Abstract
Insulin detemir is a long-acting insulin analogue and may represent a valuable treatment option for diabetic cats. So far, only one study addressing detemir treatment of diabetic cats has been published, and this was based on an intensive blood glucose monitoring protocol. The aim of the current, retrospective study was to evaluate the effect of detemir therapy in diabetic cats in a general clinical setting. Fourteen diabetic cats with a follow-up period of at least 3 months were included. Data were collected from medical records at the University Hospital for Companion Animals, University of Copenhagen, Denmark. Thirteen of 14 cats achieved moderate or excellent control of clinical symptoms within the initial 3 months of detemir therapy, including five cats previously treated unsuccessfully with other types of insulin. Clinical improvements were noted after 1 month of therapy and continued over time. Three cats achieved remission within the initial 3 months and none experienced a diabetic relapse during the study period. One cat achieved remission after 13 months of therapy. Improvements in clinical symptoms were markedly better than indicated by blood glucose and serum fructosamine concentrations. The safety of detemir was very high, with only two reported episodes of clinical hypoglycaemia, neither of which required veterinary attention. Based on these results detemir can be recommended for the treatment of diabetic cats, including cats previously treated unsuccessfully with other types of insulin.
Introduction
Diabetes mellitus (DM) is a common endocrine disease in domesticated cats, with a prevalence of approximately 0.5% in studied populations.1,2 Most cases of feline DM closely resemble human type 2 DM, which is characterised by a decreased insulin sensitivity in peripheral tissues in combination with an inadequate insulin production in the pancreatic islets (due to beta-cell failure). 3 Clinical symptoms of DM become evident when insulin secretion is no longer able to compensate for the increased insulin requirement. 4
Lente insulin (Caninsulin; Intervet) has been widely used to treat feline diabetes. The duration of effect in cats is approximately 8 h (8–14 h), which necessitates at least twice-daily administration to achieve acceptable glycaemic control.5–7 Even with this protocol, many cats will experience periods of hyperglycaemia during the day, leading to an increased risk of glucose toxicity and disease progression.6,8 Therefore, lente insulin may be suboptimal for the treatment of certain diabetic cats, and assessment of other types of insulin is reasonable.
In recent years, long-acting human insulin analogues (eg, glargine [Lantus; Sanofi Aventis] and detemir [levemir; Novo Nordisk]) have been used to treat feline diabetes. Glargine has a significantly longer duration in cats than lente insulin and has provided encouraging results in the treatment of feline DM.5,9 Glargine, in combination with a high-protein, low-carbohydrate diet, has resulted in higher diabetic remission rates than previously reported for other types of insulin.9–11 To achieve maximum glycaemic control and to increase the chance of remission, administration of glargine twice daily is recommended.10,12
In humans, detemir is slowly absorbed from the subcutaneous injection site and displays a prolonged action compared with other types of insulin. 13 Formation of dihexamers and the binding of detemir to albumin are believed to be the mechanisms behind the slow absorption of detemir.13,14 These features could be relevant for use in the treatment of feline DM. So far, detemir has only been the subject of a limited number of feline studies.15,16 In healthy cats, the pharmacodynamics of detemir and glargine were comparable. 16 The only published study involving diabetic cats reported a high remission rate within the first 5 months of treatment with detemir, and only 1/18 cats experienced mild hypoglycaemia. 15 However, this study involved a protocol aimed at intensive blood glucose (BG) monitoring, with multiple (3–7) daily BG measurements at home; therefore, it may not be representative of the standard capability of owners of feline diabetic patients. 15 The objective of this study was to report the treatment outcome in a group of diabetic cats treated with a protocol based on the guidelines from the American Animal Hospital Association (AAHA). 17 We believe that this study may provide a more realistic reflection of the results to be expected when using detemir in a general veterinary practice setting.
Materials and methods
Medical records for cats diagnosed with DM at the University Hospital for Companion Animals (UHC), University of Copenhagen, Denmark, between January 2007 and May 2013 were reviewed. Diagnosis of DM was based on clinical symptoms, including polyuria and polydipsia, and the combined results of blood and urine samples (hyperglyacaemia, increased serum fructosamine and glycosuria). The following were required for inclusion: (i) at least 3 months of follow-up from initial diagnosis; (ii) a minimum of three hospital visits (initial visit followed by a minimum of two follow-up visits) and/or three serial BG measurements during the first 3 months of detemir therapy. Cats were included if they had been treated consecutively with detemir for 3 months, regardless of any previous therapy with another type of insulin. The cut-off for data collection was June 2013, regardless of disease status at that time.
The following data were collected from the medical records: signalment, history and physical examination findings, as well as the results of complete blood counts, biochemical profiles and urinalysis from each visit. Information regarding cause of death and occurrence/duration of remission was also collected when applicable. Diabetic remission was defined as at least 4 weeks of glycaemic control without exogenous insulin therapy. 18 Where available, data from home monitoring (eg, BG curves [BGCs]) were also included.
The applied treatment protocol for detemir was in agreement with the AAHA diabetes management guidelines for treatment with insulin glargine and based on recommendations by Rucinsky et al and Rand and Marshall.17,19 The starting dose of detemir was based on BG concentration: an initial dose of 0.5 U/kg twice daily if the BG was >20 mmol/l (360 mg/dl) and 0.25 U/kg twice daily if the BG was <20 mmol/l (360 mg/dl). This protocol was also applied to cats previously treated with other types of insulin, as there can be individual variations in the time–action profile of detemir, and it was unknown how the cats would react to the change in insulin type. 16 For all cats, the dose was further adjusted based on preinsulin BG, BG nadir during a 12 h serial BGC and the clinical symptoms. BGCs were generated at home or in the hospital using a portable glucometer (Contour; Bayer). Several handheld glucometers have been shown to provide clinically acceptable readings. 20 During the 12 h BGC the BG was measured every 4 h from immediately before the morning insulin injection and ending just before the evening dose of insulin. 17 A mean BG (MBG) was calculated for each BGC as the average of the four measurements. During the first week of treatment no dose adjustments were made unless hypoglycaemic episodes were confirmed or suspected. Conducting a 12 h serial BGC was recommended at home or in the hospital every 10–14 days until acceptable glucose control was achieved. A preinsulin BG between 10 and 15 mmol/l (180–270 mg/dl), and a nadir between 5 and 10 mmol/l (90–162 mg/dl) were considered to show an appropriate level of glycaemic control.17,19
Glycaemic control was also assessed based on clinical symptoms and serum fructosamine level. Normalisation of clinical symptoms was considered excellent control, moderate control was the improvement of clinical symptoms without resolution (eg, still some degree of polydipsia and polyuria) and poor control was lack of improvement in clinical symptoms. 21 Excellent and moderate glycaemic control were expected to be associated with a serum fructosamine level of <450 µmol/l or <550 µmol/l, respectively. 22 Dose adjustments were kept in the range of 0.5–1.0 U/injection. In the case of clinical hypoglycaemia, the dose was reduced by 50%.
Owners were encouraged to feed a high-protein, low-carbohydrate diabetes diet as an integral part of the treatment plan unless a renal diet was indicated by concurrent kidney disease. 17 Cats were fed at the time of insulin administration or had unlimited access to food. The necessity of maintaining an optimal body condition was emphasised and weight loss, if required, was encouraged.
Statistical analysis
Quantitative descriptive data are presented as the median and range. Measurements of serum fructosamine concentration, preinsulin BG and nadir BG from each cat were divided into three categories based on time of measurement from initiation of detemir treatment: baseline; day 25–60; and day 61–105. To evaluate any change in these measurements over time, the time categories were compared using the Wilcoxon matched pairs signed rank sum test. The Kruskal–Wallis test was used for comparison of unpaired measurements such as starting dose of insulin. Differences were considered significant at values of P ⩽0.05. Data are illustrated with individual profile plots, showing the individual response over time.
Results
Signalment
Thirty diabetic cats treated with detemir were identified from the medical records from the UHC. Of these, 14 cats fulfilled the inclusion criteria and were included in the study. Sixteen cats were excluded: 4 owing to euthanasia within the initial 3 months, 7 were lost to follow-up within the initial 3 months and 5 had incomplete follow-up data; three of the four cats were euthanased within 12 days of diagnosis (owing to owners’ wishes or concurrent disease), and one cat was diagnosed with acute pancreatitis/cholangitis and renal failure 60 days after receiving a diagnosis of DM. The remaining five cats had incomplete follow-up data.
Eleven of the 14 included cats were domestic shorthairs. The other cats were Maine Coon (n = 1), Somali (n = 1) and Norwegian Forest (n = 1). Twelve cats were males (two intact and 10 neutered) and two cats were neutered females. The median age at time of initiation of detemir treatment was 11.0 years (range 3.5–16.9 years). The median total hospital follow-up time was 13.4 months (range 3.0–56.0 months).
Cats given prior insulin treatment
Five cats had been treated with another type of insulin prior to treatment with detemir. Four cats had been treated with lente insulin (Caninsulin; MSD) for a period of between 3 weeks and 3 months. For one cat the type and duration of prior insulin therapy was unknown. All five cats were second opinion cases owing to persistent polyuria/polydipsia (n = 4), lethargy (n = 2), weight loss (n = 1), polyphagia (n = 1), anorexia (n = 1), vomiting and diarrhoea (n = 1) and hindlimb weakness (n = 1). Following change to detemir, all five cats showed clinical improvements within the first 3 months and achieved either moderate or excellent clinical control of their DM. One cat achieved remission within 3 months of detemir treatment.
Newly diagnosed cats
Two of the nine newly diagnosed cats were diagnosed with DM during routine laboratory work for other indications (screening for feline heart disease and a pre-anaesthetic senior profile prior to dental treatment) and the owners had not noticed any clinical signs of DM. The remaining seven cats had clinical symptoms prior to the diagnosis of DM: polyuria/polydipsia (n = 7), weight loss (n = 3), polyphagia (n = 3), lethargy (n = 2), dry fur and scaling fur scale (n = 1), and anorexia (n = 1).
Within the initial 3 months of detemir therapy, 7/9 cats no longer showed any clinical symptoms of DM according to the owners. Improvements were noted after approximately 1 month of therapy, and continued to improve for the following 2 months. The clinical condition of one cat had improved, with milder signs of polyuria, polydipsia and polyphagia, while the clinical condition of one cat did not change despite insulin treatment. Two cats achieved remission within the first 3 months of detemir therapy.
Diabetic remission
A total of three cats achieved remission during the first 3 months of detemir therapy. One had been treated with lente insulin for 3 weeks before detemir therapy was started, while the other two cats were newly diagnosed. The time to achieving remission in these cats was approximately 70, 30 and 40 days, respectively. At the end of the study, all three cats had been euthanased for reasons other than DM (chronic kidney disease [CKD] n = 2, feline asthmatic bronchitis n = 1). Remission had lasted for 47, 30 and 15 months, respectively, without diabetic relapse.
Eleven cats did not achieve remission in the first 3 months of detemir therapy. However, one of the 11 cats achieved remission after 13 months of therapy. At the end of the study, remission had lasted for 4 months in this cat with no relapse.
The median starting dose of detemir was 0.3 U/kg twice daily in cats achieving remission within 3 months and 0.38 U/kg twice daily in cats not achieving early remission (P = 0.64).
Cats not in remission at time of data collection
At the end of the study, three cats were still alive, diabetic and receiving treatment with detemir. They had been treated for a median of 16 months (range 3–56 months), with two cats having achieved excellent control of their disease, while one cat had moderate control of disease. Five cats had been euthanased without achieving remission. For one cat, the date and cause of death could not be determined. The remaining four cats had been treated with detemir for 9.0, 9.5, 15.1 and 21.5 months, respectively. The causes of euthanasia were insufficient control of DM (n = 2), hepatic neoplasia (n = 1) and acute pancreatitis/CKD (n = 1). Two cats were lost to follow-up.
Fructosamine concentrations in all cats
The fructosamine levels were measured at the time of initiation of detemir treatment in 12 cats. All 12 cats had elevated serum fructosamine levels; the median fructosamine level was 685 µmol/l (range 577–1090 µmol/l, reference range 221–341 µmol/l). For 10 cats, repeated measurements of serum fructosamine levels during the first 3 months of detemir therapy were available (Figure 1). The serum fructosamine concentration was significantly reduced 25–60 days (P = 0.047) and 61–105 days (P = 0.031) after the initiation of detemir treatment. There was no significant difference in fructosamine concentration at 61–105 days compared with after 25–60 days (P = 0.25) of detemir treatment. Three cats had at least one fructosamine measurement <450 µmol/l (Table 1). In 7/10 cats the fructosamine levels did not fall below 450 µmol/l, with five cats consistently having a fructosamine level >600 µmol/l. The level of clinical control in these five cats was excellent (n = 3), moderate (n = 1) and poor (n = 1).

Mean serum fructosamine concentration in nine diabetic cats following initiation of detemir treatment. Time 0 is initiation of treatment, time 1 is 25–60 days and time 2 is 61–105 days after the initiation of treatment with detemir. P <0.05 between time 0 and 1. P = 0.03 between time 0 and 2
Assessment of glycaemic control based on serum fructosamine concentration, mean blood glucose (MBG) and nadir during a 12 h blood glucose (BG) curve, and owner observations in 14 cats treated with detemir for 3 months

MBG is the mean value of BG concentration measured every 4 h for 12 h
Owner observations: good control is normalisation of clinical symptoms, moderate control is improvement of clinical symptoms without resolution (eg, still some increased thirst and urination) and poor control is lack of improvement in clinical symptoms
BGCs in all cats
Thirteen cats had at least two BGCs performed in the first 3 months of detemir therapy. In one cat a complete BGC had not been performed, but preinsulin BG was monitored sporadically at home. This was one of the three cats achieving early remission.
The measurements of preinsulin BG and nadir BG for each cat over time are shown in Figures 2 and 3. The preinsulin BG decreased in 6/9 cats after 61–105 days compared with after 25–60 days of detemir treatment, but the difference was not statistically significant (P = 0.30). In the same period of time, the nadir BG decreased in 7/10 cats (P = 0.32).
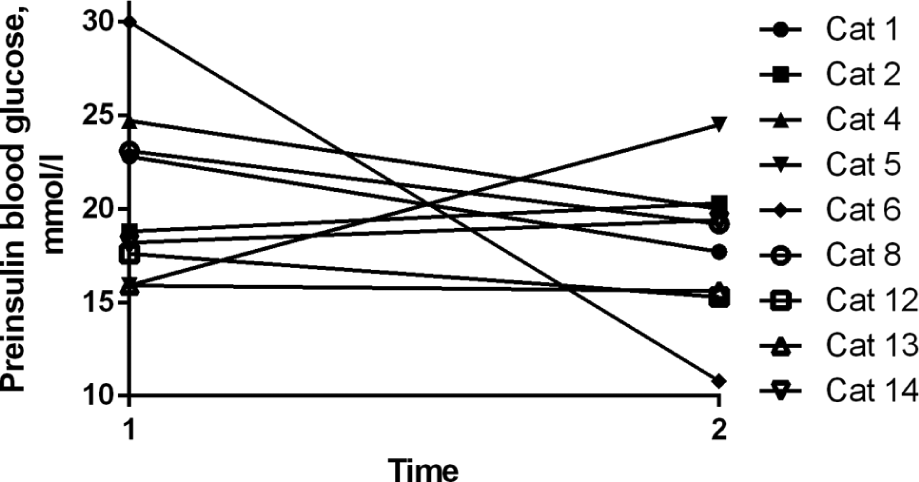
Mean preinsulin blood glucose (BG) concentration during a 12 h BG curve in nine diabetic cats following initiation of detemir treatment. Time 1 is 25–60 days and time 2 is 61–105 days after the initiation of treatment with detemir. P = 0.30 between time 1 and 2

Mean nadir blood glucose (BG) concentration during a 12 h BG curve in 10 diabetic cats following initiation of detemir treatment. Time 1 is 25–60 days and time 2 is 61–105 days after the initiation of treatment with detemir. P = 0.32 between time 1 and 2
Seven cats reached an MBG between 5 and 10 mmol/l (90–180 mg/dl), three cats between 10 and 15 mmol/l (180–270 mg/dl) and three cats consistently had an MBG >15 mmol/l (270 mg/dl). Nine cats achieved a BG nadir of <10 mmol/l, three cats between 10–15 mmol/l and one cat never had a nadir of <15 mmol/l. Nine cats achieved a preinsulin BG of <10 mmol/l, three cats of 10–15 mmol/l and for one cat the preinsulin BG never fell below 15 mmol/l.
Hypoglycaemia
Two out of 14 cats experienced a single episode with clinical symptoms of hypoglycaemia (muscle twitches, lethargy and/or weakness). None of the cats required hospitalisation or veterinary attention during the episode, and both cats were taken care of at home by their owners. The incidents occurred at detemir doses of 0.5 U/kg and 0.4 U/kg twice daily, respectively. Further, two medical records reported episodes of hypoglycaemia (both with a BG of 2.5 mmol/l) during the BGC. Those cats did not show any clinical symptoms. The BG of both cats was measured at home by the owner using a handheld glucometer (Contour; Bayer). The detemir doses at the time were approximately 0.5 U/kg and 1 U/kg twice daily, respectively.
Diet
Six cats were fed a high-protein, low-carbohydrate diet (m/d Feline dry; Hill’s Prescription Diet or Diabetic DS 45; Royal Canin Veterinary Diets) during the follow-up period. The remaining eight cats were fed a combination of a high-fibre (w/d Feline dry; Hill’s Prescription Diet) and high-protein, low-carbohydrate diet (n = 1), a renal diet (n = 2) or an unknown diet (n = 5). The three cats achieving early remission were fed a renal diet (n = 2) and a high-protein, low-carbohydrate diet (n = 1).
Discussion
Detemir is a long-acting insulin analogue with a pharmacokinetic profile that may render it suitable for treatment of diabetic cats. A previous report indicated a high rate of diabetic remission and a good safety profile of detemir in diabetic cats. 15 However, the study involved a very intensive monitoring protocol. In the current study, the effect of detemir was evaluated in a setting more closely resembling a general veterinary practice situation. The majority of our diabetic cat population was neutered males, consistent with previous studies. 23 The median age and presenting symptoms in the cats newly diagnosed with DM were also consistent with previous reports.23,24
A minimum follow-up period of 3 months was used as an inclusion criterion based on previous studies showing that clinical symptoms improved in diabetic cats treated with other types of insulin within this timeframe.21,25,26 Thus, in a study with protamine zinc insulin, 90% of owners reported improvement or resolution of clinical signs by day 45 of treatment. 25 With lente insulin the clinical signs were significantly resolved within 3 months. 26 In the current study, the clinical symptoms were improved after 1 month of detemir treatment in 13/14 cats and continued to improve during the following 2 months. Therefore, when using a protocol based on AAHA recommendations, detemir does not apparently lead to a faster control of clinical symptoms compared with other types of insulin. However, further studies with more stringent timing of follow-up visits are needed in order to fully evaluate this.
The majority of cats in our study achieved moderate or excellent control of clinical signs within the initial 3 months of detemir therapy, regardless of the duration of their disease and possible previous treatment with other types of insulin. Hence, detemir appears to be equally able to improve the clinical symptoms compared with other types of insulin.25–27 Furthermore, five cats previously treated unsuccessfully with other types of insulin all achieved either moderate or excellent control following a change to detemir. Four of the five cats were initially treated with lente insulin, which has too short a duration of effect for twice-daily dosing in some cats.5,7 Because no BGCs were available from the prior treatment period, no conclusions could be made about the cause for the initial unsuccessful insulin treatment in those cats.
According to previous reports, the chance of remission is greatest during the initial 2–4 months of insulin therapy.9,28,29 In the current study, 21% of the cats achieved remission within this timeframe, which is considerably lower than previously reported for detemir, where 67% of treated cats achieved remission. 15 However, some of these cats achieved remission after more than 3 months of therapy, and this makes a direct comparison difficult. Still, the median time to achieving remission was comparable in the two studies (1.3 VS 1.7 months) and also comparable with previous reports with various types of insulin.10,26,27,30
The previous study with detemir involved a very tight monitoring protocol with between three and seven daily measurements of BG, which was far more rigorous than recommended by the AAHA and the protocol applied in our study. 17 This highlights the importance of fastidious monitoring, as it might improve the chance of remission, which is consistent with the results of previous studies.9,28,29 However, we believe that the results from our study may be a more realistic representation of the chance of remission to be expected in a general veterinary practice using detemir. Previous retrospective reports have found a remission rate comparable with ours (16.7% gained remission after a median time of 6 months) in cats treated with various types of insulin. 21
Initially, all cats presented with an elevated serum fructosamine level, which decreased significantly over time. Based on this parameter alone, only 3/10 cats in our study had excellent glycaemic control (fructosamine <450 µmol/l) and none had moderate glycaemic control (fructosamine <550 µmol/l). 22 However, the majority of the cats had excellent or moderate glycaemic control based on their clinical symptoms (Table 1). These findings are consistent with previous reports, indicating that fructosamine alone is suboptimal for evaluating diabetic control in cats, and should be combined with other parameters such as clinical symptoms.31–33
Most owners were able to perform BG measurements at home using a portable glucometer. Despite improvement in clinical symptoms, the preinsulin BG and nadir BG was only moderately decreased after 105 days of treatment. This may reflect that calculations were based on the mean preinsulin BG and mean nadir BG for each cat in each period of time. Consistent with previous reports, many cats showed a large variation in BG values, even when the BGCs were only separated by a few days. 34 However, it may also indicate that the BGC intervals were too defensive, resulting in suboptimal glycaemic control. In the studies by Roomp and Rand,15,28 the intensive BG monitoring may have led to a faster reduction of BG, which may have contributed to an increased rate of remission.15,28 A direct comparison is not possible because no BG values were presented in the above mentioned article. 15
In accordance with previous studies, glycaemic control based on evaluation of BG levels did not represent the degree of control perceived by the owners. 26 Likewise, in a study with 54 diabetic cats, only six were well controlled based on BG levels, whereas 31 owners believed that their cat responded well to treatment. 21
Only a few cases of clinical hypoglycaemia were identified in our study, and none required acute veterinary attention. This is comparable to the findings in the previous study with detemir, where 1/18 cats experienced mild hypoglycaemia. 15 Studies with other types of insulin have reported higher frequencies of clinical hypoglycaemia wherein some required intravenous glucose infusion.25,35 Therefore, the safety of detemir appears to be high.
There are several limitations to this study. First and foremost the retrospective design. In some instances, the medical records were incomplete, and the information gathered at each visit was not strictly standardised but depended on the preference of the responsible veterinarian. This could have affected our results in multiple ways; for example, by over-/underestimating the clinical symptoms or by concealment of lack of compliance. The number of included cases is low, which may reflect the fact that some owners choose euthanasia over treatment after diagnosis of DM; unfortunately, this could not be assessed owing to the design of our record system. The low number of cases may also reflect that most veterinary practices are confident in treating feline DM, which limits the amount of referrals to our hospital.
Several of the included cats had attended far fewer control visits than recommended at our facility, most often owing to the owners not adhering to the recommended timing of control visits. Had the owners adhered to our protocol more stringently, we may have experienced a higher rate of remission. However, we believe that this level of owner compliance is not uncommon and therefore feel that our results are a fair representation of the outcome that owners and veterinarians can expect, when dealing with clients in a general practice. Suboptimal owner compliance was also noted in the study by Roomp and Rand, 15 where 43/61 cats were excluded, 15 of these because of lack of compliance. 15 There may be several reasons for the owners to have inadequate motivation. Based on our experiences, in some owners’ opinion the regular BGCs are too time-consuming and the ear-pricking is considered stressful for both cats and owners. Furthermore, some owners have financial concerns and hence skip the regular veterinary follow-up visits. It may also be that some owners fail to comprehend the possible benefits of regular monitoring, and that this must be further emphasised by the attending veterinarian.
Finally, it would have been interesting to compare the included cats with cats treated with other types of insulin during the same period; unfortunately, this was not possible because insulin detemir is first-choice insulin at our hospital and the number of cats being treated with other insulin types was extremely low.
Conclusions
For the diabetic cats in this study, treatment with detemir resulted in moderate or good control of clinical symptoms in 93% of cats; even in cats previously treated with other types of insulin. The safety of detemir was excellent. The overall remission rate in this study was quite low, compared with results from a study using a protocol for intensive BG monitoring. This has led us to suspect that the treatment and monitoring protocol proposed by the AAHA may be too defensive, and that further studies in this area are relevant.
Footnotes
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Funding
This study was funded by a PhD fellowship from the University of Copenhagen.
