Abstract
Objectives
Rectal temperature (RT) is routinely used to assess body temperature in cats but has limitations and can be poorly tolerated. Axillary temperature (AT) and tympanic membrane temperature (TMT) are reported alternatives. This study aimed to determine the differences between RT and AT, and between RT and TMT in cats. Additional aims were to examine the effect of environmental and patient factors on these differences and to assess patient tolerance to each technique.
Methods
AT, TMT and RT were measured in immediate succession. Measurement order was randomised, as was the choice of left or right axilla and tympanic membrane. A digital thermometer and a veterinary infrared ear thermometer were used. The subjective tolerance of each procedure was recorded.
Results
One hundred and fifty cats were included. Significantly more conscious cats were tolerant of AT (90.6%) than TMT (81.2%) and RT (53.0%). The rectal–axillary temperature difference ranged from −1.2°C to 1.4°C (median 0.1°C) and was within ±0.5°C in 78.0% of cats. On multivariable analysis the difference was larger in overweight cats, neutered cats, cats in which the right axilla was used and as the RT increased. The rectal–tympanic membrane temperature difference ranged from −1.6°C to 3°C (median −0.3°C) and was within ±0.5°C in 51.3% of cats, significantly fewer than for AT (P <0.001). The rectal–tympanic membrane temperature difference increased as the RT increased.
Conclusions and relevance
TMT and AT should not be used interchangeably with RT in cats. When RT measurement is not possible, AT is recommended over TMT as it is better tolerated and significantly fewer cats had clinically unacceptable differences (>0.5°C). AT may more closely reflect RT in normal or underweight cats than it does in overweight cats.
Introduction
Assessment of body temperature is an important part of the physical examination and the information obtained guides clinical decisions. Invasive contact devices such as oesophageal and pulmonary artery thermistors are considered the gold standard for assessing core body temperature;1–3 however, these are not suitable for conscious veterinary patients and are reserved for anaesthetised or critical care cases.2,4 Therefore, most feline and canine patients have their temperatures measured using contact thermometers against the rectal mucosa. 4
Good agreement has been shown between rectal temperatures (RTs) and core body temperature in dogs,2,3 and it is suggested that RT is a convenient and useful indicator of core temperature in other domestic mammals, 5 including cats. This technique can be difficult to perform, particularly in fractious patients or in those with rectal or perianal disease.2,3 It can be stressful for the patient (particularly if serial measurements are needed) and a potential source of cross-contamination and rectal injury.6,7 In addition, the accuracy and repeatability of RT can be negatively affected by depth of measurement, the presence of faeces and conditions affecting local blood flow.3,6,8
Axillary temperature (AT) measurement is an established technique in people, in which numerous studies have been performed.8–14 It is particularly convenient in paediatrics as little cooperation is required and RT assessment can be a frightening experience.9,12 The thinner skin and subcutaneous tissues of less mature infants is thought to give more reliable AT readings than in older children. 15 Advantages are that it is minimally invasive and readily available digital thermometers can be used. AT measurement is described in veterinary texts,16–18 but it has only been investigated in one study in cats. 19 This study only included 31 cats and, using statistical correlation, suggested AT was not interchangeable with RT. It has been suggested, however, that this is not an appropriate statistical tool to assess the agreement between two methods of clinical measurement. 20
Another alternative to RT is the measurement of tympanic membrane temperature (TMT) using an infrared ear thermometer. These thermometers use pyroelectric sensors to measure infrared radiation emanating from the tympanic membrane (TM).21–23 As the TM is well vascularised by the internal carotid arteries, which also perfuse the hypothalamus, TMT is thought to accurately represent core body temperature.22,24 The advantages of this technique include the ability to obtain a reading within 1 s of device activation, 21 and better tolerance in cats when compared with RT.6,25 Reported disadvantages include greater variability between measurements than for RT, 3 the necessity of using a specifically designed device and patient resentment of probe placement.3,26 TMT measurement has been the subject of several studies in cats,6,7,21,25,27 with varying conclusions; some suggest TMT may be used interchangeably with RT,7,21,25 while others suggest it should not.6,27 The largest of these studies included only 48 cats, and only two studies used veterinary devices.6,21
The aims of this study were to determine the differences between AT and RT, and TMT and RT in a large group of cats. In addition, we examined the effect of environmental and patient factors on the magnitude of these differences and assessed the subjective tolerance of cats to each technique.
Materials and methods
Animals
Cats presenting to a veterinary teaching hospital were eligible for inclusion, and the relevant welfare committee granted ethical approval prior to the study. The study population was a convenience sample of cats requiring RT measurement as part of their investigations. Cats were excluded if they had any disease or injury that could have been exacerbated by AT, TMT or RT measurement. Cats were included only once and were excluded if any of the temperature readings could not be obtained due to patient demeanour.
Age, breed, sex, neuter status, weight, body condition score (BCS) and coat length (short or long) were recorded, as were the environmental temperature and the time required to collect all three measurements. BCS was assessed using a nine-point scoring system (Purina Body Condition Tool, www.purina.co.uk/content/docs/downloadable-pdfs/bodycondsytempdf_cat.pdf?sfvrsn=2). Cats were classed as underweight (BCS 1–4), ideal weight (BCS 5) or overweight (BCS 6–9). The cats’ level of consciousness was described as conscious, sedated (this included cats recovering from anaesthesia) or anaesthetised.
Temperature measurements
Cats had AT, TMT and RT measured in immediate succession. The order in which readings were collected was randomised (Research Randomizer, www.randomizer.org), as was the choice of left or right AT and TMT using two coin tosses. The order and side used was recorded. An assistant gently restrained the cats whilst a single observer obtained the temperatures. The RT was used to describe the cats as hypothermic (<37.8°C), normothermic or hyperthermic (>39.2°C). 3
AT and RT were measured using a single digital thermometer (Kruuse Digital Thermometer; Jørgen Kruuse), which measures temperatures between 32.0°C and 43.9°C. The reported accuracy is within ±0.1°C between 35 and 39°C, and ±0.2°C outwith these limits. For RT measurement, the digital thermometer was placed in a disposable cover (Probe Covers for Digital Thermometer; Bunzl Retail and Healthcare Supplies) coated in lubricant (K-Y Lubricating Jelly; Johnson and Johnson, Santé Beautéé France SAS). The tip was inserted a minimum of 1.5 cm into the rectum and gently positioned in close contact with the mucosa. For AT, the thermometer tip was placed as proximally as possible in the axilla in close contact with the skin, approximately midway from the cranial and caudal margins (Figure 1). The foreleg was then gently held against the chest wall. For AT and RT, the thermometer was held in position until the alarm sounded. After each use the thermometer was cleaned with an alcohol swab.

Thermometer position for axillary temperature measurement
A single, commercially available veterinary infrared ear thermometer (Pet-Temp Model PT 300; Advanced Monitors) was used to measure TMT. The device measures TMT between 34.0°C and 43.0°C, with a reported accuracy of ±0.2°C. Prior to use, the lens was cleaned with an alcohol swab if required. A disposable, single-use protective cover was placed onto the thermometer tip. The cat’s ear was gently pulled caudolaterally to align the vertical and horizontal canals, and the thermometer was inserted into the ear canal directing it towards the angle of the opposite jaw. When the activation button was depressed, an audible beep indicated completion of the temperature measurement. If an error reading occurred, the measurement was attempted again. If, after three attempts, no reading was obtained, the animal was excluded from the study.
For each method of temperature measurement, cats were subjectively assessed as tolerant of the procedure if they were calm, compliant or quiet, or intolerant if they were struggling, agitated or vocal. 28
Statistical analysis
Statistical analysis was performed using Minitab (version 17.1.0). Variables were tested for normality using the Kolmogorov–Smirnov normality test, and appropriate parametric and non-parametric tests were used. The rectal–axillary temperature difference (RATD) was calculated by subtracting AT from RT. The rectal–tympanic membrane temperature difference (RTMTD) was calculated by subtracting TMT from RT. The range of RATD and RTMTD between which 95% of the cases fell and the number of cats in which the RATD and RTMTD varied by less than ±0.5°C was determined. Bland–Altman plots were created to provide visual assessment of agreement between RT and AT, and between RT and TMT.
Univariable linear regression was used to identify variables affecting RATD and RTMTD. Those with a P value <0.2 were considered for inclusion in the multivariable models. Variables were ordered by P value (smallest to largest) and r2 (largest to smallest) prior to sequential insertion into the multivariable models. Variables were retained in the models if P <0.05.
McNemar’s test was used to compare the proportion of conscious cats that were tolerant of AT, TMT and RT, and also the proportion of cats in which the RATD was less than ±0.5°C to the proportion of cats for which the RTMTD was less than ±0.5°C. A P value of <0.05 was considered significant.
Results
One hundred and fifty cats were included: 89 (59.3%) males and 61 (40.7%) females. The majority were neutered: 84 neutered males (94.4%) and 57 neutered females (93.4%). Ages ranged from 3 months to 18 years (median 8 years, mean 7 years 8 months). Most cats were domestic shorthairs (n = 110). Others included domestic longhairs (n = 4), Bengal (n = 7), Ragdoll (n = 6), Siamese (n = 5), British Shorthair (n = 4), Burmese (n = 3), Russian Blue (n = 2), Tiffany (n = 2), British Blue (n = 1), Exotic Shorthair (n = 1), Havana (n = 1), La Perma (n = 1), Maine Coon (n = 1), Norwegian Forest (n = 1) and Persian (n = 1). The cats weighed between 0.71 and 7.80 kg (median 4.10 kg, mean 4.16 kg). Fifty-nine (39.3%) were underweight, 52 (34.7%) had an ideal BCS and 39 (26%) were overweight. One hundred and forty cats had short hair (93.3%) and 10 (6.7%) had long hair.
Room temperatures ranged from 18.9–24.5°C during temperature measurement. All cats had all methods of temperature measurement performed, and no cats were excluded owing to repeated error readings while performing TMT. The median time to collect all temperature measurements in succession was 3 mins (range 2–6 mins). One hundred and seventeen cats (78.0%) were conscious, 20 were sedated (13.3%) and 13 were anaesthetised (8.7%).
One hundred and six (90.6%) conscious cats were tolerant of AT compared with 95 (81.2%) being tolerant of TMT and 62 (53.0%) being tolerant of RT (Table 1). Significantly more cats were tolerant of AT (P <0.001) and TMT (P <0.001) than RT. AT was tolerated by significantly more cats than TMT measurement (P = 0.04).
Summary of results comparing rectal, axillary and tympanic membrane temperature measurements in 150 cats
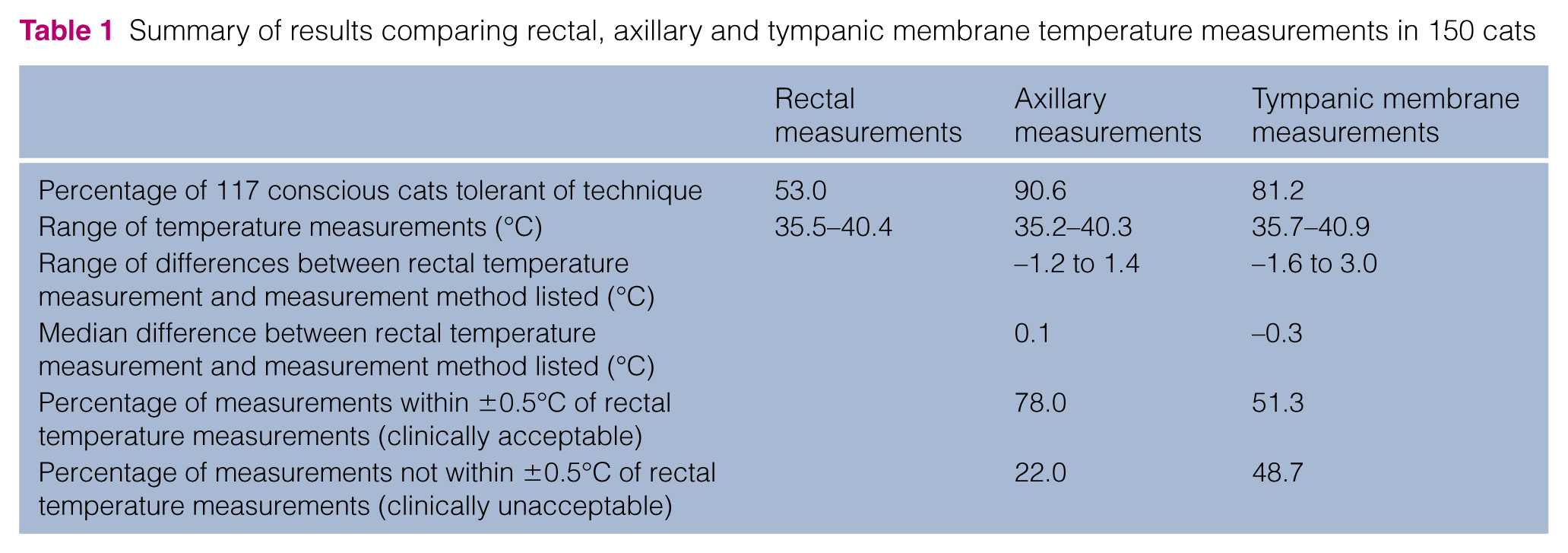
RTs ranged from 35.5–40.4°C (median 38.2°C, mean 38.06°C) (Table 1). Fifty (33.3%) cats were hypothermic, 86 (57.3%) were normothermic and 14 (9.3%) were hyperthermic.
ATs ranged from 35.2–40.3°C (median 38.0°C, mean 37.9°C). The RATD ranged from −1.2°C to 1.4°C (median 0.1°C) (Figure 2). In 95.0% of cats the RATD was between −0.7°C and 1.1°C. The RATD was within ±0.5°C in 117 cats (78.0%) (Table 1). On univariable analysis no significant associations were found between the RATD and age, sex, level of consciousness, environmental temperature or tolerance of the procedure. The BCS, weight, RT, neuter status, choice of left or right axilla, and coat length had significant (P <0.2) associations with RATD and were investigated in a multivariable model. The RATDs for normal and underweight cats were not significantly different but the RATD for overweight cats was significantly different to both groups and, therefore, overweight cats were compared with cats with BCSs of 1–5 in the multivariable model. In the final model, BCS, RT, neuter status and the use of the left or right axilla were found to be associated with the RATD (P <0.05). This model accounted for an r2 of 25.1%. The model showed that RATD was 0.38°C smaller in entire cats than neutered cats. Overweight cats had a RATD 0.34°C greater than cats with a BCS of 1–5. Excluding overweight cats from the data, 84.7% of the remaining 111 cats had a RATD within ±0.5°C. The model also showed that RATD was 0.16°C greater when the right axilla, rather than the left axilla was used, and that for every 1°C increase in RT, the RATD was 0.12°C greater.

Bland–Altman plot of the difference between rectal temperature (RT) and axillary temperature (AT) against the mean of RT and AT in 150 cats. The dashed horizontal line at 0°C corresponds to zero differences (points fell on this line when RT and AT matched). The solid horizontal reference lines correspond to ±0.5°C differences. Positive points represent an AT lower than RT. Negative points represent an AT higher than RT. The median rectal–axillary temperature difference was 0.1°C
TMTs ranged from 35.7–40.9°C (median 38.5°C, mean 38.3°C). The RTMTD ranged from −1.6°C to 3.0°C (median −0.3°C). The RTMTD was within ±0.5°C in 77 cats (51.3%) (Figure 3). Significantly more RATD were within ±0.5°C than RTMTD (117 cats vs 77 cats) (P <0.001) (Table 1). In 95% of cats the RTMTD was between −1.5°C and 1.2°C. On univariable analysis, age, RT and tolerance of RT had significant (P <0.2) associations with RTMTD and were investigated in a multivariable model. The multivariable model showed no evidence that age or RT tolerance were useful predictors of RTMTD. RT remained significant (P <0.05). The model accounted for an r2 of 3.84%. For every 1°C increase in RT the RTMTD increased by 0.16°C.
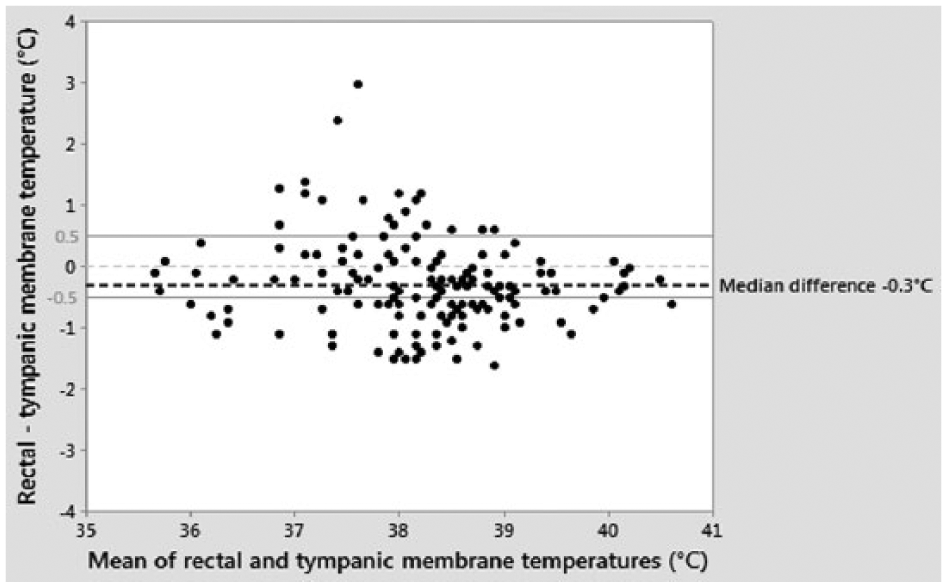
Bland–Altman plot of the difference between rectal temperature (RT) and tympanic membrane temperature (TMT) against the mean of RT and TMT in 150 cats. The dashed horizontal line at 0°C corresponds to zero differences (points fell on this line when RT and TMT matched). The solid horizontal reference lines correspond to ±0.5°C differences. Positive points represent a TMT lower than RT. Negative points represent a TMT higher than RT. The median rectal–tympanic membrane temperature difference was −0.3°C
Discussion
AT measurement was significantly better tolerated in cats than TMT or RT measurement. This has not been previously reported in cats. Similar findings have been identified in dogs; Lamb and McBrearty 29 found that assistance was significantly less likely to be required when measuring AT as opposed to RT and TMT, and Gomart et al 30 found that stress behaviour scores were lowest with AT and highest with RT. Only 81.0% of cats were tolerant of TMT measurement, which is lower than the 93.1% of cats reported in another study. 7 This may reflect the different devices used, the operators’ technique or the subjective interpretation of the cats’ behaviour by the observers.
ATs were a median of 0.1°C lower than RT, which is similar to the difference of 0.2°C reported by Goic et al. 19 ATs have also been reported to be lower than RT in dogs and humans,11–14,19,29–31 although the magnitude of the reported differences is variable. Despite the small bias between AT and RT, a large range in RATD, from −1.6°C to 1.4°C, was found. This was similar to the range of −1.1°C to 1.6°C reported in cats by Goic et al. 19 It has previously been suggested that any variation greater than ±0.5°C from the RT measurement is clinically unacceptable.2,4 Twenty-two per cent of cats had ATs that differed by more than ±0.5°C from the RT, which suggests these techniques cannot be used interchangeably.
Multivariable analysis showed that overweight cats were associated with a 0.34°C increase in RATD: a sizeable effect, when an RATD ±0.5°C is considered clinically significant. BCS was similarly found to be significantly associated with RATD by Goic et al. 19 In human patients, a high body mass index (BMI) is associated with a lower AT when compared with patients with a low BMI. 32 It is likely that the increased amount of subcutaneous fat insulates the skin from the core body temperature, therefore increasing the RATD. In contrast, the temperature of the rectum is directly related to blood flow through the splanchnic circulation.1,15 Goic et al also found that weight was significantly associated with RATD in cats; 19 however, although this was significant on univariable analysis in our study, the significance was lost when multivariable analysis was performed.
The RATD of entire cats was 0.38°C smaller than that of neutered cats. Although neutering is associated with weight gain and obesity,33–35 this was found on multivariable analysis and therefore some other factor must account for this difference between the two populations. It should be noted that the number of entire cats included was small.
For every 1°C increase in RT, the RATD increased by 0.12°C. Similarly, Goic et al reported a larger median RATD in hyperthermic cats compared with the overall mean of the population. 19 This effect has also been documented in humans.12,31 These findings could partially be explained by the physiology of thermoregulation whereby mechanisms to generate, conserve or dissipate heat are instigated as body temperature changes.9,14 For example, as a patient becomes pyrexic, there is intense peripheral vasoconstriction and the skin temperature may cool as the core temperature rises. 14 This occurs in conjunction with thermogenesis via tachypnoea and increased basal metabolic rate; these factors lead to an increased gradient between core and surface temperature. 9 It could also be possible that there is a lag between rising internal temperature changes and skin surface temperatures.
It is interesting that the RATD was higher by 0.16°C, on average, when measured in the right axilla. Variation between right and left AT has also been documented in humans, although the direction of the difference is inconsistent.10,36–38 As measurements were collected by a single right-handed investigator, it is possible that this finding reflects a better technique when measuring the temperature in the left axilla. As the reason for the difference between the left and right axilla is unclear at this time, no recommendation is made regarding the choice of axilla when using this technique.
Our model only accounted for 25.14% of the variation in RATD, which suggests that other variables that we did not investigate must play a role. Examples could include regional changes in blood perfusion, which have been hypothesised to cause disproportionate variation in temperature measurement at different sites, 15 and time of day, which has been found to affect RATD in humans. 9
TMT was a median of 0.3°C higher than RT, which is similar to the findings of Sousa et al, 7 in which the mean TMT was 0.1°C higher than RT. In contrast, Kunkle et al 6 and Michaud 25 found that the mean TMT was lower than RT by 0.07°C and 0.16°C, respectively. Although the bias between TMT and RT was small, the large range of RTMTD (−1.6°C to 3.0°C) and the fact that 48.7% of TMTs were not within ±0.5°C of RTs, suggests that these methods should not be used interchangeably in cats. This conclusion is in agreement with Garner 27 and Kunkle et al, 6 but contrasts with the conclusions of Michaud, 25 Rexroat 21 and Sousa et al. 7 Michaud 25 and Sousa et al 7 used human devices, and their conclusions were based on statistical correlation. The study by Rexroat et al 21 was conducted on behalf of the veterinary TM thermometer manufacturers. It included both cats and dogs and, as it is not possible to distinguish between the results for the two species, the reasons for their conclusions are difficult to ascertain. The large range of RTMTD may be due to differences in probe positioning, which has been reported to affect the reading obtained.6,39
On multivariable analysis, the only factor that was found to be associated with the RTMTD was RT. Similarly, Kunkle et al 6 found that the RTMTD increased in cats with temperatures considered to be febrile. For every 1.0°C increase in RT, the RTMTD increased by 0.16°C. Interestingly, this is similar to the magnitude of the variation in RATD with increasing RT.
Significantly more ATs were within ±0.5°C of RTs (78.0%) when compared with TMTs (51.3%), and although we do not recommend either method is used interchangeably with RT routinely, in those cases in which RT cannot be collected, our findings suggest that AT would be preferable to TMT measurement. This is in contrast to findings in dogs, in which the median RTMTD was smaller than RATD and more dogs had an RTMTD within ±0.5°C than had RATD within ±0.5°C. 29 Interestingly, in humans, RATD is larger in older children compared with that of neonates. 13 This difference may be because smaller patients have a greater ratio of surface area to body mass and therefore higher heat conduction rates, 40 which could result in a smaller temperature difference between the core and the periphery, and hence a smaller RATD. Another hypothesis is that it is due to the thinner skin and subcutaneous tissues of less mature infants compared with older children. 15 Either of these reasons may explain the differences found between dogs and cats as cats are obviously smaller than dogs and feline skin is thinner than that of dogs (0.4–to 2.0 mm vs 2.2–3.4 mm).41,42
As the majority of cats were conscious, invasive core temperatures were not measured. However, RT is considered the reference standard for assessing body temperatures in conscious veterinary patients. 7 Neither repeatability, nor inter-operator variability were assessed in this study. Further investigations are needed to determine how repeated AT measurements perform in individual patients to measure temperature trends.
Conclusions
TMT should not be used interchangeably with RT in cats as a wide range of RTMTD was found, and 48.7% of cats had clinically unacceptable differences between TMT and RT. Clinically unacceptable differences between AT and RT were found in 22% of cats and therefore these methods should also not be used interchangeably. In cats where RT is not possible or is likely to be inaccurate, such as in cases of aortic thromboembolism, AT is recommended over TMT as it is better tolerated, and significantly fewer cats had clinically unacceptable differences between AT and RT. In such cases, it should be considered that AT will more closely approximate RT in normal or underweight cats.
Footnotes
Acknowledgements
We would like to thank Dr Tim Parkin for his advice regarding the statistical analysis of the data, and Íñigo Sanz González for drawing the figure showing axillary temperature measurement.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Funding
This research received no grant from any funding agency in the public, commercial or not-for-profit sectors.
