Abstract
An 11-year-old spayed female domestic shorthair cat was evaluated for anorexia, lethargy and weight loss of 6 days’ duration. Bilateral mydriasis, absent menace response, slow-to-absent pupillary light reflexes, bilateral retinal detachment, intermittent horizontal nystagmus, intermittent ventral strabismus and systemic hypertension were present. Biochemical analysis revealed severe hyponatremia, severe hypochloremia and mild hypokalemia. Multifocal central nervous system disease was suspected based on optic, trigeminal sensory (ophthalmic branch), vestibulocochlear and possible oculomotor nerve dysfunction. Thoracic radiographs showed mild cardiomegaly without evidence of congestive heart failure. Ultrasound revealed mild pleural and peritoneal effusion. A cause of the severe hyponatremia was not identified, and it persisted despite fluid therapy. Syndrome of inappropriate antidiuretic hormone secretion (SIADH) was suspected as the cause of hyponatremia. Humane euthanasia was elected owing to continued clinical decline. Serum hyposmolality, urine hyperosmolality, natriuresis and lack of confirmed renal, thyroid and pulmonary disease aided in the presumed diagnosis of SIADH. Post-mortem histopathology of the brain revealed degeneration of the hypothalamus and optic tracts, along with a prominent fluid-filled craniopharyngeal duct (putative Rathke’s cleft cyst) separating the pars distalis and the pars intermedius. The hypothalamic degeneration, possibly secondary to a Rathke’s cleft cyst, was hypothesized to be the cause of presumptive SIADH in the patient. Although rare in occurrence, Rathke’s cleft cyst should be included as a differential diagnosis in dogs and cats with signs of pituitary dysfunction.
Case Report
An 11-year-old spayed female domestic shorthair cat was presented for anorexia, lethargy and weight loss. The patient had a previous history of mild degenerative mitral valve disease, atrial premature contractions and aortic dilation diagnosed on echocardiogram and electrocardiogram 3 years previously. On presentation, a systolic heart murmur was auscultated, but was unchanged from historic examinations. Systemic arterial blood pressure was 250 mmHg (doppler, systolic). Hyponatremia (130 mmol/l; reference interval [RI]: 150–160 mmol/l), low normal potassium (3.5 mmol/l; RI 3.5–5.8 mmol/l), and hypochloremia (87 mmol/l; RI 117–128 mmol/l) were identified on biochemical profile. A total T4 was normal (1.5 µg/dl; RI 1.0–3.5 µg/dl). Oral potassium supplementation and amlodipine besylate (0.625 mg PO q24h; Alkem Laboratories) were prescribed.
On presentation to a referral institution 6 days later, neurological examination revealed intermittent, spontaneous, horizontal pathological nystagmus with fast phase to the right, intermittent ventral strabismus bilaterally, bilateral mydriasis, absent pupillary light reflex in the right eye and decreased pupillary light reflex in the left eye, and absent menace response in both eyes. There were no gait abnormalities or postural reaction deficits noted. Fundic examination revealed bilateral focal retinal detachment and subretinal edema consistent with a hypertensive retinopathy. Small (0.5 mm × 0.5 mm) superficial, bilateral corneal ulcers were noted with decreased corneal sensation in both eyes. A Schirmer tear test and an electroretinogram were not performed. The combination of ophthalmic findings suggested optic, trigeminal sensory (ophthalmic branch), vestibulocochlear, possible oculomotor (general visceral efferent) nerve dysfunction and bilateral retinal detachment. A complete blood count was unremarkable, and serum biochemistry revealed severe hyponatremia, hypokalemia, hypochloremia (Table 1) and mild hyperglycemia (206 mg/dl; RI 71–159 mg/dl). A urinalysis obtained via cystocentesis demonstrated a urine specific gravity of 1.034, glucosuria (500 mg/dl), proteinuria (100 mg/dl) and an inactive sediment. Aerobic urine culture was negative. Systemic arterial pressure was 184 mmHg (Doppler, systolic). Thoracic radiographs revealed mild cardiomegaly, but no evidence of venous congestion or vessel enlargement. The patient was given lactated Ringer’s solution (USP, 90 ml/kg/day IV; Hospira) with potassium chloride (20 mEq/l) and continued on amlodipine (0.625 mg PO q12h) and mirtazapine (3.75 mg PO q72h).
Serial serum electrolytes obtained from an 11-year-old cat with syndrome of inappropriate antidiuretic hormone secretion

A serum electrolyte panel revealed slight improvement in sodium and chloride, and persistent mild hypokalemia (Table 1). Fluid therapy was changed to a combination of isotonic saline (0.9% NaCl, 48 ml/kg/day IV; Hospira) and lactated Ringer’s solution (42 ml/kg/day IV) with 40 mEq/l of potassium chloride. Despite fluid therapy, the hyponatremia, hypokalemia and hypochloremia persisted (Table 1). The fluid therapy was changed to isotonic saline with 40 mEq/l potassium chloride (90 ml/kg/day IV). Mild peritoneal effusion and mild pleural effusion were found on ultrasound; however, a cause was not identified. The liver, kidneys and adrenal glands (5 mm in diameter bilaterally) were unremarkable on abdominal ultrasound. An esophageal feeding tube (14 Fr × 13 inch Mila International esophagostomy tube) was placed in the cervical esophagus under general anesthesia. Suspected nausea and possible ileus were treated with a metoclopramide (Hospira) constant-rate infusion (1.5 mg/kg/day IV).
Four days after admission, the patient appeared depressed with little-to-no improvement in sodium, potassium, chloride (Table 1) or blood pressure (190 mmHg systolic). The patient’s weight had increased by 14% since admission (4.45 kg vs 3.9 kg on presentation). Eight percent of the increase had occurred within the previous 24 h. There was a strong suspicion of syndrome of inappropriate antidiuretic hormone secretion (SIADH) as the cause of the hyponatremia. Fluid therapy was discontinued and furosemide (2 mg/kg IV; Butler Animal Health) was given to promote free water loss. The multiple cranial nerve deficits raised the concern of underlying multifocal intracranial neoplasia or multifocal areas of infarction subsequent to sustained systemic hypertension, primarily affecting the brainstem. Other considerations included increased intracranial pressure due to edema, inflammatory disease and, less likely, infectious etiologies. Hyponatremia results in predominantly cerebrocortical deficits and was thus considered an unlikely cause of the neurological signs seen in this patient. As the owners declined magnetic resonance imaging (MRI) and the patient continued to decline, dexamethasone (Bimeda MTC, 0.25 mg/kg IV; Animal Health) was given once for empiric and palliative treatment of primary multifocal neoplastic disease.
Five days after admission, the patient’s weight decreased 6% from the previous day (4.15 kg vs 4.45 kg). Serum electrolyte evaluation showed continued hyponatremia, and progressive hypokalemia and hypochloremia (Table 1). The owners elected humane euthanasia owing to clinical decline. Serum and urine samples were submitted for osmolality and electrolyte analysis (Animal Health Diagnostic Center, Cornell University). Serum osmolality was 281 mOsm/kg (RI 280–305 mOsm/kg), urine osmolality was 678 mOsm/kg, urine sodium concentration was 90 mEq/l and serum sodium concentration was 128 mmol/l (RI 150–160 mmol/l). A presumed diagnosis of SIADH was based on persistent hyponatremia, inappropriate urine hyperosmolality in the face of serum hyposmolality, and lack of evidence to support underlying renal, thyroid, pulmonary or neoplastic disease.
The head was submitted for post-mortem histopathological examination. The pars intermedius and the pars distalis were separated by a prominent cleft, filled with homogeneous eosinophilic material and lined with cuboidal non-ciliated cells (Figure 1 a,b). As the pituitary gland was separated from the brain during brain removal, the fluid-filled space may have been larger in life, consistent with a Rathke’s cleft cyst (RCC). Ante-mortem imaging would have been required for a definitive diagnosis. Pituicytes and multifocal mineralization of small vessels were noted at the periphery of the pars distalis. Within the pars nervosa, there was a small aggregate of gitter cells. No other abnormalities were noted in the pituitary. Other findings on histopathology included bilateral moderate vacuolar degeneration of the hypothalamus (Figure 2); bilateral, severe axonal degeneration of the optic tract (Figure 3); bilateral, mid-axonal degeneration of the optic nerve; and retinal detachment and loss of ganglion cells of the eyes and retinas. Additionally, mild, multifocal lymphocytic scleritis; multifocal, mild-to-severe arteriolosclerosis, hyaline degeneration and plexiform change of the blood vessels; perivascular lymphocytes in the pons and rostral cerebellar peduncle; and focal neural degeneration of the spinal trigeminal nerve and tract were found. The oculomotor nucleus was histologically unremarkable, and the oculomotor nerve was not included in tissue sectioning.
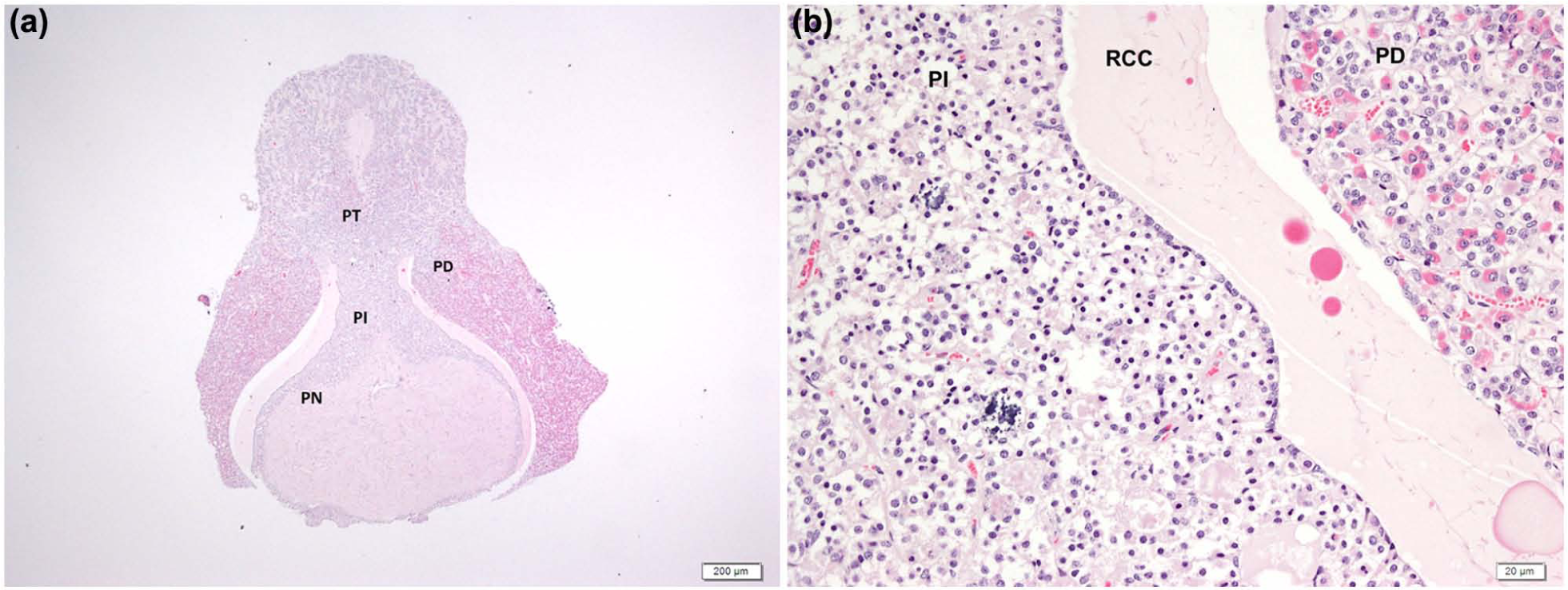
Histological section of the pituitary. (a) Cyst separating the pars distalis (PD) and pars intermedius (PI), lined by cuboidal, non-ciliated cells and filled with homogeneous eosinophilic material (hematoxylin and eosin [HE] stain, ×40). (b) Section of the cyst (Rathke’s cleft cyst [RCC]) under higher magnification (HE stain, ×400)

Histological section of the hypothalamus showing moderate vacuolar degeneration (arrows) (hematoxylin and eosin stain, ×400)

Histological section of the optic tract showing severe axonal degeneration (arrows) (hematoxylin and eosin stain, ×400)
SIADH occurs when antidiuretic hormone (ADH) is secreted without stimulus from osmoreceptors or baroreceptors. 1 Increased free water reabsorption leads to mild increases in volume, which leads to increased atrial natriuretic peptide release and causes decreased sodium reabsorption. 2 As unregulated ADH secretion continues, extracellular fluid expansion occurs and plasma becomes hypo-osmolar.1,3 Ultimately, excessive or inappropriate release of ADH results in decreased serum sodium, serum hyposmolality, increased urine osmolality and natriuresis.
SIADH secretion is one of the most common causes of normovolemic hyponatremia in humans, and can be caused by pulmonary disease, central nervous system disease, drug administration, neoplasia and endocrinopathies, or can be idiopathic.1–4 There are case reports of humans with SIADH associated with the presence of RCC.5–7 SIADH secretion is rare in small animals, with several cases in dogs associated with dirofilariasis, hypothalamic tumor, undifferentiated carcinoma, granulomatous amebic meningoencephalitis, hydrocephalus and liver disease; idiopathic cases have also been reported.8–13 There are two reports of SIADH in cats associated with vinblastine toxicity and anesthesia.14,15
Diagnosis of SIADH is based on specific inclusion and exclusion criteria. In humans, these include hyponatremia with plasma hyposmolality (<275 mOsm/kg); inappropriately high urine osmolality (>100 mOsm/kg) in the presence of plasma hyposmolality; normal renal, adrenal and thyroid functions; the presence of natriuresis (urine sodium concentration >20–40 mEq/l) despite hyponatremia and plasma hyposmolality; clinical normovolemia; lack of ascites or edema; correction of hyponatremia by fluid restriction; and no history of diuretic administration. Treatment includes water restriction and the use of arginine vasopressin antagonists.1–4 However, the only definitive treatment of SIADH is removal of the underlying cause.
A presumed diagnosis of SIADH was made in our patient owing to hyponatremia with serum hyposmolality, urine hyperosmolality, natriuresis, and lack of evidence of renal, thyroid and pulmonary disease. A serum osmolality of 281 mOsm/kg is considered hypo-osmolar, as normal plasma osmolality is higher in cats (290–330 mOsm/kg) compared with humans. 1 Serum osmolality, urine osmolality and urine sodium were measured after the patient received a single dose of furosemide. However, the urine sodium concentration and urine osmolality were still inappropriately high given the serum hyposmolality. Continued weight gain and mild pleural effusion were most likely a result of volume overload, which started to resolve with fluid restriction and furosemide administration. As hyponatremia was repeatedly documented during periods of normovolemia and prior to the development of effusion, neither congestive heart failure nor hypervolemia were a cause for this patient’s persistent hyponatremia. Metoclopramide has been reported to cause release of ADH in humans, and may have contributed to increased ADH release; however, the patient’s hyponatremia was present prior to the initiation of therapy with metoclopramide. 16
Although uncommon, hypokalemia has been documented to occur with SIADH in humans, dogs and cats. 11 The mild-to-moderate hypokalemia in this case may be explained by increased aldosterone release stimulated by hypotonicity. 17 Additionally, furosemide administration likely contributed to the decline in potassium (Table 1). Refractory hypokalemia can also occur with hypomagnesemia; unfortunately, serum magnesium was not evaluated in this patient. 18 At initial presentation to the referring veterinarian, potassium was within the normal range and sodium was severely decreased. Therefore, it is unlikely that correction of the hypokalemia would have improved the hyponatremia.
Primary hyperaldosteronism is a cause of hypertension and hypokalemia in cats, as present in this case, but was considered unlikely given the lack of evidence of an adrenocortical tumor or bilateral hyperplasia, as this patient had normal adrenal glands on ultrasound. However, ultrasound alone cannot exclude the presence of small functional neoplasms or hyperplasia within the zona glomerulosa. Plasma aldosterone concentration and/or ratio of plasma aldosterone to plasma renin activity would have been necessary for exclusion. An underlying cause for the systemic hypertension was not found in this patient, and may be idiopathic or related to undiagnosed hyperaldosteronism. Liver function tests were not performed owing to the lack of evidence of support liver dysfunction. Ideally, endogenous adrenocorticotropic hormone (ACTH) concentrations and an ACTH stimulation test should have been evaluated to assess secondary adrenal insufficiency as a contributing factor to the hyponatremia, but this was considered unlikely as the patient failed to improve with glucocorticoid administration.
In this case, histological changes were suggestive of a RCC, which is hypothesized to be the cause of inappropriate ADH release and subsequent hyponatremia. RCC, also referred to as a craniopharyngeal duct cyst, is a cystic malformation from the remnants of Rathke’s pouch.19,20 During embryological development, the oral ectoderm forms the median eminence and the pars nervosa as it extends upward from the craniopharyngeal duct to the infundibulum. Cells from the walls of Rathke’s pouch form the pars distalis, pars intermedia and pars tuberalis, and as formation of the pituitary gland is completed, a lumen known as Rathke’s cleft remains between the pars distalis and the pars intermedia.19,21,22
In humans, RCCs are typically incidental findings on autopsy, and rarely cause clinical signs of headaches, visual disturbances and pituitary dysfunction.5–8,20,23,24 RCC in dogs has been rarely associated with pituitary dwarfism, nasopharyngeal obstruction and aggressive behavior.19,24–27 To our knowledge there are no documented cases of RCC in cats.
Diagnosis of RCC in humans includes advanced imaging, such as computed tomography or MRI, and histopathology. Cysts can range from 5–50 mm in size, and can vary between ovoid to dumb bell in shape.6,20,23,28 However, CT and MRI are only suggestive of RCC, with other differentials including craniopharyngioma, pituitary adenoma, hypothalamic hamartoma and arachnoid cysts.20,23,24,29,30 On histopathology, morphology of the cystic wall varies from simple cuboidal or columnar epithelium, with or without cilia and goblet cells.7,20,23 Treatment of symptomatic RCC includes surgical drainage of the lesions and cystic wall removal via a trans-sphenoidal approach in humans.7,20,28 In dogs and cats, transsphenoidal surgery has been performed to remove pituitary tumors, and may provide a treatment option for symptomatic RCC.24,31–33
In this case report, the RCC was hypothesized to be the cause of hypothalamic or posterior pituitary dysfunction leading to inappropriate ADH secretion. RCC can cause dysfunction directly from cyst compression of surrounding tissue, or indirectly from compression of adjacent blood supply or inflammation and edema leading to hypophysitis. 19 The hypothalamic degeneration, possibly secondary to a RCC, is hypothesized to be the cause of presumptive SIADH in the patient. The axonal degeneration in the optic tracts and nerves was more severe in the tracts than the nerves and, therefore, was likely to be secondary to cyst compression rather than hypertension. In addition, the affected axons having normal cell bodies, the bilaterally symmetric nature to the axonal degeneration, and the lack of evidence of multifocal hemorrhage or hemorrhagic infarcts suggested that hypertension was less likely the cause of axonal degeneration. The abnormal pupillary light reflexes, bilateral mydriasis, and absent menace response are consistent with the optic nerve and retinal pathology noted. Given these findings and lack of pathology associated with the oculomotor never, concurrent oculomotor nerve dysfunction was unlikely. Cyst compression of the ophthalmic branch of the trigeminal nerve (as this branch courses next to the optic chiasm) may explain the decreased corneal sensation. Cyst compression may have also lead to degeneration of the spinal tract and nucleus via transneuronal degeneration. The areas of gliosis and perivascular lymphocytes within the metencephalon and myelencephalon are non-specific findings associated with degeneration and inflammation.
Histopathological examination of the brain failed to identify an underlying lesion to explain the combination of neurological abnormalities (oculomotor and vestibulocochlear dysfunction) detected on physical examination, which may have been a clinical overinterpretation. However, peripheral nerve involvement or idiopathic vestibular disease cannot be excluded.
Conclusions
We describe a potential case of RCC, presumed to be associated with inappropriate secretion of ADH, in a cat. Although rare in occurrence, RCC should be included as a differential diagnosis cats (and dogs) with signs of pituitary dysfunction.
Footnotes
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this case report.
