Abstract
Clinical signs of upper respiratory tract infection can be hard to manage in cats, particularly those in shelters. In this study, clinical data were collected from chronically ill (3–4 weeks’ duration) cats with suspected feline herpesvirus-1 (FHV-1) or feline calicivirus (FCV) infections after administration of one of two novel therapies. Group A cats were administered a commercially available formulation of human interferon-α2b at 10,000 U/kg subcutaneously for 14 days, and group B cats were administered one dose of a FHV-1 and FCV intranasal vaccine. Molecular assays for FHV-1 and FCV were performed on pharyngeal samples, and a number of cytokines were measured in the blood of some cats. A clinical score was determined daily for 14 days, with cats that developed an acceptable response by day 14 returning to the shelter for adoption. Those failing the first treatment protocol were entered into the alternate treatment group. During the first treatment period, 8/13 cats in group A (61.5%) and all 12 cats in group B (100%) had apparent responses. The seven cats positive for nucleic acids of FHV-1 or FCV responded favorably, independent of the treatment group. There were no differences in cytokine levels between cats that responded to therapy or failed therapy. Either protocol assessed here may be beneficial in alleviating chronic clinical signs of suspected feline viral upper respiratory tract disease in some cats that have failed other, more conventional, therapies. The results of this study warrant additional research involving these protocols.
Introduction
Upper respiratory tract disease (URTD) is a major problem for cats housed in shelters around the world and is very common at least in part due to the stress inherent to large captive feline populations. Owing to constraints in space, time, and other resources, URTD is a leading cause for euthanasia in cats at many shelters. 1 Feline herpesvirus-1 (FHV-1) and feline calicivirus (FCV) are the two most common causes of URTD in most reports, with recent studies showing a combined prevalence of ⩾ 60%.2–4 Other infectious agents that have been commonly isolated from shelter cats with URTD include Mycoplasma species, Bordetella bronchiseptica, Chlamydia felis, Pasteurella species and coagulase-negative Staphylococcus species. Unlike viral etiologies, these bacterial infections can usually be treated successfully with appropriate antibiotic therapy or at least provide partial clinical improvement. 2
Although a number of vaccines against FHV-1 and FCV are available and recommended as core vaccines, none has been shown to block infection and so even well-vaccinated cats can develop chronic or recurrent viral rhinitis. 5 Frequently, clinical manifestations of acute viral rhinitis will often resolve with stress relief and supportive care. However, illness in some cats can persist, necessitating other interventions. Administration of lysine or famciclovir orally has been used as an attempt to lessen clinical signs of suspected FHV-1-associated disease, with variable responses.6–8 While there are currently no specific treatments for FCV, interferons (IFNs) may be effective as non-specific antiviral agents and may also modulate cytokine cascades associated with inflammation to lessen clinical signs of disease associated with FHV-1 or FCV. 9 Although the oral IFN administered in the aforementioned study was associated with clinical improvement, it is not available worldwide, encouraging the study of other IFNs and alternate routes of administration when studying other viral infections like feline immunodeficiency virus (FIV). 10 In another study, administration of IFNs into the conjunctival fornix of cats (both feline recombinant IFN-ω and human recombinant IFN-α2b) with suspected viral disease failed to improve clinical signs and viral shedding in cats with naturally occurring viral keratoconjunctivitis. 11 However, the parenteral IFN protocol we chose to study herein has not been evaluated in large numbers of cats with or without proven viral infection.
Administration of a FHV-1, FCV and feline panleukopenia virus (FVRCP) vaccine for intranasal (IN) use results in significant protection against FHV-1 as soon as 4 days after delivery of one dose. 12 This effect was recognized prior to the development of specific FHV-1 immune responses, suggesting that the IN route of administration may have induced innate immune responses that lessened clinical signs when the cats were exposed to virulent FHV-1. This hypothesis was explored further in another study where cats administered an IN vaccine containing FHV-1 and FCV had lessened clinical signs compared with controls after challenge with B bronchiseptica, which also suggested that innate immune responses were induced by the IN vaccine. 13 These studies led to our hypothesis that the innate and specific immune system-stimulating properties associated with FHV-1 and FCV containing IN vaccines could be used as a treatment to lessen the clinical signs in cats with suspected viral URTD. The primary objective of this study was to gather clinical data on cats with suspected viral URTD before and after administration of one dose of an IN modified live bivalent FHV-1 and FCV vaccine or an empirical human IFN-α2 subcutaneous (SC) protocol.
Materials and methods
Experimental design
Cats used in this study were all owned by one of three open admission shelters in north central Colorado, and the treatment protocols to be studied were approved by each shelter and the Institutional Animal Care and Use Committee at Colorado State University. Cats were evaluated and entered into the study between March 2011 and January 2012. On entry to the shelters, all cats were vaccinated with a modified live parenteral FVRCP vaccine. Based on dentition and lack of lenticular sclerosis, all kittens ⩾16 weeks of age and adult cats with URTD (sneezing, nasal discharge, fever) that were initially recognized by a shelter staff member were considered for the study. Once a potential cat was identified, a complete physical examination was performed by the study veterinarians (JF and AF) and a veterinary student (KC). Cats with evidence of systemic disease (ie, fever, lethargy), moderate-to-severe dental disease, facial deformities or moderate-to-severe ocular signs were then excluded. The cats that initially qualified for the study were housed in an isolation room at the respective shelter and administered treatments following the facilities’ standard operating procedures.
The prestudy treatment protocols varied slightly amongst the shelters and cats but all cats were administered lysine (Viralys oral gel; Vetoquinol) by dose at either 250 or 500 mg orally (PO) q24h and doxycycline 5 mg/kg PO q12h or 10 mg/kg PO q24h (frequency of administration determined by the respective shelters) for a minimum of 7 days. For the majority of cats, doxycycline and lysine were administered concurrently but, owing to persistence of clinical signs, a number of cats were started on lysine after doxycycline treatment was finished. If mucopurulent nasal discharge was still observed after administration of doxycycline and lysine, cats were then administered amoxicillin–clavulanate (Zoetis Animal Health) 13.75 mg/cat PO q12h for a minimum of 7 days. For those cats noted to be dehydrated or to have ocular involvement, subcutaneous fluids and topical ocular therapy were provided. At that time, a cumulative clinical score was determined for each cat using a modification of the system previously used for scoring FHV-1/FCV vaccine challenge studies that has been used in other peer-reviewed studies (Table 1).12,13
Scoring system used to monitor cats for clinical evidence of disease

Supplemental assay method for scoring feline rhinotracheitis virus in cats following challenge, supplemental assay method for scoring feline calicivirus in cats following challenge, Center for Veterinary Biologics and National Veterinary Services Laboratories, Ames, IA, 2001
Cats with persistent sneezing and nasal discharge with a cumulative clinical score of ⩾6 were considered to have failed conventional treatments (lysine and two antibiotics) and were not considered suitable for adoption by the study veterinarians and so remained as study candidates. These cats were then evaluated for abnormalities by complete blood cell count, serum biochemistry panel, feline leukemia virus (FeLV) antigen, and FIV antibody test (SNAP FeLV/FIV; IDEXX Laboratories) to evaluate for conditions that could potentiate URTD. Cats that were negative for FeLV antigen and FIV antibody that also had no significant abnormalities on complete blood cell count and serum biochemical panel were transferred to the research facility at Colorado State University. For samples with adequate volume, the remnant blood sample in EDTA were placed in RNAlater (Qiagen, Germantown, MD, USA) for the performance of cytokine panels, as noted in the ‘Cytokine and PCR assays’ section.
Treatment groups and monitoring
On admission to the research facility, mucosal cells were collected from the caudal pharynx of each cat by gently rolling a swab against the mucosa in the region; total DNA and RNA were extracted, and FHV-1 PCR and FCV reverse transcription (RT)-PCR were performed as described in the ‘Cytokine and PCR assays’ section. 3 Additionally, a temperature-sensing microchip was placed in each cat for daily temperature readings. 14 The cats were group housed within a specific pathogen-free facility at Colorado State University, with all cats having access to similar perches and toys serving as enrichment. A maximum of four cats were housed in the gang room during the study. The gang room (17 ft × 12 ft) was partitioned by mesh walls into three separate sections (section 1 = 7 ft × 12 ft; section 2 = 4 ft × 6 ft; section 3 = 6ft × 12 ft) and all cats housed within these areas were visible to one another at all times. Cats that were intolerant of being group housed (withdrawing from activity, lack of interaction with other cats, fighting behaviors) while in the gang room would have been moved to a single-cat room with similar enrichment if needed. To acclimate the cats to the new environment, all cats underwent an initial equilibrium period of 3 days after entering the research facility. During that time, supportive care, which consisted of cleaning crusted debris from the eyes and nose, subcutaneous fluids, oral buprenorphine and topical ophthalmic antibiotics were the only treatments instituted if cats exhibited signs of dehydration or ocular discomfort.
After the equilibrium period, cats with a cumulative clinical score of ⩾6 were randomly assigned by coin flip into two treatment groups. Group A was administered human IFN-α2b (Intron A; Schering Plough) at 10,000 U/kg SC q24h on days 0–13. Group A cats were also administered 0.1 ml sterile saline into each nostril on day 0 to simulate the stress associated with IN vaccine administration. Group B cats were administered one dose of a commercially available FHV-1- and FCV-containing vaccine for IN administration (Feline Ultranasal FVRC; HESKA) on day 0, following the manufacturer’s guidelines. Group B cats were also administered 1 ml sterile saline SC q24h on days 0–13 in order to simulate the stress associated with administration of IFN-α2b.
A clinical score was determined at approximately the same time in the morning, daily for each cat by individuals who had been trained at the same time and who were masked to the treatment groups. As part of the clinical scoring system, the trained individuals would offer a quarter of a can of a palatable commercial diet (Science Diet A/D; Hill’s PetFood) to each cat every morning in order to monitor appetite and attitude for a 1 h observation period. After this time period, dry food was offered ad libitum until 17:00 each evening. Supportive care was provided as needed as described. When the total cumulative score was <3, the cats were deemed adoptable and returned to their respective humane societies. The study veterinarians arbitrarily set a cumulative clinical score of <3 as the level that would be acceptable to new adoptive owners. Cats with a cumulative clinical score ⩾3 on day 13 were entered into the alternate treatment group on day 14 and then monitored from day 14 to day 27. For all cats that failed to respond to the primary treatment on day 14 and from a group of cats that responded to primary therapy, a pharyngeal swab was collected as described for performance of FHV-1 PCR and FCV RT-PCR. Cats failing the first two treatments or cats that had persistent ocular disease were administered famciclovir at 62.5 mg/cat PO q12h for 14 days (days 28–41) as an attempted rescue therapy while being scored clinically. Side effects of the IFN-α2b such as vomiting, diarrhea, skin irritation and pruritus were to be recorded if observed.
Cytokine and PCR assays
Total DNA and RNA were extracted from the blood in EDTA and the pharyngeal swabs as previously described. 3 Endpoint FHV-1 PCR and FCV RT-PCR assays were performed as previously described on the nucleic acids extracted from the pharyngeal swabs; PCR for GAPDH was used as a DNA control, with negative cats excluded. The mRNA of IL2, IL4, IL6, IL10, IL12, IFNG and TNF were amplified by previously reported quantitative real-time PCR using published assays.15,16 Amplifications were carried out in duplicate in 25.0 μl reactions containing (final concentration) 12.5 μl Mastermix, 0.5 μl (400 nM) each primer, 0.2 μl (80 nM) probe, 6.3 μl PCR-grade water and 5 μl template cDNA. Relative quantification was carried out using comparative Ct method (Applied Biosystems 2001) with feline GAPDH as the reference gene.
Statistical analysis
Percentages of cats that developed apparent clinical responses after primary treatment were compared by Fisher’s exact test. To evaluate speed to resolution, Kaplan–Meier recovery curves were constructed for the cats in each group that had a cumulative clinical score <3 by day 14, and the log rank test was used to evaluate for differences between groups. The Shapiro–Wilk test was used to evaluate for normality between other test results (cytokines; PCR assay) and group. The majority were non-normal and this, combined with the relatively small samples for some assay results, led to the selection of the Wilcoxon rank sum test for the analyses. Significance was defined as P <0.05.
Results
Clinical responses
Overall, approximately 700 cats identified as potentially having URTD were evaluated by physical examination. Of those cats, the majority of those initially treated with doxycycline and lysine responded to therapy. Ultimately, 47 cats had clinical signs that persisted after the administration of amoxicillin–clavulanate and met the other entry criteria and so were transferred to the research facility. The majority of cats (n = 45) moved to the research facility were from one shelter with two other shelters entering one cat apiece. All cats were estimated to be between 16 weeks and 3 years of age. The duration of URTD prior to initial treatment of doxycycline and lysine was not consistently available but all cats had clinical signs of disease for at least 3–4 weeks prior to entering the research facility.
During the equilibration period, a number of the 47 cats were excluded from the study for spontaneously developing a total cumulative clinical score of <6 (16 cats), for developing severe ocular disease (four cats) or for being euthanized due to severe URTD before completing 2 days of primary treatment (two cats). All of the cats tolerated the gang housing. Ultimately, a total of 13 cats were entered into group A (Table 2) and 12 cats were entered into group B (Table 3). The median clinical scores of the two groups on entry to the study were not significantly different (P = 0.6818). Of the 13 cats in group A, eight (61.5%) had a cumulative clinical score of <3 by day 14 (mean 7.9 days; median 8 days; range 4–12 days) and five failed therapy (Table 2). All 12 of the group B cats had a cumulative clinical score of <3 by day 14 (mean 8.3 days; median 8 days; range 4–12 days) (Table 3). The speed to recovery was not different between groups (P = 0.55). However, the percentages of cats having apparent responses to the initial therapy was significantly greater for group B cats (P = 0.04). The five cats that did not respond to IFN-α initially were administered one dose of the IN vaccine and four had apparent responses within an additional 14 days (mean 4.75 days; median 4 days; range 3–6 days). The cat that failed both primary therapies also failed famciclovir as a rescue therapy; the cat was subsequently euthanized with 3 ml pentobarbital administered intravenously. A chronic, multifocal, severe rhinitis was revealed on necropsy.
Cats with suspected chronic viral upper respiratory infection administered interferon-α at 10,000 U/kg, daily from day 0 to day 13 (group A)

Cats with a clinical score >2 on day 13 were considered protocol failures and transferred to the alternate treatment group
Two assays: one for feline herpesvirus (FHV) DNA and one reverse transcriptase PCR for feline calcivirus (FCV) RNA. PCR 1 = positive for FHV-1 alone; PCR 2 = positive for FCV alone
CS = clinical score; FI = female intact; DSH = domestic shorthair; FS = female spayed; MC = male castrated; DMH = domestic mediumhair; MI = male intact; NS = no sample
Cats with suspected chronic viral upper respiratory infection administered one dose of an intranasal vaccine on day 0 (group B)

Cats with a clinical score >2 on day 13 were considered protocol failures and transferred to the alternate treatment group
Two assays: one for feline herpesvirus (FHV) DNA and one reverse transcriptase PCR for feline calcivirus (FCV) RNA. PCR 1 = positive for FHV-1 alone; PCR 2 = positive for FCV alone; PCR 3 = positive for both FHV and FCV
CS = clinical score; FI = female intact; DSH = domestic shorthair; FS = female spayed; MC = male castrated; DMH = domestic mediumhair; MI = male intact; NS = no sample; DLH = domestic longhair
Buprenorphine was administered to 5/13 group A cats (3, 6, 11, 13 and 15 days’ duration) and 6/12 group B cats (4, 7, 8 [two cats], 12 and 15 days’ duration). All of the group B cats administered buprenorphine responded to the initial therapy, while 2/5 cats in group A administered buprenorphine failed the initial therapy. There was no difference between overall response treatment rates in cats that were or were not administered buprenorphine (P = 1).
Molecular diagnostic test results for FHV-1 and FCV
Pretreatment (day 0) PCR assay results were available for 11 group A cats and 10 group B cats (Tables 2 and 3). In group A, FHV-1 DNA alone was amplified from two cats and FCV RNA alone was amplified from one cat (overall positive = 27.3%). In group B, FHV-1 DNA and FCV RNA were amplified concurrently from two cats, FHV-1 DNA alone was amplified from one cat and FCV RNA alone was amplified from one cat (overall positive = 36.4%). All of the cats that were positive for FHV-1 DNA or FCV RNA prior to the initial treatment had apparent responses independent of treatment group. The cat that failed all treatments was negative for nucleic acids of FHV-1 and FCV prior to treatment, after failing IFN therapy and after failing vaccine immunotherapy. Of the four cats that crossed over from IFN-α2b therapy to IN vaccine therapy and had apparent responses (clinical score <3), two were FHV-1 DNA negative and FCV RNA negative on day 0 of the study but FCV positive on day 14 of the study after 14 doses of IFN-α2b. The PCR assays were not performed on the cats in group B on day 14 as all cats had responded to therapy and the PCR assays used cannot distinguish between vaccine strains and natural strains of the organisms. 17
Cytokine assay results
Pretreatment mRNA expression results for IL2, IL4, IL6, IL10, IL12, IFNG and TNF were stratified by response to therapy (Figure 1), and by FHV-1 and FCV molecular assay results for cats with adequate samples available for study (Figure 2).
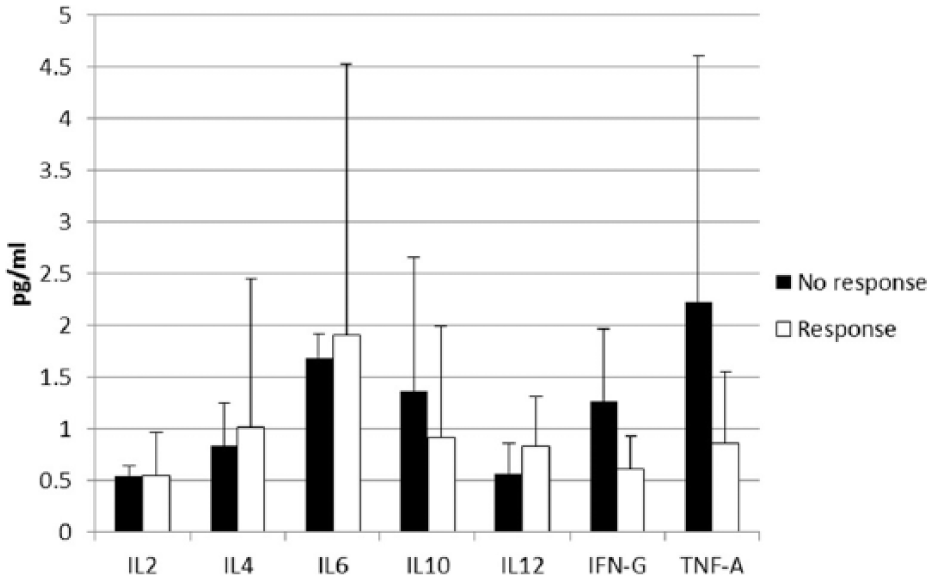
Pretreatment cytokine panel results in cats stratified by whether treatment responses were detected by day 14 of the study. None of the results were statistically significant between the treatment failures (three cats; all on IFN-α2b) and treatment successes (all in the IN vaccine group)

Cytokine distributions stratified by the presence or absence of feline herpesvirus-1 or feline calicivirus nucleic acids on pharyngeal swabs from cats with rhinitis
While no significant differences were noted (P = 0.508) between the three cats that failed therapy with IFN-α2b and the 12 cats with samples available that were successfully treated with IN vaccine or IFN-α2b, the group mean TNF expression for the non-responders was numerically greater than for the responders (Figure 1). This difference was related to the finding of a TNF expression level in 1/3 non-responders that was 6.2 times greater than the mean of the responders.
When the results from three cats that failed to respond to IFN-α2b were compared with the results from four cats that responded to IFN-α2b, expression of IL2 (P <0.03) and IL6 (P <0.01) were significantly higher in the non-responders than in the responders. When the cytokine results were stratified by the presence or absence of FHV-1 or FCV nucleic acids on pharyngeal swabs, no significant differences between groups were noted (P >0.05 for all seven cytokine comparisons) (Figure 2). However, cats that were positive for nucleic acids of FHV-1, FCV or both had numerically greater mean IL6 expression. For 2/5 cats that were positive for viral agents, IL6 expression was 3.1 times (IL6 = 4.3) and 5.7 times (IL6 = 8.0) higher, respectively, than the mean of the eight virus-negative cats tested (IL6 = 1.4).
Discussion
Of the 700 cats that were screened for URTD in this study, the majority were believed by the attending veterinarians to most likely have FHV-1 or FCV infections with either primary or secondary bacterial infections that responded to administration of lysine and doxycycline, or resolved spontaneously. The cats that were still clinically affected were then administered amoxicillin–clavulanate in an attempt to rule out potential bacteria that could have been resistant to doxycycline like some anaerobic bacteria and Streptococcus species. It was the clinical impression of the attending veterinarians that the physical examination and clinical findings combined with failure to respond to these three compounds suggested chronic FHV-1 or FCV infections. Hence, this study focused on two novel therapies and the efficacy in improving clinical signs in cats with chronic upper respiratory infection due to a suspected viral etiology.
Of the 13 cats that received the human IFN-α2b, eight had a clinical score <3 by day 14. All cats tolerated the daily injections well and no clinical side effects from the drug administration (vomiting, diarrhea, skin irritation, pruritus) were appreciated. There is mounting evidence for use of IFNs in the treatment of viral infections in cats and dogs. Some cats with FHV-1- or FCV-associated illnesses have had potential positive responses noted after administration of either feline recombinant IFN-ω (Virbagen Omega; Virbac) or IFN-α2b made for use in humans.18–20 In this study, we chose to use IFN-α2b rather than the feline recombinant product as the feline product is not available in the USA. We also selected the dosage of 10,000 U/kg SC q24h empirically based on unpublished opinions from veterinary clinicians working in this field. Unfortunately, there are no controlled pharmacodynamic, pharmacokinetic, or treatment efficacy studies in administering IFN-α2b parenterally to cats. Thus, therapeutic failures reported in the four IFN-α2b-treated cats that then had an apparent response to vaccine immunotherapy may have merely reflected this not being the optimal protocol. Whether a different IFN-α2b protocol or use of feline IFN-ω would have had different results is unknown. It is also possible that therapeutic failures could have resulted because viral infections were not part of the pathogenesis of disease, as seen with the one therapeutic failure cat that was ultimately proven to have a space-occupying proliferative rhinitis. Adequate time for recovery or the combination of the two therapies should also be considered with regard to why the four cats initially in the IFN group had an eventual response.
Although the human IFN-α2b treatment protocol was well tolerated by the study cats, the parenteral route of administration is labor intensive, especially if the protocol is being considered for outpatient use. In addition, it is possible that repeated use of a product designed for humans could lead to antibody production against this IFN inactivating it or potentially resulting in immunological side effects such as those that may occur with the parenteral use of human recombinant erythropoietin in cats. 21 Low-dose IFN-α2b given orally (10 IU/kg PO q24h) has been used successfully in some cats with FIV infection. 10 In that placebo-controlled study, treated cats survived longer and secondary problems resolved rapidly. The positive effects from oral IFN therapy are believed to be from regulation of inflammatory cytokine cascades. Oral IFN therapy should be further evaluated in cats with FHV-1 or FCV, as oral therapy would be a better option for some cats, some shelter employees and most owners. However, as previously discussed, IFN therapy will not always be effective, as seen in one study of cats with suspected viral disease that were treated with either feline or human IFN in the conjunctival fornix and failed to show significant responses. 11 It is also possible that the apparent treatment failures still had a bacterial component to the pathogenesis of disease.
All cats in this study administered the FHV-1- and FCV-containing IN vaccine as potential immunotherapy ultimately responded within 14 days, including 4/5 IFN-α2b-failure cats. Based on the timeline of the study, the investigators believe it is unlikely that these cats all had spontaneous resolution of disease; however, without an untreated control group, it is not possible to conclude definitively that the vaccine was solely responsible for the clinical improvement observed. Prior to beginning the study, the primary concerns were whether the cats would tolerate vaccine administration and whether the vaccine strains of virus could contact nasal mucosa because of the severe nasal discharge and congestion. However, the cats tolerated the vaccine administration easily, and the responses suggest the vaccine was administered appropriately. There was also concern that the vaccine administration could worsen clinical signs of disease, but this was not noted in any of the cats. The findings of this study support a therapeutic effect from the IN vaccine administration, and there are at least two potential mechanisms of action. It is possible that FHV-1- and FCV-specific local immunity was potentiated by replication of the modified live viruses contained in the vaccines, putting potential pathogenic strains of FHV-1 or FCV into a more solid remission. This theory is potentially supported by the fact that it took a median of 8 days to have the cumulative clinical scores decrease to <3 in IN vaccine-treated cats. As discussed in the introduction, it is also possible that IN administration of vaccines containing modified live strains of FHV-1 and FCV stimulate non-specific immune reactions that could result in lessened clinical signs. The results of the study showing the induction of cross protection against B bronchiseptica in cats vaccinated only for FHV-1 and FCV supports this theory. 13 A non-specific immune stimulation or immune modulation induced by IN vaccine administration might also explain why some cats that were negative for FHV-1 or FCV using molecular methods might respond to therapy. In addition to its therapeutic effect, the ease with which the vaccine was administered compared with the parenteral IFN-α2b may be an important consideration for owners of cats with URTD.
Use of molecular methods is more sensitive than previous techniques (ie, culture) to document the presence of FHV-1 or FCV in materials collected from cats. PCR assays for FHV-1 and RT-PCR assays for FCV are now widely available for clinical use in many countries. However, positive test results do not always document the presence of strains with pathological potential, and vaccine strains are also amplified. 17 In addition, while currently available molecular assays for FHV-1 and FCV PCR are extremely sensitive under optimal conditions, the potential for false negative results still exists for a number of reasons, including PCR inhibitors in oronasal secretions, utilizing only one PCR assay even though multiple FCV strains exist, and specific immune responses causing decreased viral shedding at the time of sampling. In addition, while one study showed that the nasal and pharyngeal sampling sites had high levels of agreement, the results did not agree 100% of the time. 22 Thus, a positive cat could be missed if only one site is sampled. Lastly, PCR performed on conjunctival tissue snips may be more sensitive than PCR performed on samples collected by swabs in cats with FHV-1 conjunctivitis. 23
Based on these caveats concerning molecular assay results, we believe it is difficult to align our clinical response results accurately with those of the FHV-1 PCR assay and the FCV RT-PCR assay. While the clinical suspicion for viral infection was high in our study, nucleic acids of FHV-1 or FCV were amplified from only 27.3% and 36.4% of cats in group A and group B, respectively. However, in this study, all of the cats that were positive for FCV or FHV-1 on the pretreatment sample responded to the primary treatment with presumed antiviral activity. As antiviral therapies do not eliminate FHV-1 or FCV from cats, performing repeat PCR assays is unlikely to be of clinical benefit.
The panel of pro- and anti-inflammatory cytokines was performed on the pretreatment samples from some cats to evaluate for trends that might predict either severity of disease or response to therapy. Although one non-responder had a tumor necrosis factor-α level that was 6.2 times greater than the mean of the responders and interleukin (IL)-2 and IL-6 levels were significantly higher in the non-responder group compared with the responders, the cytokine distributions failed to add much information concerning the potential for therapeutic responses or failures. However, it is possible that the results were affected by the small sample size and a greater numbers of cats should be assessed. Perhaps cytokine distributions may yield more information if performed on upper respiratory secretions or upper respiratory tissue vs peripheral blood, as evaluated in this study.
Of the 47 cats that were transported to the research facility, 16 developed a cumulative clinical score <6 by the end of the equilibration period, which disqualified them from the study prior to being randomized into a primary treatment group. This apparent clinical improvement over time emphasizes that cats with suspected viral URTD should be given ample time to respond (several weeks) if possible, especially prior to making the decision to perform euthanasia. It is possible that the housing at the research facility, which may have had a larger living space, increased human interactions and increased enrichment, was less stressful than at the shelters, which could have potentiated the lessening of clinical signs. As most of the cats that were moved to the research facility improved, rather than worsened, over time suggests that the stress associated with transport was unlikely to have affected the results. However, it is possible that by administering supportive care to the cats that developed severe ocular involvement, stress could have been alleviated, thereby improving clinical signs. In particular, use of pain medications (buprenorphine) in some cats, but not all, may have influenced the results. However, there was no difference between overall response treatment rates in cats that were or were not adminstered buprenorphine.
The primary limitations to this study were the potential for spontaneous resolution of clinical signs in some cats after entry into a treatment group, lack of information concerning optimal dosing of IFN-α2b for FHV-1 or FCV infections in cats, the subjectivity of the clinical scoring system used, the housing of the cats close to one another potentially allowing for transfer of the IN vaccine viruses amongst the cats and the lack of a negative control group. While it cannot be determined whether the IFN-α2b- or IN vaccine-treated cats would have spontaneously resolved without treatment, all of the cats had moderate clinical signs that were present for at least 1 month in the face of classical therapies prior to the majority having potential responses to one of the novel therapies. In order to address the subjectivity associated with the clinical scoring system utilized, individuals were initially trained and utilized daily for scoring of the cats to keep the scoring consistent. While there is potential for the IN vaccine viruses to be shared amongst cats and affected the results, we believe the cats with direct inoculation received the largest dose and were most likely to have a therapeutic effect. In this study, use of a negative control group was not possible based on the presence of relatively severe, chronic illness, and the direction of the respective shelter boards and the institutional animal care and use committee to protect animal welfare. In the absence of a negative control group, additional studies are warranted regarding further evaluation of these novel therapies.
Conclusions
IFN-α2b and topical immunotherapy may be viable treatment options for those cats with persistent chronic viral URTD that have failed conventional therapies. Given there was a significant difference in the initial response between the two treatment groups it may be that the ease of administration of the vaccine over parenteral IFN may be more advantageous as the price may also be cheaper for the vaccine (approximately $3.50 per dose) vs finding and purchasing human IFN-α2b and making the appropriate dilutions. However, additional studies are needed to compare topical immunotherapy with oral IFN treatment in cats suspected to have chronic viral rhinitis. Further studies are also warranted regarding how a combination of these novel therapies could further treat affected cats or perhaps increase the speed to recovery in comparison with cats receiving supportive care alone.
Footnotes
Conflict of interest
The authors have no potential conflicts of interest to declare.
Funding
Funded by a grant from the Morris Animal Foundation.
Preliminary results were presented as an abstract at the annual American College of Veterinary Internal Medicine Forum, New Orleans, LA, May 30–June 2, 2012.
