Abstract
A 7-year-old domestic shorthair cat was evaluated for hyporexia and weight loss following endoscopic placement of an intragastric pH monitoring device. Physical examination of the cat was unremarkable, and its blood work was notable for a mild hypoalbuminemia. The cat’s acute hyporexia and weight loss was attributed to discomfort associated with the intragastric pH monitoring device, as has been reported in humans. Endoscopic removal of the intragastric pH monitoring device resulted in gastric perforation. The cat underwent exploratory laparotomy for surgical resection and repair of the perforated area. To our knowledge, this is the first report of gastric perforation secondary to removal of a Bravo pH capsule. Caution may be advised when considering intragastric pH capsule removal in cats.
Case Report
A 7-year-old neutered male, domestic shorthair cat was evaluated for hyporexia and weight loss that developed following placement of a Bravo pH capsule (Given Imaging) in the stomach 4 days prior to evaluation. The cat was part of a larger study, approved by our institution’s animal care and use committee, evaluating the efficacy of commonly used oral acid suppressants in cats. Although the cat was slightly thin (body condition score of 2/5), it had a normal appetite and no clinical signs or biochemical abnormalities suggestive of gastrointestinal disease prior to study onset. On day 4 of placebo treatment (250 mg lactose capsules), the cat was anesthetized to facilitate placement of a Bravo pH capsule. A delivery device containing the pH capsule (Figure 1a) was introduced into the stomach under endoscopic guidance. Vacuum suction was applied to the delivery device to suction gastric mucosa into the well of the capsule. A spring-loaded pin was then deployed to engage the capsule to the gastric fundus. The delivery device was removed, leaving the capsule adhered to the gastric fundus (Figure 1b). Fourteen days after capsule placement, the cat was excluded from the study owing to the development of hyporexia for >48 h and weight loss (both clinical signs fulfilled study exclusion criteria). The cat had lost approximately 10% (0.4 kg) of its body weight since the onset of the study. On evaluation, the cat’s vital signs were normal, no pain was elicited on abdominal palpation and no other abnormalities were detected on physical examination. Laboratory data were notable for an albumin of 2.8 (normal range 2.9–4.0 g/dl) and a low normal globulin of 3.0 (normal range 2.8–4.8 g/dl). The cat’s serum chemistry was normal 3 months prior to study onset.

(a) The Bravo pH capsule delivery device is introduced into the stomach under endoscopic guidance. (b) A Bravo pH capsule can be observed adhered to the gastric fundus after removal of the delivery device
Discomfort associated with Bravo capsule placement, as has been previously reported,1,2 was considered as a possible cause for the cat’s acute hyporexia and weight loss. The patient underwent gastroscopy to remove the Bravo capsule and to evaluate for other possible sources of hyporexia. Endoscopic evaluation showed patchy mild white discoloration of the gastric mucosa. No other cause for the acute hyporexia could be identified. The Bravo capsule remained firmly adhered to the gastric mucosa. The Bravo capsule was removed as previously described, 2 by passing a polypectomy snare down the endoscope and encircling the gastric mucosa with the snare tightly opposed to the mucosa adhered to the capsule well (Figure 2). Following removal, a small mucosal defect and associated hemorrhage was observed. On completion of the procedure, gastric perforation was suspected because of persistent abdominal distension, despite deflation of the stomach. Pneumoperitoneum consistent with gastric perforation was confirmed by abdominocentesis and abdominal radiographs. The patient underwent emergency laparotomy. Stay sutures were placed around the greater curvature. A 2 mm full-thickness perforation was identified on the greater curvature of the stomach at the level of the short gastric arteries and resected with a two-layer closure of the wound. Histologic examination of the resected area revealed eosinophilic inflammation with edema, fibrosis and glandular atrophy (Figure 3). The patient recovered uneventfully and, at the time of publication, was receiving ongoing steroid therapy and diet modification.
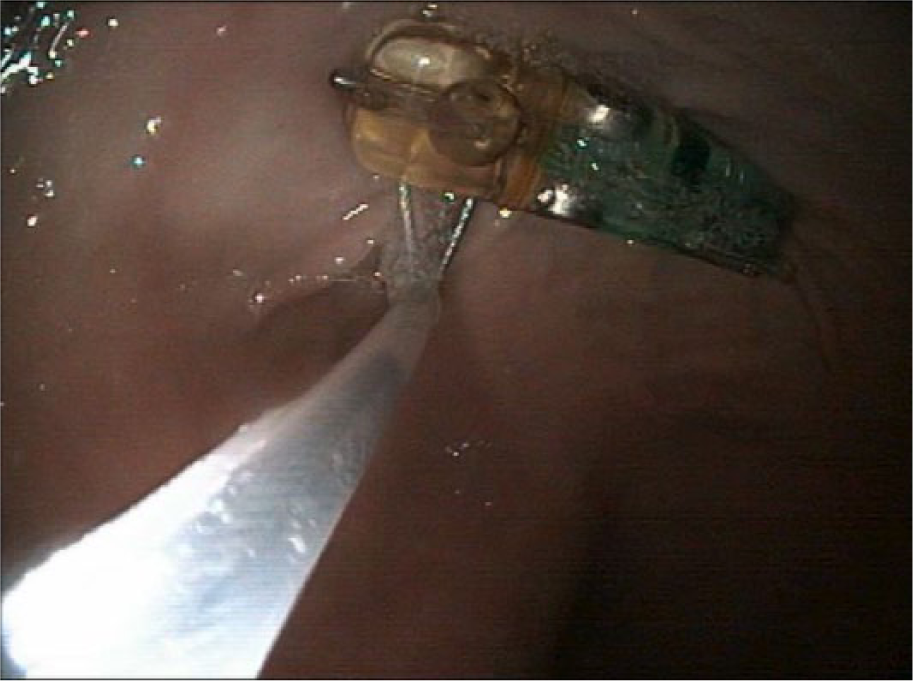
Polypectomy snare encircled around a Bravo intragastric pH capsule adhered to the mucosa of the gastric fundus
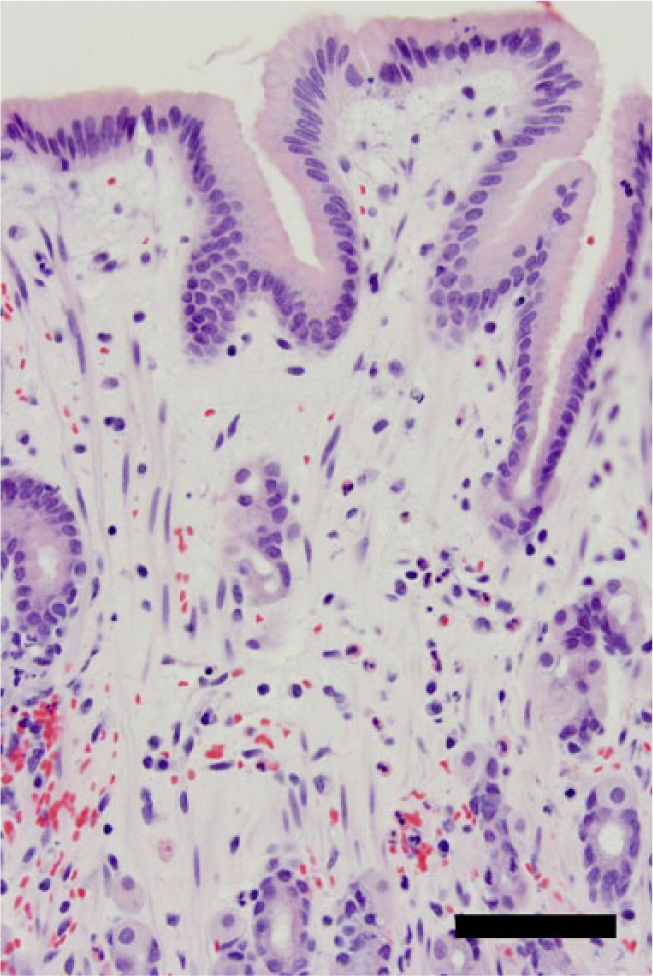
Photomicrograph of gastric mucosa showing expansion of the lamina propria by edema and numerous eosinophils. Hematoxylin and eosin stain. Scale = 50 μm. Magnification ×400
Continuous pH monitoring is used to diagnose acid-related disorders and to evaluate efficacy of acid suppressant therapy in both humans and animals.1–4 The Bravo pH monitoring system utilizes small, catheter-free capsules (26 mm × 6 mm × 6 mm) that radiotelemetrically transmit pH data every 6 s to a receiver placed on the patient or bedside. The Bravo pH monitoring system is designed to measure intragastric pH over a pH range of 0–8. This system provides several advantages compared with conventional catheter-based monitoring systems or techniques based on aspiration of gastric contents. The Bravo system is wireless and provides prolonged monitoring. pH monitoring is guaranteed for 48 h but our experience is that it provides >96 h of monitoring in the cat. The capsule is designed to slough off of the gastroesophageal mucosa with normal epithelial turnover and pass in the feces, causing no harm to the patient. Bravo monitoring is more accurate than catheter-based systems, which are uncomfortable and can become displaced from their intended location, 5 or gastric juice aspiration, which may not accurately reflect the intragastric pH. Infrequently, Bravo pH capsules are placed at an incorrect location, experience premature transmission failure or result in patient discomfort in humans, necessitating their removal.6–8 The use of a cold polypectomy snare is one of the techniques that has been used at our institution (four healthy cats and two dogs) and others for removal of Bravo pH capsules.7,8 This is the first report of gastric perforation secondary to removal of a Bravo pH capsule. Bravo capsules have been successfully removed from the stomachs of normal cats without complication. However, it is possible that the feline gastric wall, which is thin compared with that of humans and dogs, cannot tolerate Bravo pH capsule removal.9–11 Thus, we would caution against removal of capsules and advise allowing them to detach from the feline mucosa with natural epithelial turnover.
Conclusions
Continuous pH monitoring is likely to be increasingly utilized in veterinary medicine and will lead to identification of heretofore under-recognized acid-related disorders in cats and dogs. Caution may be advised when considering Bravo pH removal in patients, especially cats.
Footnotes
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this case report.
