Abstract
The normal sonographic thickness of the individual layers (ie, mucosa, submucosa, muscularis and subserosa-serosa) of the intestinal wall was evaluated in 20 clinically healthy cats. The mean thickness of the wall was 2.20, 2.22, 3.00 and 2.04 mm for duodenum, jejunum, ileum (fold) and ileum (between folds), respectively. The mean thickness of the mucosal layer was 1.27, 1.20, 0.46 and 0.49 mm for duodenum, jejunum, ileum (fold) and ileum (between folds), respectively, and its contribution to wall thickness was significantly greater than that of the other layers in the duodenum (57.7%) and jejunum (55.2%). The mean thickness of the submucosal layer was 0.36, 0.36, 1.49 and 0.53 mm for duodenum, jejunum, ileum (fold) and ileum (between folds), respectively, and its contribution to wall thickness was greater than that of the muscularis in the duodenum (16.3%), jejunum (16%) and ileum (fold) (49.8 %). The mean thickness of muscularis was 0.28, 0.35, 0.66 and 0.65 mm for duodenum, jejunum, ileum (fold) and ileum (between folds), respectively, with a corresponding contribution to wall thickness of 12.7 %, 14.4%, 22% and 31.6%. Finally, the mean thickness of serosa was 0.29, 0.31, 0.38 and 0.38 mm for duodenum, jejunum, ileum (fold) and ileum (between folds), respectively, with a corresponding contribution to wall thickness of 13.3%, 14.4%, 12.7 % and 18.7%. These values can provide baseline information that might be useful in evaluating intestinal disorders affecting preferentially some of the intestinal layers.
Introduction
Abdominal ultrasonography can be a useful tool in the diagnostic work up of small animals affected by gastrointestinal diseases.1–3 The ultrasonographic appearance of the normal feline gastrointestinal tract has been described and reference ranges for wall thickness of different segments have been reported.4,5 Changes in echogenicity and/or thickness of intestinal wall involving selectively some intestinal layers have been described in feline gastrointestinal disorders.6–9 In particular, an ultrasonographical pattern characterised by a diffusely thickened muscularis propria of the small intestine has been associated both with gastrointestinal lymphoma and inflammatory bowel disease (IBD) in cats. 8 In addition, chronic partial obstruction of intestinal loops due to foreign body or intestinal mass can result in hypertrophy of the muscular layer, producing the same ultrasonographical feature. 9
Intestinal muscularis thickening has been reported when the width of the muscular layer is equal or greater in thickness to the mucosal layer 1 or when the width of the muscular layer is greater than half the thickness of the submucosal layer. 8
At the time of collecting our data, no reports establishing the normal ultrasonographical thickness of each small intestinal layer were available, except for an abstract reporting only the relative contribution (%) of each gastrointestinal layer to the overall wall thickness. 10 Therefore, the aim of this study was to report the normal ultrasonographical thickness of the individual small intestinal layers and to provide the relative proportion of each layer to overall wall thickness in the duodenum, jejunum and ileum of clinically healthy cats.
Materials and methods
Twenty clinically healthy client-owned domestic shorthair cats were used in this study. There were eight intact males, six neutered males, one intact female and five neutered females. The age of the cats ranged from 1 to 7 years (mean ± SD age, 3.4 ± 2 years); the cats’ bodyweight ranged from 3 to 6 kg (mean ± SD age, 4.4 ± 0.9 years). Cats were considered clinically healthy, and had no history of hepatobiliary, pancreatic or gastrointestinal disease for the previous 12 months, or since birth of the youngest two cats). Physical examination, complete blood count, routine serum biochemical analyses, urinalysis and faecal examination for intestinal parasites were normal. All cats were also negative for feline leukaemia virus antigen and feline immunodeficiency virus antibody. Informed owner consent was obtained, and the ethical committee of the University of Bologna approved the protocol of the study.
The cats were fasted overnight (at least 12 h) prior to imaging. All ultrasonographical examinations were conducted by the same sonographer (AD), using a real-time ultrasound machine equipped with a high frequency (13 MHz) linear array transducer (Mylab 70 VETXV ultrasound system; ESAOTE). The cats were awake and restrained manually during the examination. The gastrointestinal tract was scanned with the cat in dorsal, left lateral and right lateral recumbency. In particular, jejunal, duodenal and ileal images were obtained with the cat in right lateral, left lateral and dorsal recumbency respectively. The duodenum was scanned at the level of mid-descending segment.
The images were recorded as cine-loops in DICOM format and transferred to a computer. An imaging processing software (OsiriX Imaging Software; Pixmeo) was used to view the images and to export the selected frames for subsequent analysis. The images were displayed on calibrated, 2-megapixel high-brightness monitor (Eizo RX240; Cusano). The matrix of the images was 800 × 555 pixel and the ratio pixel/cm was 162.5; therefore, one image point (pixel) in the digitised ultrasound image was 0.06 mm. Electronic callipers were placed at the outside edge of each individual intestinal layer, and leading edge to leading edge measurements were made from the serosal interface to the luminal interface of the mucosa. Mucosal, submucosal, muscular and serosal thickness was measured. Three consecutive measurements of each of the layers (ie mucosa, submucosa, muscularis and serosa) were obtained in both longitudinal and transverse plane for the duodenum and jejunum (Figures 1 and 2). Measurements for the ileum were only performed on transverse images as we considered that the layer demarcation was more reliably assessed on this plane. In particular, two measurements were obtained: one at the level of the fold and one between folds (Figure 3). The thickness of the entire intestinal wall was obtained by the sum of the measurements of each layer.
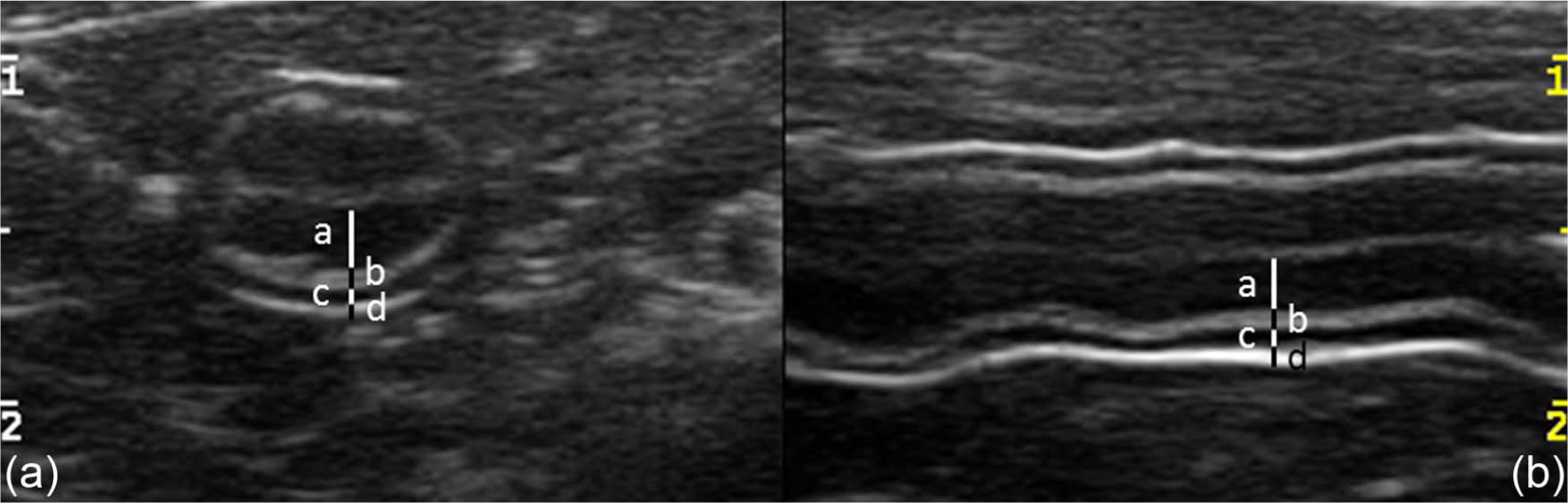
Transverse (a) and longitudinal (b) images of the duodenum illustrating the method for measurement of intestinal wall layers. Black and white bars indicate the thickness of each layer. a = mucosa; b = submucosa; c = muscularis; d = serosa
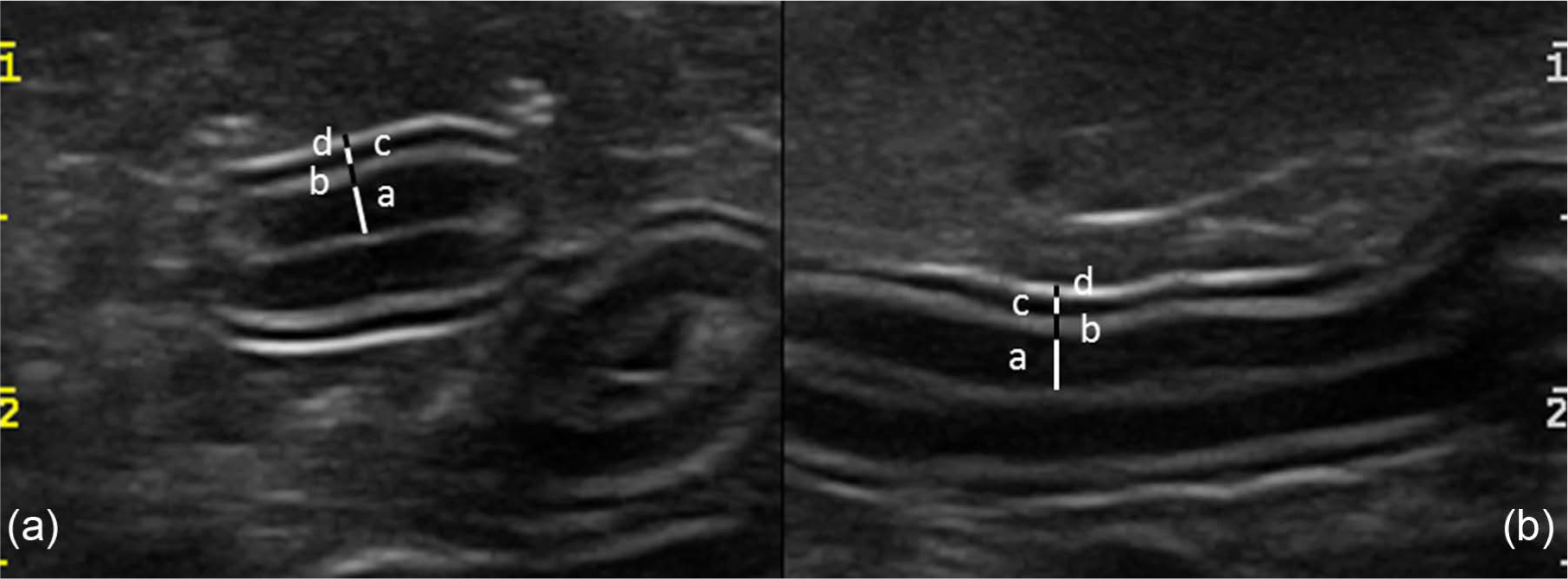
Transverse (a) and longitudinal (b) images of the jejunum illustrating the method for measurement of intestinal wall layers. Black and white bars indicate the thickness of each layer. a = mucosa; b = submucosa; c = muscularis; d = serosa
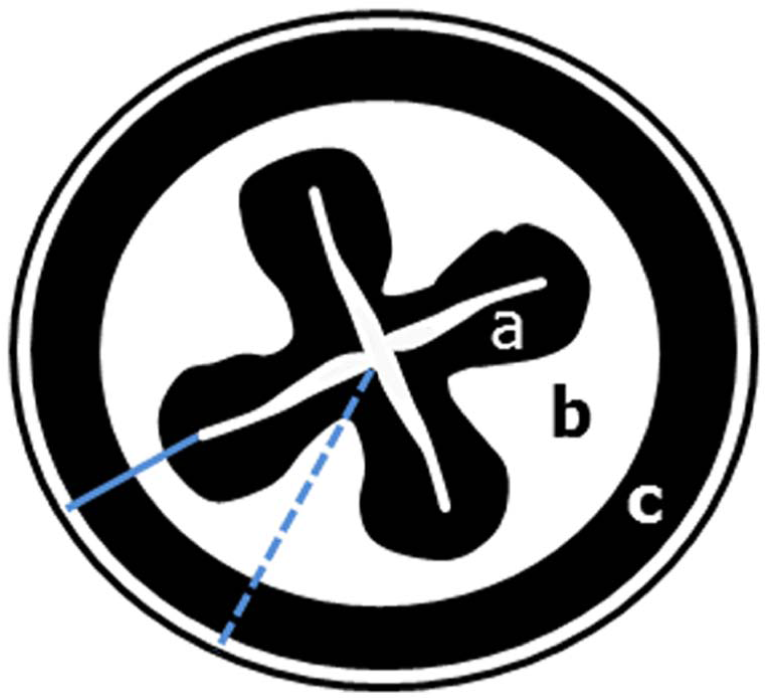
Diagram of the transverse section of the ileum of a cat illustrating the measurements of ileum at level of fold (light blue line) and between folds (dotted light blue line). a = mucosa; b = submucosa; c = muscularis
Measurements of entire wall and layers obtained from each cat were subsequently averaged, and these results were used to generate the average percentage of the contribution of each individual layer thickness to overall wall thickness. The averaged values were also used as data for statistical comparisons, and measurements were reported as mean and SD. Statistical analyses were performed using Statistica for Windows (Statsoft). In order to assess the limit of agreement between measurements obtained in longitudinal and transverse planes, Bland–Altman analysis was performed, followed by calculation of the British Standards Institution (BSI) repeatability coefficient, obtained by multiplying by two the SD of the differences between paired values. Two measurement methods are considered in agreement if 95% of the absolute differences between paired measurements is less than the BSI repeatability coefficient. 11
The intra-observer variability for all measurements of each cat was calculated and expressed by the coefficients of variation (CV), determined by dividing each SD by mean. The CV values were reported as percentage.
Before statistical analysis, the normality of the data was tested using a Shapiro–Wilk test, and all data were normally distributed. A one way analysis of variance for repeated measurements by using body weight as a covariate, and a post hoc comparison (Tukey multiple comparison test) was used in each intestinal segment (duodenum, jejunum, ileum fold and ileum between folds) to compare the thickness of the different layers of the wall. The same test was used to compare the thickness of the entire intestinal wall, mucosal layer, submucosal layer, muscularis and serosal layer among the different intestinal segments (duodenum, jejunum, ileum fold and ileum between folds). P-values <0.05 were considered statistically significant.
Results
The measurements of the small intestinal wall segments and of each intestinal layer are summarised in Table 1 and Figure 4. No statistical difference was found between measurements of duodenum and jejunum obtained in transverse and longitudinal planes. The duodenal wall thickness ranged from 1.78 to 2.51 mm. The thickness of the mucosa was significantly greater (P <0.01) than that of the other duodenal layers contributing 57.7% to the overall wall thickness. The submucosal contribution was 16.3%, significantly thicker (P <0.05) than muscularis contribution (12.7%). The serosa was 13.3% of the total wall thickness. No differences were found between the submucosal layer versus serosal layer, or between muscular layer versus serosal layer.
Intestinal thickness and layer measurements (mean ± SD, range and percentage) of different segments of the small intestine in 20 clinically healthy cats

Percentage contribution of each layer to the overall wall thickness. In each row, values with different superscript letters differ significantly: a = P <0.01 compared with duodenum, jejunum, ileum between folds; b = P <0.05 compared to duodenum and jejunum; c = P <0.01 compared to ileum fold and ileum between folds; d = P <0.01 compared to duodenum and jejunum; e = P <0.05 compared to duodenum

Average contribution (%) of each intestinal layer (identified by a different colour) to total wall thickness at the different sites of the small intestine (duodenum, jejunum, ileum at level of fold and ileum between folds)
The jejunal wall thickness ranged from 1.96 mm to 2.67 mm. The thickness of the mucosa was significantly greater (P <0.01) than that of the other jejunal layers, contributing 55.2% to the overall wall thickness. The contribution of the submucosal, muscularis and serosal layers was 16%, 14.4% and 14.4% of the entire jejunal wall, respectively. No statistical differences were found between the thicknesses of the jejunal submucosa, muscularis or serosal layers.
The ileal wall thickness at the level of the fold ranged from 2.52 mm to 3.59 mm. The contribution of the mucosal layer was 15.5%. The thickness of the submucosal layer was 49.8% of the total wall and was significantly higher (P <0.01) than that of the other layers. The muscularis was 22% of the entire wall and was significantly higher (P <0.01) than the mucosal and serosal layer. The thickness of the serosa was 12.7% of entire wall. No difference was found between the mucosal and serosal layer. The ileal wall thickness between folds ranged from 1.66 mm to 2.27 mm. The contribution of the mucosal layer was 24% and was significantly higher (P <0.01) than that of the serosa. The thickness of submucosal layer was 25.7% of the total wall and was significantly higher (P <0.01) than that of the serosa. The muscularis was 31.6% of the total wall and was significantly higher (P <0.01) than the other layers. The thickness of serosa was 18.7% of the entire wall.
The thickness of ileum at the level of the fold was significantly higher (P <0.01) than the other intestinal segments. The mucosa of the duodenum was significantly thicker (P <0.01) than that of the ileum (both at fold and between folds), whereas no difference between mucosal layer of duodenum and jejunum was found. The submucosal thickness of the ileum at the level of the fold was significantly (P <0.01) higher than that of the other intestinal segments. The muscularis of the ileum (both at fold and between folds) was significantly thicker than that of duodenum and jejunum. Finally, the serosa of the ileum was significantly (P <0.01) thicker than that of the other segments.
The results of intra-observed CV for all intestinal measurements are reported on Table 2. Higher CV values were found for ileal measurements (both at fold and between folds).
Intra-observer variability expressed as coefficients of variation (CV) of intestinal measurements in 20 healthy cats

Discussion
Our results of the feline intestinal wall thickness were in agreement with the current literature.4,5 As previously reported, 5 the ileum is the thickest intestinal segment, with a mean thickness of 3 mm. In our study, measurements of ileum were performed only on transverse section owing to the unique anatomical morphology of this intestinal segment. The presence of folds reduced the distinction of the wall layering in longitudinal section making difficult the measurements. This explains why our data included both the ileal wall thickness at the level of the fold and between folds. The values obtained for the thickness of the ileal fold (range, 2.50–3.59 mm) are similar to previously reported ranges for normal cats, 5 whereas the measurements between two folds were significantly lower (range, 1.66–2.27 mm).
The choice of the transverse plane for ultrasonographical measurement of the intestinal wall and layer thickness was made based on the reported limitations of the longitudinal plane. Oblique measurements of the bowel cause errors of either over-estimation or under-estimation of the wall thickness. 12 In small animals, the use of a single section, transverse 5 or longitudinal,13,14 has been reported. Other authors have measured the wall thickness either in transverse or longitudinal sections adding the values obtained from both sections. 4 In the present study, we also used both sections for the measurements of duodenum and jejunum. As no statistical difference was found from the two sections, the values were subsequently added for statistical analysis.
Concerning the different small intestinal layers, the mucosa was the thickest layer in the duodenum and jejunum (mean 1.27 and 1.20 mm, respectively) and accounted for more than 50% of the overall intestinal thickness. Conversely, the ileum had a thin mucosal layer (mean 0.46 and 0.49 mm at fold and between folds, respectively) and its contribution to the overall thickness was 15.5% and 24.0% for the ileum at folds and between folds, respectively.
A recent morphometric study on the histological thickness of the different intestinal layers in the dog reported the mucosa as the thickest layer in the duodenum, with a progressive decrease from proximal to distal parts of the small intestine. The size of the villi and the presence of duodenal glands and lymphoid tissue in the lamina propria are likely responsible for the high thickness of the duodenal mucosal layer. 15 Conversely, the lack of intestinal glands associated with low secretory activity is most likely responsible for the thinner mucosal layer of the ileum. 16 In the present study, the submucosa of the ileum (mean 1.49 mm and 0.53 mm at fold and between folds, respectively) was significantly thicker compared with that of the duodenum (mean 0.36 mm) and jejunum (mean 0.36 mm) and accounted for 49.8% and 25.8% of the total wall thickness at the fold and between folds, respectively. In the small intestinal wall of the dog, the mucosa and submucosa accounted for approximately 70% of the intestinal thickness, but their proportions showed differences between duodenum and ileum. In particular, the thickness of submucosa of the ileum was significantly thicker in comparison to other intestinal segment owing to an increase of lymphoid aggregates.15,16 In the cats in the present study, we found a significant difference between the values of submucosal layer obtained at the level of fold and between folds. This may be explained by the physiological role of ileum, creating an anatomical barrier between the small and large intestine. For this reason, the ileum is constantly contracted in vivo and the connective tissue of the submucosa follows the mucosal folds creating a higher thickness at this level. 17
The contribution of the muscularis layer of the ileum to overall intestinal wall thickness was 22.0% and 31.6 % at the level of fold and between folds, respectively. Their absolute values (mean 0.66 and 0.65 mm at fold and folds, respectively) were significantly higher than those obtained at the level of duodenum (mean 0.28 mm) and jejunum (mean 0.35 mm). The functional specialisation of ileum in generating vigorous peristaltic movements overcoming the physical resistance of the ileo-ceco-colic junction has been reported as a possible cause for the higher thickness of the muscular layer of ileum in dogs. 15 Finally, the serosal layer contributed to the total wall thickness for 13.1%, 14.4%, 12.5% and 18.7% in the duodenum, jejunum, and ileum at the level of fold and between two folds, respectively. These values may not represent the real thickness of the serosal layer, but may be an overestimation due to a summation of echoes produced by the internal structure of the tissue layer and those produced by interfaces between the serosal layer and surrounding mesenteric fat.18,19 In humans, differences between ultrasound and histological layer thickness have been reported.19,20 It is speculated that the discrepancy may be due to a variable degree of tissue shrinkage and expansion occurring during histologic sample processing. Additionally, the ultrasonographical five-layered appearance of the intestinal wall corresponds to the echoes produced by the tissue layers and those produced by layer interfaces. 20
A good morphological correlation between the ultrasonographical appearance and the histological layers of the intestinal wall has also been reported in veterinary medicine. 1 Nevertheless, to the best of our knowledge, there are no reports that have described the correlation between the ultrasonographical and histological thickness of each intestinal wall layers in small animals.
The results of intra-observed CV for all intestinal measurements showed the highest variability for the ileum measurements. The inability to consistently visualise the bright mucosal interface in the centre of the ileum reduces the possibility of accurate measurements of mucosal and submucosal thickness. The lower number of measurements compared to duodenum and jejunum may also be the reason.
The percentage values of each intestinal layer thickness of the present study differed from those reported in a recent abstract. 10 In the latter, the mucosal layer provided the greatest contribution to overall gastrointestinal wall thickness in all segments of the gastrointestinal tract, while in our study the mucosa was the thickest layer only in the duodenum and jejunum. However, the absence of detailed information on technical aspects (ie the frequency of transducer and type of scan employed) and measurement methods made a comparison difficult between our results and those reported in the abstract. 10
This study has some limitations that need to be emphasised. First, the population of cats prospectively recruited was considered healthy only on the basis of clinical and laboratory findings. No endoscopic or surgical biopsies were taken and, therefore, histological confirmation of the absolute normality of the intestinal wall was not available.
Second, the measurements of each individual layer of feline intestinal wall can be challenging because the placement of callipers on very small structures is difficult. Values obtained could be influenced by observer’s experience, and the evaluation of inter-observer variability was not performed in this study. Furthermore, the accuracy of these measurements is directly related to the axial resolution of the transducer and, therefore, a high imaging frequency probe is advised.
The accuracy of the thickness of layers of ileum can be questioned owing to the lower number of these measurements and to the difficulty to clearly distinguish the layers for the presence of folds. Therefore, these values should be validated in a wider feline population.
Conclusions
The ultrasonographical features of feline gastrointestinal inflammation and neoplasia can often overlap. 2 In particular, a selective thickening of the muscular layer has been frequently reported in IBD,3,6–8 and in lymphoplasmacytic and eosinophilic enteritis, but this feature has also been associated with other gastrointestinal neoplasias, such as lymphoma 8 and mast cell tumours, 21 or as a consequence of intestinal mechanical obstruction. 9 However, no reports have described the normal thickness of the muscular layer and its range to more accurately determine thickening of this layer relative to other layers. Therefore, we believe that the relative proportion values provided in this study can be useful as a baseline reference when evaluating feline intestinal disorders, such as IBD and round cell tumours, that can have different degrees of intestinal layer involvement.
Footnotes
Acknowledgements
We wish to thank Dr Calogero Vaccaro from the Department of Animal Medicine, Production and Health, University of Padua, Italy, for his technical support with the digital image analysis.
Addendum
At the end of the review process, we became aware of a paper (Winter et al 2013 22 ) in which the relative gastrointestinal layer thickness in cats without clinical evidence of gastrointestinal tract disease was evaluated using ultrasonography.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
