Abstract
Objectives
The objectives were to determine the resistive index (RI) in normal cats and in cats with various renal diseases, and to evaluate the effect of age on RI.
Methods
The subjects were cats that had ultrasonography (US) of the urinary tract and RI measurement at our centre between January 2003 and April 2014. Based on clinical evaluation, biochemical and haematological tests, urinalysis and US, the cats were classified as healthy or diseased. RI measurements were made from the interlobar or arcuate arteries. Data were analysed for differences between the right and the left kidney, the two sexes, different age groups in healthy cats, and between healthy and diseased cats.
Results
A total of 116 cats (68 males, 48 females) were included: 24 healthy and 92 diseased. In the healthy cats, RI (mean ± SD) differed significantly (P = 0.02) between the right kidney (0.54 ± 0.07) and the left kidney (0.59 ± 0.08). For the left kidney, RI was significantly higher in cats with chronic kidney disease (0.73 ± 0.12) and acute kidney injury (0.72 ± 0.08) (P = 0.0008). For the right kidney, RI was significantly higher in cats with chronic kidney disease (0.72 ± 0.11), acute kidney injury (0.74 ± 0.08), polycystic kidney disease (0.77 ± 0.11) and renal tumour (0.74 ± 0.001) (P <0.0001). There was no significant effect on RI value in either kidney in terms of age or sex.
Conclusions and relevance
RI could be considered a valuable diagnostic tool in cats, useful in the differential diagnosis of diffuse renal diseases. While it does not change with the age of the cat, ultrasonographers should be aware that RI may differ between the two kidneys.
Introduction
Renal diseases occur frequently and are serious in cats. In most cases, a subclinical chronic renal disease progresses to end-stage renal failure when the cat is exposed to various stress factors. Specific methods are required for the successful diagnosis of renal diseases in cats, including diagnostic imaging. 1
Grey-scale ultrasonography (US) is still used to evaluate the kidney, and new ultrasound devices offer high-quality images. 2 Although grey-scale US is a sensitive method of assessing renal diseases, in many cases US findings alone cannot achieve a differential diagnosis. The low specificity of grey-scale US in evaluating renal diseases, particularly diffuse diseases, is still a problem for nephrologists and radiologists in both human and veterinary medicine.3,4
A number of studies have demonstrated the potential use of Doppler US to improve renal evaluation.5–11 In the mid-1970s, a Doppler index was introduced, the arterial resistive index (RI), also known as the Pourcelot index after Leandre Pourcelot, who first proposed its use. 12 The RI measures the arterial resistance in the peripheral vessels by calculating the ratio between the peak systolic velocity (PSV) and the end diastolic velocity (EDV) (RI = [PSV − EDV]/PSV). This index is independent of the angle and position of the exploratory probe, allowing the accurate and reproducible measurement of downstream vascular impedance. The RI depends on blood pressure, glomerular filtration rate (GFR), and renal blood flow (RBF).1–4 Other factors that may affect the RI values are the heart rate, urinary obstruction and hepatic function. 13 However, experimental trials have demonstrated that the main factor affecting the RI is renal tissue and vascular compliance. 3 For this reason, the RI has been proposed as a helpful tool for the diagnosis and prognosis of renal diseases. 14
In human medicine, many studies have examined RI in both healthy and diseased kidneys, and this index is currently used in the differential diagnosis of obstructive and diffuse renal pathologies and in the evaluation of renal transplants.3,15 A number of studies have reported the RI values for healthy and diseased kidneys and renal allografts in dogs and cats, but the clinical utility of this index remains controversial.1,16–26
Studies in humans have also shown that RI values are elevated in infants and the elderly.3,27,28 A study in dogs demonstrated a negative correlation between RI and age when puppies younger than 4 months were compared with adult dogs (up to 7 years old). 29 However, there are limited data on the RI values in aged dogs and in cats.
Therefore, the aim of this study was to investigate the mean RI in healthy and diseased cats, and to assess whether the RI is affected by age in healthy cats.
Materials and methods
Cats referred to the Interdepartmental Center of Veterinary Radiology of Naples, from January 2003 to April 2014, for US assessment of the urinary tract were included in this retrospective study. Cats were excluded if the RI was not measured. Breed, sex and age were recorded for all cats. No cat was sedated during the US examination. Based on the clinical evaluations, biochemical and haematological tests, urinalysis and US, the cats were classified as healthy or diseased, with acute kidney injury (AKI), chronic kidney disease (CKD), end-stage kidney disease (ESK), polycystic kidney disease (PKD), hydronephrosis (HN), a renal tumour (RT) or renal lithiasis (RL).
AKI was suspected when there was a sudden onset of clinical signs like vomiting and/or diarrhoea and/or sensory depression associated with increased serum creatinine levels and/or azotemia and/or proteinuria. Then, the affected cats were classified according to the IRIS grading (AKI I-V, oliguric [O] or not oliguric [NO]). 30 CKD was suspected when there was a history of at least 3 months’ duration of hypercreatininemia and/or proteinuria and classified according to the IRIS grading (CKD 1–4). 30 The association between hypercreatininemia/hyperazotemia and severe ultrasonographical lesions (ie, decreased dimensions, reduced corticomedullary junction, mild pelvic dilation) were considered as ESK.
Triplex Doppler US was performed with a device equipped with a high-frequency linear probe and a multifrequency microconvex probe (Logiq 400 MD; General Electric). Before US, the cats were fasted for 12 h; water was available throughout this time. The cat’s abdominal fur was clipped, and acoustic gel was applied to the skin. US was performed with the cats in dorsal or lateral recumbency. The transducers and frequencies used were selected based on the cat’s weight and renal depth. All US examinations were performed by the same operator (LM).
To determine the RI, the interlobar or arcuate arteries were first identified with colour or power Doppler US, after placing a gate of 1–3 mm over the artery. The wall filter and pulse repetition frequency were set to the smallest values that allowed the flow to be displayed without aliasing (Figure 1). The selection criterion for the Doppler spectrum was a minimum of three similar successive waveforms. The mean RI for each kidney was determined by averaging three waveforms from the interlobar or arcuate arteries.
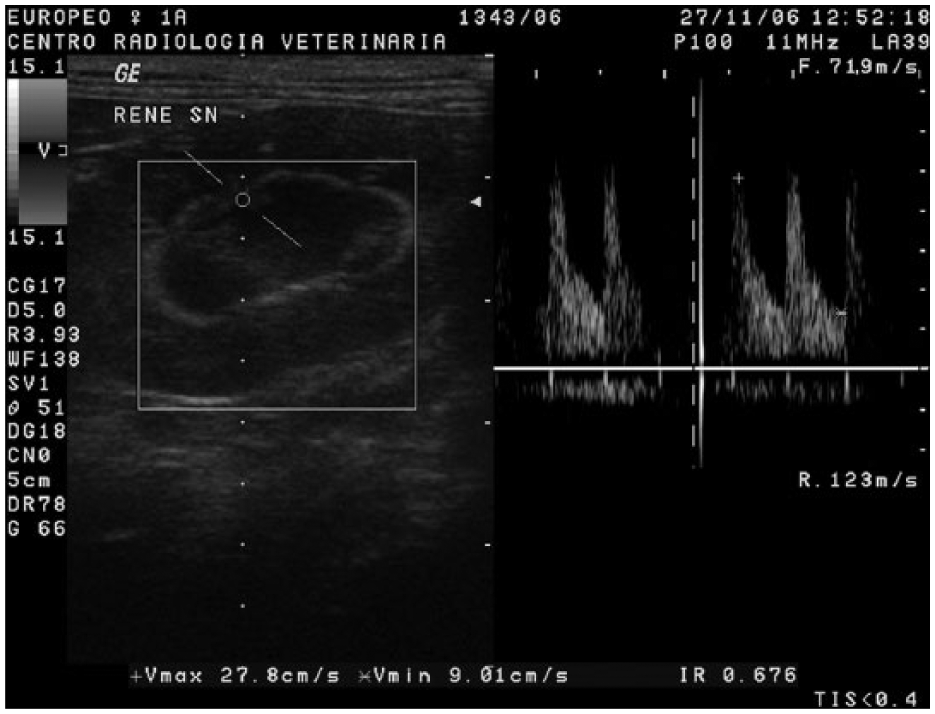
Measurement of the resistive index (RI) in an interlobar artery. After the electronic calipers were positioned on the peak systolic velocity (PSV) and the end diastolic velocity (EDV), the ultrasound scanner automatically calculated the RI using the following formula: RI = (PSV − EDV)/PSV
The data were statistically analysed with dedicated commercial software (Prism 5; GraphPad software). The normality of the distributions was tested with the Shapiro–Wilk test. Differences between the right and left kidneys were determined using the unpaired Student’s t-test for parametric data or the Mann–Whitney U-test. A least squares mean model, with the right and left kidney RI values as the dependent variables and sex as a covariate, was used to evaluate differences between male and female cats. As the data were non-parametrically distributed, comparisons between healthy and diseased cats were made with the Kruskal–Wallis test, followed by Dunn’s multiple comparisons test. The effect of age on RI in healthy cats was assessed using linear regression. One-way ANOVA was used to compare the RI values of healthy cats divided into three groups (group 1, aged ⩽2 years; group 2, aged 2–10 years; and group 3, aged ⩾11 years). Statistical significance was set at P <0.05.
All data are reported as mean ± SD.
Results
A total of 116 cats (68 males, 48 females) were included in this study: 78 European, 23 Persian, six Siamese, five Chartreux and four Birman.
The group of healthy cats comprised 24 cats (15 males, nine females). The mean age of the healthy group was 6.5 years (range 5 months to 15 years).
The group of cats with kidney diseases comprised 92 cats (53 males, 39 females), of which 20 had CKD (seven type 2; nine type 3; four type 4), 14 had AKI (three type II NO; seven type III NO; four type IV: three NO; and one O), 13 had PKD, 12 had ESK, 12 had RL, 11 had HN and 10 had a RT (seven diffuse lymphomas and three focal carcinomas).
In the healthy cats, the RI differed significantly (P = 0.02) between the right kidney (0.54 ± 0.07) and the left kidney (0.59 ± 0.08). However, sex did not affect RI in either healthy or diseased cats.
Because the RI differed significantly between the left and right kidneys, we compared the RI in the left and right kidneys of healthy and diseased cats. Regarding the left kidney, RI was significantly higher in cats with CKD (0.73 ± 0.12) and AKI (0.72 ± 0.08) (P = 0.0008) than in healthy cats. Regarding the right kidney, the RI was significantly higher in cats with CKD (0.72 ± 0.11), AKI (0.74 ± 0.08), PKD (0.77 ± 0.11) and a RT (0.74 ± 0.001) (P <0.0001) than in healthy cats, as shown in Table 1.
Resistive index (RI) values of the right (RK) and left kidneys (LK) in normal and diseased cats
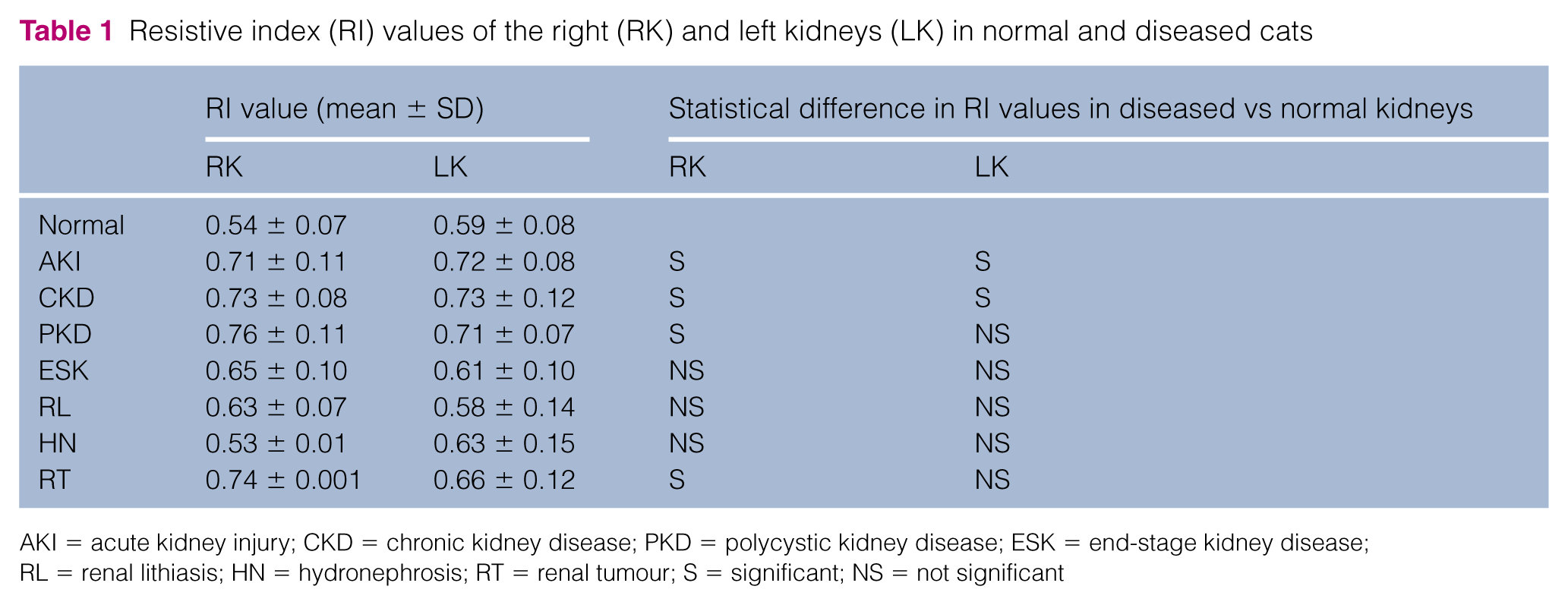
AKI = acute kidney injury; CKD = chronic kidney disease; PKD = polycystic kidney disease; ESK = end-stage kidney disease; RL = renal lithiasis; HN = hydronephrosis; RT = renal tumour; S = significant; NS = not significant
Age had a slight but significant effect on the RI of the left kidney (R2 = 0.19, P = 0.04) but no effect on the right kidney (R2 = 0.08, P = 0.17). No significant effect of age was observed when the RI values of both kidneys were analysed together. ANOVA revealed no differences among the three age categories for either the right or left kidney.
Discussion
In this study, the mean RI values differed significantly between the left and right kidneys in the healthy cats. Differences between the two kidneys have never been reported in small animals, whereas in humans differences in RI are reported in normal and hypertensive subjects.31,32 Until now, no convincing explanation has been proposed for this finding but, since the RI, among others, depend on RBF, this parameter was reported as different between the two kidneys in normal people. 33 In our study, other possible explanations could lie in the bias introduced by the ultrasonographer or in the presence of renal damages not appreciable clinically, ultrasonographically or from laboratory tests. However, in the present study, for both kidneys the RI values obtained were similar to the values reported in other studies, as shown in Table 2, and lower than the proposed cut-off value of 0.70.1,21
Kidney resistive index (RI) values reported in healthy cats

Atr = atropine; Ace = acepromazine; Ket = ketamine; Xyl = xylazine
As reported in previous studies,1,19–23 our results demonstrate the feasibility of measuring RI in conscious cats and that its values are similar to those obtained in sedated subjects.16,18 There is only one report 1 in which the use of isoflurane anaesthesia was shown to significantly increase RI, due to an increased intrarenal vascular resistance and a decreased renal blood flow.
In human medicine, RI is currently used as an indicator of vascular resistance and is correlated with the severity and progression of CKD. Several studies have shown that RI increases significantly in diffuse renal diseases, including AKI and CKD, confirming its importance in distinguishing renal failure from other prerenal diseases. 34 Likewise, in veterinary medicine, RI was shown to be significantly higher in cats with PKD, HN, 26 AKI or CKD. 18
In the present study, high RI values for the left kidney were observed in cats with AKI and CKD compared with those of healthy cats, and high RI values for the right kidney were observed in cats with AKI, CKD, PKD and a RT. Although the mean RI values were higher in cats with HN, ESK and RL than in healthy cats, the differences were not statistically significant. All the diseases considered may affect the renal arterial compliance and therefore the RI through one or more factors (GRF, RBF, etc). AKI and CKD groups showed an even higher RI than the normal group but, as there were a small number of cats with each of the different kidney diseases in this study, the correlation between the IRIS stages or the severity of the lesions and the RI values was not evaluated. However, previous studies demonstrated the presence of this correlation.25,26 Unlike a previous study in dogs, 26 our study found that RI was not significantly increased in cats with HN. This may be due to the small number of cats examined in our study or to the presence of normotensive HN. This is consistent with several studies that reported RI values within normal limits in cats with severe dilation of the renal pelvis. 35 Low RI values were also recorded in cats with ESK. As in severe HN, the low RI values recorded in ESK were attributed to reduced filtration pressure, a feature of this disease. 35
In humans, the RI is higher in children and the elderly than in other age groups. The RI is elevated in children and is correlated with plasma renin activity, tending to normalise by about 11 years of age. 28 In elderly people, increased RI values have been attributed to essential hypertension and/or intraparenchymal arteriolar damage and glomerular sclerosis. 36 In veterinary medicine, only one study has demonstrated similar, higher RI values in dogs aged <4 months. As in children, this finding was attributed to elevated plasma renin activity. 29 We observed a slight but positive correlation between the left kidney RI and age. However, no correlation was demonstrated when the healthy cats were grouped according to age. Therefore, we conclude that there is no significant correlation between age and RI in cats. However, this result could be affected by the composition and dimensions of the sample. Importantly, if the RI is not correlated with age in cats, it could be used as an independent index in cats of any age.
Conclusions
Although Doppler US is not a substitute for renal biopsy, our results suggest that the RI could be a valuable diagnostic tool, useful for the differential diagnosis of diffuse renal diseases. It appears to be reliable in cats of any age but more studies with larger samples are required to understand the effects of age on the RI. The RI may also differ between the two kidneys.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
