrasound findings were related to cobalamin concentration (P = 0.171).
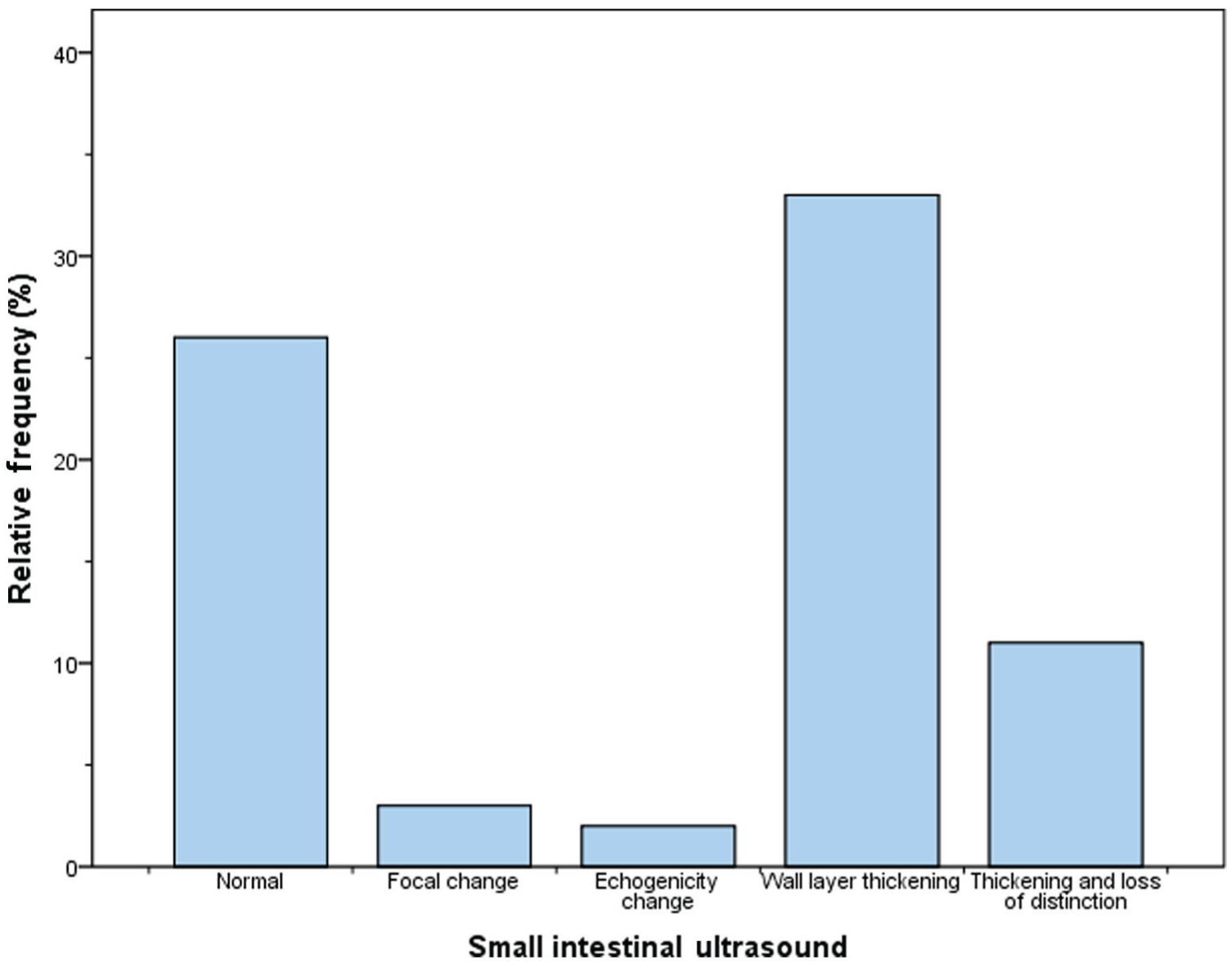
Relative frequencies of ultrasound abnormalities in the study population of cats with low or low–normal serum cobalamin and clinical signs of gastrointestinal disease

Number of cats with normal and abnormal small intestinal ultrasound examinations, subdivided by year, in the study population of cats with low or low–normal serum cobalamin and clinical signs of gastrointestinal disease
Diagnosis and cobalamin concentration
The most common intestinal diagnoses in both groups were IBD and lymphoma. IBD was diagnosed in 25/53 (47%) cats with cobalamin <300 ng/l and 8/22 (36%) cats with cobalamin 300–499 ng/l. Lymphoma was diagnosed in 22 (42%) cats with cobalamin <300 ng/l and five (23%) cats with cobalamin 300–499 ng/l. Other intestinal diagnoses included carcinoma (n= 4/75 [5%]), histoplasmosis (n = 1/75 [1%]) and mast cell neoplasia (n = 3/75 [4%]). Seven (7/75 [9%]) cats had normal histopathologic examinations, three with cobalamin <300 ng/l and four with cobalamin 300–499 ng/l. There was a significant difference in cobalamin concentration among diagnoses, with cats diagnosed with lymphoma or IBD having lower serum cobalamin than cats with other gastrointestinal neoplasia (Kruskal–Wallis H test [3] = 8.8876; P = 0.031). When evaluating diagnoses of only lymphoma and IBD, there was no difference in cobalamin concentration (P = 0.821) (Figure 4). There was no significant difference between cobalamin group (ie, low vs low–normal cobalamin) and diagnosis (P = 0.027; Pbonf. = 0.054) (Figure 5).

Box plot comparison of serum cobalamin concentration with small intestinal histopathologic or cytologic diagnosis in cats with low and low–normal cobalamin concentration and clinical signs of gastrointestinal disease. Error bars represent range, interquartile range and median cobalamin concentration within a diagnosis group. IBD = inflammatory bowel disease
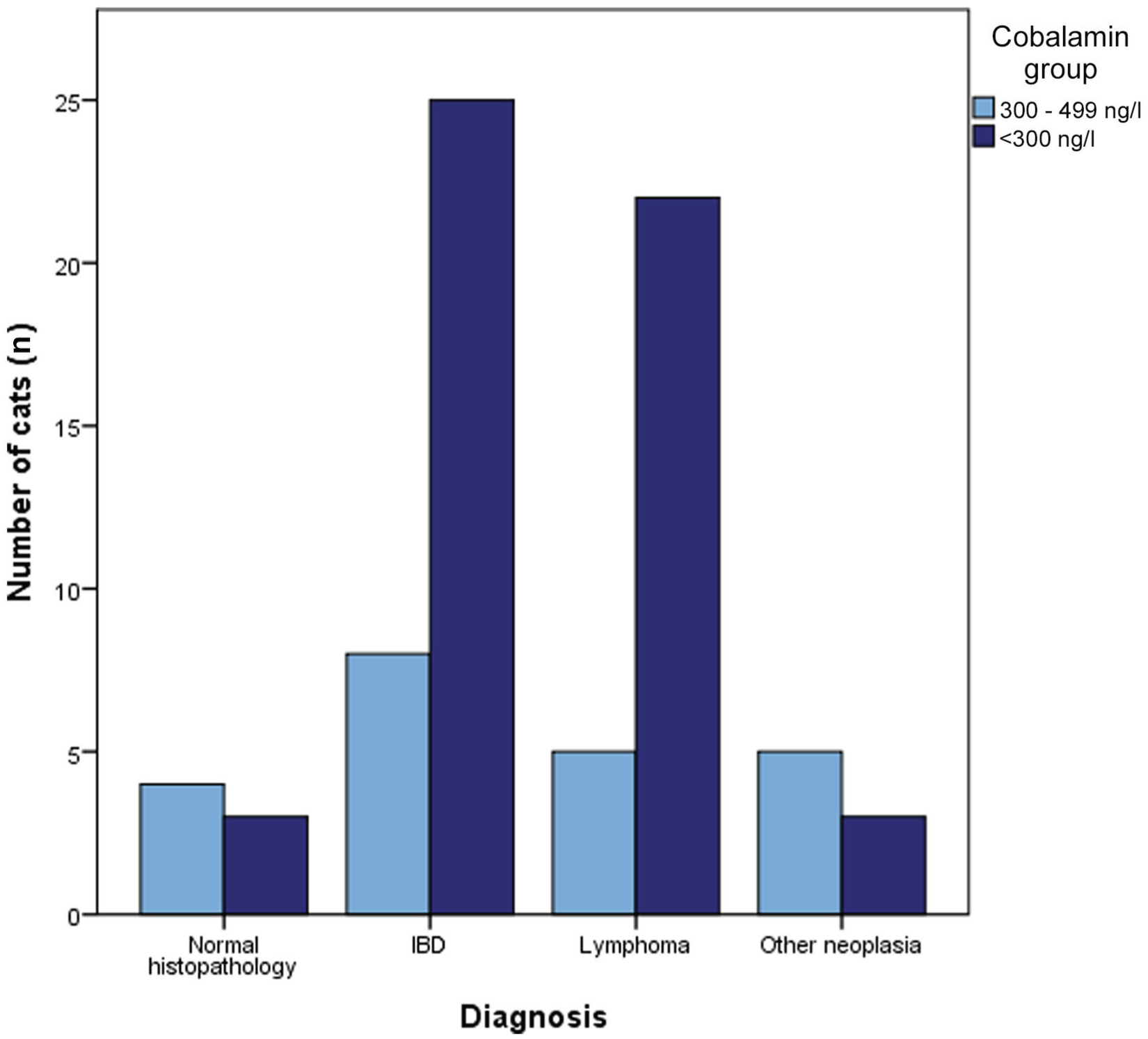
Bar chart comparison of small intestinal histopathologic diagnosis in cats with clinical signs of gastrointestinal disease, subdivided by low or low–normal serum cobalamin concentration. IBD = inflammatory bowel disease
Diagnosis and ultrasound
Twenty-six cats (26/75 [35%]) had a normal small intestinal ultrasound. Five (5/26 [19%]) cats with an ultrasonographically normal small intestine had normal histopathologic results. Fourteen (54%) cats with a normal ultrasound examination were diagnosed with IBD and four (15%) were diagnosed with lymphoma. Three cats (12%) with a normal ultrasound examination were diagnosed with intestinal neoplasia other than lymphoma. One (4%) cat with a normal ultrasound examination was diagnosed with histoplasmosis. Neoplasia was diagnosed in all three cats with a distinct mass. Intestinal wall thickening and combined thickening and loss of wall layer distinction were noted in cats with IBD and neoplasia.
Serum cobalamin and albumin
Serum albumin was measured in 47 (47/53 [89%]) cats with cobalamin <300 ng/l and 20 (20/22 [91%]) cats with cobalamin 300–499 ng/l. Twenty cats (20/67 [30%] total measurements) were hypoalbuminemic, including 17 (32%) cats with cobalamin <300 ng/l and three (14%) cats with cobalamin 300–499ng/l. In cats with hypoalbuminemia and cobalamin <300 ng/l, diagnoses included IBD, neoplasia and normal ileal histopathology. In cats with hypoalbuminemia and cobalamin 300–499 ng/l, diagnoses included adenocarcinoma and normal histopathology. There was no difference in cobalamin concentration (P = 0.438) and the presence of hypoalbuminemia.
Discussion
Serum cobalamin is used routinely as a marker of intestinal absorptive dysfunction in veterinary medicine but is a questionable marker of metabolically active cobalamin status in human patients. 15 More sensitive measurements of clinical hypocobalaminemia in human patients include methylmalonyl-CoA, holotranscobalamin, homocysteine and cystathionine, which have been investigated in cats.12,14,16–19 However, Simpson et al found that serum cobalamin may serve as a practical indicator of clinically relevant gastrointestinal disease. 1 Cobalamin concentrations for most healthy cats in previous studies were ⩾500 ng/l.12–14 While one study demonstrated higher concentrations in healthy cats (900–2800 ng/l), most cats with gastrointestinal disease had concentrations <500 ng/l. 1
Abdominal ultrasound is often used as an initial screening test for gastrointestinal disease. Evaluation includes wall layer thickness, distinction among wall layers, echogenicity of layers, mass lesions and motility. The mucosa is accepted to be the thickest layer in all segments, except the stomach. In cats, there is a range of normal segmental measurements, which overlap among studies.10,11 Studies have shown a difference in muscularis thickening between cats with gastrointestinal disease and healthy cats, as well as variation among healthy cats.7,9 One study found that the mucosa was the thickest layer in all segments, while another study found the muscularis to be the thickest layer in the ileum.7–9 Discrepancies among studies highlight relative uncertainty when examining the feline intestinal tract. Furthermore, a recent study demonstrated low sensitivity of abdominal ultrasound for detecting histopathologic intestinal abnormalities. 20
In the present study, small intestinal ultrasound changes were observed in cats with hypocobalaminemia and low–normal serum cobalamin. Abnormalities included markers frequently associated with gastrointestinal disease. However, 26/75 cats had an ultrasonographically normal small intestine. The most common change across both groups was increased wall or layer thickness, a change noted by Simpson et al. 1 Additional ultrasound changes in this population included a loss of wall layer definition, alterations in echogenicity, and a focal mass. A large proportion of cats had conflicting results between cobalamin concentration and abdominal ultrasound, and no difference was observed between cobalamin concentration or specific findings. The overlap in cobalamin range, even combined with ultrasound, precludes the ability to define a cut-off value for cobalamin and the presence of disease.
Intestinal thickening was not a specific finding, as 80% (n = 60) of cats had diagnoses of IBD or lymphoma. This is consistent with a previous study, which documented hypocobalaminemia in cats with lymphoma and IBD, as well as reports documenting ultrasound changes associated with these diseases.9,13,21 Subtle changes, such as variations in echogenicity, were documented in the study population, with lymphoma and IBD comprising the majority of final diagnoses.
Twenty-six cats had a normal ultrasound examination. Diagnoses in cats with normal small intestinal ultrasound included IBD, lymphoma, carcinoma, mast cell neoplasia and histoplasmosis. Five cats with a normal ultrasound examination had normal histopathology. Considerations for these cats include segmental intestinal disease or non-gastrointestinal illness. These observations suggest that a lack of ultrasound changes does not exclude intestinal disease in cats with low or low–normal cobalamin concentrations. Lymphoma and IBD were associated with lower cobalamin concentrations than other gastrointestinal neoplasia. There was no difference between cobalamin groups (ie, low vs low–normal) and no difference in cobalamin concentration when comparing only IBD and lymphoma, the two most common diagnoses.
Reed et al found that hypoalbuminemia was more likely in cats with gastrointestinal disease than healthy cats. 5 Twenty cats in the present study had hypoalbuminemia; the majority had serum cobalamin <300 ng/l. While concurrent hypoalbuminemia and hypocobalaminemia could increase suspicion of gastrointestinal disease, other causes of protein loss or decreased production could not be excluded in all cats. In this population, based on the presence of hypoalbuminemia, there was no difference in cobalamin concentration.
It was beyond the scope of this study to exclude concurrent disease processes. Associations have been made with hypocobalaminemia and hyperthyroidism, kidney disease, cardiac disease and hepatic disease, conditions present within this population.3,22 Two cats in this study had low trypsin-like immunoreactivity concentrations, and exocrine pancreatic insufficiency could not be excluded as the cause of low cobalamin.23,24 One study showed no difference in cobalamin concentrations in cats with gastrointestinal disease compared with other diseases. Only one cat in that study was diagnosed with hypocobalaminemia, defined as <150 pg/ml. 3 A concurrent disease process could explain some of the cats with normal ultrasound examinations and normal histopathology. Cats with normal histopathologic findings represented a small subset of the study population and do not offset the larger proportion that had histopathologic changes.
The major limitation of this study was its retrospective nature. A number of cats in the initial population were not followed with ultrasound (n = 63) or histopathology (n = 132). While a significant diagnosis was obtained in most cats ultimately included in the study population, results may be biased, as cats were being evaluated for gastrointestinal disease. Furthermore, each cat was evaluated by a variable number of clinicians, typically including at least one intern or resident and one board-certified internist. Some cats were evaluated only through the Emergency Service, and some cats were examined multiple times prior to cobalamin testing or abdominal imaging. In future studies, consistent evaluation by designated clinicians would standardize the suspicion and sequential diagnostic testing for gastrointestinal disease within the population.
The long study period allowed for variation in histopathology and radiology standards in evaluating the intestinal tract, as well as equipment sensitivity, demonstrated by the fact that there was a trend toward influence of year on ultrasound findings.10,11,25,26 Some ultrasound changes, such as echogenicity and layer prominence, inherently contain subjectivity in interpretation. Ultrasonographers were not blinded to historical or diagnostic information. Knowledge of hypocobalaminemia could result in overinterpretation of subtle changes. However, all cats were presenting for clinical signs of gastrointestinal disease, so suspicion of disease would not be unique to a particular subgroup. In addition, abdominal ultrasound was often performed on the day of presentation prior to receiving cobalamin results. Overinterpretation would likely skew the data toward positive ultrasound findings as cobalamin concentration decreased, which was not observed in this population. These sources of bias could be improved were a single radiologist, blinded to clinical presentation, to perform the examinations.
Additionally, inclusion of a normal reference population, clinical cats within the upper end of the RI and hypercobalaminemic cats would have strengthened the evidence presented. Evaluation of these cats was beyond the scope of this study, as gastrointestinal biopsy was not routinely performed. However, given recent evidence of an association with hypercobalaminemia and liver disease or solid neoplasia, further examination of these populations is likely of value. 6 These findings reinforce the importance of cobalamin in cats and monitoring abnormalities on either side of the RI.
While a larger population of cats with low–normal cobalamin might demonstrate a lower prevalence of gastrointestinal disease, cobalamin testing may have been performed early in a disease process. If close follow-up is not performed, a subset of cats with clinical gastrointestinal disease could be overlooked based on a single time point measurement. Serum cobalamin concentrations >300 ng/l, particularly at the low end of the normal RI, should not exclude the possibility of gastrointestinal pathology in cats with clinical signs, even with a normal intestinal ultrasound examination.
Conclusions
This study found that 35% of cats presenting to a veterinary teaching hospital for clinical signs of gastrointestinal disease and low or low–normal serum cobalamin concentrations had a normal small intestinal ultrasound examination. Ninety-one percent of cats had clinically significant gastrointestinal disease associated with histopathologic changes, regardless of cobalamin concentration or ultrasound changes. Lower serum cobalamin concentrations were associated with lymphoma and IBD in comparison with other gastrointestinal neoplasia; there was no difference when comparing only lymphoma and IBD, limiting the clinical usefulness of this finding. Clinical signs should be correlated with diagnostic test results, and closer evaluation of cats with low–normal cobalamin concentrations is needed. Further diagnostic testing in those patients, such as intestinal biopsy, may be indicated in the presence of moderate clinical signs or lack of response to empirical management. At the current laboratory cut-off value, we may be neglecting an important population of cats that could benefit from close monitoring of cobalamin concentrations. Future studies are needed to further examine those clinically affected cats that have normal results on standard diagnostic screening tests.
Footnotes
Acknowledgements
We would like to thank the Texas A&M Gastrointestinal Laboratory for their resources in providing cobalamin testing availability, and Dr John Bonagura for assistance with statistical analysis. This study was performed at Texas A&M University.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
