Abstract
Objectives
Diabetic kidney disease (DKD) is a frequent and serious complication in human diabetic patients, but data are limited in cats. This study was undertaken to assess whether diabetic cats are susceptible to DKD.
Methods
Kidney function was compared between 36 cats with diabetes mellitus (DM), 10 cats with chronic kidney disease (CKD) and 10 age-matched healthy cats by measuring routine kidney variables (serum creatinine [sCreat], serum urea [sUrea], urine specific gravity [USG], urinary protein:creatinine ratio [UPC]), urinary cystatin C:creatinine ratio and glomerular filtration rate (GFR). Urinary cystatin C (uCysC) was measured with a human particle-enhanced nephelometric immunoassay, validated to measure feline cystatin C, in all but two diabetic cats. GFR was evaluated by exo-iohexol clearance in 17 diabetic cats, all cats with CKD and all healthy cats.
Results
Diabetic cats had significantly (mean ± SD) lower sCreat (123 ± 38 vs 243 ± 80 µmol/l), sUrea (11 ± 3 vs 18 ± 7 mmol/l) and urinary cystatin C:creatinine ratio (6 ± 31 vs 173 ± 242 mg/mol), and a significantly higher USG (1.033 ± 0.012 vs 1.018 ± 0.006) and GFR (2.0 ± 0.7 vs 0.8 ± 0.3 ml/min/kg) compared with cats with CKD. Compared with healthy cats, diabetic cats only had significantly lower USG (1.033 ± 0.012 vs 1.046 ± 0.008). Proteinuria (UPC >0.4) was present in 39% of diabetic cats, in 30% of cats with CKD and in none of the healthy cats. However, the UPC did not differ statistically between the three groups.
Conclusions and relevance
Based on evaluation of routine kidney variables, GFR and uCysC as a tubular marker at a single time point, a major impact of feline DM on kidney function could not be demonstrated.
Introduction
Both humans and cats are frequently affected by diabetes mellitus (DM), and the prevalence is rapidly increasing.1,2 Diabetic kidney disease (DKD) or diabetic nephropathy is a common and serious complication in human diabetics, particularly in type 2 DM. Diabetic nephropathy is characterised by glomerular alterations, resulting in altered glomerular filtration rate (GFR) and micro- or macroalbuminuria, tubular damage and hypertension. In the prediabetic or early diabetic phase, GFR is often increased (glomerular hyperfiltration), whereas decreased GFR is a typical finding for patients with more prolonged diabetes.2–4 Although the detection of persistent renal (micro)albuminuria is often used as an early marker for DKD, recent findings have revealed that renal impairment without albuminuria has become an increasingly common presentation of DKD in patients with type 2 DM. Also, many patients with microalbuminuria never progress to renal dysfunction. Hence, more sensitive and specific markers for early detection of DKD are needed.2,3,5–8 Many human patients with DKD have increased concentrations of urinary biomarkers, indicating tubular damage, such as retinol-binding protein (RBP), N-acetyl-β-
As feline diabetic patients mostly suffer from type 2 DM, cats might be susceptible to developing DKD.15,16 However, evidence of whether or not feline diabetics are at risk for kidney disease is scarce. On the one hand, a high prevalence of microalbuminuria and proteinuria has been described in diabetic cats. 17 On the other, hypertension is considered to be uncommon in diabetic cats,17–19 which is in contrast to human diabetic patients. Additionally, histological lesions of the kidney were not more frequent in diabetic cats compared with cats that died from other diseases. 20 To our knowledge, data on GFR and urinary biomarkers in cats with DM are currently lacking.
This study was undertaken to assess whether diabetic cats are susceptible to DKD. Therefore, routine kidney variables (ie, serum creatinine [sCreat] and urea [sUrea] concentrations, urine specific gravity [USG] and urinary protein:creatinine ratio [UPC]), GFR and uCysC were compared between cats with DM, cats with chronic kidney disease (CKD) and healthy cats.
Materials and Methods
Animals
Cats with DM, cats with CKD and healthy cats were prospectively included. DM was diagnosed based on compatible clinical signs, persistent hyperglycaemia and glucosuria. Glycaemic control was evaluated based on the combination of history, bodyweight, blood glucose level, serum fructosamine concentration and, if available, blood glucose curve in the hospital or at home. Glycaemic control was considered good if all these parameters indicated good metabolic control (eg, absence of polyphagia, polydipsia and polyuria; stable bodyweight; blood glucose level of 10–15 mmol/l with serum fructosamine <470 µmol/l; blood glucose curve with mean blood glucose level 10–15 mmol/l). Glycaemic control was considered poor if these parameters indicated poor glycaemic control (eg, presence of polyphagia, polydipsia, polyuria, weight loss; blood glucose >15 mmol/l; fructosamine >600 µmol/l). The diabetes was considered moderately controlled in cats with several (at least two) parameters indicating poor glycaemic control and the other parameters indicating good glycaemic control. The diagnosis of CKD was based on compatible history and clinical findings, sCreat >140 µmol/l and USG <1.035. 21 Healthy was defined as the absence of clinical signs and significant abnormalities on physical examination and routine laboratory analysis (see below). Efforts were taken to age-match the healthy cats to the diabetic cats.
Routine physical examination (including thyroid gland palpation), complete blood count, serum biochemistry profile (including total thyroxine concentration in cats older than 6 years) and urinalysis (including UPC and bacterial culture) were performed to assess the general health status of all included cats. Exclusion criteria for all groups were the presence of hyperthyroidism or concurrent significant systemic disease and treatment with angiotensin-converting enzyme inhibitors and antihypertensive drugs at the time of inclusion. The presence of azotaemia was not an exclusion criterion for the DM group. Diabetic cats that were in diabetic remission and did not receive insulin therapy were not included. In the CKD group, only cats with International Renal Interest Society (IRIS) stages 2–3 were included. 21 In the healthy group, USG <1.035 as a single abnormality was not an exclusion criterion.
The study was completed at the Department of Small Animal Medicine and Clinical Biology, Faculty of Veterinary Medicine, Ghent University. All cats were privately owned, the owners were thoroughly informed about the study’s aims and protocol, and the study was approved by local and national ethical committees (EC2010_029).
Procedures
Routine kidney variables were measured in all cats, namely sCreat (modified Jaffé assay; Architect C16000, Abbott), sUrea (enzymatic assay; Architect C16000, Abbott), USG (traditional hand-held optical refractometer) and UPC (Iricell IQ, Instrumentation Laboratory). Hospital-specific reference intervals for sCreat (64.5–161.8 µmol/l) and sUrea (12.7 mmol/l) were developed using 130 healthy cats of various ages and weights. Urine bacterial culture (BioMerieux) was performed, and the urinary sediment was evaluated as previously described.22,23 The tubular marker uCysC was determined in all cats. Urine was stored at −80°C until batched analysis. The uCysC concentration was measured with a human particle-enhanced nephelometric immunoassay (Behring Nephelometer ProSpec; Siemens Healthcare Diagnostics), previously validated to measure feline cystatin C, 24 and expressed as uCysC:urinary creatinine ratio (uCysC/uCreat).
A combined plasma exogenous creatinine–iohexol clearance test (PEC–ICT) was performed in cats weighing ⩾3 kg with easy, unstressed and non-aggressive behaviour, and for which the owner gave permission. The PEC–ICT was performed as previously reported.25,26 Briefly, all cats received 40 mg/kg creatinine and 64.7 mg/kg iohexol intravenously. Blood samples were taken in tubes with EDTA as the anticoagulant before and 5, 15, 30, 60, 120, 180, 360, 480 and 600 mins after injection. Plasma was stored at −80°C until analysis. For this study, GFR was determined from exo-iohexol clearance data. Exo- and endo-iohexol concentrations were determined using a validated high-performance liquid chromatogr-aphy method with ultraviolet detection.25,27 To calculate the dose of exo-iohexol from total dose administered, the ratios of exo- and endo-iohexol stereoisomers were quantified for each Omnipaque solution administered. Pharmacokinetic analyses were performed using WinNonlin version 4.0.1 (Scientific Consulting). The plasma data were subjected to non-compartmental analysis with a statistical moment approach. The area under the plasma concentration vs time curve (AUC) was calculated by the trapezoidal rule with extrapolation to infinity, as described by Watson et al. 28 Plasma clearance of exo-iohexol was determined by dividing the exo-iohexol dose administered by AUC and indexed to bodyweight (ml/min/kg).
The study design included systolic blood pressure (SBP) measurement in all cats that underwent a clearance test for GFR determination. Whether or not SBP was measured in the other cats depended on the responsible clinicians’ decision. To measure SBP, the Doppler ultrasonic technique and a standardised procedure following the consensus statement of the American College of Veterinary Internal Medicine (ACVIM) 29 were used. Hypertension was defined as SBP >160 mmHg.29,30
Statistical analysis
All statistical tests were performed with Systat 12 (Systat Software) and at the 0.05 significance level.
The effect of the disease (DM, CKD, healthy) on age, bodyweight, SBP, sCreat, sUrea, USG, UPC, uCysC/uCreat and GFR was tested by ANOVA. In case of a global significant difference, post hoc pairwise comparisons were performed using the Tukey test. P values were adjusted for multiple comparisons by Bonferroni correction. For cats with uCysC concentrations below the limit of quantification, the uCysC concentration was considered to be ‘zero’.
Results
In total, 56 cats were included, namely 36 diabetic cats, 10 cats with CKD and 10 healthy cats. Routine kidney variables (sCreat, sUrea, USG and UPC) and uCysC/uCreat were measured in all cats, except for sUrea in one diabetic cat and uCysC/uCreat in two diabetic cats because of insufficient sample volume. The exo-iohexol GFR was determined in 17 cats with DM, all cats with CKD and all healthy cats. Owing to technical problems SBP could not be measured in three of the diabetic cats and one healthy cat that underwent a clearance test. In total, SBP was measured in 20 diabetic cats, all CKD cats and nine healthy cats.
Breed distribution consisted of 29 domestic short- or longhair cats and seven purebred cats (two Burmese, one Siamese, one Russian Blue, one Oriental, one British shorthair, one Norwegian Forest Cat) in the diabetic group; seven domestic short- or longhair cats and three purebred cats (one British shorthair, one Persian, one Siamese cat) in the group of CKD cats, and 10 domestic short- or longhair cats in the group of healthy cats. The diabetic group involved 27 male (25 neutered, two intact) and nine female (seven neutered, two intact) cats, and the CKD and healthy cat groups involved three male (all neutered) and seven female (all neutered) cats.
Eight diabetic cats were not yet treated with insulin at the time of inclusion because they were newly diagnosed, seven received insulin for a period of between 2 and 4 weeks, 15 were treated between 1 month and 1 year, and six were treated longer than 1.5 years to a maximum of 5 years. Four cats intermittently received insulin, but all were receiving insulin treatment at the time of inclusion. Glycaemic control was good in four cats, moderate in eight and poor in 24. At the time of inclusion, the mean serum fructosamine concentration in the diabetic group was 515.6 ± 160.1 µmol/l (n = 33).
The descriptive statistics for age, bodyweight, SBP, sCreat, sUrea, USG, UPC, uCysC/uCreat and GFR of the three groups are presented in Table 1. The box-plots for the GFR and uCysC/uCreat values of the three groups are shown in Figures 1 and 2. Between the three groups (DM, CKD, healthy) significant differences were not detected for age, bodyweight, SBP or UPC. Cats with DM had significantly lower sCreat and sUrea (P <0.001), higher USG (P = 0.003), lower uCysC/uCreat (P <0.001) and higher GFR (P <0.001) than cats with CKD. The same parameters also differed significantly between healthy cats and cats with CKD, which was expected based on our inclusion criteria. Diabetic cats only had significantly lower USG compared with healthy cats (P = 0.006).
Mean ± SD (median [range]) age, bodyweight, systolic blood pressure (SBP), serum creatinine concentration (sCreat), serum urea concentration (sUrea), urine specific gravity (USG), urinary protein:creatinine ratio (UPC), urinary cystatin C:creatinine ratio (uCysC/uCreat) and exo-iohexol glomerular filtration rate (GFR) for 36 cats with diabetes mellitus (DM), 10 cats with chronic kidney disease (CKD) and 10 healthy cats

Age, bodyweight, sCreat, USG and UPC were available in all cats; sUrea in all cats except for one cat with DM; uCysC/uCreat in all cats except for two cats with DM; GFR in 17 diabetic cats and in all CKD cats and healthy cats; and SBP in 20 diabetic cats, all CKD cats and nine healthy cats
Mean values differed significantly between cats with DM and CKD
Mean values differed significantly between cats with CKD and healthy cats
Mean values differed significantly between cats with DM and healthy cats
LOQ = limit of quantification
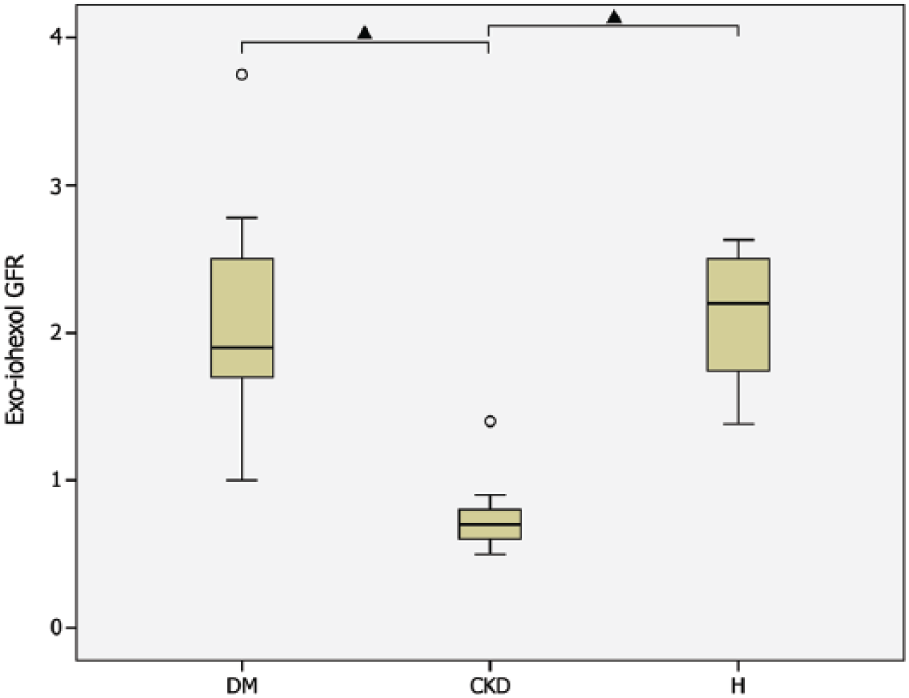
Box plots of exo-iohexol glomerular filtration rate (GFR; ml/min/kg) for cats with diabetes mellitus (DM; n = 17), cats with chronic kidney disease (CKD; n = 10) and healthy cats (H; n = 10). The triangles indicate significant differences between groups

Box plots of urinary cystatinC:creatinine ratio (uCysC/uCreat; mg/mol) for cats with diabetes mellitus (DM; n = 34), cats with chronic kidney disease (CKD; n = 10) and healthy cats (H; n = 10). The triangles indicate significant differences between groups
Four (11%) diabetic cats had an sCreat above the reference interval (>161.8 μmol/l; maximum 229 µmol/l) and nine (25%) diabetic cats had an increased sUrea (>12.7 mmol/l; maximum 20.2 mmol/l). Four (11%) diabetic, eight (80%) CKD cats but no healthy cats had USGs between 1.007 and 1.020; 17 (47%) diabetic cats, two (20%) CKD cats and one (10%) healthy cat had USGs between 1.021 and 1.035; and 15 (42%) diabetic cats, no CKD cats and nine (90%) healthy cats had USGs between 1.036 and 1.060. The only healthy cat with USG <1.035 had a normal GFR (2.4 ml/min/kg). Glucosuria was detected in 29 (81%) diabetic cats but all other cats were negative on urine dipstick for glucose. Ketonuria was not detected in any of the cats. According to the ACVIM Consensus Statement on proteinuria, 31 23 (41%) cats did not have proteinuria (UPC <0.2; 14 [39%] DM, three [30%] CKD, six [60%] healthy cats), 16 (29%) had borderline proteinuria (UPC 0.2–0.4; eight [22%] DM, four [40%] CKD, four [40%] healthy cats) and 17 (30%) had overt proteinuria (UPC >0.4; 14 [39%] DM, three [30%] CKD, no healthy cats). The urine bacterial culture was unavailable in one diabetic cat but was negative in all other cats. Urinary sediment analysis revealed microscopic haematuria (>20 red blood cells per high power field; × 40 objective) in two cats (one [3%] DM, one [10%] CKD) and a moderate amount of struvite crystals (six per low power field; × 10 objective) in one (10%) healthy cat. The urinary sediment of the other cats did not show significant abnormalities.
For the diabetic cats with increased sCreat or sUrea, two had an increase of sCreat only, seven of sUrea only and two had an increase of both sCreat and sUrea. Insulin treatment had not yet been started in one cat, one cat had been treated for <1 month, four cats had been treated for between 1 and 12 months, and five cats had been treated for >1 year. In the treated cats, diabetic control was considered good in three, moderate in three and poor in four. USG was between 1.007 and 1.020 in two cats, between 1.021 and 1.035 in six, and between 1.036 and 1.060 in three. Glucosuria was detected in seven cats. The cat with the highest sCreat had a GFR of 1 ml/min/kg and was diagnosed with CKD. In three other cats, with a mildly increased sUrea, GFR was 1.8, 1.8 and 1.9 ml/min/kg, respectively. GFR was not measured in the other cats with increased sCreat and/or sUrea.
Hypertension was present in two (10%) diabetic cats (SBP 165 and 220 mmHg), none of the cats with CKD and in two (20%) healthy cats (SBP 180 and 190 mmHg). All other cats were normotensive. The diabetic cat with a SBP of 165 mmHg also had mild proteinuria (UPC 0.5) but otherwise normal kidney parameters, including GFR. Although recommended, repeated SBP measurement was not performed. This cat was euthanased approximately 1 year after inclusion because of relapse of overt DM after a period of diabetic remission. Clinical signs of CKD were never noticed, but laboratory tests were not repeated after inclusion. The other hypertensive diabetic cat (SBP 220 mmHg) was treated with radioactive iodine for hyperthyroidism 7 years prior to inclusion. At the time of inclusion the cat had mild azotaemia with a USG of 1.020 and proteinuria (UPC 1.1), GFR was not measured. Repeated SBP measurements confirmed hypertension and further work-up 1 year after inclusion revealed iatrogenic hypothyroidism. Both healthy cats with hypertension were very anxious during the examination, which makes white-coat hypertension most likely.
Discussion
This study evaluated if there is an association between feline DM and kidney dysfunction, as there is in human medicine. Therefore, routine kidney variables, a tubular urinary marker and GFRs of diabetic cats were compared, at a single time point, with those of healthy cats and cats with CKD. Major changes in kidney function could not be demonstrated in feline diabetic patients.
Most parameters significantly differed between cats with DM and CKD, which indicates that the diabetic cats in this study did not have obvious renal dysfunction. Compared with healthy cats, the diabetic group only had significantly lower USG. As most diabetic cats of this study had glucosuria, the lower USG is probably related to osmotic diuresis. This hypothesis is strengthened by the fact that most diabetic cats of this study had USGs >1.020, which reflects renal concentrating ability in polyuric glucosuric animals. However, it should be kept in mind that marked glucosuria may falsely increase USG. 32
In the diabetic group in this study, increased sCreat and sUrea were detected in 11% and in 25% of the cats, respectively. In a recent study, 17% of cats newly diagnosed with DM or diabetic ketoacidosis had increased sCreat. 33 In the same study, having higher sCreat at diagnosis appeared to be associated with decreased survival time, but whether the azotaemia of these diabetic cats was prerenal or renal in origin was not reported. Whether the cats in the present study with increased sCreat and/or sUrea had prerenal or renal azotaemia is difficult to answer because many cats had unconcentrated urine in the presence of glucosuria, because GFR was not available in most of the cats and the majority of cats were evaluated at only a single time point. Renal azotaemia due to CKD IRIS stage 2 was confirmed in two cats, by measuring GFR in one cat and by follow-up in the other cat.
Although the mean UPC was higher in diabetic cats than in healthy cats, and approximately 40% of diabetic cats had proteinuria compared with none of the healthy cats, this did not result in a statistically significant different UPC between both groups. The prevalence of proteinuria in diabetic cats has been evaluated in two studies. Sennello et al 19 did not detect proteinuria in 12 diabetic cats, but a cut-off for proteinuria of 1 was used, which nowadays is considered high, based on the ACVIM consensus on proteinuria. 31 In contrast, in a more recent study, 75% of feline diabetics had UPCs >0.4 and the UPC of diabetic cats was significantly higher than that of healthy and sick control cats. 17 Differences in proteinuria prevalence between this study and our present study might be explained either by technical or methodological differences in UPC measurement, 34 or by differences in study population, such as duration of DM and degree of glycaemic control. The conflicting results of studies evaluating proteinuria in diabetic cats might argue for further investigation, for instance by measuring the urinary albumin:creatinine ratio (UAC). In human studies, proteinuria is more commonly quantified by measuring UAC than by UPC, both in diabetic and in non-diabetic patients. Although microalbuminuria is used as an early marker for DKD, there is no obvious evidence that UAC is superior to UPC once overt albuminuria is present.2,35 The effect of DM on UAC is poorly studied in veterinary medicine. In one study in diabetic dogs, an increase in UPC and UAC was commonly present, and some diabetic dogs only had an increased UAC with normal UPC value. Thus, UAC might have additional value to UPC for the detection of early renal damage in diabetic dogs. 36 To our knowledge, data are currently unavailable in cats. Another important question is whether this low-level proteinuria (UPC 0.4–1.0), which commonly affects feline diabetic patients, is an early marker for more severe renal dysfunction and whether these cats will develop DKD with more prolonged DM. On the one hand, in cats with CKD, it is accepted that low-level proteinuria is a negative prognostic factor.37–39 On the other, the prognostic significance of low-level proteinuria in non-azotaemic cats is less studied. However, preliminary data indicate that it might be associated with reduced survival times. 40 Borderline proteinuria was detected in 22% of diabetic cats and 40% of healthy cats. A high prevalence of borderline proteinuria in apparently healthy cats has recently been reported by our group, but the clinical significance of this finding remains unknown.22,23
Altered GFR, initial glomerular hyperfiltration and, at a later stage, glomerular hypofiltration, is typical of DKD in humans.2,41 In contrast, our diabetic cats did not show significant changes in GFR compared with healthy cats. Only one of the diabetic cats, diagnosed with DM the day before entering the study, was suspected of having glomerular hyperfiltration (Figure 1). The GFR of this cat exceeded the GFR values for healthy cats of the current study and a previous study from our group, 42 and also exceeded the mean GFR value for hyperthyroid cats (3.3 ml/min/kg). 43 Only one diabetic cat in the present study had a GFR below the low GFR cut-off (1.2 ml/min/kg) for exo-iohexol clearance in the previous study 42 This indicates that almost all our diabetic cats had normal GFR.
In contrast to human patients with DM who frequently suffer from hypertension, particularly patients with type 2 DM, 4 only 10% of our diabetic cats had hypertension. This is comparable with 15% previously found in cats. 17 In two other reports, hypertension was not detected in any of the diabetic cats, but a cut-off of 180 mmHg was used to define hypertension.18,19 Although the latter two studies are limited by this inappropriately high cut-off to define hypertension, 29 the current veterinary literature suggests that hypertension is uncommon in feline diabetic patients.
In our study, we could not detect significant differences in uCysC levels between diabetic and healthy cats. However, this parameter needs to be interpreted cautiously because most diabetic cats (n = 31) had undetectable concentrations of uCysC. Somewhat unexpectedly, uCysC also could not be found in several cats with CKD (n = 5). In contrast, in a previous study – using the same human nephelometric assay – all cats with CKD, but none of the healthy cats, had measurable uCysC concentrations. 24 The urine samples in our study were stored up to 3 years at −80°C before analysis of cystatin C compared with a maximum of 1.4 years in the study of Ghys et al. 24 Cystatin C is considered to be a stable protein in human medicine, and freezing or freeze/thaw cycles do not affect its concentration.44–46 Therefore, stability of uCysC was assumed, but the stability of feline cystatin C in serum and urine is currently under investigation at our institution. Although uCysC is a good marker for early detection of diabetic nephropathy in human medicine, it is possible that uCysC is a less ideal tubular marker in diabetic cats. Another tubular marker that has been studied in cats is RBP.47,48 In an in-house pilot study, significant differences in exo-iohexol GFR and urinary RBP:uCreat ratio were not detected between seven diabetic and five age-matched healthy cats. 49 The combined results of both urinary markers might indicate that cats with DM are less sensitive to tubular damage compared with human diabetics.
Humans with DKD may have variable structural changes of the kidneys, even before renal dysfunction occurs. The most typical structural renal changes of DKD are thickening of the glomerular and/or tubular basement membrane and mesangial expansion.50,51 Although a small study of cats with persistent hyperglycaemia detected similar changes in some cats, 52 a recent study did not detect more frequent histological glomerular, tubulointerstitial lesions or vascular lesions in the kidney of diabetic cats compared with cats that died from other diseases. 20 The latter study is in line with our findings suggesting that DKD does not seem to be of major importance in feline diabetic patients. Unfortunately, both veterinary studies only evaluated the kidneys by light microscopy.20,52 Maximum information can only be obtained by evaluating kidney biopsies with a combination of light, electron and immunofluorescent microscopy,53,54 as is routinely performed in human patients with diabetes.55–57
Several reasons might explain why cats are not as sensitive as humans to developing DKD. The shorter duration of DM in cats might be a major issue. In human patients with type 2 DM, progression to overt nephropathy usually takes >10 years and progression to end-stage renal disease >30 years. 58 In the present study population, only 6/36 diabetic cats were treated for >1.5 years with a maximum of >5 years. Genetic predisposition, which is an initiator of DKD in humans, 59 might be less important in cats, but studies are currently lacking. Several promoters of human DKD, such as hypertension and dyslipidaemia, are less common in cats, and others, such as smoking, are not relevant for cats. 59 Although hypertension is a frequent complication in human diabetic patients, 4 this does not appear to be true in cats. In this study only 10% of diabetic cats had hypertension, which is comparable to the 15% found by Al-Ghazlat et al. 17 Also, primary hypertension is a major problem in human medicine but not in feline medicine. 60 Around 18–20% of hypertensive cats are reported to have primary hypertension,61,62 but subclinical CKD and primary hyperaldosteronism may have been overlooked as underlying causes for hypertension in these studies. 60 Dyslipidaemia in humans is associated with atherosclerosis and microvascular dysfunction, leading to renal and cardiovascular disease.59,63,64 Hypercholesterolaemia has been described in diabetic cats,65,66 and obese cats have dyslipidaemia, characterised by abnormalities in lipoprotein particle number and size. 67 In addition, atherosclerosis can be induced in cats by feeding them a high-fat, cholesterol-enriched diet. 68 However, spontaneous cases of cats with atherosclerosis have not been described and atherosclerosis is not a feature of feline obesity or diabetes, which contrasts markedly with the situation in humans. The lack of an apparent systemic inflammatory reaction in response to increased fat mass in cats might explain why atherosclerosis and cardiovascular problems do not occur in obese cats. 69
Firstly, our study was limited by not measuring GFR and SBP in all diabetic cats. Secondly, the patients were evaluated only at a single time point without performing follow-up. Thirdly, we were also not able to evaluate the influence of glycaemic control on kidney function because only a small number of cats had good glycaemic control at the time of inclusion. Fourthly, the different sex and breed distribution between groups may be potential confounding factors. The final limitation is the lack of histopathological examination of renal tissue.
Conclusions
By evaluating routine kidney variables, GFR and uCysC as a tubular marker at a single time point, we could not demonstrate major changes in kidney function in cats with DM. In cats with concurrent DM and CKD, the question of whether DM (partially) causes CKD or whether both diseases are unrelated cannot be fully answered. Still, the current veterinary literature does not support a strong relationship between both diseases. However, follow-up studies, mainly to reveal the clinical significance of low-level proteinuria, are required in diabetic cats.
Footnotes
Acknowledgements
We wish to thank Ms J Lambrecht and Mrs E Lecocq for their laboratory assistance. The results of this study were presented, in part, at the 23rd European College of Veterinary Internal Medicine – Companion Animals (ECVIM-CA) Congress, Liverpool, 12–14 September 2013 and published as an abstract in 23rd ECVIM-CA Congress Proceedings p 243.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Funding
This research received no grant from any funding agency in the public, commercial or not-for-profit sectors.
