Abstract
Objectives
This study aimed to assess non-invasively the cardiovascular effects of high-dose medetomidine on healthy male cats undergoing semen collection.
Methods
Haemodynamic evaluations were assessed on the basis of clinical examination, systolic arterial pressure (SAP) and transthoracic echocardiographic examination. Eight client owned, male domestic shorthair cats were sedated with a bolus of medetomidine intramuscularly (IM; 0.13 mg/kg), and semen collection was performed. A second transthoracic echocardiographic examination and SAP measurement were carried out 15 mins after sedation. At the end of the examination, the patients received a bolus of atipamezole (0.3 mg/kg) IM.
Results
The cats were deeply sedated, relaxed and laterally recumbent during the entire procedure. No rhythm abnormalities were observed during the examinations and no significant increase in SAP was recorded. Heart rate dropped from 200 ± 33 to 92 ± 13.1 beats per min after sedation. There was a significant increase in left ventricular dimensions and the left atrial area. The parameters of left ventricular systolic function were reduced, as were systemic and pulmonary cardiac outputs. Peak diastolic wave velocities were significantly reduced, while isovolumic contraction and relaxation time of the left ventricle were prolonged. Aortic valve insufficiency was recorded for all cats, while mitral valve insufficiency was noted in five cats. None of the subjects developed systolic anterior motion of the mitral valve.
Conclusions and relevance
The protocol allowed us to collect good semen samples in healthy cats. However, high-dose medetomidine induces significant haemodynamic effects on the feline heart, mainly due to a reduced heart rate, an increased cardiac preload and impaired systolic function. The animals recovered from the anaesthesia, after antagonist administration, without showing any clinically relevant consequences.
Introduction
Medetomidine is a highly selective α2-adrenoreceptive agonist; it is widely used in veterinary medicine to induce sedation, analgesia and muscle relaxation.1–3 Medetomidine is used alone or in combination with other drugs for minor surgical and interventionalprocedures and, if used for premedication, reduces the amount of drugs necessary to induce and maintain general anaesthesia.1,3 In clinical practice, the dose used in cats varies from 0.02–0.08 mg/kg.1,3 The sedative and analgesic effects, and their depth and duration are dose dependent and can be completely reversed by using the antagonist atipamezole.2–4
α2-adrenoreceptor activation produces effects on the central nervous system, desirable for patient anaesthetic control and, at the same time, it also has several cardiovascular effects. Owing to peripheral arterial vasoconstriction, medetomidine induces responsive bradycardia mediated by ventricular baroreceptors. This effect is potentiatedby the action of the drug on the central nervous system, with subsequent reduced sympathetic tone.1–7 Cardiac output (CO) is typically reduced when medetomidine is administered in a healthy animal as a consequence of increased systemic vascular resistance (SVR) and reflex bradycardia.1,3,4,8
In dogs, medetomidine is reported to induce some electrocardiographic alterations, such as enhanced sinus arrhythmia, sinus bigeminy and atrioventricular blocks.1,3,9 In cats with hypertrophic obstructive cardiomyopathy due to the systolic anterior motion (SAM) of the mitral valve (MV), medetomidine is reported to reduce the left ventricular outflow tract gradient and to resolve the dynamic stenosis. 8
α2-adrenoreceptive agonists and medetomidine can also be used for semen collection. In horses, erection and ejaculation can be artificially produced by administering xylazine or imipramine. 10 In cats, medetomidine induces a myorelaxant effect on the deferent duct, leading to sperm ejection and the release of spermatozoa within the urethra.11,12 This method of semen collection in the feline is feasible and apparently safe; however, the dose of medetomidine needed to produce a good semen sample is much higher than the dose reported for anaesthetic and pain management procedures, and varies in cats between 0.13 and 0.14 mg/kg.12,13 The cardiovascular effect of such a dose of medetomidine in a population of feline patients has never been evaluated.
The aim of the present study was to assess non-invasively the cardiac and vascular sequelae of high-dose medetomidine in a group of healthy male cats undergoing semen collection.
Materials and methods
Animals
Privately owned intact male cats undergoing semen collection at the small animal reproduction service of the Veterinary Teaching Hospital were used for the study. All the owners were informed about the study and were asked to sign a written informed consent form. The experiment was conducted in accordance with the provisions of European Economic Community Council Directive 86/609, adopted by the Italian Government (DL 27/01/1992 n° 116). The study was approved by the local and national ethical committees (SVC 43/2013).
In order to exclude cats having major systemic abnormalities or cardiac murmurs, all the animals underwent a complete clinical examination by an anaesthetist and a cardiologist. The patients were included if no clinical signs (gastrointestinal, neurological, respiratory or cardiovascular disorders) were detected. Thus, all the animals needed to be classified as American Society of Anesthesiologists’ (ASA) I, according to its classification of physical status. The patients were hospitalised for the time required by the procedure and until complete recovery from sedation. At the end of the protocol, all the animals underwent a second clinical examination, and were dismissed in accordance with the judgement of the anaesthetist.
Sedation protocol and semen collection
Each patient received a single bolus of medetomidine (0.13 mg/kg; Sedastart [Esteve SpA]) intramuscularly (IM) and was placed into a cat carrier sheltered from light and noise but strictly monitored in order to evaluate sedation status and possible side effects. After 15 mins, the patients were taken out of the carriers and cardiovascular examination was performed as described below. When all the procedures were completed, semen collection was performed by urethral catheterisation after pharmacological induction (URCAPI), as previously described;12,13 the semen collection required approximately 5 mins. During the entire procedure, the cats received oxygen supplementation through an anaesthesia mask. At the end of the examination, the patients received an atipamezole bolus (0.3 mg/kg IM; Sedastop [Esteve SpA]). Ten minutes after the injection of the antagonist the grade of sedation and the heart rate (HR) were evaluated.
After animal sedation, the entire procedure took approximately 30 mins.
Cardiovascular examination
Haemodynamic evaluations, assessed on the basis of clinical examination (femoral pulse evaluation, mucous membranes and capillary refill time), systolic arterial pressure (SAP) and transthoracic echocardiography (ECG) were carried out before sedation with awake, manually restrained cats, and then repeated 15 mins after medetomidine administration.
Systolic arterial pressure was measured using a Doppler device (Minidop ES-100 VX; Hadeco) as reported by Binns et al. 14 In detail, the Doppler probe was placed over the common digital branch of the radial artery on the palmar aspect of the foot. The hair was clipped before placing the probe. Ultrasound transmission gel (Aquasonic 100; Parker Laboratories) was placed between the probe and the skin to improve ultrasonic contact, and the volume of the Doppler machine was adjusted to obtain a clearly audible signal. The cuff was inflated until flow sounds were no longer audible, and then gradually deflated until clear flow sounds became audible. The manometer reading at the reappearance of flow sounds was recorded as the SAP, as described by Jepson et al. 15 Five repeated measurements were carried out; the highest and the lowest values were excluded, and the mean value was calculated.
Transthoracic ECG was performed by the same experienced operator (MBT) using an ultrasound unit (M7Vet ultrasound unit; Shenzhen Mindray Bio-Medical Electronics) equipped with a multifrequency phased array probe using second harmonic settings (7–3 MHz) and continuous ECG tracing. All echocardiographic examinations were carried out with the cats in both right and left lateral recumbency, using a dedicated table, and following a standard approach as described elsewhere. 16 Complete M-Mode, two-dimensional, colour, pulsed wave and continuous wave Doppler analysis were carried out. The images and video clips were stored in the internal hard disc memory of the ultrasound unit and used subsequently for off-line analysis. The echocardiographic variables measured and calculated for this study are listed in Table 1. All the measurement criteria and the formulae used to calculate some variables were obtained from the veterinary literature and are detailed elsewhere. 17
Haemodynamic variables in eight male cats sedated with high-dose medetomidine

Values are given as mean ± SD. Significant values are in bold
Ao = aorta; CO = cardiac output; CSA = cross sectional area; DT = deceleration time; E:A = ratio between the mitral valve E wave peak velocity and the mitral valve A wave peak velocity; EF = ejection fraction; EDV = end-diastolic volume; ESV = end-systolic volume; FS = fractional shortening; IVCT = isovolumic contraction time; IVRT = isovolumic relaxation time; IVSd = interventricular septal thickness in diastole; IVSs = interventricular septal thickness in systole; LA = left atrium; LV = left ventricle; LVET = left ventricular ejection time; LVIDd = left ventricular internal diameter in diastole; LVIDs = left ventricular internal diameter in systole; LVPWd = left ventricular posterior wall thickness in diastole; LVPWs = left ventricular posterior wall thickness in systole; MV = mitral valve; RV = right ventricle; SAM = systolic anterior motion of the mitral valve; SV = stroke volume; VTI = velocity time integral
Statistical analysis
Data are reported as mean ± SD. Descriptive statistical analyses were carried out using Microsoft Excel. Continuous data were tested for normality using a D’Agostino-Pearson test. Haemodynamic variables before and after medetomidine administration were compared using a paired t-test for repeated measurements. P <0.05 was considered significant. This part of the analysis was run using dedicated software (Prism 5; GraphPad Software).
Results
Animals and sedation
Eight cats were enrolled in the study on the basis of the above-mentioned criteria. All animals were domestic shorthair cats with an age ranging from 6–96 months; they had a mean body weight of 3.6 ± 0.9 kg.
All patients vomited gastric secretions once within 15 mins after medetomidine injection. All the cats were deeply sedated, relaxed and laterally recumbent during the entire procedure. Semen collection was feasible in all animals. Atipamezole administration produced a visible decrease in the quality of sedation within 10 mins of injection; sternal recumbency was achieved within 20 mins in all cats, and complete recovery without any grade of sedation was recorded; the discharge was possible not more than 1 h after the end of the procedure. The owners did not notice any abnormalities at home.
Cardiovascular examination
Arterial blood pressure and echocardiographic examination were performed before and after sedation in all cats. No rhythm abnormalities were observed during the examinations. All the recorded haemodynamic variables are listed in Table 1. Variables with a statistical difference before and after sedation are indicated. HRs dropped significantly from 200 bpm to 92 bpm (as a mean value) after medetomidine administration (P <0.01). SAP increased after medetomidine administration but the difference was not statistically significant. Preload increased, as expressed by increased left ventricular diastolic dimensions and atrial area, while left ventricular systolic function, and its related variables, including CO, appeared reduced due to the medetomidine effect (Figure 1). Some diastolic variables, such as E wave, representing the peak velocity of the early left ventricular diastolic refill wave, and A wave, the peak velocity of the late left ventricular refill wave due to active atrial contraction, and isovolumic relaxation time (IVRT) were also affected in terms of reduction of the first two and an increase in the last one. Left ventricular wall thickness and great vessel diameter did not change before and after sedation. Only left ventricular posterior wall thickness in systole decreased after sedation. Interestingly, aortic valve insufficiency was recorded for all cats, while MV insufficiency was noted in 5/8 cats as an effect of the medetomidine. None of the subjects developed SAM.
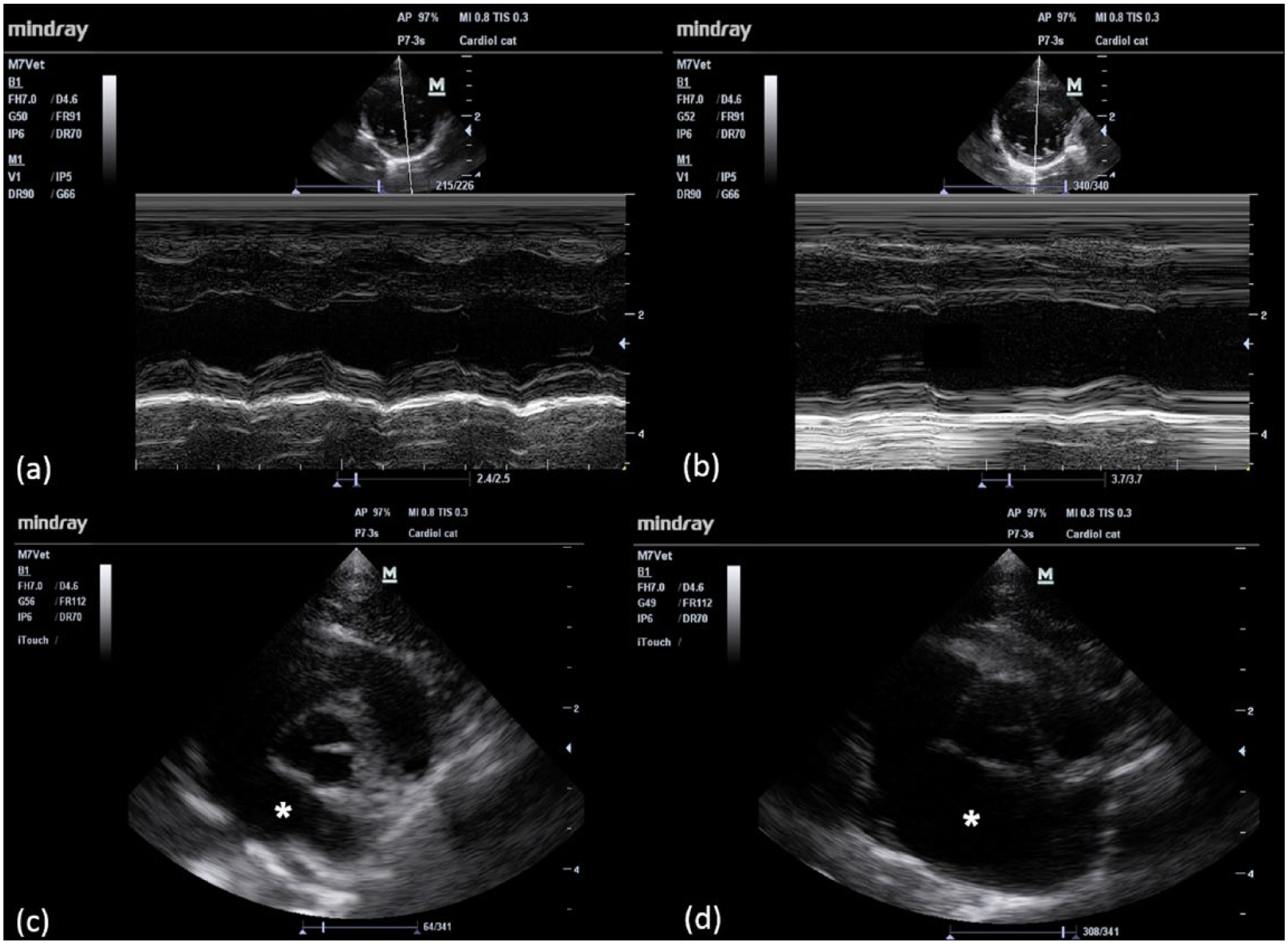
Echocardiographic images of two cats enrolled in the study before (left panels) and after (right panels) the intramuscular administration of high-dose medetomidine (0.13 mg/kg) for sperm collection. In the upper panels, a two-dimensional (2D) guided M-mode analysis of left ventricular function over time obtained from one cat enrolled in the study is displayed. Note the ventricular diameter (a) before and (b) after medetomidine administration. There is a clear decrease in heart rate and an increase in both diastolic and systolic dimensions with a subsequent reduction in systolic function. In the lower panels, 2D images of the heart base and left atrium (asterisk) of another cat enrolled in the study group are shown. An obvious difference in the left atrial area is visible between the (c) baseline state and (d) after sedation
Discussion
In the present study, we echocardiographically evaluated the haemodynamic effects of high-dose medetomidine in healthy cats undergoing semen collection using URCAPI.12,13
The main finding observed was that high-dose medetomidine induces significant haemodynamic effects on the feline heart.
The semen collection using URCAPI did not seem to produce any pain in cats. The introduction of a urinary 3F tomcat catheter (Portex Jackson Cat Catheter; Jorgensen Laboratories) in the urethra for semen collection did not evoke any response in cats, and changes in cardiac or respiratory frequency were not recorded. 12 The procedure was feasible in all animals, and the patients recovered within 30 mins of the end of the protocol, and only vomiting was observed as an adverse effect. However, several haemodynamic parameters, as assessed by transthoracic ECG, changed significantly after sedation, leading to a reduced HR, increased preload and reduced systolic function of the left ventricle (LV). As an expression of this, CO also decreased significantly after sedation, mainly owing to the reduction in HR.
Some of these findings have previously been described in cats receiving low doses (0.02 mg/kg) of medetomidine. 4 CO tends to be reduced by 50–70% from the starting values, owing both to the bradycardia effect and suppressed myocardial contractility. This degree of reduction is similar between our study and previous studies in which lower doses of medetomidine were tested.2,4 Systolic blood pressure tended to increase in our study but no statistical difference was observed. In previous studies, no relevant variation was seen inSAP in the first 15 mins after administering the medetomidine but there was a progressive decrease later on.4,18 In the study of Lamont et al, 4 only mean and diastolic systemic pressure increased 15 mins after drug administration. In our study, we were only able to measure the systolic values using a Doppler device; therefore, our accuracy in detecting pressure variations during monitoring could have been underpowered. In fact, Jepson et al found that the Doppler device in cats yields superior results than an oscillometric machine for determination of systolic blood pressure only, 15 while the determination of the diastolic blood pressure is not reliable and more difficult to obtain.
Systolic function of the LV as assessed by fractional shortening, ejection fraction, end systolic volume, left ventricular internal diameter in systole, and forward aortic and pulmonary flows were significantly reduced in the cats in the present study after medetomidine was administered. These parameters are not HR-dependent, as is CO, and their reduction represents clear evidence of suppressed systolic function and therefore likely reduced contractility of the ventricle. The clearest explanation for this is the abrupt increase in SVR mediated by the medication, which induced an increase in afterload and subsequent depressed contractility due to a baroreceptor reflex. 8
However, in our cats, a significantly increased preload was found in response to the medetomidine administration. Both left ventricular and atrial dimensions increased, as seen by increased LV internal diameter and volume in diastole, and increased left atrium area. Lamont et al recorded an increase in pulmonary capillary wedge pressure (PCWP) in cats sedated with medetomidine. 4 Similarly, Pypendop and Verstegen reported a transient increase in PCPW in dogs after intravenous administration of medetomidine, and this effect appeared to be dose dependent. 5 As PCPW approximates LV preload, a possible explanation for this finding could be a transient response to an acute circulatory stasis related to bradycardia and to an increased LV afterload, resulting in blood stasis in the pulmonary capillaries. Another explanation could be the reduction in myocardial compliance secondarily to drug-related decreased myocardial perfusion.4,5 Similarly, an increase in the SAP expressed by an increase in central venous pressure has been described in dogs receiving medetomidine as an expression of reduced venous capacitance and CO, and subsequent blood stasis on the venous side. 5
Some LV diastolic variables are known to be affected by HR, such as left ventricular ejection time, IVRT, isovolumic contraction time and LV inflow waves. This could have affected the significance of our results as we found a statistical difference for some of these variables in our cohort of cats before and after medetomidine administration. However, in a study conducted on healthy cats, the authors concluded that, even if a weak correlation is present between HR and MV E and A wave peak velocity, and IVRT, this effect has only minimal influence on the haemodynamic variables, and is unlikely to produce clinical relevance. 19
Aortic and MV insufficiency were highly prevalent in the cats in our study after sedation. This might partially be explained by the increased blood pressure, which led to stress on the aortic valve with subsequent mild leakage during diastole. However, the LV dilation could have produced a mitral annulus stretch with the subsequent loss of complete closure of the mitral leaflets during LV contraction.
In our study, the cats were judged to be healthy, and recovered completely after sedation. At the end of the study vomiting was the only adverse clinical effect seen after medetomidine administration. Vomiting could be a cause of mortality in sedated animals because of aspiration.
However, in our cohort, it consisted of only a small quantity of gastric secretions, and the mouth was inspected to exclude the presence of foreign material. Other relevant clinical disturbances were not reported by the anaesthetist and the owners. This suggests that, despite the considerable effect on the cardiovascular function we observed, these changes are not clinically relevant and dangerous in healthy cats. Moreover, similar negative effects on the cardiovascular system have already been observed in cats even with conventional doses of medetomidine, meaning that some cardiovascular sequelae are intrinsic in the mechanism of action of the molecule and are not necessarily dose related. It should, however, be noted that, in another study conducted on cats, such significant echocardiographic changes in heart function were not detected when using a standard dose of medetomidine. 8 Whether high-dose medetomidine might significantly impair the cardiovascular system, leading to subtle consequences for the animal, cannot be assessed on the basis of our study.
Some limitations of the present study must be highlighted. First, cats were not re-examined after administration of atipamezole to evaluate the presence of residual myocardial damage or cardiovascular alterations secondary to medetomidine injection. However, cats were judged to be clinically normal by the anaesthetist at the time of discharge from the hospital, and owners did not note any abnormality at home. Second, even if cats were considered healthy on the basis of baseline cardiovascular examination, a subtle myocardial alteration could not be excluded for sure, as some cardiomyopathies in cats might be missed until they produce haemodynamic sequelae. Third, LV volumes were calculated using also the Teichholz formula, which has not been validated in cats, while other two-dimensional values might be more appropriate in this species. 20 However, the Teichholz method was used as an arbitrary way to evaluate the drug-induced cardiovascular alterations in the cats of the present study, and these alterations remained statistically relevant, even when using different techniques to measure CO (ie, Doppler-derived stroke volume) or just measuring the LV diameters during systole and diastole. Finally, a single dose of medetomidine was used. This was done as the purpose of our study was to evaluate this specific sedative protocol, which has been standardised in previous studies as a method for semen collection in cats.12,13
The cats used in the present study were judged to be healthy on the basis of a clinical examination and baseline ECG. The medetomidine dose used seemed to be well tolerated. However, it cannot be ruled out that a significant negative effect on cardiac function might be produced in cats with heart disease. A careful cardiovascular examination and ECG should be carried out before sedation with a high dose of medetomidine.
Conclusions
In the present study, the cardiovascular effects of high-dose medetomidine (0.13 mg/kg) in intact male cats undergoing semen collection were evaluated non-invasively. Significant modification in both systolic and diastolic cardiac function, and in SAP were observed. The animals were stable during the procedure and completely recovered from the anaesthesia after antagonist administration. Whether this sedative protocol might be harmful in cats affected by heart disease remains to be clarified. However, as medetomidine can significantly impair cardiac function, a cardiovascular examination and ECG should be performed before the procedure,and the protocol should be reserved for cats assessed as ASA I only.
Footnotes
Acknowledgements
We express our gratitude to Dr Ilaria Juka, DVM, Foschi srl (Rome, Italy), for her technical support and for providing the fully equipped ultrasound unit.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Funding
This research was supported by a Ricerca Fondamentale Orientata grant from the University of Bologna.
