Abstract
The objective of this study was to evaluate the clinical usefulness, in terms of analgesic efficacy and safety, of ultrasound-guided pudendal nerve block performed with bupivacaine in cats undergoing perineal urethrostomy. Eighteen client-owned male cats scheduled for perineal urethrostomy were enrolled in the study and assigned to one of two treatment groups. The pudendal nerve block was performed under general anaesthesia, as described elsewhere, with 0.3 ml/kg of either saline (group C) or 0.5% bupivacaine (group B) – the total injection volume being split equally between the two sites of injection (left and right). Intra-operatively, assessment of nociception was based on the rescue analgesics requirement, as well as on the evaluation of changes in physiological parameters in comparison with the baseline values. Postoperative pain assessment was performed using three different pain scales at recovery and then 1, 2 and 3 h after recovery. Cats in group B showed lower heart rates and required fewer analgesics during surgery than group C. Postoperatively, group B had lower pain scores and needed less rescue buprenorphine than group C. Iatrogenic block-related complications were not observed. In conclusion, the ultrasound-guided pudendal nerve block can be considered clinically useful in feline medicine as it provides reliable analgesia in cats undergoing perineal urethrostomy.
Introduction
Urethral obstruction is a common disease affecting the male feline population and, as a consequence, perineal urethrostomy (PU) is a widely performed surgical procedure in cats.
Traditionally, the best ways to provide effective pain management during PU are either systemic opioids administration or lumbo-sacral epidural analgesia.
Intravenous administration of opioids at constant-rate infusion is a safe and commonly used method to control visceral pain.1,2 However, the need for early post-operative evaluation of urinary function, together with the potential for uncorrected hyperkalaemia and consequent cardiac dysrhythmias, makes the systemic side effects of opioids, such as bradycardia and urinary retention,3,4 undesirable in cats affected by urethral obstruction.
Similarly, neuroaxial block has some drawbacks, such as transient bilateral hind limb motor blockade and, when opioids are administered epidurally, the potential for urine retention,5,6 which may not only cause pain and discomfort to the patient, but also render postoperative assessment of urinary function particularly challenging.
In light of these considerations, a more selective, targeted loco-regional anaesthetic technique may represent an attractive alternative to provide pre-emptive analgesia to cats undergoing PU and other surgical procedures involving the urethra.
An ultrasound-guided approach to the feline pudendal nerve has been recently described in cat cadavers and was found to be technically feasible by anaesthesists with average expertise in ultrasonography. 7
The aim of this prospective, randomised, blind, placebo-controlled clinical investigation was to evaluate the analgesic efficacy of the pudendal nerve block (PNB) in client-owned cats undergoing PU.
We hypothesised that PNB performed with bupivacaine would result in satisfactory peri-operative analgesia in cats undergoing PU.
Materials and methods
Animals
The study was performed with the permission of the local committee for animal experimentation (licence number: 117/11) and with signed informed owner consent. Eighteen male castrated cats undergoing PU during the period January 2012 to June 2013 were enrolled in the study and randomly allocated to one of two treatment groups by a simple randomisation method based on the flipping of a coin, where the side of the coin determined the assignment of each subject (heads = control, tails = treatment). The number of animals was decided on the basis of a sample size calculation performed prior to commencing the trial.
Anaesthetic protocol and pudendal nerve block
After cephalic vein catheterisation, the cats were premedicated with intravenous (IV) methadone (0.1 mg/kg; Methadon Streuli, Streuli) and acepromazine (0.01 mg/kg; Prequillan, Fatro). Twenty minutes later, general anaesthesia was induced with IV midazolam (0.2 mg/kg; Dormicurm, Roche) and propofol (Propofol, Fresenius Kabi), titrated to effect. The trachea was intubated with an appropriately sized endotracheal tube; the latter was connected to a neonatal circle breathing system (1.6 M neonatal breathing system; Intersurgical) and isoflurane (Isoflurane; Abbott) delivered in a mixture of air/oxygen (1:1). Lactated ringers’ solution (Ringer-Lactat; Baxter) was administered throughout the procedure at the infusion rate of 10 ml/kg/h. The animals were instrumented with a multiparametric monitor (Maquet SC7000; Siemens) and a doppler (Model 811B; Parks Medical Electronics), and physiological parameters were manually recorded every 5 mins. A constant end-tidal isoflurane concentration (FE’Iso) of 1.3% was targeted during anaesthesia. In each cat, as soon as the level of anaesthesia was judged adequate on the basis of clinical parameters (decreased tone of the lower jaw and of the skeletal musculature, and absence of palpebral reflex with the corneal reflex still present), the rectum was manually emptied and the urethra catheterised with a polyurethane urinary catheter (Winged Cat Catheter 1.0 × 130 mm; Burtons). Thereafter, a bilateral, ultrasound-guided, anaesthetic PNB was performed, always by the same operator blind to the treatment (CA), with a deep dorso-lateral approach, as described elsewhere. 7 In cats allocated to group C (control group), the PNB was performed with saline (0.15 ml/kg injected at each of the two injection sites, to reach a total volume of injectate of 0.3 ml/kg), whereas cats assigned to group B (bupivacaine) were injected with an equal volume of bupivacaine (Carbostesin 0.5%; AstraZeneca).
The anus was sutured only after performing the block, in order to avoid ultrasonographical artefacts due to the accumulation of gas into the rectum. In all cats, the time elapsed between the completion of the nerve block and the beginning of surgery was 30 mins.
Intraoperative assessment of nociception
For each cat, baseline values for systolic arterial blood pressure (SAP), heart rate (HR) and respiratory rate (RR) were determined before performing the nerve block and prior to surgical stimulation, when a stable, adequately deep anaesthetic level was achieved. Intra-operatively, increases in SAP, HR and RR of 30% above baseline values were considered indicative of nociception. When two of these three parameters increased over the defined value, nociception was treated with repetitive administration of rescue fentanyl (3 µg/kg−1 IV; Fentanyl, AstraZeneca), every 5 mins until the physiological variables returned to baseline values. Rescue analgesic requirement and changes in cardiorespiratory parameters during surgery were used to evaluate the efficacy of intra-operative analgesia.
Postoperative pain assessment
Postoperative pain was evaluated using a composite pain scale (CPS), 8 the Colorado State University Feline Acute Pain Scale (CSUFAPS), 9 and a visual analogue scale (VAS). 10 Pain assessments were performed at awakening, when the cats were judged by the anaesthetist to be sufficiently conscious to be evaluated on the basis of clinical parameters (ability to maintain sternal recumbency and to lift up the head, responsiveness to tactile stimulation and to call), and then every hour, always by the same anaesthetist (CA). Cut-off values selected to administer rescue analgesia (buprenorphine, 10 µg/kg IV; Temgesic, Reckitt Benckiser) were scores exceeding 40% of the maximum value of the pain scales; an increase in score above the aforementioned value for at least one of the employed pain scales was sufficient to proceed with pain treatment. After the last pain evaluation, occurring at 3 h from recovery from anaesthesia, each cat received IV buprenorphine (10 µg/kg every 7 h for 48 h) in order to avoid an analgesic gap overnight.
Assessment of postoperative complications
In all cats, the urinary bladder was manually emptied at the end of anaesthesia; subsequently, the time elapsing between recovery and the occurrence of the first spontaneous urination was recorded. Decreased or absent reaction to clamping of the toes of the hind limbs, as well as proprioceptive deficits, were considered indicative of motor blockade. For 3 days after surgery the cats were examined daily by an observer unaware of the treatment (CA) to rule out the emergence of block-related complications. The puncture areas were investigated to detect local infection or haematoma. Increases in body temperature beyond 39.5°C and/or the presence of blood in the faeces were considered potentially indicative of iatrogenic puncture of the rectum.
Statistical analysis
Normality was tested with the Kolmogorov–Smirnov test and the Shapiro–Wilk test. Intra-operative variables (HR, SAP and RR) measured at four different time points, namely baseline (B), immediately after the nerve block was performed (PNB), at incision (I), and at the beginning of the suture of the urethral mucosa (S), were used for repeated measures analysis of variance (ANOVA). Similarly, postoperative variables (CPS, CSUFAPS, VAS) recorded at other four time points, namely at recovery (0) and then 1, 2 and 3 h after recovery (1, 2 and 3, respectively), were used for statistical analysis. Repeated measures ANOVA was used for the numerical variables whose data were normally distributed, whereas the non-parametric Friedman repeated measures analysis of variance on ranks, with treatment (group) and time of data collection as factors, was used for not normally distributed data. The total doses of intra-operative fentanyl and post-operative buprenorphine received by groups C and B were tested using Kruskal–Wallis ANOVA; additionally, the proportions of cats requiring peri-operative rescue analgesics within each group were analysed using Fischer’s exact test. Commercially available software [NCSS-2007, NCSS (SigmaStat) and SigmaPlot 12 (Systat Software)] was used; P-values <0.05 were considered statistically significant.
Results
Normally and not normally distributed data are presented as means and SD, and medians and ranges [min–max], respectively.
The sample size calculation revealed that eight animals per group were necessary to detect a minimum difference in means equal to 10% of the maximum value of the CPS (with the expected SD of residuals set at two thirds of the minimum detectable difference, and with an 80% power and 95% level of confidence). Groups C and B were composed of eight and 10 animals, respectively.
Duration of general anaesthesia was 129 (± 28) mins, with surgery lasting 64 (± 21) mins. The time required to perform the pudendal nerve block was 14 (± 6 mins). The age of the cats was 6.1 [2.0–13.0] years and the body weight was 5.8 (± 1.3) kg. Data were normally distributed for all variables except age, SAP and peri-operative rescue analgesics doses.
Intra-operatively, group C received a significantly higher total dose of fentanyl than group B (4.5 [0–36] and 0 [0–3] µg/kg IV, respectively; P = 0.036) and also showed higher HR (P = 0.002; Figure 1). The proportion of cats requiring rescue intra-operative fentanyl was significantly greater in group C than group B (65% and 10%, respectively; P <0.001; Figure 2). No statistically significant differences in SAP, RR or VAS scores (Figure 3) were observed over time between treatment groups. In both groups, SAP and HR tended to decrease slightly after the nerve block compared with baseline values, but these changes did not result in clinically significant bradycardia or hypotension.

Intra-operative values of heart rate (HR) [beats per minute (bpm)] at four time points corresponding to different events. B = baseline values recorded prior to performing the pudendal nerve block; PB = immediately after the pudendal block was performed; I = at incision; S = at the beginning of the suture of the urethral mucosa. The dots and the whiskers indicate means and SDs, respectively. P = 0.002

Proportions (%) and number (n) of cats receiving intra-operative fentanyl and postoperative buprenorphine within each group. P <0.001

Postoperative visual analogue scale (VAS) scores at four different time points: 0 = recovery; 1 = 1 h after recovery; 2 = 2 h after recovery; 3 = 3 h after recovery. The box and the line represent the interquartile ranges and the median, respectively
Postoperatively, group C showed significantly higher CPS (P <0.001; Figure 4) and CSUFAPS (P <0.001; Figure 5) scores, and needed a greater buprenorphine total dose (20 [10–30] µg/kg IV, P <0.001) than group B. The majority of the cats in group C (87%), but none of the cats in group B, required buprenorphine during the time elapsing from recovery until the last pain assessment; this difference was found to be statistically significant (P <00.1; Figure 2). All but one cat assigned to group B, in which the urinary bladder was emptied manually under sedation 4 h after the end of surgery, urinated spontaneously after 2.8 (± 1.2) h after recovery. Neither motor blockade nor block-related complications were observed in the postoperative period, and all cats were discharged from the intensive care unit of the hospital 3 days after surgery in good clinical condition.

Postoperative Composite Pain Scale (CPS) values at four different time points: 0 = recovery; 1 = 1 h after recovery; 2 = 2 h after recovery; 3 = 3 h after recovery. The box and the line represent the interquartile ranges and the median, respectively. The whiskers indicate minimum and maximum scores, and the dots the outliers. P = 0.00006
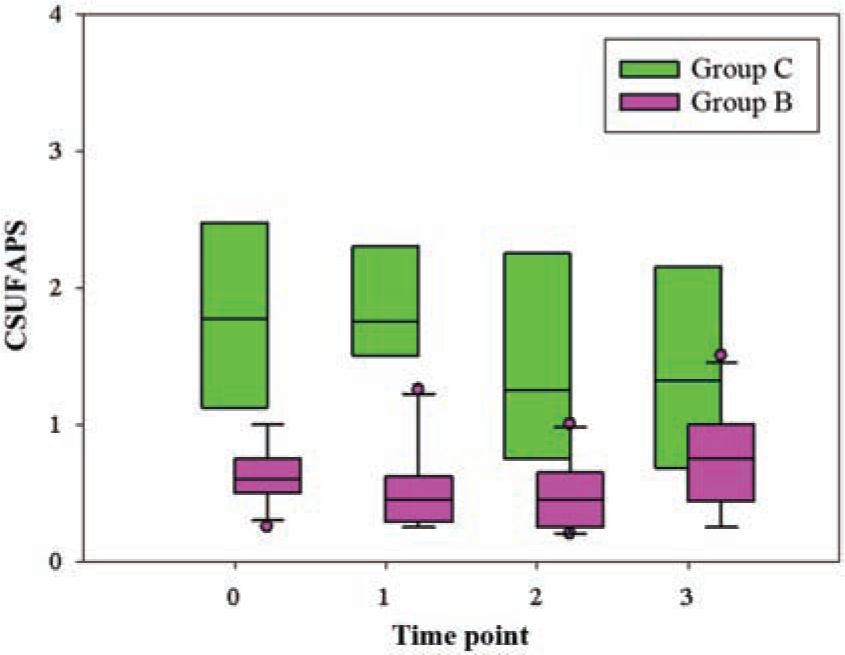
Postoperative Colorado State University Feline Acute Pain Scale (CSUFAPS) values at four different time points: 0 = recovery; 1 = 1 h after recovery; 2 = 2 h after recovery; 3 = 3 h after recovery. The box and the line represent the interquartile ranges and the median, respectively. The whiskers indicate minimum and maximum scores, and the dots the outliers. P = 0.000008
Discussion
Our results show that ultrasound-guided PNB provides adequate peri-operative analgesia in cats undergoing PU and can therefore be a useful tool in the clinical setting.
Owing to the method used for randomisation, groups C and B were composed of different numbers of animals. Simple randomisation was chosen in this study owing to its ease and simplicity but, in small sample size clinical research, this method carries the potential for an unequal number of participants among groups. To overcome this disadvantage, we should have selected another randomisation technique, such as block randomisation. Alternatively, the group sizes could have been equalised by excluding two cats from group B, but this would have resulted in a selection bias. Nonetheless, because in none of the groups the number of cats was lower than the value obtained with the sample size calculation, we do not expect this inequality to have significantly affected the reliability of our findings.
The majority of the cats in the control group required intra-operative fentanyl. This finding demonstrates the invasive nature of PU, and further highlights the need for adequate treatment of nociception when such a procedure is carried out. However, the fact that only one animal in the bupivacaine group required rescue analgesics during surgery bears out our hypothesis and indicates that the PNB was effective in providing reliable analgesia even in the presence of invasive manipulation of the urethra.
Cats in group C showed a trend to higher physiological values than group B. Nonetheless, there were no statistically significant differences between treatments in SAP and RR. This is not surprising, considering that cardiorespiratory variables were used in this study to evaluate the nociceptive responses and to titrate the rescue analgesics requirement during anaesthesia, which implies that fentanyl was administered in response to changes in the aforementioned physiological parameters. Therefore, it is likely that fentanyl contributed to lowering of the SAP and RR values in the control group owing to its analgesic effect, which is expected to suppress the autonomic response to nociception.
The analgesic effect of the PNB extended to the postoperative period, as demonstrated by the considerably low pain scores in the bupivacaine group, although the scores tended to progressively increase during the postoperative evaluation period. Considering that the anaesthetic block was performed immediately before surgery and that the latter had an average duration of 1 h, it is reasonable to assume that the analgesia provided by the PNB — performed with 0.5% bupivacaine — may last approximately 4–5 h. However, extending pain assessment to further time points would have allowed a more precise estimation of the duration of the analgesic effect.
Interestingly, in this clinical trial, the VAS showed poor sensitivity in detecting differences in postoperative analgesia between treatment groups. This is in contrast with previous similar studies, which found the VAS to be a reliable, though subjective, test for quantifying pain.11,12 Owing to its subjective nature, assessment of nociception in animals is particularly challenging and this is one limitation of all studies whose main purpose is the evaluation of analgesia. In the present clinical trial, the reliability of our observations should have been increased by the inclusion of three different pain scales.
We did not observe the occurrence of major block-related iatrogenic complications. However, it should be emphasised that this clinical trial was designed to evaluate the efficacy — and not the safety — of this novel technique, and that the sample size calculation was performed accordingly. As a result, one limitation of this study is that the small number of animals enrolled does not allow any conclusion to be drawn about the incidence of block-related complications. One of the cats treated with bupivacaine showed a minor complication, namely transient urinary dysfunction, which required manual emptying of the bladder under sedation.
Neurophysiological studies performed in cats demonstrated that, depending on the stimulation frequency, electrical stimulation of the pudendal nerve could elicit a urinary bladder response of either inhibitory or excitatory nature.13–15 Stimulation frequencies of 20–40 Hz seem to result in sustained contractions of the detrusor muscle and consequent urination, and have been proposed for treating urinary continence in humans. 16 Accordingly, it is reasonable to assume that, in the aforementioned cat, the anaesthetic blockade of the branches of the pudendal nerves may have provoked transient urine storage owing to bladder hypomotility. As an alternative hypothesis, the anuria could have been a side effect of systemic methadone; 17 however, considering the very low dose administered and the long time between premedication and recovery, this explanation seems to be unlikely. Nevertheless, the urinary continence resolved spontaneously in this cat and did not result in long-term complications.
Besides analgesic efficacy and safety, a novel clinical technique should fulfill other requirements to become an integral part of routine client-owned patient management. Some important prerequisites are the possibility for a clinician with average experience to perform the technique in a short, reasonable time, and a steep learning curve characterised by a rapid increase in technical skills. Although the time needed for the block was relatively short in this study and seemed not to interfere with the clinical routine, it should be highlighted that intensive and time-consuming training was required for the anaesthesists to achieve adequate technical skills. Therefore, one factor limiting the applicability of this novel technique in the clinical setting may be a slow learning curve owing to the fact that in many facilities PU is not an everyday scheduled surgical procedure.
Conclusions
Owing to the reliable analgesia it provides, ultrasound-guided PNB can be suggested for managing peri-operative pain in cats undergoing PU, although the training required to achieve sufficient technical skills may represent a limitation to its clinical application. Further investigation is needed to evaluate the safety of this novel technique and the incidence of block-related complications.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
