Abstract
Objectives
Feline intestinal mast cell tumours (FIMCTs) are rare and reportedly characterised by poor differentiation, aggressive biological behaviour and lack of reliable therapeutic aids. KIT proto-oncogene-activating mutations have never been investigated in these tumours. This study describes the main clinicopathological and microscopic features observed in 17 FIMCTs.
Methods
Tumour degree of differentiation, proliferative activity, Kit protein expression and KIT mutations were evaluated and correlated with survival to assess their prognostic relevance.
Results
Ten tumours were located in the small intestine, two in the ileocaecocolic junction, and five in the large intestine. Survival times ranged from 3–538 days. Fifteen tumours were evaluated histologically, and there were six well-differentiated, six moderately differentiated and three poorly differentiated FIMCTs. The last showed a medium-to-large deposition of collagen tissue (P <0.001), and significantly higher mitotic and Ki67 indexes compared with more differentiated tumours (P = 0.011). On survival analysis, tumour degree of differentiation (P <0.001) and a mitotic index >2 (P = 0.022) were significantly associated with decreased survival times. Twelve cases showed Kit protein immunoexpression. The Kit pattern was membranous in five cases (33.3%), focal paranuclear in five (33.3%) and diffuse cytoplasmic in two (13.3%). Cytoplasmic Kit patterns were associated with a lesser differentiation (P = 0.015). Mutation analysis was successfully performed on 12 primary tumours and four lymph node metastases; however, no encoding mutation was detected.
Conclusions and relevance
Contrary to reports in the literature, FIMCTs seem to have an extremely variable biological behaviour. We propose a classification based on tumour degree of differentiation and proliferative activity. These findings need to be confirmed in larger series, and exploration of further genomic regions of KIT is warranted to clarify its role in the development and progression of these neoplasms.
Introduction
Globally, mast cell tumours (MCTs) account for 2–15% of feline neoplasms, and are classified anatomically into cutaneous, splenic and intestinal forms.1,2
While cutaneous and splenic MCTs bear many histological and behavioural similarities, including ‘typical’ morphology, abundant cytoplasmic granulation, generally low malignancy and potential concomitant occurrence, primary alimentary tract MCTs show strikingly distinctive features.2,3 Little information is available about these neoplasms in the veterinary literature and they are rarely encountered in clinical practice, although they are considered the third most frequent intestinal tumour in cats.2–5 They may present as either a single nodule or multifocal lesions, and most commonly affect the distal small intestine or colon. 6 Clinical signs are aspecific, and include inappetence, vomiting, weight loss, depression and abdominal distension. Metastases are extremely frequent and predominantly involve mesenteric lymph nodes and liver. 2 Surgery currently represents the standard of care but postsurgical prognosis is poor. This is especially true for the sclerosing variant of feline intestinal MCTs, with survival times rarely exceeding 2 months.1,7 Histologically, these tumours show highly atypical features, including spindle/polygonal cell morphology and faint cytoplasmic granulation; this may complicate differential diagnosis from other intestinal diseases (lymphoma, neuroendocrine carcinoma, sarcomas, sclerosing enteropathy).2,7
The KIT proto-oncogene encodes the receptor tyrosine kinase kit, which has been shown to regulate normal mast cell survival, proliferation, differentiation and migration.8,9 In canine MCTs, juxtamembrane domain KIT mutations and aberrant cytoplasmic Kit protein localisation have been associated with increased cellular proliferation and with both reduced progression-free and overall survival rates.10,11 As a consequence, Kit receptor tyrosine kinase inhibitors (TKIs) have entered as an active part of MCT treatment in dogs.12,13 Aberrant cytoplasmic Kit immunohistochemical expression has been reported in 29–67% of feline cutaneous MCTs, and a higher frequency was observed in tumours with unfavourable outcome.14–16 KIT proto-oncogene mutations in exons 6, 8, 9 and 11 have also been detected in feline cutaneous and splenic MCTs, and there are anecdotal reports of beneficial response to molecular-targeted therapy.17,18 To date, Kit receptor dysregulations have never been investigated in feline intestinal MCTs. A thorough understanding of the role that Kit plays in the development of feline intestinal MCTs may provide help in the clinical approach to and treatment of this disease.
This study reports the main clinicopathological features observed in a series of primary intestinal feline MCTs. For each case, proliferative activity and Kit dysregulations (ie, cytoplasmic immunohistochemical expression and KIT mutations) were further investigated.
Materials and Methods
Inclusion criteria and histological parameters
Formalin-fixed and paraffin-embedded (FFPE) surgical or biopsy samples of feline primary intestinal MCT (FIMCT) were retrieved from the archives of the Department of Veterinary Medical Sciences (University of Bologna, Italy) and IDEXX Laboratory Alfort (Alfortville, France). Histologic sections (4 μm) stained with haematoxylin and eosin were reviewed, and a metachromatic stain (toluidine blue) was applied in all cases to confirm the diagnosis of MCT and to evaluate the amount of granules in the neoplastic cells. Masson’s trichrome was used to highlight the presence of dense stromal collagen (sclerosis) within the neoplastic mass.
The proliferation marker Ki67 (MIB-1 antibody) and CD117 immunohistochemistry (IHC) were applied to evaluate tumour proliferative activity and Kit protein expression pattern, respectively.
Table 1 summarises the quantitative and qualitative histological parameters evaluated for each included case.
Histological parameters evaluated in 15 feline intestinal mast cell tumours

Mouse monoclonal
Rabbit polyclonal
HE = haematoxylin and eosin; IHC = immunohistochemistry; HPF = high-power field
Immunohistochemical analysis
Tumour sections were immunolabelled for Ki67 and Kit protein by using commercial antihuman antibodies (MIB-1 [mouse monoclonal] and CD117 [rabbit polyclonal]; Dako), with validated reactivity in feline tissues. 15
Endogenous peroxidase activity was blocked by incubation for 30 mins with 0.3% hydrogen peroxide in methanol (MIB-1) or 3% hydrogen peroxide in distilled water (CD117). For antigen retrieval, slides were microwaved in citrate buffer (pH 6.0), for two (CD117) or four (MIB-1) cycles of 5 mins, at 750 W. Sections were incubated overnight at 4°C in a humid chamber with the primary antibody; MIB-1 was diluted 1:600 in a blocking solution (10% goat serum in phosphate-buffered saline [PBS]); CD117 was diluted 1:100 in PBS.
Sites of primary antibody binding were identified using a commercial streptavidin–biotin–peroxidase kit (LSAB; Dako) and 3,3′-diaminobenzidine was used as chromogen. Sections were counterstained with Papanicolaou’s haematoxylin, dehydrated and mounted.
Feline intestinal mucosa was used as an internal positive control for MIB-1. For CD117, a feline cutaneous MCT was used as positive control. Negative controls were obtained by substituting the primary antibody with an unrelated serum.
Table 1 summarises the methods for the evaluation of immunopositivity.
Mutation analysis
Genomic DNA was purified from 10 µm sections (n = 3) of FFPE tumour samples by using a commercial kit (AllPrep DNA/RNA FFPE kit; Qiagen) following the manufacturer’s instructions. The extracted DNA concentration and purity were measured using Nanodrop ND-1000 spectrophotometer.
The entire sequence of exons 8, 9, 11 and 17 of feline KIT were amplified by PCR using the oligonucleotides already described. 17 Furthermore, for exons 8 and 9, whenever the full-length amplicon could not be obtained using those primers, the target was amplified using different combinations of the same outer and novel inner primers, as schematically indicated in Figure 1. Detailed primer sequences are given in Table 2.

Schematic representation of the strategy used for feline KIT exons 8 and 9 amplification. The position of outer and inner primers used and the length of the different PCR products obtained are reported
Primers used for the amplification of exons 8, 9, 11 and 17 of feline KIT

Two microlitres of diluted DNA (up to 50 ng/µl) were used as a template for the PCR amplification. Reactions were carried out in a T-Personal thermocycler (Biometra GmbH) by using 0.5 µM of each primer (Tema Ricerca), 0.02 U/µl of Q5 High Fidelity DNA polymerase (EuroClone), 200 μM deoxyribonucleotide triphosphate mix and 1× Q5 buffer (final concentrations). The following PCR conditions were used: an activation step at 98°C for 45 s; 35 cycles of 15 s at 98°C; 15 s (exons 8 and 9 shorter fragments) or 30 s (full-length amplicons) at 56°C (full-length exons 9, 11, 17) or 58°C (for all the remaining amplicons); 20 or 40 s at 72°C (for exons 8 and 9 shorter fragments and full-length amplicons, respectively); and a final extension step of 2 mins at 72°C. Amplicons were visualised in a 1% agarose gel electrophoresis. All the PCR products were sequenced on an automated sequencer by BMR Genomics (Padua, Italy) and Macrogen Europe (Amsterdam, the Netherlands). Amplicons were sequenced starting from both forward and reverse primers used for the PCR amplification and from at least two different PCR products.
Obtained electropherograms were analysed by the FinchTV tool. The alignment of the wild-type sequence available in the database Ensembl Genome Browser with sequences obtained in this study was performed using Multalin interface page (http://multalin.toulouse.inra.fr/multalin/).
Medical records
Patient records were reviewed to collect signalment and tumour location.
Referring veterinarians were contacted for additional information, including history, clinical stage, laboratory findings, ultrasound/surgical findings, treatment details and outcome.
The availability of these data was not strictly considered among the inclusion criteria.
Statistical analysis
Data were analysed using a commercial software program (SPSS Statistics v 19; IBM); P values ⩽0.05 were considered significant.
When appropriate, data sets were tested for normality by use of the D’Agostino and Pearson omnibus normality test. Values were expressed as mean ± SD in cases of normal distribution, or as median with a range in cases of non-normal distribution. Correlation between mitotic index (MI) and Ki67 index was evaluated by means of the Spearman’s rank correlation coefficient. Differences in qualitative and quantitative variables according to the degree of differentiation of the MCTs were assessed by Fisher’s exact test and analysis of variance, respectively.
Overall survival was defined as the time (days) from the date of diagnosis to the last reported date that the patient was seen alive. The patient status was recorded as alive, dead because of non-MCT-related causes or dead because of MCT-related causes.
The log-rank test was used to compare survival distributions. Variables analysed for prognostic significance were tumour degree of differentiation, MI, Ki67 index and Kit pattern. For MI and Ki67 index, the median was used as the cut-off value.
Results
Medical records
Seventeen cats (aged 7–15 years [mean 11.8 ± 2.5 years)] with a histologically confirmed intestinal MCT were included in the study (Table 3). Breeds included domestic shorthair (n = 15) and Persian (n = 2). There were eight males (47.1%) and nine females (52.9%). Vomiting, dysorexia and abdominal pain were the most common clinical signs at presentation. A palpable abdominal mass was present in three cases.
Signalment, location, histology and clinical course of 17 cases of feline intestinal mast cell tumours (FIMCTs)

MI = mitotic index; NA = not available; DSH = domestic shorthair; M = male; F = female
Peripheral hypereosinophilia was detected in three cats. Mastocytemia was not referred. On ultrasound/gross examination, all tumours presented as a non-circumeferential, eccentric wall thickening. Ten tumours (58.8%) were located in the small intestine, two (11.8%) were in the ileocaecocolic junction and five (29.4%) were in the large intestine.
At presentation, five patients had histologically confirmed metastases to the regional lymph nodes and one had liver metastases.
Two cats did not receive any therapy. Three were treated with a TKI (masitinib, n = 2; toceranib, n = 1); another three cats were treated with corticosteroids. Surgical excision was performed in nine cases; among them, five had non-infiltrated margins, one had infiltrated margins and in three cases margins were not evaluated histologically.
Follow-up information was available in 13 cases. Of the cats receiving surgery, four died within 1 month for postoperative complications. One cat was lost to follow-up after 90 days, one was deceased at 402 days and another cat was alive after 472 days. In the cats treated only with TKIs, survival times were 538 days with masitinib and 488 with toceranib. The cats treated exclusively with corticosteroids had a median survival time of 400 days. The two cats receiving no treatment survived 285 and 372 days. Overall, six cats survived >1 year.
Histological examination
Histological samples were available for further examination in 15/17 cases (Table 3). Nine surgical samples and six laparotomy biopsies were examined.
A complete section of the intestinal wall was available in 12 cases. The majority of tumour cells were seen in the submucosa layer (eight cases; 66.7%); in three cases (25.0%) cells were almost equally distributed within the submucosa and the muscularis; whereas in one case (8.3%) tumour cells were predominantly located in the muscularis layer. Serosa was secondarily involved in nine cases (75.0%). In six cases (50.0%) there was minimal mucosa infiltration. Mucosal ulceration was never observed.
Six tumours (40.0%) were classified as well-differentiated FIMCTs and were composed by sheets and nests of round-to-oval cells, 10–15 µm in diameter, with distinct cell borders. Cytoplasms were moderate to abundant, pale eosinophilic, occasionally showing eosinophilic granules. Nuclei were round to oval, with condensed chromatin and inconspicuous nucleoli. The size of cells and nuclei within the neoplasm was uniform (variations <50%) (Figure 2).
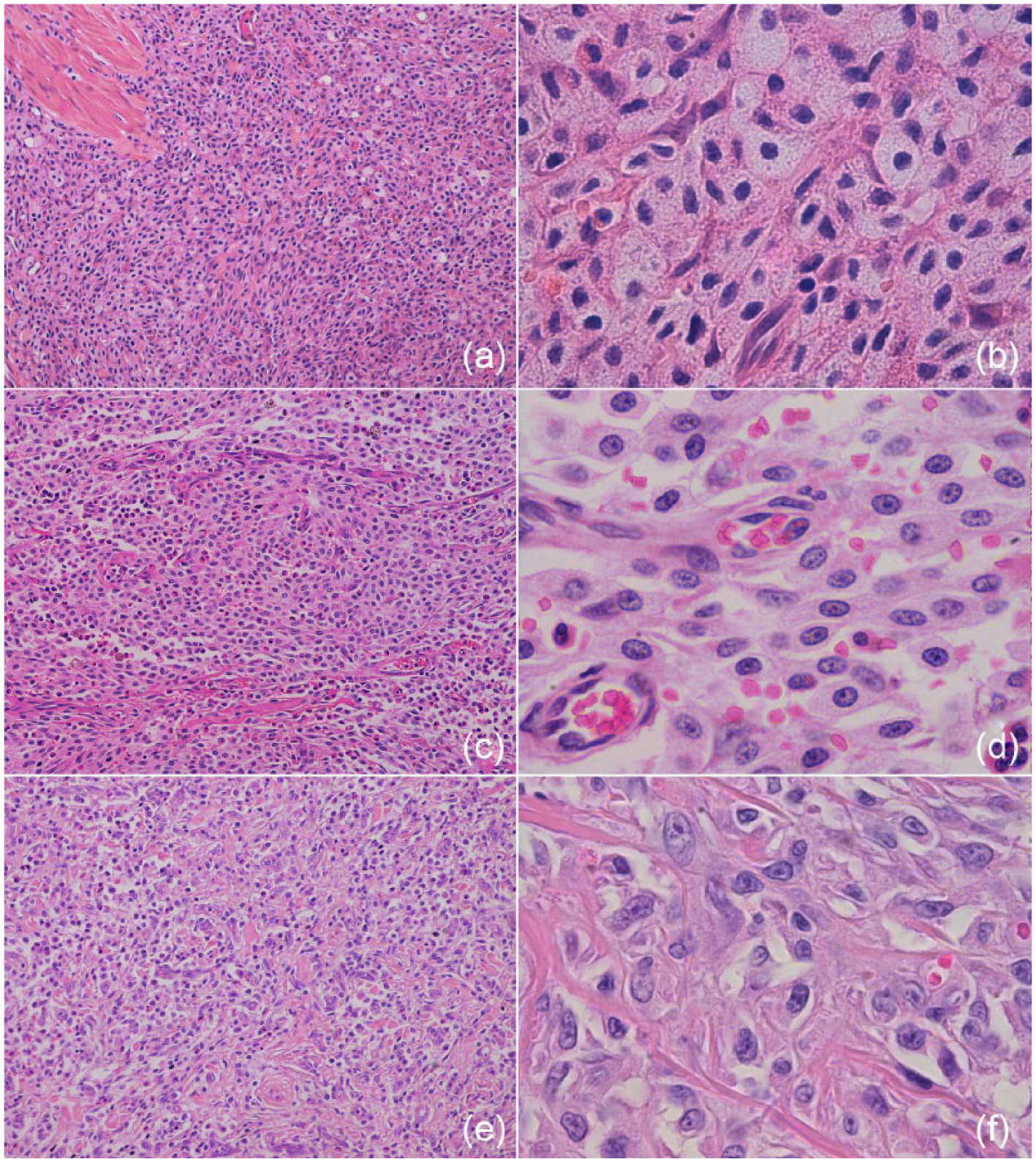
(a,b) Well-differentiated intestinal mast cell tumour. Sheets and nests of round-to-oval cells, 10–15 µm in diameter, with distinct cell borders. Cytoplasm is moderate to abundant, occasionally showing eosinophilic granules. Nuclei are round to oval, with condensed chromatin and inconspicuous nucleoli. Anisocytosis and anisokaryosis are low. Haematoxylin and eosin, (a) × 200 and (b) × 1000. (c,d) Moderately differentiated intestinal mast cell tumour. Sheets and streams of oval-to-spindle-shaped cells, 10–20 µm in diameter, with mostly distinct cell borders and moderate eosinophilic cytoplasm. Nuclei are oval shaped, with finely stippled chromatin and a small single central nucleolus visible in most cells. Anisocytosis and anisokaryosis are low to moderate. Haematoxylin and eosin (c) × 200 and (d) × 1000. (e,f) Poorly differentiated intestinal mast cell tumour. Pleomorphic, polygonal-to-spindle-shaped cells, arranged in small cords and bundles interspersed in abundant collagenous stroma. Cells are 20–40 µm in diameter, with indistinct borders and little, amphophilic cytoplasm. Nuclei are large, elongated and vesicular, with prominent nucleoli. Anisocytosis and anisokaryosis are marked. Haematoxylin and eosin (e) × 200 and (f) × 1000
Six tumours (40.0%) were classified as moderately differentiated FIMCTs and characterised by sheets and streams of oval-to-spindle-shaped cells, 10–20 μm in diameter, with mostly distinct cell borders and moderate eosinophilic cytoplasm. Nuclei were oval shaped, with finely stippled chromatin and a small single central nucleolus visible in most cells. A variation of 50–100% in the size of cells and nuclei was present (Figure 2). Three tumours (20.0%), classified as poorly differentiated FIMCTs, were composed of areas of pleomorphic, polygonal-to-spindle-shaped cells, arranged in small cords and bundles interspersed in abundant collagenous stroma. Cells were 20–40 µm in diameter, with indistinct borders and small, amphophilic cytoplasm. Nuclei were large, elongated and vesicular, occasionally horseshoe-shaped or bizarre, with prominent nucleoli. A variation of cellular and nuclear size of >100% was present. These cells resembled, in their architectural pattern and morphology, histiocytic cells, activated fibroblasts or sarcoma cells, and metachromatic stains allowed identification of them as mast cells (Figure 2).
The density of cytoplasmic granules was low in four cases (26.7%), moderate in nine (53.3%) and high in three (20.0%). There was no statistical relationship between the degree of tumour differentiation and the amount of cytoplasmic granules. Median MI was 2 (range 0–12). MI was significantly different among well-differentiated (median 0), moderately differentiated (median 2) and poorly differentiated (median 6) FIMCTs (P = 0.011). Eosinophilic infiltrate was absent to occasional in six cases (40.0%), moderate in four (26.7%) and abundant in five (33.3%). Presence of sparse lymphoid aggregates was observed in five cases (33.3%). Moderate-to-high amounts of sclerosis were recorded exclusively in the three cases classified as poorly differentiated (P <0.001). Necrosis was seen in one case.
On survival analysis, poorly differentiated FIMCTs (P <0.001) and a MI >2 (P = 0.012) were significantly associated with reduced survival times.
Ki67 index
Median Ki67 index was 5.03% (range 1.07–18.06%). Ki67 index was significantly correlated with MI (R = 0.839; P <0.001). A higher Ki67 index was observed in poorly differentiated FIMC (median 14.48%; P = 0.007). There was no statistical relationship between Ki67 index and survival.
Kit pattern
Five tumours (33.3%) had a membranous expression of Kit (Kit pattern 1), five (33.3%) showed a focal paranuclear expression (Kit pattern 2) and two (13.3%) had a diffuse cytoplasmic expression (Kit pattern 3; Figure 3). In three cases the immunoreactivity was too faint to be evaluated. All the moderately-to-poorly differentiated FIMCTs showed Kit pattern 2 or 3 (P = 0.015). There was no significant relationship between Kit pattern and survival.

Intestinal mast cell tumour. (a) Faint membranous positivity for Kit protein (Kit pattern 1). (b) Focal paranuclear positivity (Kit pattern 2). (c) Diffuse cytoplasmic positivity (Kit pattern 3). Kit immunostaining. Haematoxylin counterstain (× 1000)
Mutation analysis
Mutations analysis was carried out on all samples. Exons 8, 9, 11 and 17 were successfully amplified in 12 primary tumours and in four metastatic lymph nodes.
No mutation resulting in amino acid changes in the protein were found. Only silent single nucleotide polymorphisms were detected in exon 9 (n = 2) and in exon 17 (n = 7).
Discussion
Demographic data were fairly consistent with published information. All cases were diagnosed in cats aged 7–15 years, with no breed or sex predisposition, and tumours were most commonly located in the small bowel.2,4,5
The most frequently described paraneoplastic syndrome associated with cutaneous and splenic MCTs in cats is gastrointestinal ulceration related to histamine release.2,5 This lesion is thought to be the result of vasoconstriction caused by the release of histamine from neoplastic mast cells. Interestingly, ulceration did not occur in any of the intestinal MCTs in this or in other studies, suggesting that intestinal neoplastic mast cells may either be deficient in or entirely lacking vasoactive substances. 5 This is not surprising, as there is morphological and histochemical evidence that mast cells in the intestinal tract differ from those in other locations, and that they synthesise and store mediators qualitatively and quantitatively different from those of mast cells at other sites. 5 This parallels the relatively lower number of cytoplasmic granules encountered in FIMCTs compared with cutaneous and splenic MCTs, which, in some cases, contributes to hindering the differential diagnosis from other intestinal neoplasms. 4
Eosinophils are uncommonly found in high numbers in feline cutaneous and splenic MCTs, while being more frequent in FIMCTs, both as tumour infiltrates or in circulating blood. Mast cells synthesise and release eosinophilic chemotactic factor and other substances, such as interleukin (IL)-3, IL-5 and granulocyte macrophage colony-stimulating factor, which attract eosinophils to the site of inflammation. Eosinophils inactivate mediators released from mast cell granules and function to modulate hypersensitivity reactions associated with degranulation of mast cells.5,19,20 The interaction of mediators contained within mast cells and eosinophils can cause a vicious cycle of tissue destruction and extensive proliferation of both mast cells and eosinophils. Additionally, the release of fibrogenic cytokines like fibroblast growth factor and transforming growth factor β by mast cells and/or eosinophils suggests a possible mechanism of mast cell-induced sclerosis. In the present study, three tumours characterised by high eosinophilic infiltrate also showed a variable amount of interstitial fibrosis. All of them were classified as poorly differentiated FIMCTs and had histological features that were strikingly similar to those reported in a recent paper by Halsey et al, 7 describing the so-called ‘sclerosing’ variant of feline intestinal MCT.
According to other authors, the observed spindle cells prevailing in these lesions are morphologically consistent with fibroblasts and not with neoplastic mast cells, warranting a diagnosis of feline gastrointestinal eosinophilic sclerosing fibroplasia. In fact, spindle cells are positive for vimentin and smooth muscle actin, indicating myofibroblastic differentiation.20,21
Lesions densely populated by mast cells and eosinophils are common in both feline mast cell neoplasia and eosinophilic enteritis.7,20 These mixed lesions are extremely hard to differentiate. According to our experience, IHC with mesenchymal markers should not be used to aid differential diagnosis, as even the more typical forms of FIMCT are usually positive for vimentin, and smooth muscle actin-positive cells may represent remnants of the intestinal muscularis cells. Anti-mast cell-specific tryptase antibody and metachromatic stains should rather be used to estimate the proportion of mast cells within the lesion. Indeed, while not all of the mast cells may show a high amount of cytoplasmic granules, it is usually possible to recognise a progression from well-differentiated intestinal mast cells to more elongated cells, and a small amount of granules is almost always discernible in oil immersion, even in lesser differentiated cells, which are morphologically identical to fibroblasts. Additionally, in all of the three cases of poorly differentiated FIMCTs identified in this study, there was histologically confirmed metastasis to regional lymph nodes, characterised by effacement and replacement of normal tissue architecture with features similar to those of the primary tumour, and survival times were significantly shorter. This is in agreement with the paper by Halsey et al, 7 which reports a more malignant biological behaviour of these tumours. Notably, a histiocytic (atypical, spindeloid) variant of feline mast tumour is also reported in the skin. In a previous study, we observed a generally worse prognosis of this variant, which presents more typically as single or multiple subcutaneous masses. 18 Further research to evaluate similarities in morphological, biochemical and functional characteristics between these two forms is recommended.
FIMCTs are regarded to be very aggressive, despite a generally low MI.3,7 In this study, a generally low mitotic activity was confirmed. In order to avoid underestimation, IHC for the cell proliferation marker Ki67 was also carried out, and the Ki67 index showed a high degree of correlation with MI. The limited mitotic activity of MCT is well acknowledged in both feline and canine species. However, this study demonstrated a correlation between mitotic activity, tumour differentiation and survival, suggesting that MI may actually represent a prognostic indicator, as previously demonstrated for feline cutaneous MCT. 18
The results of survival analysis may, indeed, have been affected by the poor statistical power, as the total number of cats in this study is very low and treatment was extremely heterogeneous. Nevertheless, this is inevitable when dealing with an uncommon tumour type. An objective fact is that, in seven of the cats in this study, survival times exceeded 1 year. These patients were treated with surgery alone, or corticosteroids, or they did not receive therapy at all. This is quite surprising, as the literature reports a rapid progression of these neoplasms, with survival times generally <4 months.
In the present study, a clear correlation was not found between Kit protein expression and tumour biological behaviour, although a cytoplasmic pattern was most frequently associated with lesser differentiated tumours. Halsey et al also noted a similar membrane distribution in cells of round morphology, with an increasing cytoplasmic distribution in cells of polygonal-to-spindle morphology. 7 No apparent correlation was found between Kit pattern and mutations, as all the tested samples were wild type. Cats treated with TKIs showed good survival times; however, these were not different from those of cats receiving corticosteroids. In a previous study, a statistically significant association between KIT mutations and Kit pattern was not demonstrated for feline cutaneous MCTs, although a tendency was observed (P = 0.066). 18 The absence of an association between Kit expression, protein localisation and mutations has also been observed in canine MCTs, and it has been hypothesised that they may represent three independent events.10,22
Considering the role of Kit receptor in the development and progression of other forms of canine and feline MCT, it seems unlikely that it may be completely irrelevant in FIMCTs. Possibly, mutations in this study may have been underestimated owing to the higher prevalence of patrimonial cells in the intestinal sections compared with the cutaneous nodules; or, alternatively, mutations in FIMCTs are located at other sites. Furthermore, the extraction of nucleic acids from canine FFPE blocks remains problematical. Owing to the time of fixation in formalin and the persistence of inhibitors in the extract following DNA isolation (ie, paraffin, xylene, ethanol), which could interfere with the success of PCR amplification, the extracted DNA is often degraded and fragmented.
Conclusions
Contrary to reports in the literature, FIMCTs seem to have a variable biological behaviour; additionally, proliferative activity and the degree of differentiation appear to be relevant in assessing tumour behaviour. To confirm these findings and clarify the role of Kit in feline intestinal MCTs, additional studies must be performed on a larger number of cases and include a mutation analysis of the whole nucleotide sequence of KIT, preferentially starting from surgical biopsies stored in RNAlater solution.
Footnotes
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
