Abstract
Clinical challenges:
Anaesthesia for dental and oral procedures in cats can be challenging and many factors need to be taken into consideration. Often it is older patients requiring these procedures and, while old age itself is not a contraindication for general anaesthesia, older patients tend to have limited homeostatic reserves and are, therefore, more prone to anaesthesia-induced insults of vital organs. Most sedative and anaesthetic agents have cardiovascular side effects, which may result in detrimental consequences in older patients in which organ reserves are likely reduced.
Aims:
The need for good patient assessment and management during the entire perianaesthetic period cannot be overemphasised. This article describes how both anaesthetic protocol and intravenous fluid therapy should be tailored to the individual cat’s needs. Application of a multimodal analgesic protocol (the combination of different analgesic agents) and a balanced anaesthetic technique incorporating local nerve blocks is highly recommended and a particular focus of the review. The use of local anaesthetic agents for the latter not only provides optimal pre-emptive analgesia, but also reduces the amount of anaesthetic agents needed to maintain an adequate level of anaesthesia and, therefore, limits their side effects. Other key aspects of perianaesthetic care of the feline dental and oral patient include airway protection, monitoring and maintenance of body temperature, eye protection, and analgesia extending well into the post-anaesthetic period.
Evidence base:
The authors draw on their clinical experience and the referenced literature to provide a practical overview of this critical but often-overlooked aspect of feline dentistry.
Challenges of the perianaesthetic period
In 2008, the Confidential Enquiry into Perioperative Small Animal Fatalities (CEPSAF), in which sedation and general anaesthetic records of over 79,000 cats were analysed, identified risk factors associated with perioperative mortality. 1 Overall, risk of sedation-and anaesthetic-related death in cats was 0.24%. Mortality risk was found to be 0.11% in healthy cats (American Society of Anesthesiologists [ASA] grade 1–2) and 1.40% in sick cats (ASA grade 3–5).
Compared with dogs, cats have a higher risk of complications, which can be related to their smaller body size predisposing them to hypothermia and (relative) drug overdose. Risk factors identified in the CEPSAF study were poor health status (ASA classification), old age (>12 years), extremes of body weight (<2 and >6 kg), increased procedural urgency and complexity, endotracheal intubation and the use of intravenous (IV) fluid therapy. Monitoring of the peripheral pulse reduced the risk of anaesthetic-related death by 70%; in combination with pulse oximetry, the risk was reduced by 80%. 2 More than 60% of deaths occurred in the immediate postoperative period (first 3 h after termination of the procedure); in 20% of these cases the cause of death was unknown, indicating that patients were not closely monitored. Cardiovascular or cardiopulmonary problems resulted in over 63% of all deaths. 1 This emphasises the need to improve the quality of anaesthetic care and the level of monitoring of our feline patients, not only during the general anaesthetic period itself but also during recovery from anaesthesia.
Although general anaesthesia for oral and dental procedures may be required in young and healthy patients (for tooth extraction, fracture repair, etc), often older patients are involved. Old age itself is not a disease and not a contraindication for general anaesthesia. Older patients will, however, have limited homeostatic reserves and are, therefore, more prone to anaesthesia-induced insults of vital organs such as the kidneys, liver, heart and central nervous system (CNS). General anaesthesia for oral and dental procedures carries an increased risk of aspiration of blood, saliva and irrigation fluids; and surgery on the head can hinder the application of monitoring equipment such as a pulse oximeter. All these factors combined make anaesthesia for these kinds of procedures in cats more challenging and underline the need for good patient assessment and management during the entire perianaesthetic period.
Pre-anaesthetic preparation
Special emphasis should be placed on assessment of renal, cardiovascular and thyroid function. Obtaining a good history and performing a thorough clinical examination are of equal, if not greater, value than routine haematology and biochemistry screening.
The authors’ recommendation is to perform pre-anaesthetic bloods (haematocrit, total proteins, electrolytes, urea and creatinine) in all patients over 8 years of age and in younger patients when there are reasons for concern. Cats may appear to be clinically healthy with no signs of azotaemia, but still may already have lost 50–75% of functioning nephrons. It is perhaps wise to presume that all geriatric cats, defined as those having reached 75% of their anticipated life span, have a degree of renal impairment and to treat them accordingly, especially by means of appropriate supportive care and monitoring. Trends in creatinine values over time provide good information on renal function when levels are still within normal range; an increase over time is very likely the result of a decrease in renal function.
Urinalysis adds easily accessible and significant additional information about renal function; a specific gravity >1.040 is adequate in cats. Although feline urine should not contain any proteins under normal healthy conditions, urine dipstick tests may indicate a trace of protein (1+). This is because dipsticks are made for analysis of human urine, which is more dilute than feline urine; the buffer capacity of the reagent strip is often insufficient. Normal urine protein:creatinine (UPC) ratio is <0.2; values of 0.2–0.4 are borderline proteinuric and values >0.4 are proteinuric; values of >0.4–0.5 are predictive of shorter survival rates.
As both UPC ratio and plasma creatinine concentration have been shown to be predictors of azotaemia development in cats, monitoring of both parameters has been recommended as part of geriatric screening in cats. 3 More than 30% of cats aged over 10–15 years are estimated to have chronic kidney disease, of which up to 65% may also suffer from chronic hypertension.4,5 Patients with some degree of chronic renal failure may benefit from fluid therapy started at least several hours before anaesthesia to optimise their circulating blood volume. Often a rate of 1–2 x maintenance is sufficient (2–4 ml/kg/h). Compared with dogs, cats are more prone to fluid overload because of their lower circulating blood volume (60–65 ml/kg vs 80–90 ml/kg), especially when occult cardiac disease is present. Ideally, IV fluids should be administered by means of an infusion pump or syringe driver to avoid accidental fluid overloading. Special paediatric fluid administration sets exist for free-flowing, gravity-based fluid administration, either delivering 60 drops/ml instead of the usual 20 drops/ml or with a burette that can be primed with the desired volume of fluid to be administered (Figure 1).
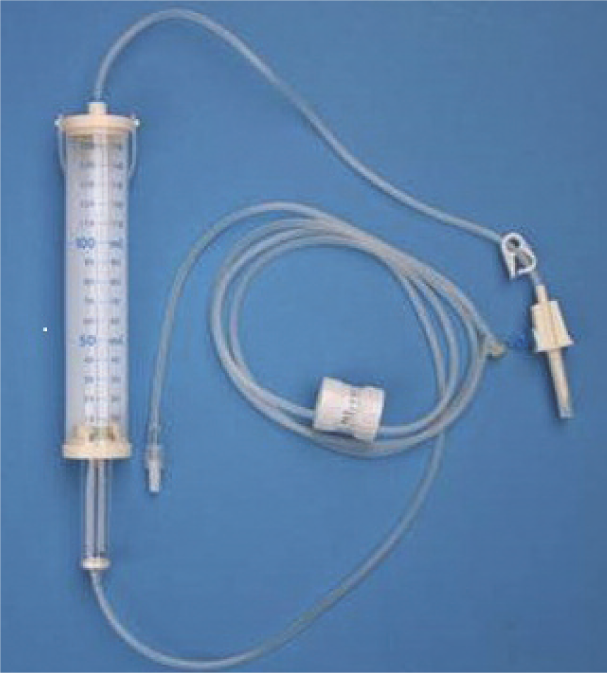
Paediatric fluid administration set. A burette is incorporated that can be primed with the desired amount of fluid, to avoid accidental fluid overload. With a flow dial the desired fluid rate can be set
Hypokalaemia, especially levels <3.0 mmol/l, should be corrected by the administration of fluids spiked with potassium. The authors usually administer IV fluids (Hartmann’s solution) spiked with potassium to a concentration of 25–30 mmol/l (Table 1). Care has to be taken not to bolus these fluids and not to exceed the maximum recommended potassium infusion rate of 0.5 mmol/kg/h, although this is rarely encountered in general practice. Potassium-spiked fluids should always be administered by means of an infusion pump or syringe driver to avoid accidental overdose.
Guidelines for intravenous potassium supplementation in cats

Fluids should still be administered at the guidelines rate of 3–5 ml/kg/h, such that K+ overdose is highly unlikely.
From DiBartola SP. Disorders of potassium: hypokalemia and hyperkalemia. In: Fluid, electrolyte, and acid-base disorders in small animal practice, 4th ed. Elsevier, 2012, p 107
The risk of hyperthyroidism in cats increases with age; approximately 10% of cats aged over 9 years are hyperthyroid, while a prevalence of 50% has been found in cats over 15.5 years of age in one UK study. 6 Associated cardiovascular changes may include tachyarrhythmias, gallop rhythm, systolic murmurs and hypertension. Preoperative stabilisation of the hyperthyroidism is desirable before elective procedures are undertaken.
An IV catheter should be placed in all patients, not only to enable IV fluid administration but also to allow IV drugs to be titrated to effect instead of administered as a calculated bolus. Titration to effect results in less drug administered and thus fewer side effects. A good alternative site to the cephalic vein for IV catheter placement in cats is the medial saphenous vein (Figure 2). The application of EMLA cream 5% (AstraZeneca), a eutectic mixture of the local anaesthetic agents lidocaine and prilocaine, over the site of catheter placement will facilitate catheter penetration of the skin. After clipping of the skin, a small amount can be applied, after which a synthetic, non-absorbable dressing (cling film or part of a rubber glove) is loosely wrapped around the area and kept in place for 20–30 mins to increase uptake of local anaesthetic agents by the skin. Sometimes sedation is needed in uncooperative cats to achieve catheterisation.
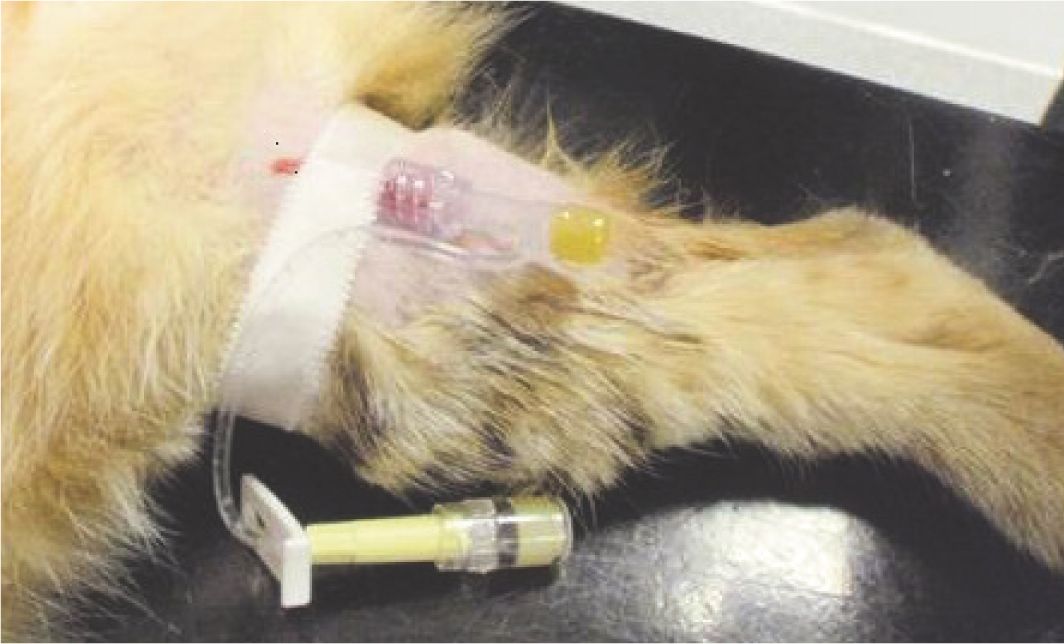
The median saphenous vein provides a good alternative site for intravenous catheter placement in cats
Anaesthesia
Some important considerations for a patient undergoing anaesthesia are highlighted in the box below.
Pre-anaesthetic medication
Acepromazine is a long-acting sedative with no analgesic properties; it is metabolised by the liver and cannot be reversed. Its blockade of peripheral alpha-1 receptors results in vasodilation, which may contribute to intraoperative hypotension and hypothermia. By contrast, the alpha-2 adrenoceptor agonists dexmedetomidine and medetomidine provide reliable sedation, contribute to analgesia and can be reversed with atipamezole. Their use results in characteristic cardiovascular side effects such as reduced cardiac output and possible decreased perfusion of peripheral tissues; they are best avoided in older cats with potentially decreased renal function or cardiovascular instability. In patients diagnosed with left ventricular outflow tract (LVOT) obstruction, which is typically seen in hyperthyroid cats with a systolic murmur, medetomidine rather than acepromazine may be the drug of choice, as a reduction of peripheral vascular resistance by acepromazine may worsen the LVOT obstruction. 8 Intramuscular (IM) alfaxalone, at 1–2 mg/kg, combined with midazolam and an opioid is a good alternative for sedation of fractious cats; onset of sedation is within several minutes. Unfortunately, the volume of injection is rather large and may result in pain on injection and potentially some degree of muscle trauma.

When a painful condition already exists or a painful procedure is planned, an opioid should form the mainstay of the pre-anaesthetic protocol, especially in patients in which non-steroidal anti-inflammatory drugs (NSAIds) are contraindicated. Opioids do not exert major cardiovascular side effects and will provide some degree of sedation. In fact, some cats, even hyperthyroid ones that may be difficult to handle, can become more compliant with just buprenorphine (0.02 mg/kg), for which the transmucosal/sublingual route is a good alternative to intramuscular administration in this species. 9
Midazolam, a benzodiazepine, can be combined with an opioid for pre-anaesthetic medication in older, debilitated patients; its use is devoid of cardiorespiratory side effects. In young and healthy animals, however, it may result in disinhibition and less cooperation.
Induction and maintenance of anaesthesia
For induction and maintenance of anaesthesia, ideally drugs that exert minimal cardio-respiratory depression should be used. However, familiarity with both the anaesthetic drug and technique is at least as important in providing ‘safe anaesthesia’ as the choice of a theoretically ideal but to the veterinary surgeon unfamiliar drug or technique. In order to reduce the cardiovascular side effects induced by the induction agent, a co-induction technique with a benzodiazepine or an opioid such as fentanyl can be applied. Drug doses and routes of administration are given in Table 2.
Drugs used for (co-)induction of anaesthesia in cats

IV = intravenous
Both propofol and alfaxalone are suitable induction agents.10,11 Their IV administration should be titrated to effect as these drugs depress the cardiovascular system and can result in induction apnoea. The use of ketamine is not recommended in patients with conditions for which an increase in myocardial oxygen demand should be avoided (eg, hypertrophic cardiomyopathy).
Because of the high risk of aspiration, all patients should be intubated with a cuffed endotracheal tube (ETT). To ensure the ETT is the appropriate length it should first be measured against the patient (Figure 3): the tip of the tube should reach the level of the shoulders, with the proximal part of the tube not protruding much in front of the incisors. An ETT that is too long will result in increased mechanical dead space and rebreathing. Also, there is the risk that the tip of the tube enters one of the bronchi, resulting in one-lung ventilation with consequent difficulties in maintaining an adequate plane of anaesthesia, ventilation/perfusion (V/Q)-mismatching and shunt formation, leading to hypoxaemia and hypercapnia.
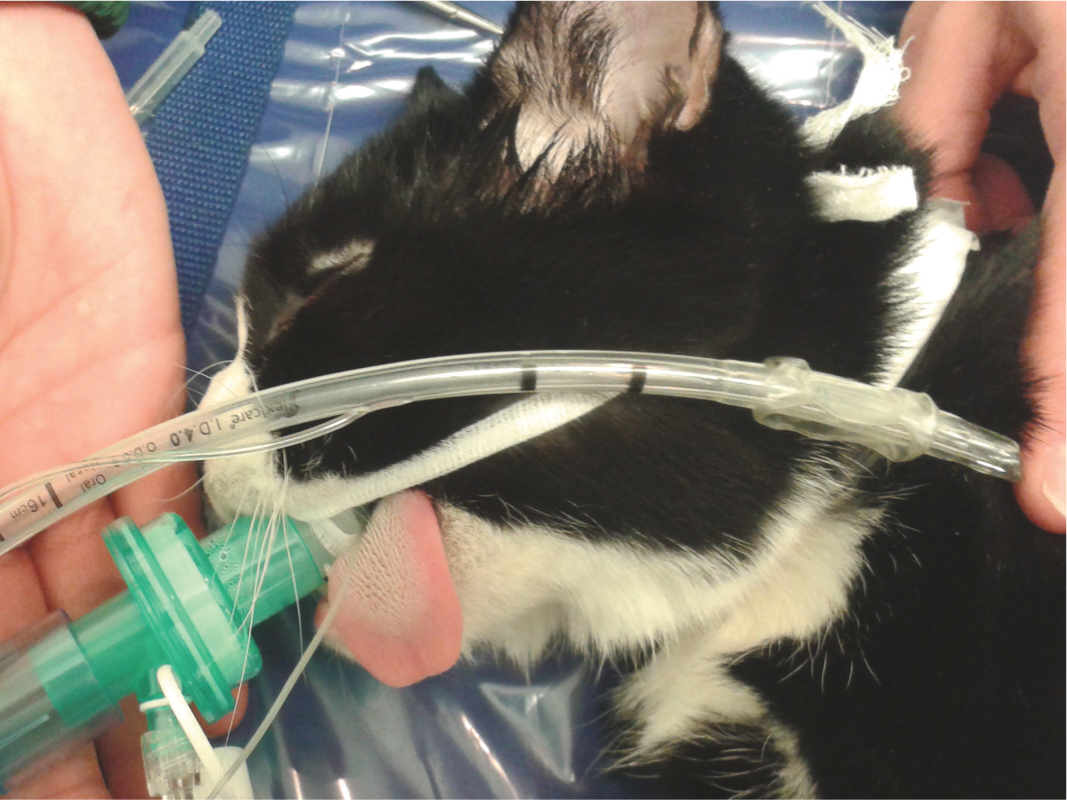
The endotracheal tube held against this patient would have been too long. The correct length of an endotracheal tube is checked by measuring it against the patient: the tip of the tube should reach the level of the shoulders, with the proximal part of the tube not protruding much in front of the incisors
The feline upper respiratory tract is very sensitive and prone to damage and laryngeal spasm. To facilitate endotracheal intubation, the larynx should be desensitised by spraying lidocaine topically on the vocal cords. Intubeaze (Dechra Veterinary Products) is licensed in the UK for cats for this purpose and contains 20 mg/ml of lidocaine; one good squirt (0.1–0.2 ml) results in the application of 2–4 mg. Because lidocaine is well absorbed through the mucous membranes, it is important to consider the total dose applied, especially if local anaesthetic agents will be part of the anaesthetic protocol at a later stage (ie, nerve blocks). The maximum recommended dose of lidocaine in cats is 6 mg/kg; application of Intubeaze should preferably be limited to one or two sprays. Care should be taken not to use xylocaine, a topical lidocaine spray used in human anaesthesia, as this contains preservatives known to cause life-threatening laryngeal oedema in cats. 12

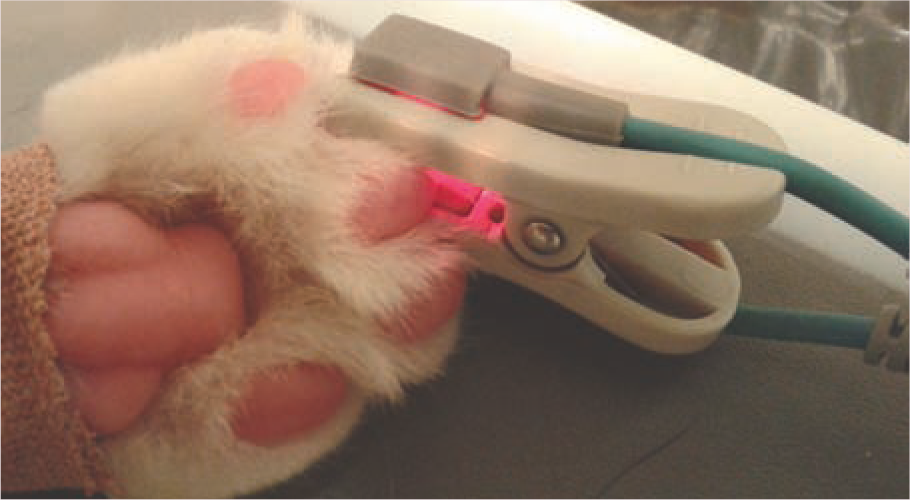
An alternative site for the pulse oximeter probe

Doppler probe and blood pressure cuff placed around the base of the tail, with the probe positioned ventrally
Both sevoflurane and isoflurane can be used for maintenance of anaesthesia, and there is no real benefit of one volatile agent over the other. These agents are potent vasodilators and given, therefore, that they can contribute significantly to intraoperative hypotension, 13 their administration should be reduced to the lowest possible level.
It is recommended that IV fluids are provided during anaesthesia, not only to compensate for ongoing losses and potential haemorrhage but also to counteract any relative hypovolaemia resulting from anaesthetic agent-induced hypotension. Although a ‘surgical fluid rate’ of 10 ml/kg/h has long been recommended, lower rates such as 3–5 ml/kg/h are often sufficient, with a reduced risk of fluid overload. Depending on the cardiovascular status of the patient this rate can be adjusted if needed. 14 If required and no contraindication exists, a crystalloid fluid bolus of 5 ml/kg can be infused over 5–10 mins, and repeated if necessary (although care has to be taken with the total amount infused).
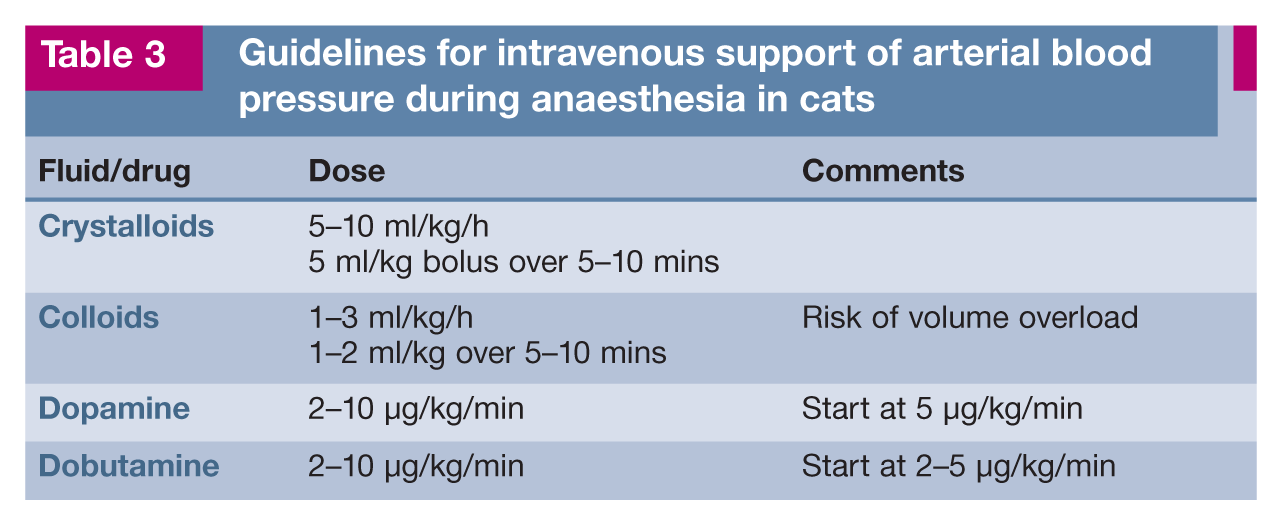
The use of local anaesthetic techniques will allow relatively low vaporiser settings; additional intraoperative analgesia can be provided by the IV administration of fentanyl (2–5 µg/kg bolus or 5–10 µg/kg/h continuous rate infusion) or ketamine (0.2–0.5 mg/kg bolus or 10 µg/kg/min continuous rate infusion). Administration of these analgesic agents, especially as a bolus, may result in respiratory depression and the need for temporary (manual) ventilatory support. If hypotension persists despite a reduction in vaporiser setting and the administration of IV crystalloids, either IV colloids or a positive inotrope such as dopamine or dobutamine can be infused (see Table 3).
Guidelines for intravenous support of arterial blood pressure during anaesthesia in cats
Pain generation pathways relevant to oral and dental procedures
To effectively prevent and treat pain a good understanding of the mechanisms involved is essential. Activation of nerve endings by noxious stimuli (chemical, mechanical or thermal) results in generation of an electrical impulse. These electrical impulses are generally transmitted via afferent nerve fibres to the dorsal horn of the spinal cord, through which they reach the cerebral cortex where pain perception takes place. Afferent neural impulses from the oral cavity are transmitted via the trigeminal nerve (cranial nerve V; see box on page 29) and synapse with neurons located in the nucleus caudalis of the medulla.
Noxious stimuli initiating this pathway originate either directly from oral manipulation or from inflammation triggered by tissue damage. Inflammatory mediators such as prostaglandins, potassium and hydrogen ions, adenosine triphosphate, bradykinin and nerve growth factor all reduce the threshold for stimulation of sensory nerve endings. Consequently more impulses arrive at the nucleus caudalis – a process called peripheral sensitisation. This results in increased stimulation of N-methyl-D-aspartate (NMDA) receptors within the CNS and, in turn, an exaggerated response (central sensitisation or ‘wind-up’). This hyperexcitable state within the CNS can remain present even after the original painful stimulus has disappeared. Characteristic for central sensitisation is the presence of allodynia (in which a normally non-painful stimulus is experienced as painful) and hyperalgesia (a painful stimulus elicits an abnormally strong pain reaction). Central sensitisation can last for days to weeks, or even a lifetime. 17

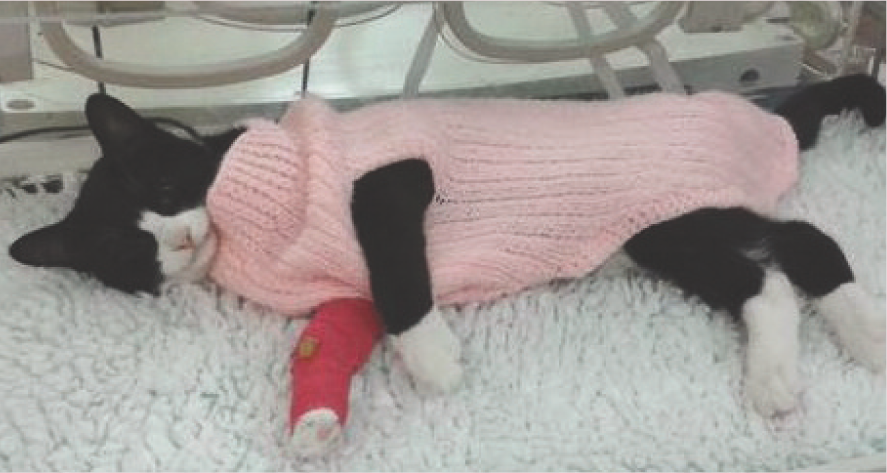
A knitted woollen jumper is a very effective way to keep a cat warm during the entire perianaesthetic period


Lateral view of the skull showing landmarks for regional anaesthesia. A = infraorbital foramen, B = mandibular foramen (on the medial aspect of the mandible), C = mental foramina
Desensitised structures after successful nerve blockade. The principal nerve blocks are shown in bold

Once pain is established, it becomes more difficult to control. Thus pre-emptive administration of analgesic agents before a painful stimulus is the ideal, the aim being to prevent the development of central sensitisation. Inadequate levels of analgesia may result in extended hospitalisation and fluid support, and the requirement for additional analgesics and feeding assistance. 18
Multimodal analgesia
There are several classes of analgesic agents that can be used as part of a multimodal analgesic protocol:
Recommended doses and routes of administration of these various analgesic agents are given in Table 5.
Analgesic agents for use in cats

IV = intravenous, IM = intramuscular, SL = sublingual, PO = oral, SC = subcutaneous
Local anaesthetic techniques
Local anaesthetic techniques should be part of the balanced anaesthetic protocol as they ensure optimal analgesia by blocking sensory nerves (see box on page 29). Application of these techniques results in a reduction in the amount of anaesthetic agents needed to maintain an adequate level of anaesthesia; this thereby improves patient safety by reducing side effects. Administration of long-acting local anaesthetic agents also ensures analgesia into the postoperative period. Contraindications for local anaesthetic techniques are infection and inflammation at the injection site. Performing a local anaesthetic technique in or around a location of suspected neoplasia is also a contraindication, to avoid spreading of neoplastic cells.
For extraoral approaches, the fur can be clipped and the skin surgically prepared before inserting the needle. The authors highly recommend that readers review the pertinent anatomy and practise the approaches using skulls.
There is no need for special instruments; a 1 ml syringe with a 25–27 G needle is all that is required to apply regional nerve blocks. While preloaded cartridges (glass cylinders that fit into a special dental syringe) are useful in larger animals, it is difficult to accurately administer the small volumes required in cats with these ready-to-use devices. It is advisable not to draw up more than the maximum recommended dose of local anaesthetic agent, to avoid over-dosing. In general, a maximum volume of 0.2–0.3 ml is recommended per injection site in cats. 19 To limit penetration of the periosteum, the needle should be positioned so that the bevel remains parallel to the bony surface and is directed towards the nerve to maximally expose the nerve to the anaesthetic agent.
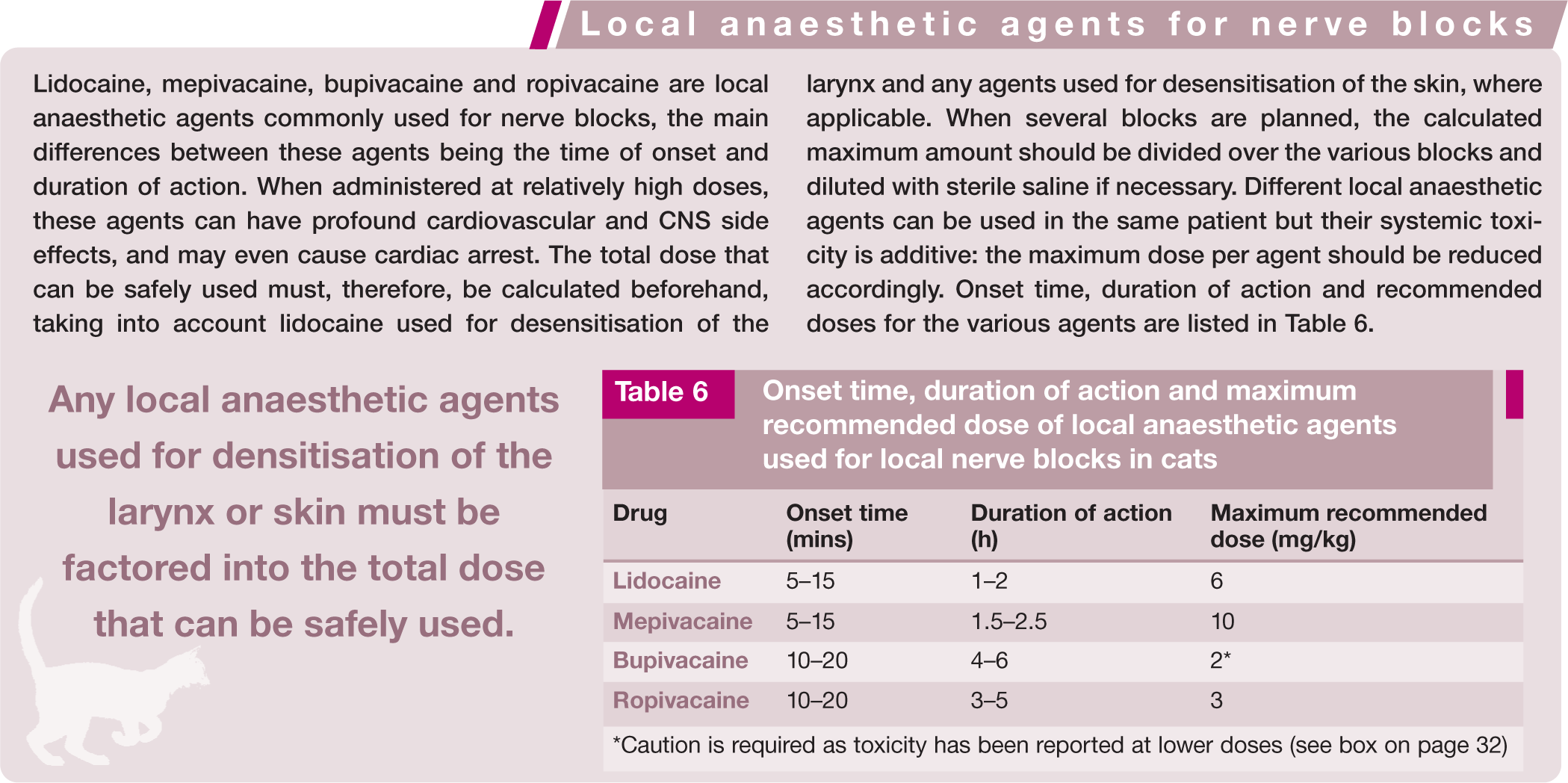
Onset time, duration of action and maximum recommended dose of local anaesthetic agents used for local nerve blocks in cats

Caution is required as toxicity has been reported at lower doses (see box on page 32)
Infraorbital nerve block
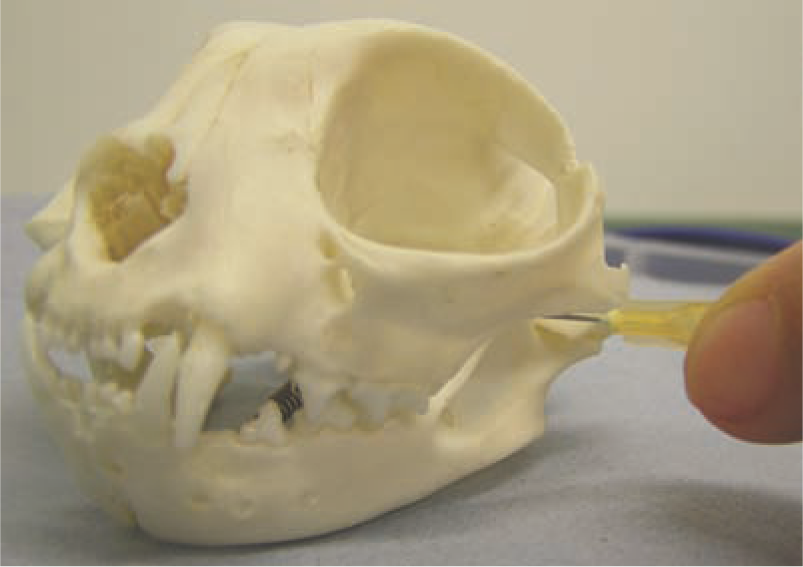
In cats, the infraorbital foramen is located just ventral to the ventral margin of the orbit, where a clear bony ridge (the lateral wall of the foramen) can sometimes be palpated above the third premolar (107/207) (Figure 8). The infraorbital nerve can be blocked within the rostral aspect of the infraorbital canal by aiming ventrally at about a 45° angle and for a very short distance (2–3 mm). However, this canal is very short in cats (up to 3–4 mm), 19 so there is the significant risk of ocular damage when a needle is inserted into the canal. Deposition of the local anaesthetic agent at the infraorbital foramen, followed by firm digital pressure applied over the injection site for 30–60 s, is anecdotally reported to stimulate diffusion of the agent into the canal.

A needle at the infraorbital foramen of a skull: the landmark for the approach to the infraorbital nerve block. The foramen is always dorsal to the third premolar
The transcutaneous or extraoral approach is preferred for the infraorbital block in cats as the tight upper lip often makes it difficult to insert the needle through the vestibular mucosa at the correct angle. 19
Unfortunately, this nerve block technique may not result in the desired desensitisation of all relevant structures, as injection of local anaesthetic agent on the nerve as it leaves the canal will generally not anaesthetise the teeth rostral to the foramen but only the skin and upper lip (Table 4). A carefully performed maxillary nerve block may, therefore, be an alternative technique.
Maxillary nerve block
The maxillary nerve can be blocked via either an extraoral or intraoral approach. For the extraoral approach, which is the preferred method, the needle is passed through the skin perpendicularly to the long axis of the palate, at the rostroventral aspect of the zygomatic arch, at the dorsocaudal limit of the hard palate (Figure 9). For the intraoral approach, the maxillary tuberosity (the thin caudal margin of the maxilla covering the roots of the molar tooth) is palpated and the needle is inserted underneath the finger, behind the maxillary molar (109/209), slightly caudomedial to the tooth. The needle is advanced at a slightly rostral angle towards the maxillary foramen, as perpendicularly as possible to the hard palate or in a slightly rostral direction, with the bevel directed rostrally. Care must be taken not to enter the retrobulbar space.
Extraoral approach for the maxillary nerve block
Mental nerve block
The middle mental foramen (the largest of the mental foramina) in cats is located equidistant between the mandibular canine and the third mandibular premolar (ie, between 304 and 307, and 404 and 407) (Figure 10). As with the infraorbital nerve block, deposition of local anaesthetic agent rostral to the foramen results in desensitisation of only soft tissues and not dental structures (Table 4). Identification of the middle mental foramen is very difficult in cats given its location medial to the lip frenulum; and concerns exist that penetration of the foramen with a needle may cause trauma to the nerve. 20 Therefore, the mandibular nerve block is a better alternative to this technique, and will also desensitise all ipsilateral mandibular teeth and associated bone and soft tissues.

Landmark for the approach to the mental nerve block
Mandibular nerve block
The mandibular nerve enters the mandibular canal via the mandibular foramen at the ventromedial aspect of the ramus of the mandible. The foramen and associated neurovascular bundle can be palpated on the medial aspect of the mandible on a straight line between the distal aspect of the mandibular molar (309/409) and the angular process of the mandible, and equidistant between these two landmarks. As for the maxillary nerve block, both an extraoral and an intraoral approach can be used.

For the extraoral approach the needle is inserted through the skin and advanced dorsally along the medial aspect of the mandible. A finger in the mouth at the mandibular foramen confirms that the needle is placed at the foramen for injection of the local anaesthetic agent (Figure 11).
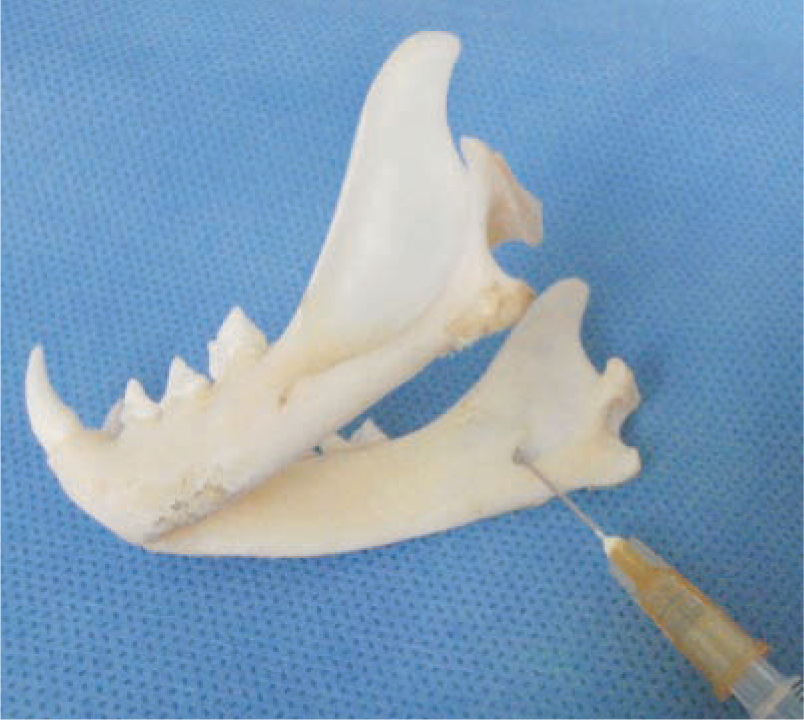
Landmark for the extraoral approach to the mandibular nerve block
For the intraoral approach, the needle is inserted between the distal aspect of the mandibular molar (either 309 or 409) and the molar salivary gland, and advanced on the medial aspect of the mandible, aiming for the angular process. It is important not to stray too medially with the needle, to avoid accidental block of the lingual nerve(s). Again, a finger in the mouth at the location of the mandibular foramen confirms accurate location of the tip of the needle and, if performed correctly, swelling of the tissues can be detected (Figures 12 and 13).

Landmark for the intraoral approach to the mandibular nerve block
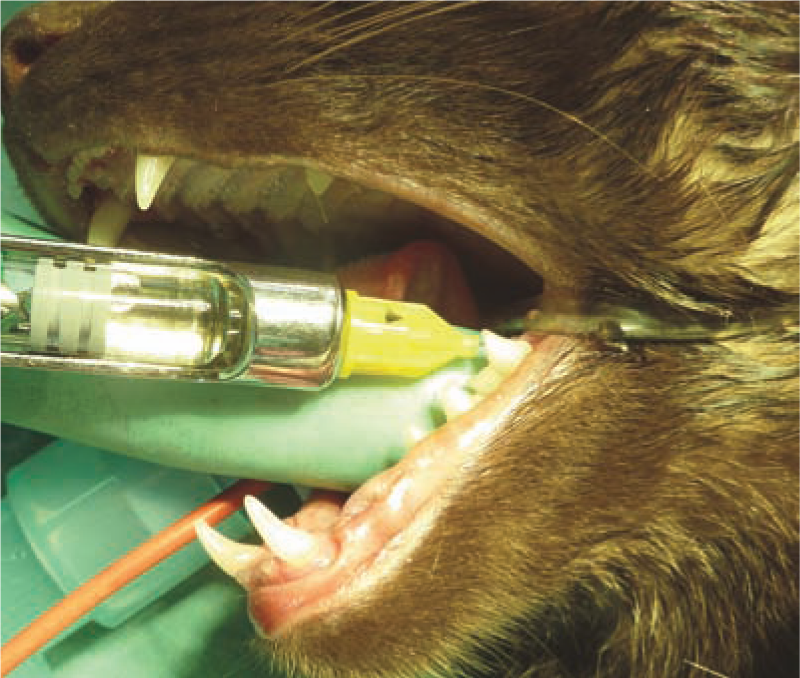
Intraoral approach to the mandibular nerve block. A gloved finger is in position to palpate the tip of the needle and ensure correct positioning at the mandibular foramen on the medial aspect of the mandible
Splash block
If a regional anaesthetic technique is not possible or unsuccessful (eg, the patient reacts to a painful stimulus in the affected area), a splash block can be performed with 2% lidocaine or 0.5% bupivacaine by bathing the alveolus or incision before closing. The agent should be left in place for 2 mins for adequate penetration into the nerve endings. This does not provide pre-emptive analgesia, but may still provide analgesia.

Opioids
Mu opioid receptor agonists provide excellent analgesia for moderate to severe pain. Methadone, morphine and buprenorphine are all suitable for use in cats.
Morphine may result in vomiting, especially in non-painful cats; its IV administration can potentially result in histamine release. Slow IV administration or IM injection is, therefore, advisable. Methadone is similar in action and duration to morphine but less likely to induce vomiting; it also has some antagonistic effects on NMDA receptors. Buprenorphine is a partial mu agonist and suitable for the treatment of mild to moderate pain. It is 100% bioavailable after oro-transmucosal/sublingual administration; the subcutaneous route is not recommended for its administration because of unreliable uptake and therefore effect. The multidose preparation of buprenorphine has been shown to be less favourable for the oro-transmucosal route than the 1 ml vials; its administration resulted in hypersalivation and less cooperative behaviour in one study. 24
Tramadol
Tramadol is a synthetic derivative of codeine but, strictly speaking, not an opioid. Its main action is via inhibition of serotonin and nor-adrenaline reuptake; some of its metabolites have (weak) mu opioid receptor agonist activity. Compared with dogs, cats produce high concentrations of O-desmethyltramadol, which has a high affinity for the mu receptor. 25 As tramadol is metabolised in the liver via demethylation and glucuronidation before excretion through the kidneys, a lower dose with a longer dosing interval is recommended in cats than in dogs. Unfortunately, not much information on the clinical use of tramadol in cats is available yet. Its use combined with the NSAID vedaprofen for analgesia in cats undergoing ovariohysterectomy has been described. 26 In another study, IM tramadol provided superior analgesia compared with pethidine in cats undergoing ovario-hysterectomy. 27 Its analgesic contribution to an anaesthetic protocol for orchiectomy in cats has recently been published. 28
NSAIDs
NSAIDs inhibit COX-1 and COX-2 enzymes, which are involved in prostaglandin synthesis. A variety of these drugs are licensed for use in cats, although none specifically for dental procedures.
NSAIDs vary in their selectivity for inhibition of the two COX enzymes. The most common adverse effects related to their use are gastrointestinal disturbances; these range from vomiting and diarrhoea to erosions, perforations and even death. Renal toxicity is another side effect because NSAIDs can alter renal haemodynamics, resulting in regional hypoperfusion and subsequent renal failure. Their use is contraindicated in hypovolaemic and hypotensive patients; and particular care has to be taken in patients with decreased liver and renal function.
The use of NSAIDs in cats with renal disease remains controversial as there is still a relative paucity of information on their chronic use in this species. Fear of toxicity exists, which may be due to the unique metabolism of the cat; its relative deficiency of glucuronyl transferase enzymes may result in a prolonged half-life for certain drugs. Meloxicam, piroxicam and robenacoxib are less likely to accumulate in cats, as their metabolism does not involve these enzymes. Long-term treatment with meloxicam for degenerative joint disease has been described in cats with both normal renal function and chronic renal disease.29,30 Life span was not reduced in the latter group compared with cats without renal impairment, while quality of life clearly had improved.
Patients should be carefully selected when NSAIDS are prescribed for long-term use and regularly assessed for adverse effects. If in doubt, withholding the administration of the NSAID until the patient is fully recovered from anaesthesia and normal hydration status and normotension are ensured is advisable. An excellent review and guidelines on the longterm use of NSAIDS in cats are available.31,32
Alpha-2 adrenoceptor agonists
Alpha-2 adrenoceptor agonists provide analgesia, sedation and muscle relaxation and act synergistically with opioids. Besides these desirable effects, these drugs also exert profound cardiovascular side effects – an increase in systemic vascular resistance, bradycardia and a significant reduction in cardiac output are common. Alpha-2 agonists should not be used in patients with cardiovascular compromise. Often, doses much lower than the manufacturer recommended doses are sufficient when these agents are used as part of pre-anaesthetic medication. However, even doses as low as 5 µg/kg IV result in a profound reduction in cardiac output.
NMDA-receptor antagonists
Ketamine and amantadine are, as mentioned, examples of NMDA-receptor antagonists that can be used for the treatment of chronic pain states. Ketamine can either be administered as a bolus or as an IV infusion. Amantadine is administered orally. Unfortunately, available capsules contain 100 mg and thus have to be split into more suitable amounts for use in cats. An oral solution exists as well (1 mg/ml); however, cats often resent this formulation so treatment via this route can be a challenge.
Post-anaesthetic care
As highlighted early on in the article, a significant percentage of feline fatalities occurs during the recovery period. Cats should, therefore, be closely monitored until fully recovered from anaesthesia. Pulse oximetry is invaluable during this period to detect hypoxaemia; this can develop after disconnection from the anaesthetic machine when the patient starts breathing room air instead of an oxygen-enriched gas mixture. Patients often still hypoventilate in the immediate postoperative period because of residual anaesthetic agents and hypothermia, which further increases the risk of hypoxaemia.
Ideally, dental patients should be kept intubated for as long as possible to maintain a patent airway and reduce the risk of aspiration. On the other hand, the feline upper respiratory tract is very sensitive and laryngeal spasm develops rapidly. A good time for extubation is when the ‘ear flick reflex’ (an ear twitch in response to gentle tickling of the inner pinna) is re-established, which is often slightly before the gag and cough responses to the ETT in situ occur. It is important to check the mouth, pharynx and larynx for any debris, blood, saliva and fluid before extubation. Throat packs or swabs should already have been removed by this stage.
Analgesia should be continued in the post-operative period (see Table 5). If not yet administered and no contraindications exist, an NSAID should be given. Shivering in response to hypothermia may increase oxygen requirements by as much as 400% and these patients benefit from oxygen supplementation and active warming until their body temperature has been sufficiently restored and shivering has ceased.
Anaesthesia-related complications
Cortical blindness
Post-anaesthetic blindness and CNS deficits have been described in cats.33,34 Potential causes are ischaemic optic nerve neuropathy, central retinal artery occlusion or undefined cerebral cortical ischaemia. Hypotension and hypoxaemia may also play a role, resulting in decreased perfusion of the optic nerve and/or inadequate oxygen supply. Other contributing factors to cortical blindness may include hypoventilation, apnoea, inadequate oxygen flow meter settings, inadequate oxygen: nitrous oxide ratio, incorrect set-up of the breathing system, monitoring device failure and enrofloxacin. Enrofloxacin is potentially retinotoxic in cats, and can result in acute and diffuse retinal degeneration.
Anaesthesia-related cortical blindness has a guarded prognosis, although vision may return after 1–2 weeks.
A recent study identified use of a mouth gag as a risk factor for post-anaesthetic blindness in cats. In 16 out of 20 cats that became blind after general anaesthesia, a mouth gag had been placed for either dental procedures or upper gastrointestinal endoscopy. 35 Fourteen cats regained vision, most within the first few days. One of the proposed theories for this acute blindness is that use of a spring-held mouth gag impairs blood flow to the brain through the maxillary artery by stretching of the vasculature and/or adjacent musculature, resulting in vascular compromise.35–37 Hypotension plays an important role in this too. It is strongly recommended that spring-held mouth gags are avoided in cats and that efforts are made to limit maximal opening of the mouth. Detection and treatment of hypotension is paramount, and use of IV fluids will be invaluable in this regard.
An alternative to a spring-held mouth gag is a 1 ml syringe cut to the appropriate length (Figure 14), or to use no mouth gag at all and maintain the mouth open with the non-dominant hand.

A cut 1 ml syringe is used as an alternative to a spring-loaded mouth gag to avoid excessive opening of the mouth
Tracheal damage
Damage to the trachea, especially after dental procedures, has been described in cats. 38 Disconnecting the ETT from the anaesthetic breathing system is recommended when changing the patient’s position, because rotation of the ETT with an inflated cuff can cause tracheal damage. Care must be taken not to overinflate the cuff of the ETT because high pressures within the cuff will impair tracheal mucosal blood flow and may result in post-intubation tracheal stenosis. In general practice, the safest way to ensure secure airways without overinflating the ETT cuff is to inflate the patient’s lungs with the valve on the breathing system (partially) closed while listening for the presence of a leak around the ETT. If a leak is indeed present, the ETT cuff should be gently inflated until the leak is no longer audible. It is important not to inflate the cuff without testing for a leak as sometimes a relatively snug fit is achieved when a large ETT is used; consequently there is a risk of overinflating the cuff when a standard amount of air is injected in the cuff. It should always be ensured that the valve of the breathing system is fully opened again immediately after leak testing.
Key Points
General anaesthesia is not contraindicated in older cats, as long as good perianaesthetic care is provided.
Use of an intravenous catheter is highly recommended in order to administer intravenous fluids. Intravenous anaesthetic agents should be titrated to effect.
Fluid therapy should form part of the (pre-)anaesthetic protocol to ensure adequate circulating blood volume and organ perfusion.
A multimodal analgesic protocol should be applied, including local anaesthetic techniques when indicated.
Complications of local nerve blocks are rare and should not dissuade the practitioner from incorporating these techniques in the anaesthetic protocol.
Because of the high risk of aspiration associated with oral and dentistry procedures, patients should be carefully intubated with a cuffed endotracheal tube.
Perianaesthetic care should include monitoring of body temperature, eye protection and appropriate provision of analgesia.
Footnotes
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this article.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
