Abstract
Lagochilascariasis, a parasitic disease little known in Brazil, is caused by an ascarid nematode that has a peculiar life cycle, with a predilection site for the cervical region in the final hosts: humans, cats and dogs. We aimed to record the occurrence of Lagochilascaris minor in domestic cats from rural areas in the Municipality of Pelotas, Rio Grande do Sul State, Brazil, with reports of clinical signs and the treatment applied.
Lagochilascariasis is a parasitic disease that occurs in dogs, cats and humans. Its etiologic agent is an ascarid nematode of genus Lagochilascaris. Five species have been described: Lagochilascaris minor Leiper, 1909, first described to parasitize humans; Lagochilascaris major Leiper, 1910, a natural parasite of the African lion Felis leo sabakiensis; Lagochilascaris turgida (Stossich, 1902) Travassos, 1924, recorded in the American opossum Didelphis crassicaudata; Lagochilascaris buckleyi Sprent, 1971, parasite of the American cougar Felis concolor; Lagochilascaris sprenti Bowman, Smith and Little, 1983, a parasite of the American opossum Didelphis virginiana.1,2
L minor is considered to be the most important ascarid species, with dozens of cases reported in humans. It occurs in several countries, including Mexico, Costa Rica, Venezuela, Suriname, Trinidad and Tobago, Colombia, Bolivia, Paraguay, Ecuador and Brazil. The majority of cases occur in the Legal Amazon, where lagochilascariasis is related to human populations of low socioeconomic status inhabiting rural areas with the habit of consuming bush meat, especially wild rodents, which act as intermediate hosts of the parasite and therefore serve as sources of infection for humans. 1
Case reports of this parasite in cats are rare, and to date there has been no mention of the disease in southern Brazil. It was first described in Brazil by Dell’Porto et al 3 in São Paulo State; by Fraiha et al, 1 Amato et al 4 and Sudré et al 5 in Rio de Janeiro State; in Argentina by Led et al, 6 Sprent 7 and Romero and Led; 8 and in Uruguay by Sakamoto and Cabrera, 9 and Castro et al. 10
The life cycle of L minor seems to be heteroxenous. Cats become infected by ingesting cysts containing third-stage larvae present in organs and tissues of rodents. The larvae are released into the cat’s stomach and migrate to the upper portion of the digestive tract, and can subsequently be found in several locations of the head and neck, where they form abscesses containing larvae, adults and eggs. When the abscesses occur as fistulas the eggs are discarded into the environment; however, when the abscesses are located in the oropharynx the eggs are found in the feces (Figure 1). 11 Cases of pulmonary and central nervous system infection can lead to death before the diagnosis of the disease becomes clear. Pathogenesis and symptomatology of lagochilascariasis in humans are similar to those observed in cats. 12 The efficacy of ivermectin was tested by Barbosa et al 13 on third-stage larvae, in encysted and migratory phases in mice after experimental infection with L minor.

Life cycle of Lagochilascaris minor. (a) Definitive host, (b) intermediate host. 1 = unembryonated egg; 2–4 = eggs undergoing division; 5 = first-stage larva inside the egg; 6 = second-stage larva inside the egg; 7 = third-stage larva inside the egg, which is being swallowed; 8 = larvae hatched from the egg and penetrates in the intestine wall; 9 = larvae in the liver; 10 = larvae in the lungs; 11 = encysted larvae in the muscles; 12 = infected mouse ingested by the definitive host; 13 = larvae hatching from the cysts in the stomach; 14 = larvae in the esophagus; 15 = adult worms in the rhino- and oropharynx; 16 = larvae and adults worms can be find in the trachea. Parasitologia Humana 11th ed. ilustration. São Paulo: Atheneu; 2011 17
We report the occurrence of L minor in naturally infected domestic cats in southern Brazil, from rural areas of two districts in the Municipality of Pelotas, Rio Grande do Sul State: Quilombo (31º56’24,2’’S–52º 45’78,7’’W) and Vila Nova (31º44’19,5’’S–52º21’54,5’’W), 7th District; and Cascata (31º62’31,8’’S – 52º50’39,5’’W), 5th District.
The first cat diagnosed had anorexia, progressive weight loss and an abscess in the cervical area. It was treated with oral enrofloxacin for 5 days and showed improvement. This drug is not considered to be a good choice for treating cat fight abscesses owing to its poor activity against obligate anaerobes; however, it was the only drug available at the Federal University of Pelatos Veterinary Hospital. After a few days, another abscess appeared in the same area. The cat was then treated with oral trimethoprim and sulfa for 7 days, and again it showed improvement. However, after 2 weeks, two abscesses appeared and suppuration was detected. One hundred specimens of nematodes were found alive inside the abscesses and identified as L minor. The identification was based on Sprent, 7 Costa et al 14 and Bowman et al 15 ; the morphological characteristics of L minor and L major are similar, but the eggs of L major have approximately 33–45 pits around the circumference, whereas the eggs of L minor have approximately 15–25.
The cat was treated subcutaneously with injectable ivermectin at a dose of 400 µg/kg. The lesions healed rapidly, and after a 7-day interval the cat received another dose of ivermectin with no recurrence. This was the first case diagnosed in southern Brazil of a domestic cat parasitized by L minor. Seven other cats also had this parasitosis with the following signs and lesions: one with unilateral suppurative otitis that evolved into an abscess located below the affected ear; three with anorexia and progressive weight loss, until cachexia, showing an abscess in the pharynx area; one with myiasis in one of the ears evolving to internal otitis; one with an abscess on the neck; and one with cachexia. All animals were treated with injectable ivermectin at a dose of 400 µg/kg. The first four cats received a second dose of the medication after 7 days. In cases where the abscesses gave rise to fistulas, the parasites were removed, and the animal received enrofloxacin injectable for 7 days. All animals had an excellent recovery; however, one of them had sequelae of the internal otitis. The feces of some cats were examined and eggs of L minor were found. The eight cats were from three sites located in Quilombo and Cascata.
In Vila Nova, the death of four cats at different times was reported. According to the owner, the animals showed difficulty in feeding, lost weight and small lesions appeared on the neck, healed and recurred. The fifth cat was taken to the Laboratory of Wild Animals Parasitology, Federal University of Pelotas, because it had a 4–5 cm long lesion on the neck (Figure 2a). The animal was examined, and pus in the lesion was collected. The presence of parasites and eggs confirmed suspicion of lagochilascariasis (Figure 2b). The cat received a first dose of injectable ivermectin (400 µg/kg); however, it died before receiving the second dose. This was the 13th case in 2012, but we believe that more cases occurred without being diagnosed.
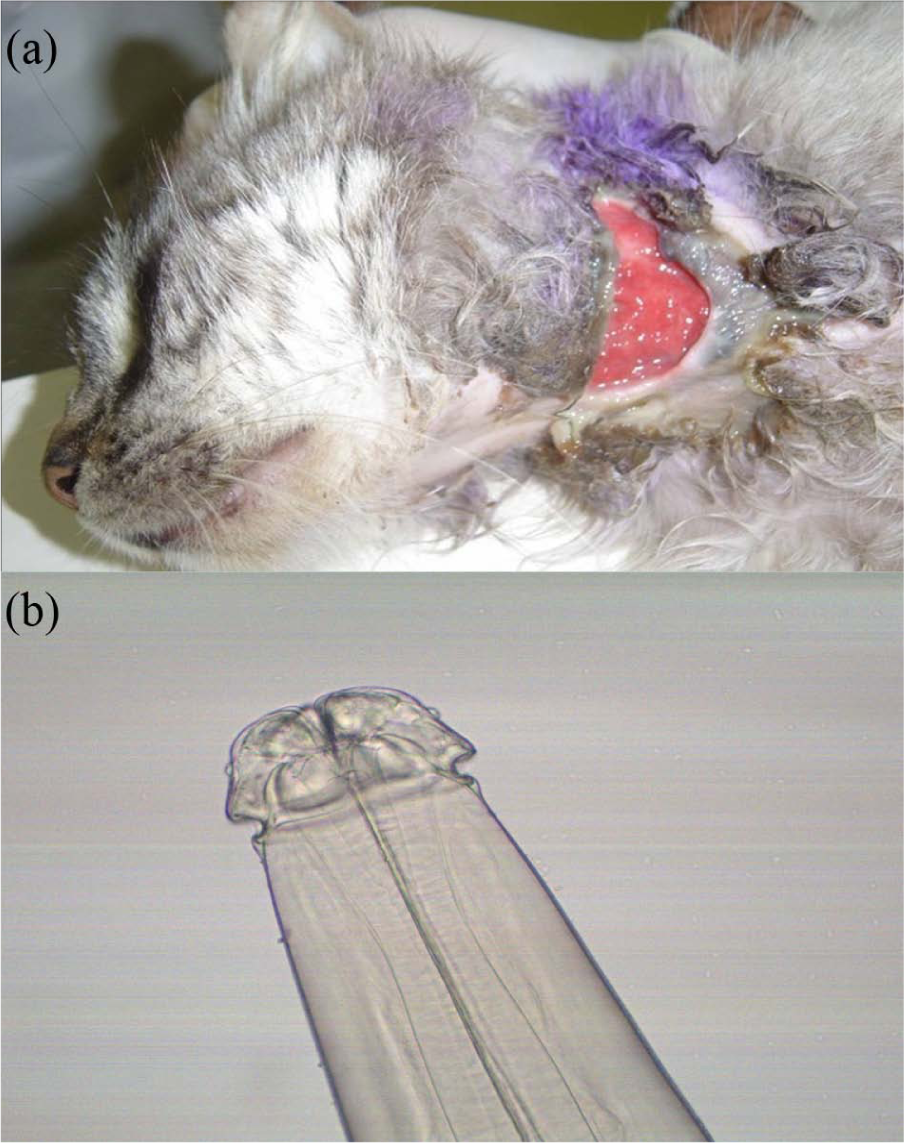
(a) Lagochilascariasis lesion in a domestic cat. (b) Lagochilascaris minor collected from the lesion in a domestic cat.
Conclusions
Lagochilascariasis is considered a rare disease.2,12,16 In this study we observed that clinical signs in cats are not always characteristic, and this disease is not easy to diagnose, it can be confused with viral and bacterial infections, and many infected animals die before the diagnosis is established. Perhaps because of this, few reports in cats have been published.
Because lagochilascariasis is an unknown disease in southern Brazil, and considering the zoonotic potential of this parasite, studies in this region began in July 2013 to clarify the biological cycle in domestic cats and their wild rodent intermediate hosts.
In the light of the foregoing we report the presence of L minor in southern Brazil. Lagochilascariasis illness is not always easy to diagnose in cats. We also confirm the effectiveness of the treatment with ivermectin.
Footnotes
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
