Abstract
Objectives
The objectives of this retrospective study were to investigate the actual incidence of postobstructive diuresis after relief of urethral obstruction in cats, as well as to identify changes in blood and urine parameters that might be associated with postobstructive diuresis (POD), and to assess the impact of fluid therapy.
Methods
The medical records of 57 male cats with urethral obstruction that were treated with an indwelling urinary catheter were retrospectively analysed. Absolute urine output in ml/kg/h every 4 h and the incidence of cats with polyuria (urine volume >2 ml/kg/h) at any time point over a 48 h period after the re-establishment of urine flow were investigated. In addition, postobstructive diuresis in relation to fluid therapy (PODFR) was defined as urine output greater than the administered amount of intravenous fluids on at least two subsequent time points. Polyuria and PODFR were investigated for their association with blood and urine laboratory parameters.
Results
After 4 h, 74.1% (40/54) of the cats had polyuria, with a urine output of >2 ml/kg/h. Metabolic acidosis was present in 46.2% of the cats. Venous blood pH and bicarbonate were inversely correlated with urine output in ml/kg/h after 4 h. The overall incidence of POD within 48 h of catheterisation was 87.7%. There was a significant correlation between intravenous fluid rate at time point x and urine output at time point x + 1 at all the time points except for the fluid rate at time point 0 and the urine output after 4 h. PODFR was seen in 21/57 cats (36.8%).
Conclusions and relevance
POD is a frequent finding in cats treated for urethral obstruction, and can be very pronounced. Further studies are required to determine whether or not a change in venous blood pH actually interferes with renal concentrating ability. The discrepancy between the frequency of cats with polyuria and PODFR (87.7% vs 36.8%) in the present study indicates that administered intravenous fluid therapy might be the driving force for the high incidence of polyuria in some cats with naturally occurring obstructive feline lower urinary tract disease.
Introduction
Urethral obstruction in male cats due to feline lower urinary tract disease (FLUTD) is a frequent and potentially life-threatening emergency in veterinary practice.1–3 It can be caused by urethral plugs, urolithiasis, or urethral inflammation and urethral sphincter spasm.1,4 Patients may be severely compromised, dehydrated, hypovolaemic and bradycardic on presentation. Initial management includes obtaining blood samples, including electrolyte and venous blood gas levels, administering intravenous (IV) fluid therapy, electrocardiography examination and re-establishment of urine flow. Further work-up includes urinalysis, as well as diagnostic imaging to rule out stones.2,3,5 The most important aspects of postobstructive care are considered to be analgesia and fluid therapy.2,3,5
Postobstructive diuresis (POD) has been defined, rather vaguely, as a massive and inappropriate output of urine following a state of functional urinary tract obstruction (UTO), with urine output exceeding the physiological maximum of 2 ml/kg/h.6,7 Studies on obstructive uropathy in rats, dogs and humans have shown that POD is a complication in the treatment of urethral or ureteral (unilateral, but more so bilateral) UTO.3,8–12 In humans, POD with urine output >4 l/day after UTO is reported in up to 54% of cases. 12 The impact of UTO on renal function has also been studied in experimental models of UTO by bilateral ureteral ligation in rats,6,13,14 and POD has been found to occur in most cases within the first 2–3 h of relief of the obstruction, regardless of the administered fluid therapy. In rats, POD is reported to last up to 30 days after experimentally induced UTO. 15 The pathogenesis of POD is still not fully understood.
Although obstructive FLUTD is one of the most common emergencies veterinary medicine, 2 there is only one study that investigated the incidence of POD in 28 cats with naturally occurring obstructive FLUTD, 7 and there are no studies regarding the severity of POD in natural cases of feline UTO. Apart from that, the somewhat poor characterisation of POD presents some difficulties, especially in the face of fluid therapy. The role of fluid therapy in the pathogenesis of POD has not yet been properly investigated, making it hard to differentiate true POD from iatrogenically driven polyuria. Thus, the aims of the present study were to assess the incidence of POD in cats with naturally occurring UTO, taking into consideration the impact of fluid therapy, and to investigate associations with laboratory parameters in the urine and blood.
Materials and methods
Patients
The database of the Clinic of Small Animal Medicine was retrospectively searched for cats that were treated for obstructive FLUTD during the period 2005–2012. Inclusion criteria were hospitalisation and documented treatment according to a written standard operating procedure (SOP) for the management of cats with obstructive FLUTD. The SOP included intravenous (IV) fluid therapy based on volume and hydration status of the individual cat, and the placement of an indwelling urinary catheter, as well as emptying of the bladder prior to connection to a closed urine collecting system. According to that protocol, the initial fluid rate was calculated with the aim of rehydrating the cat within 8 to 12 h. It also included the adjustment of the fluid rate every 4 h to replace urine output plus one-third of the maintenance requirement (0.7 ml/kg/h, for insensible fluid losses). The minimum initial fluid rate was a maintenance rate (2 ml/kg/h); according to hospital policy, fluid rates exceeding 10 ml/kg/h should not be administered. For fluid therapy, isotonic crystalloid fluids (Sterofundin 1/1; B Braun) were chosen. If initial fluid boluses were given, they were included in the initial fluid rate. Only cats with biochemical analysis, blood gas analysis and urinalysis results available on admission were included. Furthermore, documentation of urine output in ml/kg/h every 4 h over the duration of catheterisation was required. Cats were excluded if they had other known concurrent diseases influencing urine concentrating ability, such as diabetes mellitus, hyperthyroidism, liver disease or chronic kidney disease, or had been treated with steroids or diuretics. Diagnostic imaging (abdominal radiographs and/or abdominal ultrasound) was performed in all cats included into the study.
Urine output and incidence of POD
Urine output was measured in ml/kg/h every 4 h for each cat and the mean urine output over 48 h was calculated. In order to compare the results of the present study with those of prior studies, POD was defined as urine output >2 ml/kg/h. Each cat was evaluated for the presence of POD at each time point and throughout the entire surveillance period.
With the intention of accounting for the impact of fluid therapy, IV fluid rate at time point x was analysed for correlation with the following urine output at time point x + 1, and an additional definition for postobstructive diuresis in relation to fluid therapy (PODFR) was used: PODFR was defined as urine output greater than the administered amount of IV fluids on at least two subsequent time points.
Evaluated parameters
Laboratory parameters of interest included a complete blood count, venous blood gas analysis (pH, bicarbonate, sodium, ionised calcium and potassium), serum chemistry (phosphate, blood glucose, bilirubin, blood urea nitrogen [BUN] and creatinine) and urinalysis, including urine specific gravity (USG), dipstick analysis, urine sediment and bacterial culture.
Laboratory changes on admission were defined as follows: acidaemia (venous blood pH <7.27), 16 hypobicarbonataemia (bicarbonate <15 mmol/l), hyperkalaemia (potassium >5.6 mmol/l), hyperphosphataemia (phosphate >2.36 mmol/l), hypocalcaemia (ionised calcium <1.2 mmol/l), hyperglycaemia (blood glucose >7.9 mmol/l), hyponatriaemia (sodium <141 mmol/l) and postrenal azotaemia (BUN >11.3 mmol/l and/or creatinine >169 µmol/l). On urinalysis, haematuria was defined as >12 erythrocytes per high-power field (HPF), pyuria as >5 leukocytes/HPF, proteinuria as protein ⩾1+, bilirubinuria as ⩾1+, low USG as <1.035 and aciduria as urine pH <5.5.
Statistical analysis
For statistical analysis, commercially available statistical software was used (Microsoft Excel 2010, IBM SPSS PASW Statistic 18, Prism Graph Pad 5). P <0.05 was considered significant. Descriptive statistics were performed for the ages of the cats, laboratory parameters on admission and urine output at each time interval. Univariate correlation analysis was performed to determine laboratory variables associated with the development and degree of POD. Parameters with a P <0.05 and R >0.350 as determined by univariate analysis were then tested for covariance with backward linear regression analysis. Parameters with high covariance were excluded. The χ2 test was applied to assess whether defined laboratory abnormalities were significantly correlated with the development of POD and PODFR.
Results
Patients
Out of 292 cats that were presented with obstructive FLUTD in the investigated time period, 57 male cats fulfilled the inclusion criteria. The majority were domestic shorthair cats (n = 34; 59.6%), followed by domestic longhair (n = 6), British Shorthair (n = 4), Maine Coon (n = 4), Chartreux (n = 3), Persian (n = 2), Turkish Angora (n = 1), Russian Blue (n = 1), Siamese (n = 1) and Norwegian Forest Cats (n = 1). The mean ± SD age of the affected cats was 7.18 ± 3.65 years (range 1.00–15.00 years, median 7.00 years). In 38/57 (66.7%) cats, the obstruction was idiopathic. In 10/57 (17.5%) and 5/57 (8.8%) cats, urethral plugs and uroliths were diagnosed. Based on a positive aerobic urine culture, four cats (7.0%) were diagnosed with bacterial urinary tract infection.
On admission, 82.4% (42/51) of the cats were assessed as being dehydrated, with 28 cats (54.9%) showing only mild to moderate dehydration (up to 6.0%). More severe dehydration (7.0–9.0%) was observed in 10 cats (19.6%), while 4 cats were found to be severely dehydrated (≥10%). In the remaining six cats, information on hydration status was not available. The results of blood gas and biochemistry analysis on admission are shown in Table 1, and those of urinalysis are shown in Table 2.
Laboratory parameters on admission and their association with absolute urine output in ml/kg/h within 4 h after re-establishment of urine flow
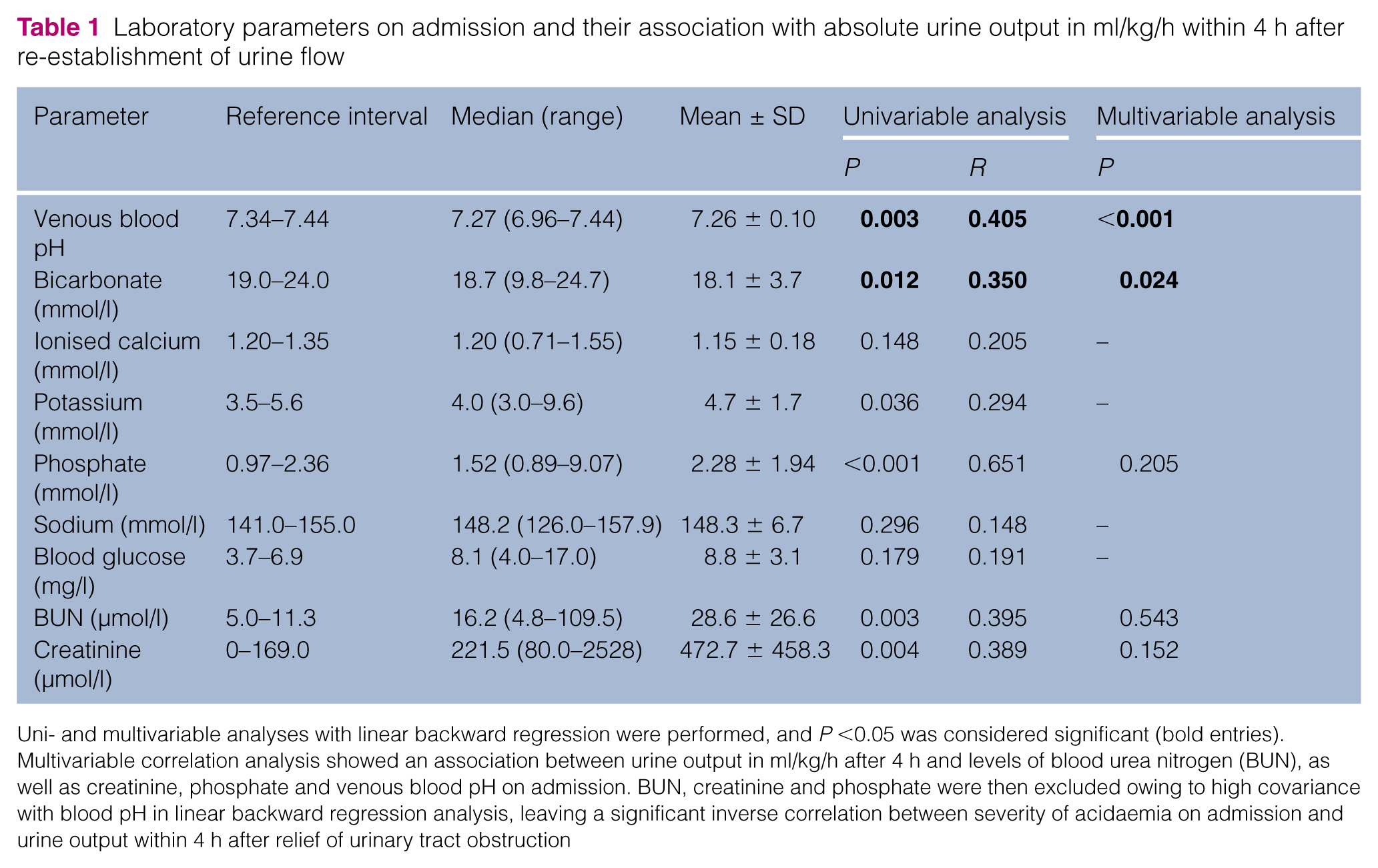
Uni- and multivariable analyses with linear backward regression were performed, and P <0.05 was considered significant (bold entries). Multivariable correlation analysis showed an association between urine output in ml/kg/h after 4 h and levels of blood urea nitrogen (BUN), as well as creatinine, phosphate and venous blood pH on admission. BUN, creatinine and phosphate were then excluded owing to high covariance with blood pH in linear backward regression analysis, leaving a significant inverse correlation between severity of acidaemia on admission and urine output within 4 h after relief of urinary tract obstruction
Results of urinalysis in cats (n = 57) with obstructive feline lower urinary tract disease on admission, proportion of cats with abnormal results and association of severity of laboratory changes with mean urine output within 48 h of re-establishment of urine flow

Uni- and multivariable analyses with linear backwards regression were performed, and P <0.05 was considered significant. Severity of haematuria on admission was significantly correlated with overall severity of postobstructive diuresis
Measured using a diagnostic urine dipstick
HPF = high-power field
Absolute urine output and incidence of POD and PODFR
The mean ± SD urine output at the investigated time points ranged from 4.8 ± 3.5 ml/kg/h after 4 h to 7.0 ± 4.1 ml/kg/h after 24 h (Table 3). The lowest and highest urine outputs of the individual patients were 0.1 ml/kg/h and 26.8 ml/kg/h, respectively. Patients with polyuria with a urine output of >2 ml/kg/h within the first 4 h were more likely to have a higher mean urine output over the entire period of catheterisation (P <0.001). The overall incidence of POD within 48 h of catheterisation was 87.7%, indicating that 50/57 cats had POD at some point during catheterisation. The incidences at the different time points varied from 74.1% (n = 40/54) after 4 h up to a maximum of 95.5% (n = 21/22) after 44 h (Table 3).
Urine output in ml/kg/h (median and mean) of 57 cats with obstructive feline lower urinary tract disease after relief of obstruction, and number and proportion of cats with postobstructive diuresis (POD)

PODFR, was seen in 21/57 cats (36.8%). In nine cats (15.8%) urine volume was higher than fluid rate after 4 h and 8 h. In two cats PODFR was observed after 12 h and after 16 h, while the remaining five cats had PODFR within 24 h.
Influence of fluid therapy on urine output
The mean ± SD initial fluid rate was 6.0 ± 2.3 ml/kg/h (range 2–10 ml/kg/h). There was no correlation between the fluid rate over the first 4 h (fluid rate 0) and the subsequent urine output after 4 h (urine output 1) (P = 0.514, R = 0.095). There was a significant correlation between urine output and the preceding fluid rate at any time point (P ⩽0.001), except for the fluid rate at time point 0.
Association with laboratory parameters in blood and urine
Urine output within the first 4 h was significantly correlated with venous blood pH (P <0.001) and levels of bicarbonate (P = 0.024) on admission. Levels of BUN, creatinine and electrolytes were not correlated with urine output at any time point or mean urine output within 48 h. Baseline acidosis was not correlated with PODFR in general or with PODFR within the first 8 h.
The incidence of POD after the first 4 h was significantly higher in the cats with acidaemia (P = 0.024) but its overall incidence was not associated with the overall incidence of acidaemia on admission (Table 4). No other blood or urine parameters were correlated with the incidence of POD at any time point or with the overall incidence of POD over 48 h. PODFR was not correlated with any parameter in blood.
Association between laboratory abnormalities on admission and the incidence of postobstructive diuresis (POD) 4 h after re-establishment of urine flow and overall incidence of POD

Acidaemia was defined as venous blood pH <7.27. P was calculated using multivariable regression analysis, and P <0.05 was considered significant (bold entry). BUN = blood urea nitrogen; PODFR = postobstructive diuresis in relation to fluid therapy
Regarding the urinalysis results, no correlations between POD or PODFR could be identified (Table 2).
Discussion
The clinical importance of POD after relief of obstruction has been shown in numerous human studies, and UTO is a common feline emergency. Yet there is only one clinical study that has investigated the incidence of POD in cats with natural UTO and its association with laboratory parameters. 7 So far, there are no data regarding the severity and duration of POD, and their associations with laboratory parameters, and there are no studies investigating the impact of fluid therapy in naturally diseased cats. Humans in danger of developing POD are often transferred to an intensive care unit because of the dangers of dehydration and further organ damage. This is especially important in patients who are in any way physically or mentally compromised because they might not be able to replace the loss of fluids sufficiently by oral intake. Pet cats also cannot be expected to drink sufficiently to compensate for excessive POD, especially if they are still weakened. Investigating the actual incidence, severity and risk factors of POD in feline patients is thus important, especially with regard to further optimising therapy for affected cats.
It proved to be quite challenging to find a suitable definition for a condition that can actually truly be regarded to be POD in patients receiving fluid therapy. Historically, POD has been characterised as a urine output >2 ml/kg/h,7,8 but arguably this seems insufficient to describe a state of renal concentration disorder in a patient receiving IV fluid therapy. Most studies on animals undergoing experimental urinary tract obstruction by unilateral or bilateral ureteral ligation are focused on the renal function regarding various solutes,6–10, 51, 52 and urine output is not evaluated in the face of fluid therapy. Moreover, most of the animals included in those studies received specifically composed IV solutions in order to examine the postobstructive function of the kidney, which makes the results less helpful in understanding natural urethral obstruction and thus natural POD. In one study on experimentally obstructed cats, POD, defined as a urine output >1–2 ml/kg/h, occurred in all animals. 8 However, those cats received initially high fluid rates of 20 ml/kg/h over the first 6 h post-obstruction and 15 ml/kg/h for the following 6 h. As investigating POD was not the main aspect of that study, the possible impact of fluid therapy was not discussed. In the only other study on naturally occurring POD in cats, 46% of cats had POD with urine output >2 ml/kg/h within the first 6 h and 64% after 12 h. 7 In that study, cats also received IV fluid rates higher than maintenance. The fluid rate was included into the multivariable regression analysis and not found to be correlated with the frequency of POD.
The cats included in the present study were treated with fluid therapy according to the clinic’s standard SOP protocol for cats with UTO, requiring the tending veterinarian to adjust the fluid rate every 4 h according to urine output plus a third of maintenance rate.
In the present study, POD with a urine output >2ml/kg/h was seen in 74.1% of cats within the first 4 h after catheterisation. After 8 h, 90.9% of the animals had POD. The high incidence of POD led to the investigation of urine output in relation to fluid therapy. For this purpose, PODFR was assessed, defined as a urine output higher than fluid input on at least two subsequent time points. We decided on the 8 h interval because that seemed a sufficiently long enough period to be considered a concentration disorder rather than a coincidental finding. Within the first 8 h, 15.8% of cats had PODFR. More than 70% of the cats had urine output >2 ml/kg/h throughout the entire surveillance period of 48 h, while PODFR was only seen in 36.8% of cats.
The high correlation between fluid rate at time point x with urine output at time point x + 1 suggests that, firstly, fluid rates were adapted to urine output as required by SOP protocol, and, secondly, that fluid rate does apparently play a role in urine output and thus cannot be neglected when discussing incidence and severity of POD. The only exception was the initial fluid rate 0, which did not correlate with urine output within the first 4 h. This might be owing to the fact that the cats were still dehydrated at that time. This discrepancy between the frequency of POD and PODFR in the present study might indicate that the calculation of fluid rates, as suggested in literature and performed in this study, exceeds the needs, and might be the driving force for the high incidence of POD in many cats with naturally occurring obstructive FLUTD.
The incidence of POD after the first 4 h was significantly higher in the cats with acidaemia on admission, which is in accordance with a previous study by Francis et al, 7 reporting that cats with acidaemia on admission were five times more likely to develop POD. This correlation, however, could not be confirmed with those cats showing PODFR within 8 h of catheterisation.
In the present study, after 8 h, there was a strong correlation between fluid rate and urine output throughout the investigated period, which indicates that fluid therapy is a major driving force for urine production in these cats. However, as there was no correlation between fluid rate and urine output within the first 4 h, the correlation between blood pH and bicarbonate with urine output might be accurate. However, it might be coincidental as acidaemia due to metabolic acidosis has been reported to be one of the most common laboratory changes in patients with UTO.3,8,17–19 While acidaemia was associated with POD in the only previous clinical study on POD, 7 in the present study a correlation was only observed after 4 h. Acidaemia did not seem to influence the further course or severity of POD, and there was no correlation at all with PODFR.
There is evidence that POD is partially caused by downregulation of aquaporin-2 (AQP2).20–23 Aquaporins (AQP) are active water carriers that are mainly responsible for the transport of water in the renal medullary collecting duct (MCD),24,25 and this downregulation is probably caused by temporary resistance against the antidiuretic hormone arginine vasopressin (AVP).15,20–23,26–32 Consequently, POD could be considered a form of acquired nephrogenic diabetes insipidus. 33 The pathophysiology of AVP resistance is complex and includes the release of atrial natriuretic peptide (ANP) induced by atrial stretch due to hypervolaemia34,35 as well as an obstruction-induced increase in cyclooxygenase-2 in the kidney,36–38 which leads to the elevated production of prostaglandin E2 (PGE2),10,14,39–41 and to the impairment of the cyclic adenosine monophospate-regulated expression of AQP and urea transporters (UT) located in the MCD.27,42 Changes in intrarenal haemodynamics due to obstruction apparently play an important role,14,43 as do increases in nitric oxide and catecholamines.44,45 Whether a change in blood pH also has a direct effect on the expression of AQP in the mammalian MCD has not yet been researched. However, there is evidence that AQP in certain plants reacts to changes in the intracellular pH.46,47 Besides, acidaemia has recently been suggested to worsen brain oedema by playing a role in AQP1 downregulation. 48 Thus, acidaemia might causally contribute to the pathogenesis of POD by its suspected effect on the translocation of AQP2 to the apical membrane of the MCD, 49 but this remains to be confirmed.
Of the other circulating factors that have often been discussed to play a part in the pathogenesis of POD, particular emphasis has been set for sodium and BUN.6,50–53 Owing to the impaired ability of the obstructed kidney to reabsorb sodium, natriuresis has often been found to occur simultaneously with diuresis in POD. However, neither hyper- nor hyponatriaemia were found to be correlated with urine output or incidence of POD or PODFR in the present or prior studies. The measurement of fractured excretion of sodium in the urine of cats with FLUTD is not routinely performed in the hospital and thus analysis was not possible in the present study.
Historically, a postrenal increase in plasma urea has been considered to be responsible for the onset of POD,6,51–53 but recent studies have not obtained similar results in natural cases of UTO.7,12 In the present study, the BUN level was not associated with either urine output at any time point or the incidence of POD or PODFR. However, both directly and indirectly AVP-regulated UTs that are necessary for the active moderation of urea distribution alongside the MCD have been found to be downregulated in UTO.42,54 In experimental studies, knockout mice with reduced numbers of UT were able to concentrate their urine properly as long as their plasma BUN level was normal but exhibited polyuria when the BUN level was increased by IV urea administration. Thus, one would expect that the combination of reduced UT (caused by UTO) with higher levels of plasma BUN (caused by postrenal azotaemia) would lead to more pronounced POD.
It is not clear whether the downregulation of AQP and UTs is also dependent on the duration of UTO. The urine flow of animals used in experimental studies is usually blocked for 24 h straight, while the duration of UTO in natural cases can only vaguely be estimated and is near impossible to evaluate. While the rate of IV fluid administration did not affect POD in a previous clinical study on cats, 7 and apparently had no effect on POD in experimental studies on rats and dogs, the results of the present study suggest that fluid therapy might have a more important effect on POD than previously postulated.
In our opinion, the general lack of correlation between POD, as well as PODFR, and practicably assessable biochemical parameters suggests that circulating potentially diuretic factors are not the only or main factors influencing the renal concentration disorder in UTO. Finally, it should be stated that it is still not clear whether POD in itself – presuming it is compensated for with adequate fluid therapy – is a harmful condition or rather a sign of the kidneys recovering from prostrenal damage. A recent study in human medicine has found POD to be a positive sign associated with remission, while lack of POD was associated with higher morbidity. 12 Whether this finding can be transferred to feline patients, has not been investigated.
The retrospective characteristic of this study entails some limitations. Firstly, not all patient parameters were available for all cats at all the time points. Data were checked for accuracy, but it cannot be excluded that there were miscalculations that could not be identified retrospectively. Secondly, not all underlying diseases that might have had an effect on urine concentration could be ruled out completely for all patients. However, as most patients were young cats with no prior history of illness, undetected chronic diseases are not very likely. Thirdly, a major limitation is the lack of a control group of physically healthy cats, treated with an indwelling urinary catheter for monitoring urine output, while receiving IV fluids. However, this cannot be performed in a clinical setting.
The cats actually included in the study were a small proportion of those presented to the hospital for UTO; a larger number was excluded from the study for various reasons but mostly because of unavailable medical data. We still think that it is a representative sample, as the cats were randomly chosen, simply based on the availability of their medical records.
There was a loss of data over the time of catheterisation, which has to do with the fact that many cats did not tolerate the catheter any longer, or there was trouble with blocking catheters. If those cats that lost their catheter prematurely were able to urinate, they were not re-catheterised because that would not have been justified in a clinical setting.
Conclusions
The present study confirms that POD is a frequent finding in cats treated for UTO. It also shows that POD is influenced by the administered fluid therapy, which might increase the incidence of polyuria. While the presence and severity of metabolic acidosis on admission was correlated with urine output within the first 4 h, it was not correlated with incidence or overall severity of POD. It is thus possible that the pathophysiological changes leading to the downregulation of AQP in the MCD, as well as the rate of IV fluid therapy, have a greater impact on urine concentration and POD than changes in circulating factors that are assessable in blood and urine. It appears that fluid therapy calculated based on urine output plus insensible losses might actually exceed the needs of cats with obstructive FLUTD.
Footnotes
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
