Abstract
A 13-year-old female domestic shorthair cat presented with polyphagia and weight loss. Marked systolic hypertension was found on examination. Elevated total thyroxine levels confirmed hyperthyroidism, and hypokalaemia was also documented. A euthyroid state and normotension were achieved following 4 weeks of treatment with carbimazole and amlodipine. Despite potassium supplementation, the hypokalaemia worsened. Abdominal ultrasonography revealed left adrenomegaly. Plasma aldosterone concentrations were initially in the lower half of the reference interval and, when repeated 2 months later, were undetectable. Urea and creatinine remained in the lower half of the reference interval throughout treatment, and urine specific gravity suggested good urine concentrating ability. The fractional excretion of potassium confirmed a renal source of potassium loss. Blood gas analysis was unremarkable. It was theorised that an aldosterone precursor was causing signs of mineralocorticoid excess and undetectable plasma aldosterone levels. Treatment with an aldosterone receptor antagonist successfully increased the serum potassium concentration. Owing to difficulties administering medication and associated effects on life quality the cat was euthanased. Adrenal hyperplasia was apparent on post-mortem histopathology
Case Report
Feline hyperthyroidism is the most common endocrine disorder diagnosed in cats over the age of 8 years. 1 More than 98% of cases have benign adenomatous hyperplasia. 2 Hypokalaemia has been reported in up to 17% of hyperthyroid cats, 3 and is thought to be related to increased adrenergic stimulation promoting potassium uptake by cells due to increased activity of the Na+, K+-ATPase. 4 Hyperthyroidism is not now thought to be a common cause of hypertension; in some studies there have been no significant differences between systolic blood pressure (SBP) in control and hyperthyroid cats, 5 and in others only mild hypertension has been documented. 6 Here, we document a case in which hypertension was marked, and hypokalaemia did not respond to treatment of the primary diagnosis of hyperthyroidism. Subsequent investigation indicated adrenal hyperplasia and suspected non-aldosterone mineralocorticoid excess. The clinical signs most closely resembled the non-tumourous form of hyperaldosteronism in cats.
A 13-year-old female domestic shorthair cat presented with weight loss and polyphagia of 2 months’ duration. The cat was anxious and tachycardic (heart rate 240 beats per minute) with no audible murmur or dysrhythmia. Direct ophthalmoscopy revealed retinal vessel tortuosity, but no evidence of haemorrhage or focal detachment. Mean SBP, measured using the Doppler technique, was 280 mmHg. With the presence of only mild ocular lesions, white coat syndrome was thought to be partially responsible.
Serum biochemistry showed moderate hypokalaemia (2.9 mmol/l, reference interval [RI] 3.5–5.8 mmol/l) and elevated thyroxine (T4) (179 nmol/l, RI 10–60 nmol/l).
Carbimazole (Vidalta 15 mg q24h; MSD Animal Health), amlodipine (Norvasc 1.25 mg q24h; Pfizer) and potassium gluconate (Kaminox oral solution 2 mEq q12h; Vet Plus) were started immediately. β-blockers, such as atenolol, are recommended for the tachycardia that accompanies hyperthyroidism, but are not reliable antihypertensive agents in these patients. 7
After 10 days, mean SBP was 160 mmHg. T4 reduced to 45.7 nmol/l at 4 weeks. Despite supplementation the potassium was lower after 4 weeks of treatment (2.7 mmol/l). The dose was increased to 2.2 mEq q12h, and 4 weeks later the potassium was 3.1 mmol/l. At 8 weeks T4 was 13.4 nmol/l with no dose adjustment, urea was 6.9 mmol/l (RI 5.7–12.9 mmol/l) and creatinine was 100 μmol/l (RI 71–212 μmol/l).
At 8 weeks an abdominal ultrasound examination revealed left adrenomegaly (14.7 mm × 4.6 mm; RI 8.9–12.5 × 3.0–4.5) 8 (Figure 1). The right adrenal gland was smaller (7.2 mm × 2.3 mm) (Figure 2). There was no evidence of invasion into the caudal vena cava, suggestive of malignancy, and no calcified areas. The cat had ultrasonographically normal kidneys.

Ultrasound image of the left adrenal gland using an 8 MHz curvilinear transducer. The length was 14.7 mm and the width of the caudal pole was 4.6 mm

Ultrasound image of the right adrenal gland. The right adrenal gland was significantly smaller than the left one and was 7.2 mm long with a caudal pole width of 2.3 mm
Serum aldosterone was 218 pmol/l (RI 195–390 pmol/l). 9 A value in the lower half of the RI is inappropriate in a hypokalaemic cat as the low potassium concentration would be expected to suppress aldosterone secretion. The aldosterone renin ratio (ARR) is the gold standard for screening for primary hyperaldosteronism (PHA), but a large blood sample and instant freezing of the separated plasma are needed. Also, a single ARR within the RI does not exclude PHA in cats with idiopathic bilateral nodular hyperplasia. 10 A urine sample collected by cystocentesis showed good concentrating ability (specific gravity 1.046) and no abnormalities. Some cats with early chronic kidney disease (CKD) can still concentrate urine above 1.035, 11 but urea and creatinine were still in the lower half of theRI following normalisation of T4, which was not consistent with chronic renal failure.
Fractional excretion of potassium (FEK) was 51.7%, confirming a renal source of potassium loss; values >6.0% are abnormal in potassium-depleted animals with normal renal function. 12 Renal tubular acidosis is rare in cats, 13 and was ruled out with venous blood gas analysis (pH 7.37, RI 7.24–7.4).
The initial hypertension, unilateral adrenomegaly and hypokalaemia were so suggestive of PHA that aldosterone levels were measured again 8 weeks later. In cats with idiopathic hyperplasia, the plasma aldosterone concentration (PAC) may be only slightly elevated or within the upper limit of the RI, 10 and it was also necessary to rule out sampling or laboratory error. The PAC at this point was undetectable (<20 pmol/l). As renal potassium loss was established and adrenal enlargement was noted, it was theorised that an aldosterone precursor was causing signs of mineralocorticoid excess. Ideally, renin would also have been measured to demonstrate concurrent suppression of renin activity, but costs precluded this.
Spironolactone (Prilactone 10 mg orally q24h; Ceva Animal Health) was started to inhibit competitively the mineralocorticoid receptors. Plasma potassium levels were measured 2 weeks later and, despite inconsistent dosing, the potassium had increased to 3.4 mmol/l. In the previous 8 weeks, the cat had shown a very slow response to oral potassium supplementation; while it is possible that it was starting to respond to the continued oral therapy, it is more likely that this increase was related to the spironolactone administration.
Unfortunately, the owner found medicating the cat difficult and felt it was affecting its quality of life, and requested euthanasia. A limited post-mortem examination was performed. The left adrenal gland measured 15 mm in length and the right was 10 mm in length. They were both sent for histopathology. The sections of the two glands showed similar histological appearance. In each case the capsule was unremarkable. The cortex and the medulla were distinct and clearly demarcated from each other (Figure 3). In the cortex, the zona glomerulosa, the zona fasiculata and the zona reticularis were recognisable; there was hyperplasia of the cortical tissue, particularly of the zona fasciculata, The cortical hyperplasia was diffuse. The cells were swollen and showed fine cytoplasic vacuolation (Figures 4 and 5). The medulla was occupied by normal-looking chromaffin cells. There was bilateral diffuse hyperplasia of the adrenal glands, and the lesion appeared to be most prominent in the zona fasciculata. There was no evidence of active inflammatory reaction, adrenal carcinoma, phaeochromocytoma, malignant phaechromocytoma or any other type of neoplasia.
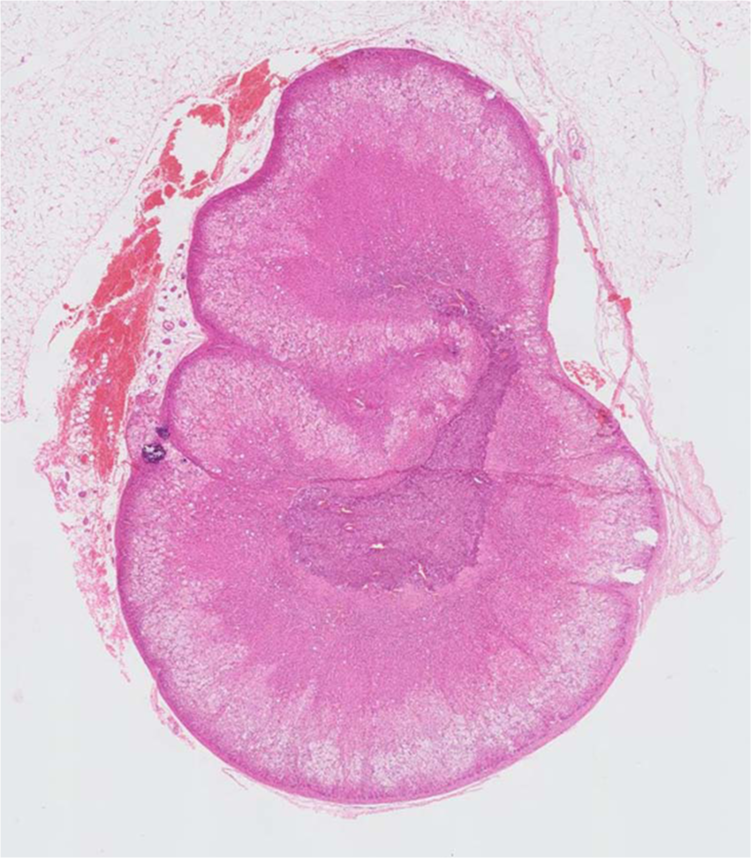
Left adrenal gland stained with haematoxylin and eosin at × 1 magnification. The cortex and the medulla are distinct and clearly demarcated from each other

Left adrenal gland stained with haematoxylin and eosin at × 10 magnification showing swollen cortical cells with fine cytoplasmic vacuolation
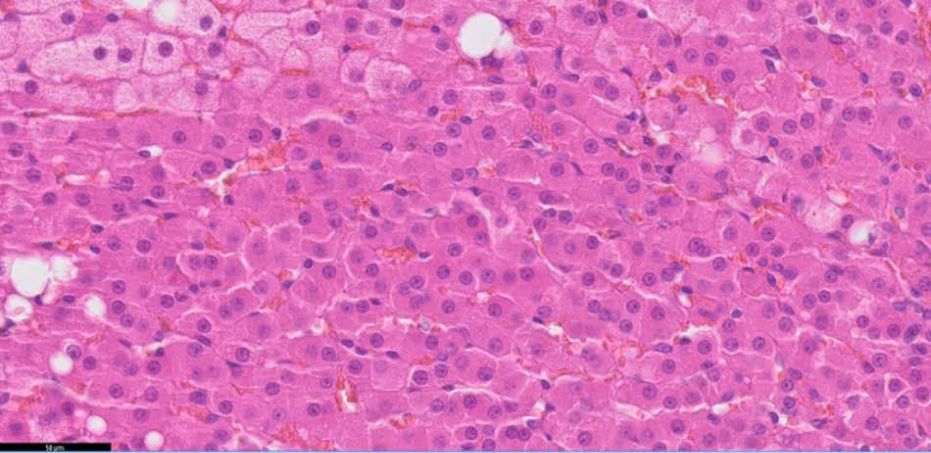
Left adrenal gland stained with haematoxylin and eosin at × 40 magnification showing the fine cytoplasmic vacuolation in more detail
The continued presence of hypokalaemia, despite effective management of hyperthyroidism, and the significant hypertension, suggested another cause in this cat. Causes of hypokalaemia in cats are decreased intake (anorexia or potassium deficient fluids), translocation into cells (metabolic alkalosis, hyperthyroidism and hyperinsulinaemia), increased gastrointestinal losses (vomiting or diarrhoea) and renal loss (including mineralocorticoid excess). A small reduction in plasma potassium concentration following amlodipine administration has been reported in cats, 14 but the mechanism is unknown. This may explain some of the potassium drop after 4 weeks of treatment, but not the initial hypokalaemia.
The effect of hyperthyroidism on adrenal size is controversial. 8 One study reported adrenal glands up to 20% larger in hyperthyroid cats, 15 while another found no statistically significant difference. 16 The asymmetrical adrenal enlargement was not consistent with a non-adrenal trophic effect. Adrenal masses may exhibit variation in steroidogenic production, and signs of mineralocorticoid excess can be caused by increased 11-deoxycorticosterone production leaving low, or undetectable, circulating aldosterone concentrations. 17 Functional tumours producing excessive amounts of an intermediary adrenocortical steroid are rare in dogs and cats. 18 Deoxycorticosterone has mineralocorticoid activity and acts on the same receptor as aldosterone. Two cases of deoxycorticosterone-secreting adrenocortical carcinomas have been reported in dogs. One with hypertension, increased plasma deoxycorticosterone and non-detectable PACs, 19 and the other with all the former signs, but also hypokalaemia and increased FEK. 20 In this case, it is possible that idiopathic hyperplasia of the zona fasciculata resulted in production of 11-deoxycorticosterone or other hormones with mineralocorticoid activity. Response to treatment with the aldosterone antagonist spironolactone supported the suspicion of an aldosterone precursor causing signs of mineralocorticoid excess and undetectable plasma aldosterone levels.
Unfortunately, we were not able to find a laboratory to measure feline deoxycorticosterone; however, we were able to measure the other adrenal steroids, all of which were in the normal range (Table 1).
Adrenal steroids measured in this study

The laboratory had no feline RIs available for androstenedione. Samples were taken from one 2-year-old and one 4-year-old female neutered domestic shorthair cat for comparison. This sample size is far too small to enable an RI to be generated, and the cats used for comparison were younger, but it does suggest that there was no overproduction of androstenedione
RI = reference interval
Basal cortisol was also measured, and was elevated at 428 nmol/l (RI up to 270 nmol/l).
Adrenal function has been reported to be altered by hyperthyroidism, resulting in significantly higher cortisol levels before and after stimulation with adrenocorticotropic hormone (ACTH) than other groups. 21 Hyperadrenocorticism has a guarded-to-grave prognosis, and without treatment most cats succumb to complications within a few weeks to months of diagnosis. This cat underwent treatment for 5 months and still did not have clinical signs consistent with hyperadrenocorticism, so it is most likely that the elevated cortisol was a result of the hyperthyroidism. However, without performing further tests, hypercortisolism causing hypokalaemia and low aldosterone levels could not be definitively ruled out. Phaeochromocytoma could present as a unilateral adrenal enlargement, but this was ruled out by histopathology; an ectopic phaeochromotoma could not be definitively ruled out.
Another cause of low aldosterone and signs of mineralocorticoid excess is a deficiency of 11-b-hydroxysteroid dehydrogenase allowing mineralocorticoid receptors to be occupied by cortisol due to decreased conversion to inactive cortisone. Congenital 11-b-hydroxysteroid dehydrogenase deficiency has been described in humans,22–24 but not in cats. In humans, high doses of furosemide, 25 licorice 26 and cholestasis 27 have been reported to inhibit 11-b-hydroxysteroid dehydrogenase. An acquired deficiency of 11-b-hydroxysteroid dehydrogenase has not been reported in the cat, but, in this case, a deficiency of 11-beta-hydroxysteroid dehydrogenase could have been responsible for the clinical signs.
A deficiency of 11-b-hydroxylase could lead to increased production of 11-deoxycorticosterone. 11-b-Hydroxylation is necessary for cortisol synthesis in the zona fasciculata, and defects in this step lead to increased production of 11-deoxycorticosterone. 28 This has been reported as a congenital deficiency in a cat causing adrenal hyperplasia with high levels of serum ACTH, testosterone, androstenedione, progesterone, 17-hydroxyprogesterone, 11-deoxycortisol and deoxycorticosterone. 29 There are no reports in the literature of cats with an acquired enzyme deficiency; interestingly, in cats with CKD and idiopathic hypertension the cortisol-to-cortisone shuttle has been reported to be more effective. 30
Aberrant expression of functional hormone receptors in the adrenal cortex has been reported in a dog causing adrenocorticotrophic hormone-independent hyperadrenocorticism associated with meal-induced hypercortisolaemia. 31 This could explain the changes seen on histopathology along with the low aldosterone, but signs consistent with hyperadrenocorticism would have been expected.
The adrenal hyperplasia noted on histopathology most closely resembles non-tumourous PHA, where hyperplasia is seen extending from the zona glomerulosa into the zona fasciculata and zona reticularis. 32 In this case, the lesion was most prominent in the zona fasciculata, where deoxycorticosterone is predominantly synthesised. 33 At present, histological findings for PHA in cats indicate that adrenal carcinoma occurs more often than unilateral and bilateral adenoma, or bilateral nodular hyperplasia. 10 This differs markedly from humans, where bilateral hyperplasia of the zona glomerulosa accounts for 60–65% of cases. 34 The non-tumourous form of PHA in cats has many similarities with ‘idiopathic’ PHA in humans, 32 and often presents with milder signs. Idiopathic bilateral nodular hyperplasia probably occurs more frequently in cats than suggested by histopathology data, 10 and PHA should no longer be considered a rare condition. 6
Conclusions
To the best of our knowledge, this is the first reported case of feline adrenal hyperplasia causing possible non-aldosterone mineralocorticoid excess, but a definitive cause of the adrenal hyperplasia is unknown. It would be beneficial to establish feline RIs for other mineralocorticoids such as deoxycorticosterone. Increased awareness of PHA in practice may also lead to increased recognition of adrenal hyperplasia and other cases of non-aldosterone mineralocorticoid excess.
Footnotes
Acknowledgements
We thank Mark Obwolo at IDEXX Laboratories for the histopathology report and histopathology photographs.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this case report.
