Abstract
Objectives
Two hundred and thirty-four cats removed from an institutionalized hoarding facility (IHF) demonstrated severe, atypical pyogenic infections. The objective of this study was to document the various syndromes and determine the etiology of the infections.
Methods
All cats were evaluated initially after removal from the IHF and on a daily basis for at least 15 months. Samples were collected and sent for culture/susceptibility and histopathology to commercial laboratories or stored at −20oC. PCR was performed using universal bacterial primers to amplify the 16S–23S rRNA intergenic spacer region. PCR products were sequenced to determine the identity of the bacteria.
Results
Multiple pyogenic syndromes were documented, including abscesses of the paws and carpal/tarsal regions in 82 cats, acute rhinitis with profuse purulent nasal discharge in 68 cats and cervical lymphadenitis with abscessation unassociated with any wounding in 51 cats. Many cats exhibited septic arthritis with total joint destruction, necrotizing fasciitis, meningitis, otitis and septic shock, often leading to death. These infections appeared to be caused by beta-hemolytic streptococci (BHS) based on initial culture results (n = 10), though speciation was unclear and some samples (n = 6) produced no growth. Based on PCR results (n = 26), Streptococcus canis was the only bacterial species or the dominant species identified in each sample, and was the only species present in all the regions associated with the pyogenic infections.
Conclusions and relevance
Horizontal gene transfer and loss of the cell wall may account for the discrepancy between the culture and PCR results and the highly pathogenic nature of S canis in this particular population of cats. A large-scale hoarding situation with multiple animal species, overcrowding, stress and mixing of animals from many geographical regions created ideal conditions for these events to occur. The specific virulence factors present may be more useful in predicting the pathophysiology of BHS infections than the species of Streptococcus found in the host per se.
Introduction
Beta-hemolytic streptococci (BHS) are gram-positive cocci, commonly found in pairs or chains, that have the ability to lyse erythrocytes completely to create a zone of clearing around colonies growing on blood agar plates. They can be further characterized through serological detection of cell wall, or capsular antigens and classified in various Lancefield groups.
In humans, group A BHS (predominantly Streptococcus pyogenes) are responsible for a variety of diseases, including pharyngitis, otitis, sinusitis, cellulitis and abscesses, endocarditis, septic arthritis, pneumonia, meningitis, necrotizing fasciitis, sepsis and toxic shock syndrome. 1 After clearing the infection, poststreptococcal glomerulonephritis, due to antigen–antibody complex deposition within the kidneys, or rheumatic fever may occur. 2
Group C BHS include two main species with great significance to equine health: Streptococcus equi subsp equi (S equi) and Streptococcus equi subsp zooepidemicus (S zooepidemicus). S equi is a highly contagious pathogen of horses that causes fever, enlargement of cervical lymph nodes that is often followed by abscessation and drainage, pharyngitis and rhinitis that can be accompanied by profuse purulent nasal discharge. 3 Purulent ocular discharge and empyema of the guttural pouch can also occur. The purulent discharge becomes a source of infection to other horses, with S equi entering the new host via the mouth or nose.
In some horses, S equi infections extend beyond the head and neck, causing abscesses in organs throughout the body, pneumonia, myocarditis and septic arthritis.4,5 As with the group A BHS infections of humans, immune-mediated diseases can also result from S equi infection. The main difference is that antigen–antibody complex deposition most likely occurs on blood vessel walls, resulting in vasculitis (purpura hemorrhagica) rather than leading to glomerulonephritis. 6
Even though S equi has 98% DNA homology to S zooepidemicus, the latter is a commensal of mucosal surfaces in horses, and is considered to be an opportunistic pathogen that causes infection secondarily to stress or concurrent viral infection. A significant number of horses with pneumonia are found to have S zooepidemicus infections,7,8 and it has been associated with hemorrhagic pneumonia characterized by acute dyspnea, hemorrhagic nasal discharge, rapid deterioration and death. 9
There have been sporadic reports of humans becoming infected with S zooepidemicus and developing respiratory disease, pneumonia, endocarditis, septic arthritis, meningitis, septicemia and streptococcal toxic shock syndrome.10,11 A case of cervical lymphadenitis was also documented. 12 As with the group A BHS infections, antigen–antibody complex deposition within the kidneys (poststreptococcal glomerulonephritis) has also been reported secondarily to S zooepidemicus infection in humans. 13
S zooepidemicus has been recently recognized as an emerging pathogen of dogs. 14 Several severe outbreaks of streptococcal hemorrhagic pneumonia characterized by acute onset of dyspnea, rapid progression of the disease and a high fatality rate have been documented.15–18 As with S equi infections in horses, the S zooepidemicus infections were found to spread through contact with the contaminated respiratory secretions. Dogs more mildly affected presented with purulent nasal discharge, rhinitis and sinusitis.17,19
Unlike S zooepidemicus, Streptococcus canis of Lancefield group G, is a common inhabitant of the skin and mucosal surfaces of dogs. Based on the presence of S canis in healthy dogs, as well as the presence of other microorganisms found along with S canis in areas of infection, S canis has historically been considered an opportunistic pathogen.20–22 There have, however, been reports of S canis causing severe, invasive infections such as necrotizing fasciitis, sepsis and toxic shock syndrome in dogs, with striking similarities to the presentation in humans with severe S pyogenes infections.22–24
There are fewer reports available regarding streptococcal infections of cats. Goldman and Moore were the first to document group G BHS infections in cats within a random source cat colony. 25 The majority of the affected cats presented with severe cervical lymphadenitis and abscessation, although three cats had paw and limb abscesses, and two cats had nasal discharge, ocular discharge and sinusitis. All of the cats recovered after treatment; however, three cats had recurrent abscesses in the regions that were previously affected.
Swindle et al identified lymphadenitis in the head and neck, abscesses in the same regions and evidence of systemic infection caused by group G BHS. 26 When healthy adult cats were exposed to the bacteria via the oral cavity, they developed the same signs as in the naturally occurring infections, as well as tonsillitis, acute rhinitis with purulent nasal discharge, sinusitis, otitis media and bronchopneumonia.
In a different outbreak of group G BHS, the infection was found to be highly contagious, with indirect transfer occurring in four individually housed cats and a recurrence of the disease 3 months later. 27 Even more severe manifestations of group G BHS infections, including necrosuppurative bronchopneumonia and severe pyothorax, were documented in two cats from a colony of 105 cats. 28
Iglauer et al described a pathologic condition associated with a group G BHS, S canis, characterized by purulent arthritis of one joint. 29 More recently, three shelter outbreaks of S canis were documented with two distinct patterns of infection. 30 Necrotizing sinusitis and rhinitis, followed by meningitis, as well as wounds of the distal limbs and paws were documented in two of the shelters. In the other shelter, a rapidly progressive streptococcal disease resulted in ulcerated and oozing paws, necrotizing fasciitis and sudden death. 30
Another recent shelter outbreak in Israel documented cats with rhinitis associated with copious purulent nasal discharge and severe, acute and diffuse bronchopneumonia that was suppurative or necrosuppurative, as well as four cats with pyogranulomatous meningoencephalitis. 31 S zooepidemicus was isolated from the lungs of all of these cats and was either the only organism cultured or the dominant organism in mixed culture.
This report illustrates highly pathogenic and pyogenic BHS infections in a large number of cats rescued from an institutionalized hoarding facility (IHF), with a unique combination of clinical signs that were not previously described in any single facility.
Materials and methods
Over 500 animals were recovered from the IHF in March 2008. Eight dogs, six draft horses, a goat and several chickens were found in addition to the 497 cats and cat carcasses (390 living and 107 dead). The IHF consisted of a house, garage, modular trailer and several small shed-type buildings. There was some separation of groups of cats from one another within the various structures but many cats had access to the outdoors within enclosures or were free roaming. No isolation or quarantine procedures were in effect, and the environment was contaminated with purulent discharge, blood, vomitus and excrement. The proprietor’s own records indicated that hundreds of cats came into the IHF each week from more than 12 different states. The facility had been in operation for at least 10 years.
Cats were given identification numbers based on the region in which they were found in the facility. The animals were triaged at the scene, and the critically ill were examined and treated in mobile animal hospitals. Cats that appeared stable enough to travel were taken to a former animal shelter that was reopened specifically for the cats. All of the animals were examined by veterinarians and a standardized examination sheet was completed, and pictures were taken to document the physical conditions. The deceased cats that were found in freezers were initially examined and photographed in the frozen state. Necropsies were performed as time allowed, and samples were collected and stored at −20°C. All living cats in the population were tested for feline leukemia virus and feline immunodeficiency virus with the ELISA SNAP test (IDEXX) by the end of the first week of treatment. The initial protocol of benzathine/procaine penicillin (Bimeda-MTC Animal Health) dosed at 50,000 IU/kg subcutaneously (SC) q48h was used alone for wounds/abscesses, and in combination with enrofloxacin (Bayer Animal Health) dosed at 5 mg/kg q24h (either orally or SC depending on the temperament and clinical signs of the cat) if any respiratory clinical signs were present, for at least 10 days. Other treatments were administered based on the condition of the individual cat.
The veterinary team continued to document the status of each cat with a minimum of daily visual inspection, and monitoring of food and water intake, as well as stool and urine production. More clinical data were gathered (ie, rectal temperature, pain assessment, physical examination) for cats showing clinical signs of disease. While it was not practical to do full diagnostic work-ups on all of the cats, when resources allowed and the diagnostics would benefit the understanding of the disease processes for better treatment of the population, samples were taken and either analyzed in-house (blood chemistry, fecal flotation and cytology, hematocrit and blood cytology, skin scrapes, fungal cultures), sent to a reference laboratory (Antech Diagnostics, Irvine CA, USA) or sent to university laboratories (Dr Kate Hurley, University of California, Davis; Lucy Whittier Molecular and Diagnostic Core Facility, University of California, Davis; and The Ohio State University). Other samples that were not immediately analyzed were placed in formalin or stored at −20°C for later evaluation.
Twenty-six samples taken from the cats with suspected streptococcal infections were thawed, and DNA was extracted using the DNeasy Blood and Tissue Kit (Qiagen), following the manufacturer’s instructions. PCR was performed with universal bacterial primers ISTF and ITSReub, 32 which are complimentary to conserved regions of the 16S and 23S rRNA genes, to amplify the intergenic spacer region of all bacteria present in the samples. Each reaction contained 1 × PCR buffer (without MgCl2), 3mM MgCl2, 250 μM of each deoxynucleotide triphosphatase, 0.25 μM of ITSF, 0.25 μM of ITSReub, 1.5 U of Taq DNA polymerase and 300 ng of template DNA in a final volume of 50 μl. Reactions were run at 94°C for 3 mins, followed by 35 cycles of 94°C for 45 s, 55°C for 1 min, 72°C for 2 mins and a final extension at 72°C for 7 mins. PCR products were evaluated via agarose gel electrophoresis, with DNA from various bacterial species separating by size.
After purification with the Wizard Genomic DNA purification Kit (Promega), ligations of the products into plasmid pGEM-T (Promega) were followed by transformation into competent Escherichia coli JM109 cells. Plasmid DNA was purified with a plasmid prep kit (Qiagen) and evaluated by agarose gel electrophoresis. Sequencing reactions were performed using the BigDye kit (Applied Biosystems) according to the manufacturer’s instructions. Reactions were purified using Centri-Sep Columns (Invitrogen) and sequenced on the ABI Prism 3100 Genetic Analyzer. Basic Local Alignment Search Tool (BLAST) was used to determine the identity of the bacteria from previously determined sequences (GenBank, National Institutes of Health).
Results
Two hundred and thirty-four cats from this population demonstrated severe, atypical pyogenic infections. Abscesses were documented in 142 cats, predominantly in the cervical region, paws, carpal joints and/or tarsal joints. Most individuals with abscesses had either the limbs affected (82 cats) or the head and neck affected (51 cats) (Figure 1). The cervical abscesses developed spontaneously, without any trauma to the region. In several cats, cervical abscesses preceded limb abscesses. Eight samples from these abscesses were submitted for culture but produced no growth, or had scant growth of a skin contaminant. One cat (N-31) with a recurring spontaneously opening draining track in the cervical region did have a positive culture that was reported as ‘group C Streptococcus’ when speciation was requested.

Abscesses of the (a) right carpus and (b) right front paw. (c) Ventral neck wound with submandibular lymph node exposure that eventually healed. (d) Bilateral ventral neck abscesses. Cleaning, debridement and closure with surgical staples was performed (along with administration of systemic antibiotics) owing to the size of the wounds. One wound dehisced. The cat died of septic shock
Twenty-eight cats were identified to have severe septic arthritis, with complete destruction of one or more joints. All but one of the cats had involvement of the carpal and/or tarsal joints (one cat had destruction of the stifle). Based on radiographic appearance and palpation of the affected joints (Figure 2), the articular cartilage, as well as bone, was destroyed, resulting in pathologic dislocation. Again, three samples of purulent material from these regions submitted for culture produced no growth. One sample that came from a cat (O-13) with septic arthritis of both carpal joints did grow a small amount of an organism characterized as α-hemolytic Streptococcus species. Cat O-13 also had wounds of the ventral neck, indicating a previous abscess in the area, and severe pneumonia with diffusely hemorrhagic lungs grossly visible on necropsy (Figure 3a).
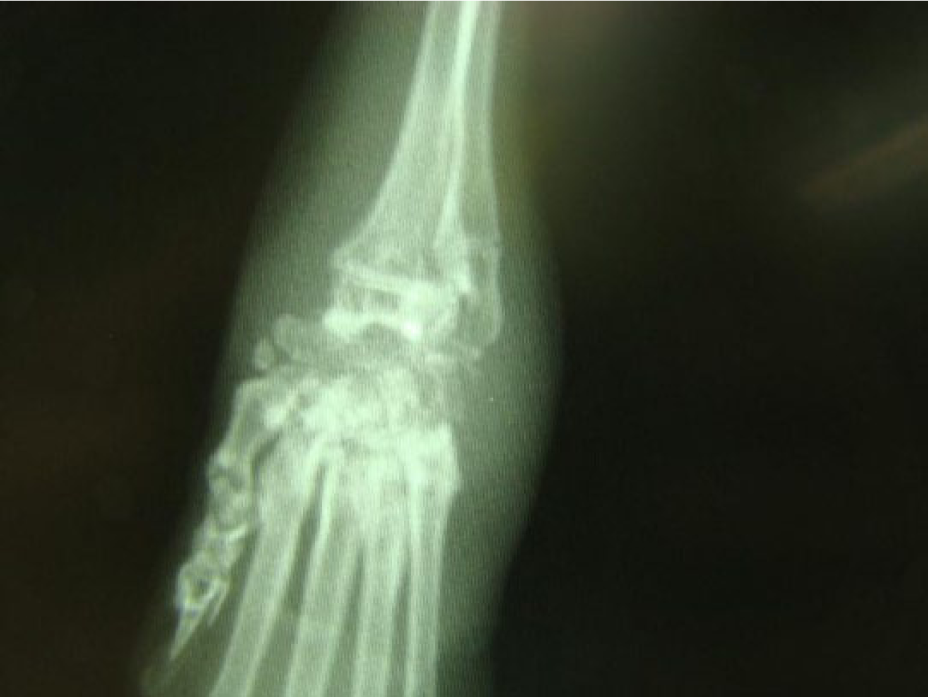
Radiograph of the carpal joint showing severe destruction of the carpal bones secondary to septic arthritis

(a) Lungs of cat O-13 with the histopathological diagnosis of severe subacute necrosuppurative pneumonia. (b) Thoracic cavity of cat L-5 showing purulent discharge and fibrinous adhesions. Histopathology revealed severe neutrophilic pleuritis with atelectasis and bronchopneumonia. (c) Severe purulent ocular discharge. (d,e) Necrotizing fasciitis of the palmar surface of the paw and nasal bridge, respectively. (f) Severe necrotizing fasciitis and myositis of the right lateral thigh
While upper respiratory infections (URIs) were found in the majority of cats (n = 294) and 71 cats were documented to be severely affected, it is difficult to ascertain how many of those cats had streptococcal infections. Cultures were positive for feline calicivirus (FCV) in 24/27 samples from cats suspected to be positive (Dr Kate Hurley Laboratory, University of California Davis), and PCR of pooled samples from the upper respiratory tract secretions were positive for FCV, feline herpesvirus-1, Chlamydophila felis, Bordetella bronchiseptica and Mycoplasma haemofelis (Lucy Whittier Molecular and Diagnostic Core Facility, University of California Davis). It is also difficult to ascertain how many cats also had concurrent lower respiratory disease as diagnostic testing of that number of cats was impractical. Based on clinical signs of dyspnea and increased lung sounds (with crackles sometimes asculted) and necropsy findings, many cats were diagnosed with lower respiratory disease. While some of the lower respiratory disease could have been viral in origin, multiple cats had pneumonia consistent with bacterial etiology. Cat O-13 (mentioned above) with bilateral carpal septic arthritis and wounds in its ventral cervical region, had concurrent severe subacute necrosuppurative pneumonia with multifocal alveolar necrosis with fibrin, cellular debris and a large number of degenerated neutrophils. The histopathology results of other cats with lower respiratory disease did not show severe necrosis but did demonstrate infiltration with neutrophils, with no evidence of viral inclusions. Based on the suppurative nature, bacterial infections were considered by the pathologist to be the most likely cause, although no organisms were seen.
At necropsy, two cats had grossly visible purulent discharge covering the visceral pleura of the lungs and associated pyothorax. During the necropsy of cat L-5, the diaphragm was noted to be bulging into the abdominal cavity. The thoracic cavity was filled with >60 ml purulent discharge and had a large amount of fibrin causing adhesions between the visceral and parietal pleurae (Figure 3b). The purulent discharge submitted for culture produced no growth. Histopathology revealed severe neutrophilic pleuritis with atelectasis and bronchopneumonia, based on expansion of the pleura with dense sheets of neutrophils, severe subpleural lung and bronchiolar infiltration by neutrophils, lymphocytes, plasma cells and macrophages. Alveoli were collapsed and the alveolar walls were expanded by neutrophils and macrophages. All of the changes were consistent with bacterial pleuritis and pneumonia, although no organisms were detected.
Of the severe cases, many of the cats had copious purulent ocular and nasal discharge with the same appearance as the purulent discharge from the abscesses seen in this population (Figure 3c) as compared with the more typical mucopurulent discharge of URIs. Additionally, a unique syndrome was recognized where cats that were previously showing no clinical signs (based on daily observations by trained veterinary technicians) would acutely develop bilateral, purulent nasal discharge. Typically, mucopurulent discharge caused by secondary bacterial infections would occur following serous nasal discharge from virally induced URIs. A primary bacterial infection appeared to cause rhinitis in 68 cats. Several cats had more severe respiratory disease that accompanied or followed the rhinitis, including severe dyspnea secondary to pneumonia.
Fifteen cats presented with recurrent otitis externa characterized by purulent discharge with diplococci seen on cytological examination of the exudate. Three cultures sent to a reference laboratory (Antech Diagnostics, Irvine, CA, USA) showed the presence of BHS. Speciation of the organism from one of the cats was reported as S equi. Unfortunately, speciation was not available for the other samples.
Other invasive syndromes were identified, including septic shock (42 cats) with acute death as a common sequella for many of the cats, necrotizing fasciitis and myositis (11 cats), and otitis and sinusitis leading to meningitis (nine cats). Of the cats with septic shock, 20 also had abscesses. One particular cat, J-86, had a left mandibular abscess on initial examination that resolved with treatment followed by abscesses on its right hindlimb 22 days later with devitalization and degloving of the skin on the limb. Despite treatment, the cat went into septic shock 3 days later but was stabilized. Another abscess developed on the left front paw the following day, a wound spontaneously developed on the bridge of the nose 4 days later and 2 days later a wound appeared on the ventral thorax. The cat was still eating and drinking, but the skin covering the entire ventral thorax sloughed off, even though the underlying tissues showed only petechial hemorrhages and no purulent discharge. Blood from this cat was frozen and analyzed along with the other samples taken from the abscesses.
Another cat, L-17, had what appeared to be mild recurrent bouts of rhinitis characterized by purulent nasal discharge. It was treated with enrofloxacin (Bayer Animal Health) dosed at 5 mg/kg q24h orally for 14 days, azithromycin (5 mg/kg q24h PO for 10 days), clindamycin (20 mg/kg q24h PO for 14 days) and benzathine/procaine penicillin (50,000 IU/kg q48h SC for 14 days) successively during these bouts of rhinitis, and appeared to be clinically normal between bouts. A week after the last rhinitis episode, L-17 became acutely neurologic with seizures, circling and anisocoria. The cat was euthanized because of concerns of meningitis and brain herniation, which were confirmed on necropsy, and purulent material was found covering the brain. Bacteria with the morphological features of Streptococci were seen microscopically, but no growth occurred on culture. Two other cats sent for necropsy to the Ohio State University were also confirmed to have meningitis and brain herniation. In these cats, the cultures did produce bacterial growth that was reported as S zooepidemicus.
While most of the necrotizing fasciitis and myositis cases produced relatively small lesions (Figure 3d,e), one cat, L-11, developed extensive lesions of both lateral thighs extending down the length of the femur that were approximately 6 × 4 cm (Figure 3f). At the time of the rescue the cat was relatively healthy, although underweight with mild nasal discharge, fleas and ear mites. Its mild URI resolved, and the cat was initially stable; however, approximately 1 month later, it developed the severe infections in its hindlimbs. The cat was singly housed, with no possible trauma to the hindlimbs, and no wounds evident until the massive skin necrosis occurred. Treatment with benzathine/procaine penicillin (50,000 IU/kg q48h SC) made no improvement and the cat’s condition deteriorated. Exploration of the wounds after euthanasia showed destruction of tissue extending as deep as the coxofemoral joints, with copious amounts of purulent discharge. The ligaments of the coxofemoral joints were destroyed, and the head of the femur was readily disarticulated from the acetabulum.
The samples representative of the described clinical syndromes, including a blood sample from J-86 and the purulent discharge associated with the necrotizing fasciitis from L-11, the brain of L-17, the carpal joints of O-13 and the thoracic cavity of L-5, all had a PCR product of approximately 375 base pairs (bps), indicating that one bacterial species was common to all of the samples (Figure 4). No other band was common to all samples. Additionally, the 375 bp band was the only visible band in three of the samples, which would be comparable to a single organism growing on culture with conventional microbiological methods. In the samples with additional bands, representing additional species of bacteria, the 375 bp band was comparatively the brightest, indicating the highest quantity of DNA. Sequencing results showed that the bands were 374 bps in length, and evaluation with BLAST indicated that the sequence had a 100% match to the intergenic spacer region of S canis (Genbank: AF324463.1).

Agarose gel electrophoresis. L = 100 base pair ladder; P = positive control; N = negative control; A = cat R-42, purulent ocular discharge; B = cat R-42, purulent nasal discharge; C = cat L-11, purulent discharge from severe necrotizing fasciitis/myositis; D = cat E-40, purulent discharge from spontaneously forming abscess on ventral neck; E = cat B-1, purulent discharge from an open wound on the plantar surface of the right hind paw; F = cat J-86, purulent discharge from the right tarsal joint; G = cat O-13, purulent discharge from the right carpal joint; H = cat L-17, purulent discharge from the brain postmeningitis and herniation
Discussion
Highly pathogenic streptococcal infections have not been widely reported in cats, and the few reports available almost always involve facilities housing multiple cats. This report also illustrates this trend but differs in that this is the largest population of cats exhibiting signs of BHS infections and is the only population in which the cats demonstrated all of the clinical signs that have been attributed to these types of infections in previous reports. Additionally, the circumstances of the situation allowed the veterinary medical team to evaluate the cats over an extended period of time on a continual basis within a closed facility (no additional animals came into the shelter). Because the primary concern of the team was to treat the patients and restore them to health, this study occurred after the population was stable and therefore samples sent to the reference laboratory were no longer available for further examination and speciation. Attempts to grow BHS from frozen tissues have not yet produced viable organisms for study. However, DNA of the various pathogens affecting the population was well preserved by freezing the samples, making the molecular detection of the pathogens the ideal way to make a diagnosis.
All of the frozen samples from the pyogenic clinical syndromes had just one bacterial species in common, S canis. Additionally, three of the samples had S canis as the only bacterial species present in the purulent discharge (ie, the PCR equivalent of a ‘pure culture’). These results, combined with the previously described streptococcal syndromes, strongly suggest S canis as the cause of the infections. Based on previous studies, 26 S canis-infected discharge is infectious to naive cats, either by direct or indirect contact, and can cause systemic disease after being introduced via the oral cavity. The oral cavity as a portal of entry is logical with the observations in this study, as no wounds preceded the infection of the cervical lymph nodes/cervical region abscesses, facial abscesses or limb abscesses. Hematogenous spread to these regions was further supported by the presence of S canis in the blood sample taken from cat J-86.
It is unclear why the septic arthritis with severe joint destruction was predisposed to affecting the carpal and tarsal joints. The one previous report of S canis septic arthritis in cats did not demonstrate this pattern. 29 It is possible that these joints were affected owing to a concurrent infection that demonstrated affinity to the carpal and tarsal joints, although more data are needed to support this hypothesis.
The episodes of rhinitis, where cats that were previously showing no signs of illness presented acutely with large amounts of purulent nasal discharge, may have been due to recrudescence of the infections or due to exposure, or possibly re-exposure, via direct or indirect contact with S canis. Recrudescence of S canis infections has been documented previously.25,27 Strict sanitation measures were enforced to minimize spreading of diseases; however, cats that appeared to have had no signs of active infectious disease for at least 4 weeks were moved to multiple cat housing (‘catteries’) to allow them more environmental enrichment. It is possible that one or more of the in-contact cats was still harboring S canis. Another observation that may support new exposure vs recrudescence of infection is that the timeframe where the cases of rhinitis were observed was rather narrow (2–4 months after rescue), which is more supportive of a small ‘outbreak’ of infection. However, not all cats in a particular ‘cattery’ developed rhinitis, and cases were interspersed throughout the shelter without a discernible pattern. Also, there were multiple cats that had multiple bouts of rhinitis, with the average time between episodes (with no clinical signs) of 2 months. Based on that timeframe, it is possible that the ‘outbreak’ of rhinitis beginning in May could have reflected recrudescence of infections that were not evident when the cats were first examined in March.
The majority of cats that presented with rhinitis (80%) did not have a history of cervical, facial or joint abscesses, and no cats developed those syndromes following rhinitis. This may indicate a different portal of entry for the pathogen, more than one strain of S canis or even different species of BHS, each with different tissue predilection, different virulence factors and/or different antibiotic susceptibility (such that treatment with antibiotics eradicated the ‘abscess-producing’ strain). It could also be that the same strain of S canis was located in an anatomical region that was less accessible to antibiotics and/or less likely to spread hematogenously.
Multiple cats with rhinitis had extension of the infection to the middle and external ear (usually only one ear was affected), which was evident by the presence of a head tilt to the side of the infection, ataxia and purulent discharge from the external ear canal. The ear infections tended to be chronic and recurrent for a period of time (2–18 weeks, mean 7.5 weeks) before cats became clinically normal; however, some infections resulted in a permanent head tilt. Several cats in the population had even more severe extension of the infection from the nasal cavity, through the cribriform plate and into the brain, resulting in marked suppurative meningitis and encephalitis, with acute cerebellar herniation. The exudate from two of these cats was cultured in-house at the Ohio State University following necropsies, and cytology was consistent with streptococcal meningitis. Based on culture followed by biochemical testing, the streptococcal species was determined to be S zooepidemicus. A sample of purulent exudate from the brain of another cat was determined by molecular means to be S canis.
Interestingly, all five of the samples from the lesions associated with BHS infections that were evaluated by the outside microbiology laboratories via culture were characterized as group C BHS, whereas the molecular methods consistently identified S canis, a Lancefield group G organism (including one sample that was categorized as group C by culture). Preliminary work with additional samples from cats found deceased in freezers at the IHF at the time of the raid and cats euthanized immediately owing to the severity of their conditions (ie, cats that had received no antibiotic treatment) have shown the presence of two additional streptococcal species, S zooepidemicus (Lancefield group C) and Streptococcus porcinus (Lancefield groups E, P, V, U, NG1 [A1, C1], NG2 and NG3 33 ). It is possible that the laboratories were actually growing S zooepidemicus but this is not consistent with the molecular findings. Alternatively, the presence of these other streptococcal species in the population provides a scenario that may have promoted horizontal gene transfer between streptococcal organisms that could affect the Lancefield antigens. Horizontal gene transfer could also be responsible for the increased pathogenicity of these infections, as multiple streptococcal species could exchange virulence factors to produce a more virulent strain. 34
As nearly all of the cats had some type of bacterial infection, the majority were treated early on with long-acting penicillin injections. It could be that the infections caused by S zooepidemicus and S porcinus were not found in the samples of the treated cats because they were susceptible to penicillin. While there is no substantial evidence to support this, one of the samples that was found by molecular testing to have S canis was found to be resistant to methicillin by the reference laboratory. Additionally, many of the samples of purulent material sent to the reference laboratory that came from the lesions consistent with the S canis infection produced no growth, and the only drug that worked to treat these infections was doxycycline. This could be due to the S canis transforming into an L-form, or cell wall-deficient (CWD) bacteria. Group A streptococci have demonstrated this tendency, and CWD streptococci have been documented on multiple occasions in the blood of humans with rheumatic fever. 35 CWD streptococci could also account for the observed recrudescence of infections, where the CWD forms persist without causing disease and later revert to the virulent wall-containing form. The revertant bacteria may have biochemically dissimilar characteristics to the parent bacteria and even among morphologically identical revertant colonies, 36 which is another potential reason for the discrepancies between the culture results and the molecular results. The α-hemolytic Streptoccocus that was cultured from the joint of cat O-13 could very well represent a revertant S canis L-variant, as streptococci capable of β−hemolysis in their parent form have been shown to produce only α-hemolysis after reverting back from an L-variant form. 37 Perhaps more support for the CWD bacteria argument is the complete lack of visualization of any microorganisms on histopathology in specimens that were reported as having all of the characteristics of bacterial bronchopneumonia and pyothorax. Dense sheets of neutrophils were seen covering the lungs of cat L-5, along with severe infiltration with neutrophils within the lungs themselves. What is striking is that there was no visualization of any bacteria mentioned in the report, although it was stated to be a ‘pattern of severe inflammation consistent with a bacterial pleuritis and pneumonia’. L-5 was not being treated with antibiotics at the time of death, as it was not exhibiting outward signs of the infection prior to that time.
Wu et al described the pathology of two cats with group G BHS infections that lead to pyothorax and bronchopneumonia, 28 and they described ‘severe, acute, diffuse, fibrinous pleuritis’ with the pulmonary pleura ‘thickened by fibrin admixed with basophilic nuclear debris and colonies of cocci’. Cocci were also seen in the septic exudate and S canis grew on culture. However, while the pathology was similar, cocci were not seen in the purulent material from L-5, and the culture produced no growth.
Several other cats in the population had severe subacute necrosuppurative pneumonia stated to have a bacterial etiology but with no bacteria visible in the sections. The pathology described by Blum et al in a S zooepidemicus outbreak was nearly identical to the cats in this current study; 31 however, cocci were seen within the lungs along with the neutrophils. Interestingly, they did find one strain of S zooepidemicus that proved to be difficult to culture, requiring anerobic conditions followed by a 5% carbon dioxide environment.
Conclusions
While much is still to be learned about streptococcal infections in cats, it is clear that these organisms are capable of producing severe, life-threating infections that can quickly spread throughout a population. Typically, outbreaks of infections that spread rapidly and result in severe pathology would be associated with a novel pathogen in a susceptible population. All of the described ‘streptococcal syndromes’ in the literature were associated with cats in large groups. Additionally, in this study, as well as several of the others, cats from various regions (in this case at least 12 different states) were brought together to form not only a new population of cats, but also a new population of their associated microorganisms.
When stress and a less than optimal immune response is added to this, an environment exists that promotes co-infections with pathogens capable of horizontal gene transmission. When virulence factors get exchanged, novel pathogens can result. In this study, all of the streptococcal syndromes previously described in cats (ie, cervical lymphadenitis and abscessation, abscesses of the limbs and distal joints, necrotizing fasciitis and myositis, rhinitis, purulent otitis, meningitis, pneumonia and pyothorax) were seen in this population. This suggests a high number of virulence factors within the merged bacterial population. With the identification of S zooepidemicus in lung tissue and S porcinus in two abscesses of untreated cats from the IHF, an exchange of streptococcal virulence factors becomes even more likely.
Striking similarities between streptococcal syndromes in the various host species are evident – septic arthritis in humans, horses and cats; rhinitis in horses, dogs and cats; necrotizing fasciitis in humans, cats and dogs; and pneumonia in humans, horses, dogs and cats. What is also becoming evident is that a particular streptococcal syndrome is not necessarily always caused by the same species of BHS. Species that were once considered to be non-human animal pathogens have crossed the line. 37 A ‘one health’ approach is needed to increase our understanding of streptococcal diseases and the ever-evolving world of pathogens.
Footnotes
Acknowledgements
We would like to thank Drs Ellie Hirshberg, Melinda Merck, Robert Reisman and Michelle Honse for their assistance in gathering the initial physical examination data; Dr Randall Fertelmes for assisting with sample analysis and providing supplies for the cats; Dr Kate Hurley, Dr Patricia Pesavento, and Michael Bannasch for Calicivirus isolation; Dr Steven Weisbrode for post-mortem examination of several cats; the students in the Duquesne University Superlab course for their contributions to the laboratory work; Shanna Best and Diane Goldbloom for caring for the cats, keeping meticulous records and collecting samples for analysis; and Dr Danielle Gunn-Moore for providing detailed edits that greatly enhanced the paper.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
