Abstract
Practical relevance:
Gastrointestinal (GI) surgical procedures are performed commonly in cats in general practice for both diagnostic and treatment purposes. It is essential that the surgeon understands and adheres to the principles of GI surgery in order that postoperative morbidity and mortality are kept to a minimum.
Audience:
This review is aimed at feline and general practitioners wanting to update their core skills in GI surgery. It discusses anatomical considerations and surgical principles, and aims to familiarise the surgeon with techniques (some well established, others newer) that will help to promote surgical success.
Equipment:
Standard general surgical equipment is required, together with the facilities to provide adequate pre-, intra- and postoperative patient care.
Evidence base:
The author draws on clinical experience and evidence from the literature, where appropriate, in reviewing the guidance and techniques under discussion.
Principles of GI surgery
Pre- and perioperative patient care
Good pre- and perioperative care of the patient is essential when dealing with the gastrointestinal (GI) tract, as for any surgery.1–5 Patients presenting for GI surgery may have fluid, acid–base and/or electrolyte imbalances, all of which must be corrected preoperatively. It is also vital that the response of the patient to fluid administration is monitored before, during and after surgery. This is especially important in cats, to prevent overhydration with its potentially life-threatening sequelae. 6
Antibiotic prophylaxis
The routine use of antibiotics is to be discouraged in order that we minimise the risks of development of multidrug resistance. Newer generation antibiotics (fourth and fifth generation cephalosporins), and the fluoroquinolones in particular, 7 should be avoided. Moreover, there are strong arguments, on both scientific and ethical grounds, that antibiotics with restricted use in human medicine (eg, imipenem, vancomycin) should never be used.

It is a well established principle that perioperative prophylactic antibiotics are warranted for some GI surgical procedures (see box, left). 8 However, there is a strong argument for not using antibiotics at all for gastric and small intestinal surgery when there is no inflammation and the potential for abdominal cavity contamination is low. 8
The choice of antibiotic(s) for antimicrobial prophylaxis is based on the most likely contaminating microorganism(s) for the GI tract and their susceptibility to the drug(s) used. Antibiotic selection is by necessity empirical but is based on clinical experience combined with published data. Antibiotic prophylaxis requires that the chosen drug is effective against at least 80% of probable pathogens (Table 1).
Pathogens associated with postoperative infection

Modified from Brown 9
Clavulanic acid potentiated amoxicillin (20 mg/kg) or cefuroxime (20–50 mg/kg), a second generation cephalosporin, are commonly used. Amoxicillin with clavulanate is useful in the treatment of Gram-positive bacilli and cocci, as well as Gram-negative bacilli including Escherichia coli. It also has an anaerobic spectrum that includes Bacteroides and Fusobacterium species and so is adequate for surgery of the colon, where Gram-negative aerobic and anaerobic bacteria predominate. Cefuroxime has activity against Gram-positive and Gram-negative organisms, with some activity against anaerobes. It is very effective against Enterobacter species, some Proteus species, E coli and Klebsiella species. For colonic surgery a combination parenteral treatment of cefuroxime and metronidazole (10 mg/kg slow IV) has also been advocated. 10



Saline-moistened laparotomy swabs (sponges) placed under the tines of a Baby Balfour retractor to protect tissues and to minimise desiccation and heat loss
Cefuroxime has a long half-life and, where indicated, a repeat dose need only be given after 3–4 h. This compares with 1.5 h for clavulanic acid potentiated amoxicillin.
Tissue handling
When performing GI tract surgery it is all too easy to overlook the fundamental requirement for gentle handling of tissue – which pertains not only to manual disruption but also to desiccation (see box below). Ensuring minimal surgical trauma is one of Halsted’s principles of surgical technique that should always be adhered to.
Surgical approach to the abdominal cavity
The most common and useful surgical approach to the GI tract of the cat is via a ventral midline laparotomy, which provides excellent access to all the intra-abdominal organs.
It is important to note that, when closing the laparotomy, suturing of the peritoneum is not needed and may, in fact, inhibit healing and predispose to adhesion formation.11–14 The peritoneum rapidly migrates and seals over a defect such as a closed laparotomy incision. Closure of the external rectus sheath provides a reliable strong closure of a laparotomy (Figure 2). 15

Cross sections of the ventral abdominal wall taken at three levels in a craniocaudal direction (a to c). Note the lack of covering of the deep layer caudally (c)
All fascial tissues heal slowly and, as extended wound support is required, it is essential to use an appropriate type and gauge of suture material. 14 The choice lies between simple interrupted sutures of non-absorbable monofilament nylon or polypropylene (eg, Prolene; Ethicon), or a simple continuous pattern (Figure 3) of a synthetic absorbable suture material which retains a high proportion of its tensile strength at 28 days. Examples of the latter include polydioxanone (eg, PDS II; Ethicon), which retains 50% strength, and polyglyconate (eg, Maxon; Covidien), which retains 41%. 16 The gauge depends on the size of the patient; 2 metric (3/0) is suitable for most cats, although 3 metric (2/0) may be considered for some of the larger breeds.

Simple continuous closure of the linea alba
While non-absorbable monofilament suture material (nylon or polypropylene) has been used for fascial closure in cats, 17 it reportedly leads to an increased risk of suture sinus formation 18 and infection, 19 and thus its use cannot be recommended. Moreover, the external abdominal fascia is no more likely to dehisce with a continuous suture pattern than when simple interrupted sutures are used. 17
The security of a continuous suture is only as good as the two end knots and, therefore, a sufficient number of throws must be used with this pattern. Polydioxanone requires five throws for the start knot and seven for the end knot. 20 The use of a barbed knotless wound closure device comprised of a copolymer of glycolic acid and trimethylene carbonate (V-Loc 180; Covidien), which has an extended absorption profile, can also be considered for the fascia. 21
Anatomy of the GI tract wall
A cross section at any level of the stomach or intestines will reveal four layers: serosa, muscularis, submucosa and mucosa (see box on the right).
Healing processes and implications for suturing
Though the GI tract is constantly exposed to food, enzymes and bacteria, by virtue of its rich vascular supply, healing is usually uncomplicated and is relatively fast due to the capacity of the epithelium to regenerate and the natural defences against leakage provided by the omentum and peritoneum. The healing process itself is arbitrarily divided into three phases, but as with all physiological events there is overlap between these, and the divisions are there for ease of understanding.

For optimal healing by first intention in the intestines, accurate apposition of wound edges is required. Proper alignment of the submucosa ensures that there is good approximation of blood vessels which carry the vital cells and factors needed during the healing process. Direct apposition promotes rapid re-epithelialisation and deposition of well vascularised collagen, and minimises the risk of reducing the bowel lumen diameter.
The large intestine poses a particular problem to the surgeon because healing is slower than for the stomach or small intestine. This has implications for suturing (see below). Also, bowel preparations (oral antibiotics, multiple enemas) are contraindicated as enemas increase the risk of peritoneal spillage by producing a liquid slurry within the colon, which then readily leaks. 10
Choice of suture pattern
One- or two-layer appositional suturing is the most logical within the GI tract. Two-layer closure is usually reserved for the stomach; single layer closure is adequate for the small and large intestines, and is less likely to result in stenosis.22,23 For two-layer closure, the inner layer is the mucosa and submucosa, while the outer layer is the muscularis and serosa. The selection of suture pattern is very much an individual choice, the options being simple interrupted (see box below), interrupted crushing suture or simple continuous.
The critical step with appositional suturing is to pass the suture material through the submucosa (Figure 4), which is the vascular and collagen-containing layer of strength within the intestinal tract. A swaged-on taper point needle is preferred. The use of eyed needles is contraindicated in GI surgery.

(a,b) Appositional intestinal closure showing a simple interrupted suture passing through the submucosal layer of the intestine
Choice of suture material
The suture gauge needs to be 2 metric (3/0) for the stomach; 1.5 metric (4/0) is preferred for the small intestine. A monofilament synthetic absorbable suture such as poliglecaprone 25 (Monocryl; Ethicon) should be used. This material is absorbed by hydrolysis and, as a monofilament, produces minimal friction and drag as it passes through tissue. It has the added advantage of having minimal memory, which allows snug, safe knots to be produced. The absorption data of poliglecaprone 25 show that at 7 days it retains 50–60% of its strength, at 14 days it retains 20–30% and at day 21 it has lost all tensile strength. This makes it ideal for gastric and small intestinal surgery where prolonged wound support is not required. By contrast it is not appropriate for fascia or other tissue requiring extended wound support. Where there is intraperitoneal sepsis or severe inflammation of the bowel wall, for example, it would be prudent to consider a suture material providing extended support, such as polydioxanone, as the healing process may be delayed. 24
Chromic catgut should never be used in feline GI tract surgery as it usually causes a severe inflammatory response, which can lead to severe fibrosis and marked intestinal narrowing.16,25,26


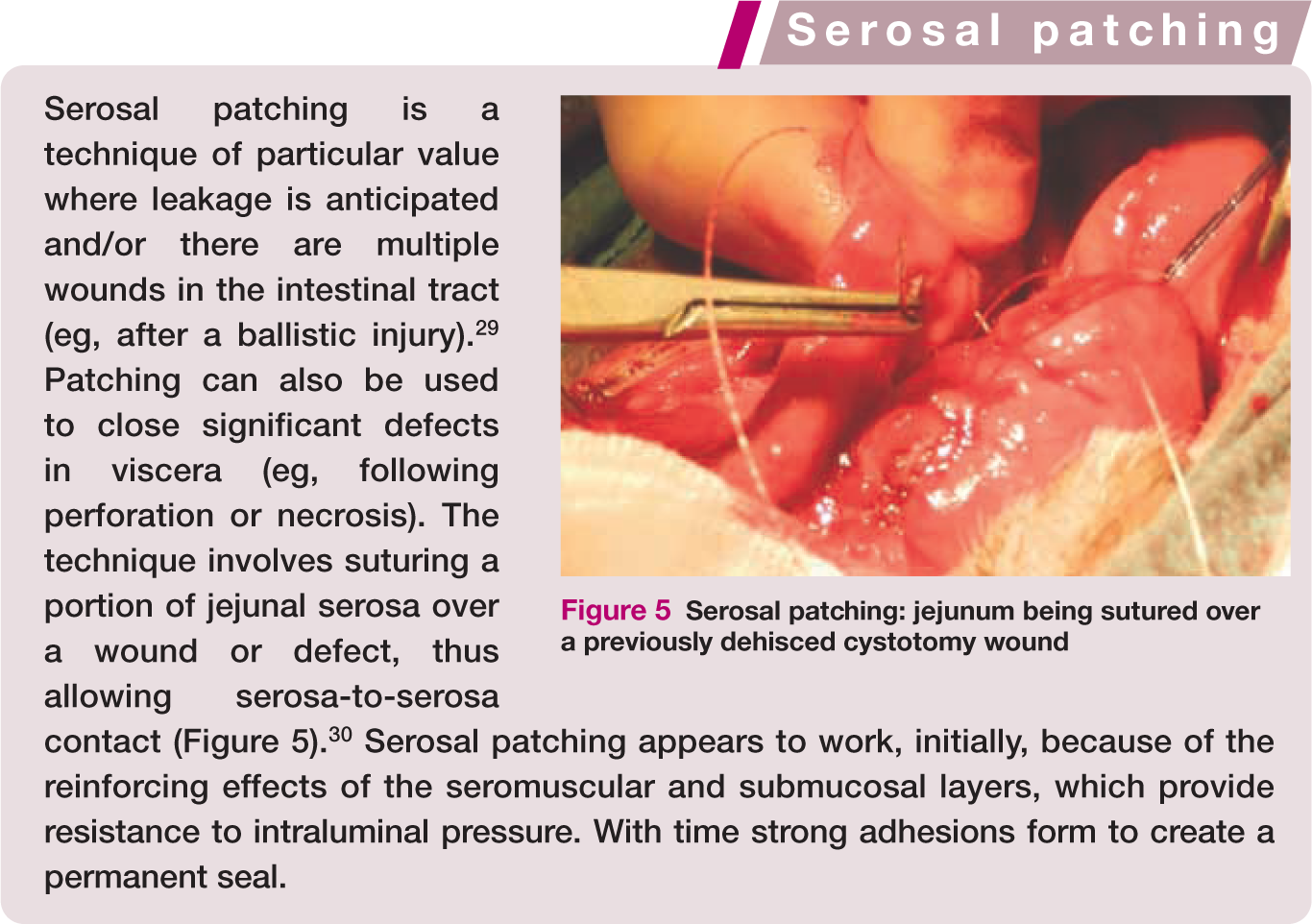

Serosal patching: jejunum being sutured over a previously dehisced cystotomy wound
Special considerations in the colon
It has been established that initially the wound strength at the surgical site within the colon is weak (around 30% of normal strength after 48 h), 27 and that this weakness will last for a minimum of 3–4 days. Subsequently, the wound is at increased risk of dehiscence due to collagen degradation by matrix metalloproteinases. Return of wound strength is slower in the colon compared with the small intestine, with around 75% of normal strength regained at 4 months post-surgery 27 versus 100% at 3 weeks, respectively. Care must be taken in apposing the tissue and a suture such as 1.5–2 metric polydioxanone is preferred, as it provides extended wound support. Vascular supply to the area can be augmented by overlying the wound with omentum (see box, above left).
An alternative technique that is extremely useful in reinforcing and sealing intestinal wounds where leakage is anticipated is serosal patching (see box, above right).
Enterotomy
An enterotomy is an incision through the intestinal wall and is usually used to remove foreign bodies, to carry out full thickness biopsy or, occasionally, to access the common bile duct via duodenotomy.31,32
When performing an intestinal procedure, it is best practice to exteriorise the bowel to be incised and to pack it off from the remainder of the abdominal cavity with saline-soaked swabs (sponges) (Figure 6). Prior to enterotomy, the intestinal contents should be milked away, cranially and caudally, from the proposed incision site. If a foreign body is to be removed, the incision is ideally made distal to the obstruction in a healthy part of the bowel. A longitudinal incision is made with a number 15 scalpel blade in the antimesenteric portion of the intestine (Figure 7). When a full thickness biopsy is required this can be modified to an elliptical incision with a blade or fine scissors (Figure 8).

Intestine isolated from the abdominal cavity with moist swabs

(a,b) A longitudinal incision is made with a number 15 scalpel blade in the antimesenteric portion of the intestine

For full thickness biopsy an elliptical incision is made with a blade or fine scissors
Prior to closure of the site any mucosa ballooning out from the wound (Figure 9) is trimmed away either with a scalpel or fine Metzenbaum scissors. If routine longitudinal closure is likely to lead to significant narrowing of the intestinal lumen, a transverse closure is preferred (Figure 10a). In the author’s experience, transverse closure is always required in cats following elliptical biopsy. 33 The defect is closed with a simple interrupted or simple continuous pattern of 1.5 metric monofilament absorbable suture (Figure 10b,c). The suture line can then be reinforced by wrapping omentum around the wound and intestine; this should be tacked to the serosa with 1.5 metric suture material.

Mucosa ballooning out from the incised wound in a very inflamed jejunum

(a–c) Transverse closure of the intestine (using I.5 metric monofilament absorbable suture material) is always required following elliptical biopsy
An alterative to creating an elliptical incision for biopsy is to use a 4 or 6 mm skin punch biopsy instrument, which is ‘pushed’ through from the serosa to the lumen (Figure 11). The resulting defect is closed, either longitudinally or transversely (depending on the width of the intestine), with simple interrupted 1.5 metric sutures. 34
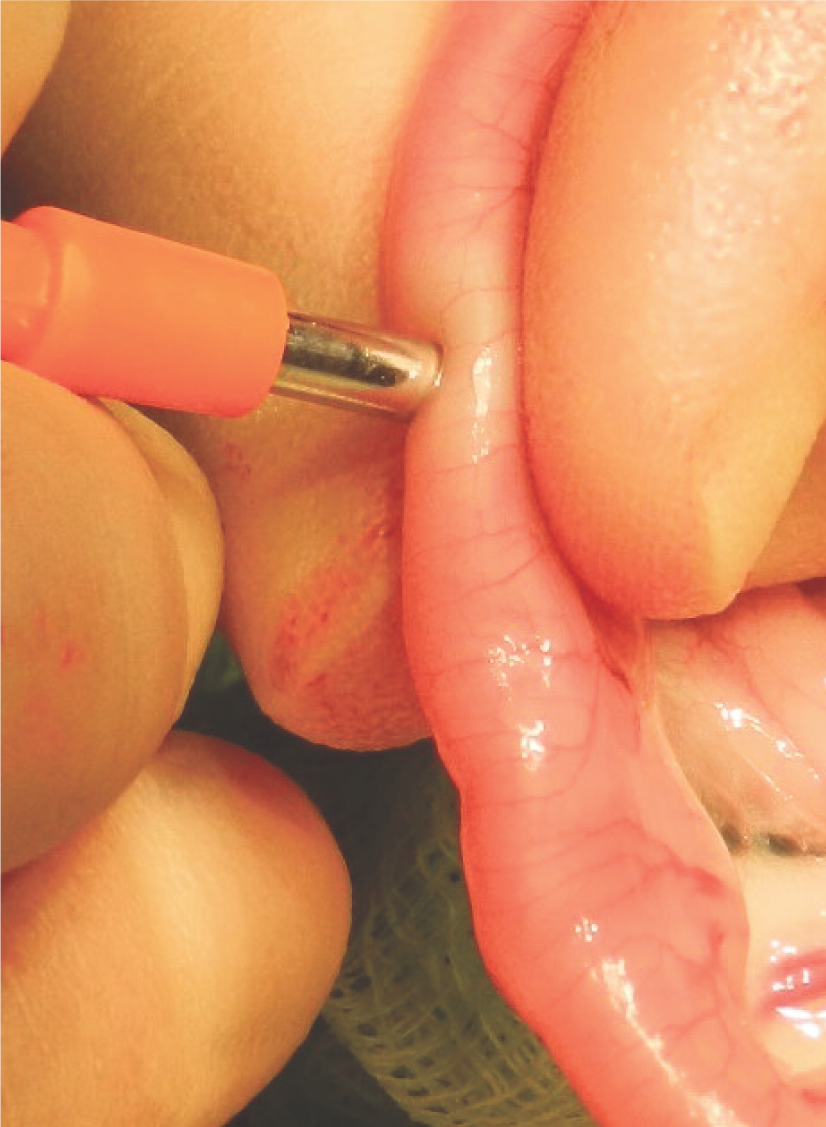
A 4 mm skin punch biopsy instrument being ‘pushed’ through from serosa to lumen
Enterectomy
Enterectomy (intestinal resection and anastomosis) is usually necessary as a result of a foreign body impaction or intussusception that has resulted in ischaemia and necrosis, or for resection of intramural tumours. Transection should be carried out with a scalpel and resected tissue submitted for histological examination.
If there is lumen disparity, this can most readily be addressed by spatulating the smaller lumen (Figure 12a). Simple interrupted sutures of 2 or 1.5 metric (3/0 or 4/0) absorbable monofilament suture should be placed at the mesenteric and antimesenteric borders initially (Figure 12b). The ends should be left long to allow their use as stay sutures; tension can be maintained on the wound edges by gentle traction on the stays. The intestine is then sutured using an appositional interrupted or continuous pattern on one side. Once closed, the intestines are rotated and the opposite side is closed. The author prefers to use simple interrupted sutures in the cat as there is less risk of creating a ‘purse string’ narrowing effect, compared with a continuous pattern.

(a) Lumen disparity is most readily addressed by spatulating the smaller lumen at its antimesenteric border. (b) Simple interrupted sutures of 2 or 1.5 metric (3/0 or 4/0) absorbable monofilament material are placed at the mesenteric and antimesenteric borders initially. The ends are left long to allow their use as stay sutures; tension can be maintained on the wound edges by gentle traction on the stays
If there is a large lumen disparity – for example, after ileocolostomy or if the pylorus is resected during a gastroduodenostomy (Bilroth I) procedure – the larger lumen (distal colon or gastric antrum) can be partially closed with a simple continuous suture pattern until its diameter matches that of the remaining colon or proximal duodenum (Figure 13).

Where there is a big lumen disparity, the larger lumen (eg, distal colon) can be partially closed with a simple continuous suture pattern until it matches that of the remaining colon or proximal duodenum
Alternatives to suturing for anastomosis
Stapling devices
Stapling devices are increasing in popularity in veterinary surgery and can be used either to form an anastomosis or to close a bowel wound. GI anastomosis (GIA) instruments create a functional rather than a true end-to-end anastomosis (EEA). The technique also requires the cut ends of the bowel to be closed either by hand suturing or by use of a suitably sized thoracoabdominal stapler. 35
The main described advantage of stapling is the ease in which intestine of different diameters can be anastomosed – for example, when the oral portion is dilated after intestinal obstruction or when anastomosing small intestine to colon. There is less tissue manipulation, and no difference in wound-bursting strength or absolute strength during the healing process compared with sutured anastomosis. 36 Disadvantages are similar to those of sutured anastomosis, with leakage, abscess formation and late foreign body obstruction at the stapled site having been described. In addition, mucosal ulceration has been described as a not uncommon complication of stapling. 36

It is essential to note that use of a GIA linear stapler is not appropriate in cats (and restricted to larger dogs), as the forks need to enter the intestinal lumen. The smaller endoscopic linear cutting stapler can be used in cats, but it is expensive to use. Costs may be offset by reduced surgical times, especially where there is luminal disparity.
Tubular EEA stapling can be carried out transcaecally 25 or transrectally 37 in cats. In 15 cats that underwent the ‘single surgical field’ transcaecal approach there were no long-term complications. 25 With a ‘dual surgical field’ transrectal approach, 2/10 cases developed strictures at the anastomosis site. Further prospective clinical evaluation of these techniques is warranted, with current data suggesting that a ‘single surgical field’ approach should be adopted in cats. 10
For the transcaecal approach the caecum and colon are exteriorised and packed off with moistened swabs. Vasa recta are ligated and divided as necessary and colonic contents are massaged away from the transection sites. Doyen intestinal forceps are placed proximal and distal to these planned sites. A Furniss purse string instrument is placed at the proximal and distal limits of the colectomy site, some 2 cm distal to the caecum and 2 cm proximal to the pelvic brim. This allows a purse string suture to be placed at both sites using 2 metric (3/0) monofilament suture material on a straight needle. It is essential that this is placed accurately to allow even inclusion of the intestine in the stapling device. The colon is transected with a scalpel blade, using the Furniss purse string instrument as a cutting guide.
After removal of the purse string instrument, a 3 cm incision is made on the antimesenteric surface of the caecum, and an ovoid sizer lubricated with sterile water-soluble gel is introduced through the caecal incision and advanced normograde to the proximal colonic margin. Ovoid sizers are essential to allow measurement of bowel diameter so that an appropriately sized staple cartridge is chosen and that the colon is dilated sufficiently to facilitate introduction of the EEA instrument. For most cats, either a 25 mm or 21 mm cartridge is appropriate.25,37 The EEA stapler is introduced transcaecally into the colon; the intestinal ends are eased over the cartridge and anvil and the purse string sutures are tied. The bowel segments are then pushed together and the EEA stapler is closed to fire the staples. The stapler is then opened slightly and rotated to ease it around the staple line so that it can be removed.
The stapled anastomosis should be inspected for gross leakage and/or haemorrhage and the caecal wound closed with either an appositional suture pattern or thoracoabdominal stapler. 25
Biofragmentable anastomosis ring
The biofragmentable anastomosis ring (BAR device, Valtrac; Covidien) is introduced after colonic resection, which is carried out using the same technique of Furniss purse string suture placement that is used for an EEA stapler. Once the colon is resected the BAR device is passed into the orad lumen of the colon using a holding device, and the purse string suture is then tied securely against the internal barrel of a 25 mm BAR ring (with a 1.5 mm gap width). Once secured the BAR ring is placed in the aboral colonic segment and the purse string suture tied. The BAR ring is snapped shut by applying digital pressure on its caps through the colonic wall. This creates an inverting serosa-to-serosa anastomosis. The wound is checked for leakage and haemorrhage before an omentum wrap is placed over the site. 38
The major disadvantages of the EEA and BAR devices are cost and size. It is clear that they are not suitable for all cats.
Key points
Perioperative antibiotics should only be used if they are absolutely necessary. They are not a substitute for poor surgical technique.
Abdominal exposure should be achieved via a ventral midline approach. Only closure of the skin, subcutaneous tissues and fascia is required; muscle and peritoneum should not be sutured.
Anatomical realignment of the intestinal wall allows for optimal healing, with simple appositional sutures being preferred.
Small gauge suture materials are favoured, with 1.5 or 2 metric sizes being ideal in cats.
Synthetic absorbable sutures cause less reaction than non-absorbable sutures. Catgut should never be used in intestinal surgery, due to the reaction it induces.
In general, feline intestines heal well and the reported risks of dehiscence are low.
Footnotes
Funding
The author received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this article.
Conflict of interest
The author does not have any potential conflicts of interest to declare.
