Abstract
This report describes a cat that presented with abdominal pain and worsening azotaemia following unknown trauma. Further diagnostic investigations and surgery confirmed bilateral ureteral trauma. The cat was initially managed surgically by bilateral ureteroureterostomy over ureteral stents. The clinical signs and biochemical parameters rapidly resolved, but 2 months later the cat developed signs consistent with sterile cystitis that was unresponsive to medical management. Removal of the ureteral stents resulted in severe azotaemia as a result of stricture formation at the previous ureteral anastomosis site. The ureteral stents were initially replaced with soft stents, but subsequently cut short owing to the persistence of clinical signs of cystitis. Following shortening of the ureteral stents severe azotaemia was again observed. The resulting pelvic dilatation allowed for placement of bilateral subcutaneous ureteral bypass (SUB) systems, which resulted in alleviation of all clinical signs 12 months after SUB placement.
Case Report
Traumatic ureteral injury is uncommon and most often results from road traffic accidents or focal blunt trauma. Surgical options described in the veterinary literature for management of ureteral rupture include unilateral ureteronephrectomy, ureteroureterostomy, neoureterocystostomy and ureteral ileal graft.1–4 Ureteral anastomosis over a ureteral stent and use of a subcutaneous ureteral bypass (SUB) system are novel options that have recently become available; however, there are no reports of their use following trauma.
A 7.6-year-old (3.2 kg) spayed female Burmilla cat presented to the referring veterinarian after 24 h of inappetence, lethargy and absence of urination. Serum biochemistry revealed azotaemia, which deteriorated despite intravenous (IV) fluid therapy, and the cat was subsequently referred. At presentation the cat had a temperature of 36.7°C, a heart rate of 150 beats per minute and a respiratory rate of 28 breaths per minute. The cat was depressed and painful, and bruising was present on the cranial ventral abdomen. A 2 cm abdominal wall defect was palpable caudal to the left costal arch. Initial serum biochemistry revealed hyperkalaemia (5.6 mmol/l, reference interval, 3.6–4.6 mmol/l), hyperglycaemia (15.8 mmol/l, reference interval, 4.2–6.6 mmol/l), elevated creatinine (1085 µmol/l, reference interval, 50–140 µmol/l) and urea that was above measurable limits of the analyser. Urine specific gravity was 1.014. While receiving ongoing intravenous fluid therapy with 0.9% sodium chloride (NaCl; Vetivex 1, Dechra), the cat was prepared for general anaesthesia and radiography of the thorax and abdomen. Repeat serum biochemistry following anaesthesia induction revealed that the hyperkalaemia had increased to 6.3 mmol/l (reference interval, 3.6–4.6 mmol/l). Calcium gluconate (calcium gluconate injection 10%; Hameln Pharmaceuticals) 1ml/kg intravenously (IV), neutral insulin (Actrapid; Novo Nordisk) 0.2 IU/kg IV and glucose (Glucose 25 g/50 ml; Hameln pharmaceuticals) 0.4 g/kg IV were administered. The cat was premedicated with methadone (Physeptone 10 mg/ml; Martindale Pharmaceuticals) 0.2 mg/kg IV and general anaesthesia was induced with alfaxalone (Alfaxan; Vetoquinol) 1.9 mg/kg IV. The patient was intubated and anaesthesia was maintained on isoflurane (Isoflo, Abbot); inspiratory oxygen was provided throughout the procedure. Thoracic radiographs were unremarkable. Survey abdominal radiographs revealed loss of detail in the retroperitoneal space. Intravenous urography was performed with iohexol (Omnipaque 300 mg/ml; GE Healthcare) 600 mg/kg IV; after 15 mins, minimal contrast was present in the renal parenchyma, pelvis or ureters bilaterally (Figure 1). Abdominal ultrasound showed normal renal perfusion, bilateral pyelectasia and the presence of retroperitoneal fluid.
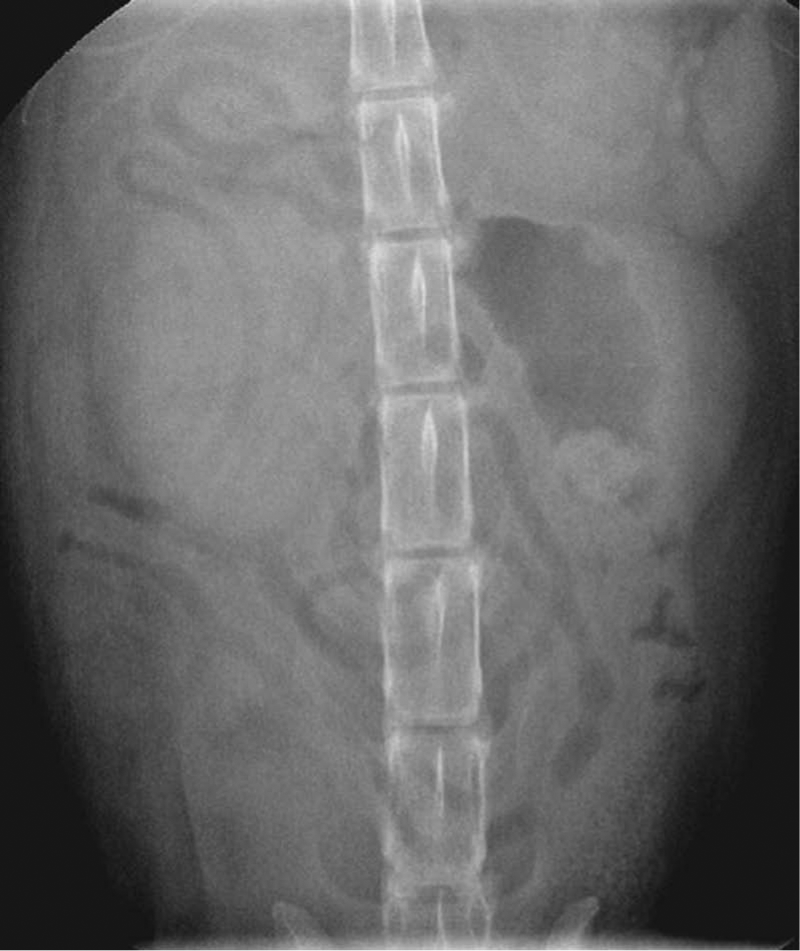
Intravenous urogram 15 mins after contrast administration. There is contrast present in the renal parenchyma, but no contrast present in the renal pelvis or ureters
Bilateral ureteral trauma was suspected and therefore an exploratory laparotomy was performed. Diffuse haematoma and fluid were present in the retroperitoneal space, and direct visualisation confirmed bilateral ureteral trauma. Transection of the left proximal to mid-ureter and a 5 cm crush injury of the mid-section of the right ureter were identified. A left-sided paracostal body wall rupture and small ventral diaphragmatic rupture were also present. A 22 gauge intravenous catheter was placed into the renal pelves bilaterally to achieve urine output; a continual flow of urine was observed from both kidneys. The crushed mid-section of the right ureter was resected; stay sutures were applied to the ends of the ureter to aid atraumatic manipulation. An angle-tipped hydrophilic guidewire (Weasel wire; Infiniti Medical) 0.018 inches in diameter and 180 cm in length was placed in an anterograde manner through the 22 gauge intravenous catheter into the renal pelvis and advanced into the proximal ureter. A 2.6 F (length: 40 cm) catheter push (ureteral dilator) was placed over the guidewire into the renal pelvis and proximal ureter. A ventral midline cystourethrotomy was performed and extended into the proximal urethra to allow visualisation of the trigone, and another hydrophilic guidewire was placed in a retrograde manner through the right ureteral orifice. The dilator in the proximal ureter was placed over the guidewire in the distal ureter and passed through the right ureteral orifice. The guidewire that had been placed through the ureteral orifice was removed and the proximal guidewire was passed through the dilator all the way to the bladder. A 2.5 F 12 cm double-pigtail stiff ureteral stent (Vet Stent-Ureter; Infiniti Medical) was placed over the guidewire and passed in an anterograde fashion immediately after the ureteral dilator as it was removed through the cystotomy. Right renal descensus was performed along with a right nephropsoaspexy and nephrocystopexy to relieve the tension on the ureteral anastomosis. The proximal and distal ends of the right ureter were sutured over the ureteral stent with 0.7 metric polydioxanone (Ethicon PDS II; Johnson & Johnson Medical). Urine flow was immediately evident through the ureteral stent into the bladder. The distal end of the right ureteral stent was cut owing to its excessive length in the urinary bladder. A 3 mm section of the left ureter was removed to debride the ruptured ureteral ends, and the same procedure was repeated on the left side; however, owing to lack of tension on the ureteral anastomosis site, left renal descensus was not performed. Postoperative radiographs documented the position of the ureteral stents (Figure 2). Urine output was matched with intravenous fluid therapy of 0.9% NaCl. Potassium concentrations were monitored and supplemented as necessary. Clinical demeanour and serum biochemistry parameters progressively improved: 7 days postoperatively, urea was 26.3 mmol/l (reference interval, 3.0–10.0 mmol/l) and creatinine 127 µmol/l (reference interval, 50–140 µmol/l) and the cat was discharged. Two months postoperatively the cat developed stanguria and dysuria. Symptomatic treatment with oral buprenorphine 0.02mg/kg provided minimal improvement. The cat urinated up to nine times a day; typically, small amounts of urine were produced. Urinalysis and bacteriology ruled out the presence of a urinary tract infection. Sterile traumatic cystitis was suspected owing to the presence of the stents, and removal of the stents was therefore planned as following ureteral healing they were perceived to be unnecessary. Haematology was unremarkable; biochemistry revealed a mild elevation of urea 18.9 mmol/l (reference interval, 6.1–12.0 mmol/l) and creatinine 196 µmol/l (reference interval, 107–193 µmol/l). Urine specific gravity was 1.030.

Postoperative radiograph following placement of bilateral ureteral stents
Diagnostic imaging was performed to rule out any other cause for lower urinary tract signs and to assess the relative contribution of each kidney to total glomerular filtration rate (GFR) and guide surgical decision making. Abdominal ultrasonography revealed mild pylectasia in the left kidney (2.5 mm) and the presence of the proximal aspect of the stents within the renal pelves bilaterally. Survey radiographs of the abdomen revealed that the position of the ureteral stents was unchanged. A retrograde vaginourethrocystogram was unremarkable. Renal scintigraphy was performed by administration of frusemide (Dimazon 5%; Intervet) 3 mg/kg IV followed by 99mTc-DTPA – technetium 99m-labelled diethylenetriamine penta-acetic acid 137.8 megabecquerel (MBq) IV. 5 Renal scintigraphy revealed that the left and right kidney were responsible for approximately 66% and 33% of total GFR, respectively, and indicated the importance of preserving both kidneys (Figure 3).

Renal scintigraphy showing a higher concentration of technetium (red areas) in the left kidney. The right kidney is located further caudally following renal descensus
Four months after the initial surgery a caudal midline laparotomy and small ventral cystotomy were performed to remove the ureteral stents. Stent retrieval was straightforward with no presence of adhesions. Bacteriology of urine and bladder wall was negative. Seventy-two hours after surgery the cat was discharged home, but represented 24 h later owing to vomiting and anuria. Repeat biochemistry revealed hyperkalaemia 7.4 mmol/l (reference interval, 3.8–5.3 mmol/l), hyperphosphataemia 5.9 mmol/l (reference interval, 0.9–2.2 mmol/l), elevated urea 82.6 mmol/l (reference interval, 6.1–12.0 mmol/l) and elevated creatinine 1564 µmol/l (reference interval, 74.5–185.3 µmol/l). The hyperkalaemia was treated and general anaesthesia was induced as previously described. Abdominal ultrasound revealed bilateral renal pelvic dilatation: the right renal pelvis was 7.5 mm and the left renal pelvis was 8 mm.
Midline ventral laparotomy and ventral cystotomy were performed to allow the ureteral stents to be replaced with 2.5 F 12 cm soft stents (Vet Stent-Ureter; Infiniti Medical) via a retrograde approach. Five days after surgery, repeat biochemistry revealed that both urea and creatinine were within the respective reference intervals. The cat made a good recovery from surgery; however, the dysuria continued intermittently, but gradually worsened 6 months after replacement of the ureteral stents. The cat was prescribed prednisolone (Prednicare 1 mg; Animalcare) 0.5 mg/kg q12h PO along with buprenorphine for intermittent treatment of the sterile cystitis. Numerous urine samples submitted for microbial culture were all negative. Clinical signs improved while on medication, but did not completely resolve. Seven months after replacement of the ureteral stents, the cat presented to the referring veterinarian with rectal prolapse, which was manually reduced. Over the following 6 months the dysuria became less responsive to prednisolone and buprenorphine, and recurrent mild rectal prolapse occurred. Examination of the rectum and colon revealed no underlying cause for the prolapse, and observation indicated that it was associated with episodes of stranguria. A decision was made to shorten the ureteral stents to prevent irritation at the proximal urethra and bladder trigone. 6 While shortening of the stents was recognised to potentially compromise their function leading to renal pelvic dilatation, it was recognised that this would then allow replacement of the ureteral stents with a SUB system (Norfolk Vet Products), providing an alternative method of renal pelvic drainage.
Following general anaesthesia and routine surgical preparation, the distal ends of the stents were cut just proximal to the ureteral orifice via a ventral cystotomy; a colopexy was performed. Ten days after surgery the cat began vomiting, but continued to void good volumes of urine. Repeat biochemistry revealed hyperkalaemia (5.65 mmol/l [reference interval, 3.6–4.6 mmol/l]), elevated creatinine (760 µmol/l [reference interval 50–140 µmol/l]) and urea that was above measurable limits (reference interval >36 mmol/l). Abdominal ultrasound revealed bilateral moderate renal pelvic dilatation. We elected to proceed with surgical placement of bilateral SUB via a midline laparotomy. An 18 gauge intravenous catheter (18G Jelco IV Catheter Radiopaque; Smiths Medical) was placed into the right renal pelvis from the caudal pole. Intraoperative fluoroscopy was used to confirm position by injecting 0.5 ml of a 1:1 solution of 300 mg/ml of iohexol and saline. A hydrophilic guidewire was placed through the intravenous catheter under fluoroscopy and allowed to coil within the renal pelvis. A 6.5 F locking loop nephrostomy catheter with a preplaced dacron cuff (locking loop kidney catheter and introducer; Norfolk Vet Products) and a hollow trocar was advanced over the guidewire until it reached the renal pelvis. The hollow trocar was removed and the catheter was advanced into the renal pelvis, the locking string was pulled taught and maintained in tension with a mosquito haemostat to prevent premature dislodgement. Sterile cyanoacrylate glue (Histoacryl; Braun) was applied between the cuff of the catheter and the kidney to secure it in place. Stay sutures were placed around the apex of the bladder just to the right and a number 11 blade was used to make a stab incision into the bladder lumen. A cystostomy catheter (bladder catheter and introducer; Norfolk Vet Products) with a hollow trocar was advanced through the hole into the bladder lumen until the cuff of the catheter was against the bladder serosal surface. A purse string suture was placed around the catheter with 1.5 metric polydioxanone. Four additional simple interrupted sutures were also placed full thickness through the bladder wall and the catheter cuff. Sterile cyanoacrylate glue was placed between the catheter cuff and bladder wall for added security. The same procedure was repeated on the left. The nephrostomy and cysostomy tubes were exited via stab incisions in the abdominal wall and connected to two ports (Bypass-Le petite bypass companion port) which were secured in the subcutaneous space. A Huber needle (PosiGrip Huber Point Needle; Norfolk Vet Products) was used to inject iohexol 1:1 dilution in saline, 5 ml of 300 mg/ml directly into the bypass port confirming there was no leakage (Figure 4). The same was performed on the left side. Seven days later the cat was discharged from the hospital. Four weeks after SUB placement, biochemistry revealed a urea of 17.8 mmol/l (reference interval, 6.1–12.0 mmol/l) and a creatinine of 243 µmol/l (reference interval, 74.5–185.3 µmol/l). One year after surgery the cat was reportedly urinating 3–4 times a day and producing larger volumes of urine. There were no more episodes of stranguria or vocalising, and there was a forceful steady flow of urine. Long-term monitoring recommended involved urinalysis for bacteriology, flushing of the ports, and haematology and biochemistry every 3–6 months. Blind cystocentesis was contraindicated owing to the possibility of damage to the nephrostomy or cystostomy tubes.
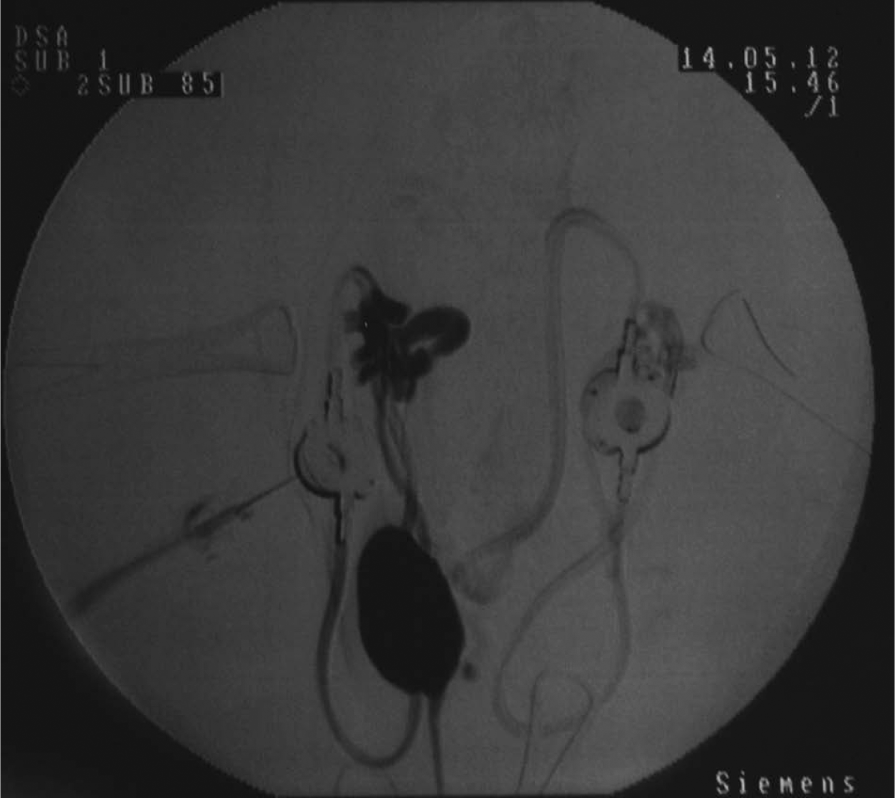
Digital subtraction fluoroscopy following bilateral subcutaneous ureteral bypass placement. Contrast was injected into the port to assess for any urinary leakage prior to closure. A dilated right renal pelvis and proximal ureter are present, and contrast is present in the bladder and proximal urethra. There is no evidence of any contrast leakage
Ureteronephrectomy was not an option in this case as both ureters were involved. The long-term kidney function following trauma was unknown and preservation of both kidneys was considered optimal. The luminal diameter of an unobstructed feline ureter is approximately 0.4 mm in diameter, 7 and primary anastomosis of the ureter is very challenging and may result in ureteral stricture.8,9 The use of a hydrophilic guidewire placed in a normograde direction was essential to allow identification of the ureteral lumen and ureteral repair over the ureteral stents. Unfortunately, the cat developed signs consistent with sterile cystitis. It has now been recognised that sterile cystitis can affect 36% of cats that have ureteral stents placed and will persist in approximately 7%.10,11 The trigone in the cat is located in the proximal urethra and, as a result, the presence of the stents exiting at this location may make sterile cystitis more likely. This is supported by the fact that only 1/9 cats developed sterile cystitis in a series where neoureterocystostomies were performed along with ureteral stent placement. 2 Although at the time of initial surgery bilateral ureteral implantation at the bladder apex with the ureteral stent was an option, it subsequently became a more challenging option owing to the adhesions and scar tissue that were present. At this time, SUB placement was considered a better option.
Removal of the stents was considered appropriate for the management of suspected sterile cystitis. The subsequent clinical and biochemical deterioration was due to the development of ureteral strictures at the site of the previous ureteral anastomosis. Predisposing factors for the development of the strictures are severe retroperitoneal bruising, previous surgery or tension present at the right ureteral anastomosis. 12 The SUB system had become available and was considered the most optimal solution to address the ureteral strictures and resolve the sterile traumatic cystitis as there are no implants located near to the bladder trigone. 13 SUB placement involved insertion of a locking loop nephrostomy catheter, a cystostomy catheter and a connecting subcutaneous port that is placed between the body wall and the skin. 14
Conclusions
Traditional surgical methods used to address ureteral injuries in this cat are likely to have been unsuccessful owing to the severity of trauma and the small diameter of the feline ureter. The advent of ureteral stenting and more recently the SUB system provided a useful alternative for treatment and management of this challenging case.
Footnotes
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this case report.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
