Abstract
The objectives of this study were (1) to measure normal thickness values of the muscularis, submucosal, mucosal and serosal layers in each gastrointestinal (GI) segment (gastric fundus, body and pyloric antrum; duodenum; jejunum; ileum; colon), and (2) to calculate a ratio of muscularis and mucosal layer thickness to aortic diameter measured at the level of the celiac artery (Musc:Ao and Muc:Ao) in each GI segment in a sample of healthy cats. Ultrasonographic examination of the GI tract was performed, and measurements of the individual layers in each GI segment were obtained in 38 healthy cats without clinical evidence of disease. The muscularis layer was significantly thickest in the ileum, compared with other segments, and it was thicker than the submucosa in all segments except the colon. The mucosa was the thickest layer in all segments, and was thickest in the duodenum and ileum. Measurements of the submucosal and serosal layers were not significantly different between all segments. Musc:Ao and Muc:Ao in each segment were 0.12 and 0.25 (gastric fundus), 0.12 and 0.18 (gastric body), 0.11 and 0.16 (pyloric antrum), 0.08 and 0.27 (duodenum), 0.08 and 0.22 (jejunum), 0.14 and 0.25 (ileum), and 0.05 and 0.08 (colon), respectively. Musc:Ao and Muc:Ao are clinically relevant values that can be used to objectively identify thickening of the muscularis and mucosal layers in response to GI diseases.
Introduction
Ultrasound (US) evaluation of the gastrointestinal (GI) wall is frequently used as a diagnostic tool for GI disease in cats.1–6 Variations in normal GI wall thickness in different GI segments (gastric fundus, body and pyloric antrum; duodenum; jejunum; ileum; colon) of the GI tract have been described in cats.7,8 In one study conducted on 14 healthy cats, the duodenum wall was thicker when cats were sedated (mean, 2.7 mm) compared with when cats were awake (2.4 mm). 7 In another study conducted on 11 healthy cats, the ileum wall was thicker (mean, 2.8 mm) than all segments of the GI tract (gastric fundus, 2.0 mm; pylorus, 2.1 mm; duodenum, 2.2 mm; jejunum, 2.3 mm; and colon, 1.5 mm). 8
While measuring total GI wall thickness in the different GI segments is an important part of ultrasound examination of the GI tract in cats, evaluation of specific GI wall layers (serosa, muscularis, submucosa and mucosa) can provide additional information of clinical relevance. Recent studies in cats have suggested that thickening of specific wall layers can occur in response to GI disease.4,6,9 In one study, diagnoses of lymphocytic–plasmocytic enteritis and foreign bodies were associated with thickening of the muscularis layer in four cats. 4 In a case report of small bowel infarction in a cat caused by an aortic thromboembolism, a series of US evaluations documented progressive, focal mucosal and submucosal layer prominence, with final loss of normal wall layering over a period of 48 h. 9 In these studies, normal GI layer thicknesses measurements were not reported, and the observation of layer thickening or prominence was subjectively assessed. In a recent study, cats with muscularis layer thickening were reported to more likely have T-cell lymphoma than inflammatory bowel disease. 6 In that study, muscularis layer thickening was defined as being present when the width of the muscularis layer of the small intestine was greater than half the thickness of the submucosal layer. 6
Layer thickness alterations have been reported as a component of feline GI diseases. However, baseline measurements of GI wall layer thickness in healthy cats with no clinical evidence of GI diseases have not been reported. In humans, conditions such as Crohn’s disease cause alterations in the thickness of multiple, adjacent layers; 10 therefore, individual layer thickness relative to adjacent layer thickness or total wall thickness may be misleading. In addition, between patients, there may be some variation in the range of thicknesses measured owing to breed or size differences. Several authors have described the feasibility of using ratios to a structure such as the aorta to normalize measurements in US examinations.11,12 The objectives of this study were (1) to measure the thickness of the muscularis, submucosal, mucosal and serosal layers in each GI segment (gastric fundus, body and pyloric antrum; duodenum; jejunum; ileum; colon) in healthy cats, and (2) to calculate a clinically useful, normal ratios of muscularis and mucosal layer thickness to aortic diameter.
Materials and methods
All procedures were approved by the Institutional Animal Care and Use Committee and the Clinical Research Committee at the University of Florida College of Veterinary Medicine (UFCVM).
Study population
Seventy healthy cats without clinical evidence of GI disease (weight loss, inappetence, vomiting, previous diagnoses of neoplasia) or evidence of other chronic disease were initially considered for inclusion. Study cats were provided by employees and students of the UFCVM. For each cat presented, a physical examination was performed, and information was collected regarding age (years), breed and diet (wet/dry/combination) using a form that was completed by each owner. Normal routine complete blood count and serum biochemical analysis were required for inclusion in the study group. Thirty-two cats did not meet the inclusion criteria and were excluded. The final enrollment included 38 clinically healthy cats.
Imaging and data collection
Study cats were fasted for at least 12 h prior to US examination. The abdomen of each cat was clipped, and each cat was placed in dorsal recumbency for a complete abdominal US examination. Sedation, consisting of a combination of ketamine hydrochloride (4 mg/kg, IV) and diazepam (0.2 mg/kg IV) was used when necessary.
A complete abdominal US examination was performed by a board-certified radiologist (MDW) using a linear 17–5 MHz broadband transducer equipped with spatial compounding and harmonics (Philips Healthcare). Additionally, images of the aorta at the level of the celiac artery were collected, including cineloops, using a microconvex 8–5 MHz broadband transducer. Axial and lateral resolutions were evaluated for each transducer using a US phantom (Multi-Purpose Multi-Tissue Ultrasound Phantom Model 040GSE; CIRS). Axial resolution was determined to be less than 0.08 mm with the linear transducer and 0.1 mm with the microconvex transducer. The US phantom used would not allow resolution determination beyond 0.08 mm. Images were stored on a picture archive and communication system (Merge PACS; Merge Healthcare) using DICOM file format for later review on calibrated, 3-megapixel grayscale display monitors.
Ultrasound images of the GI tract were reviewed by one of the authors (LL). Three measurements of overall intestinal wall, mucosal, submucosal, muscularis and serosal thickness were made in each GI segment (gastric fundus, body, pyloric antrum, duodenum, jejunum, ileum, colon) on images obtained using the 17–5 MHz linear transducer without harmonics (Figure 1a–h). Though the cats were fasted, there were occasional segments that contained ingesta; measurements were made in segments that did not contain ingesta. Calipers were placed at the outside edge of each layer to obtain each measurement. An average of the three measurements was calculated for total wall thickness and individual layer thicknesses. For GI segments in which the submucosa had normal thickness variation, such as the ileum or rugae of the stomach, the thinner inter-rugal region was measured for consistency (Figure 1a and g) Additionally, aortic diameter was measured on a transverse image of the aorta (Figure 2) in two planes in a fashion similar to that reported previously.11,12 Three measurements were made during systole, and the average of these three measurements was calculated. This measurement was used to calculate a ratio of muscularis and mucosal layer thickness to aortic diameter.
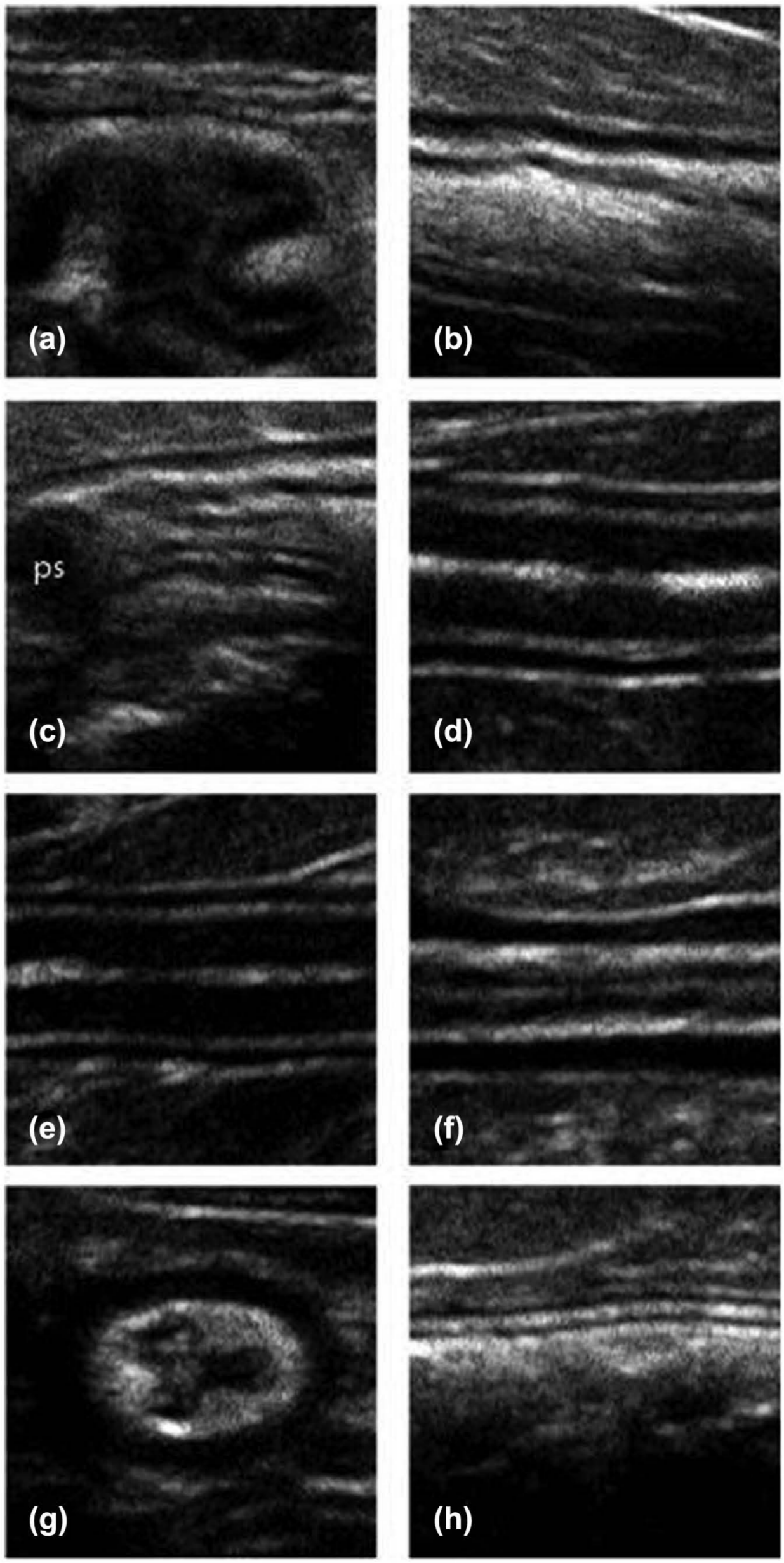
Composite ultrasound images of the gastric fundus (a); gastric body (b); pyloric antrum (c); duodenum (d); jejunum (e); ileum in longitudinal (f) and transverse (g) orientation; and colon (h). Note the visibly evident differences in individual layer thickness, and the variations in submucosal layer thickness in the gastric fundus and the ileum. Also note the pyloric sphincter (ps) in the aboral portion of the pyloric antrum. The pyloric sphincter itself was not measured in the study population
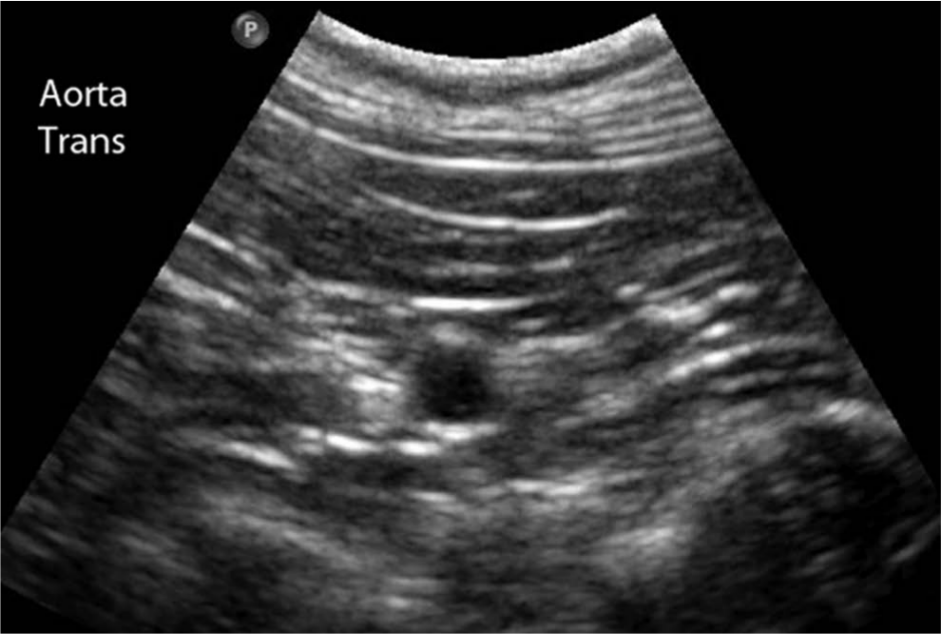
Transverse image of the aorta made at the level of the celiac artery
Data analysis
Gastro-intestinal segment and layer data were not normally distributed, as determined by the Shapiro–Wilk test. Thus, the median, first and third quartile values in mm were reported to describe GI segment and layer measurements.
Within each GI segment, the thickness of each layer was compared using the non-parametric Wilcoxon signed rank test. Between different GI segments, the thickness of each layer was also compared using the Wilcoxon signed rank test. The mucosal layer thickness-to-aortic diameter ratio and the muscularis layer thickness-to-aortic diameter ratio were compared between segments using the Wilcoxon signed rank test.
Associations between total wall thickness in different segments and diet were examined by using the non-parametric Wilcoxon rank sum test. The relationship between age and total wall thickness in different segments was examined by calculating the correlation value between these two variables. For all tests, values of P <0.05 were considered statistically significant.
Results
Thirty-eight cats were included in this study. The most common breed was domestic shorthair (n = 21), followed by Siamese (n = 7), domestic longhair (n = 3), Bengal (n = 3), Sphynx (n = 1), Persian (n = 1), Himalayan (n = 1) and domestic mediumhair (n = 1). The age of the study cats ranged from 0.5 to 16 years, with an average and a median of 5 years. The average weight was 5.2 ± 1.5 kg.
Comparisons of GI wall layer thickness within each segment in the 38 study cats are presented in Table 1. Within each segment, the mucosal layer was significantly thicker than the submucosal, muscularis and serosal layers (P <0.05). The muscularis layer was significantly thicker than the submucosal and serosal layers in all segments, and the submucosal layer was significantly thicker than the serosal layer (P <0.05). The ileum was the thickest GI segment, and the colon was the thinnest GI segment, consistent with previous findings. 8
Comparisons of gastrointestinal wall layer thickness (mm) within and between each segment in 38 healthy cats*

Within a row, values with different superscript letters are statistically different (P <0.05) (ie, gastric fundus: serosal versus muscularis or other layers)
Within a column, values with different superscript numbers are statistically different (P <0.05) (ie, mucosal: colon versus ileus and other segments)
Data are presented as median (first quartile, third quartile)
Comparisons of individual GI wall layer thickness between GI segments in the 38 study cats are also presented in Table 1. The mucosal layer was significantly thickest in the duodenum compared with other segments (P <0.05), except the ileum (P <0.05). In addition, the mucosal layer was significantly thickest in ileum compared with the gastric body, pylorus and colon (P <0.05). Measurements of the submucosal layer in the gastric fundus and gastric body were thicker compared with the duodenum, jejunum and colon (P <0.05). The muscularis layer of the ileum was significantly thicker compared with the muscularis layer thickness in all other segments (P <0.05). The muscularis layer in the gastric fundus, gastric body and pyloric antrum was significantly thicker than in the duodenum and colon. However, the muscularis layer thickness in the pyloric antrum and the jejunum was not significantly different. Measurements of the submucosal and serosal layers were similar between segments, although small, but significant, differences (P <0.05) were observed between the submucosal layer of the gastric fundus, body and pylorus (0.04 mm) and the duodenum and jejunum (0.03 mm), as well as the colon (0.02 mm). The serosal layer of the colon was significantly thinner compared with all other segments (P <0.05). The relative contributions of the submucosal and serosal layers to the total wall thickness of the pyloric antrum, duodenum, jejunum and ileum were not significantly different (P >0.05).
The ratios of muscularis layer to aortic diameter (Mus:Ao) and mucosal layer to aortic diameter (Muc:Ao) are displayed in Table 2. The Mus:Ao ratio was significantly greater in the ileum than in all other segments (P <0.05). The Muc:Ao ratio was significantly greater in the duodenum, gastric fundus, ileum and jejunum than in the colon, pyloric antrum or gastric body (P <0.05).
Comparisons of mucosal layer thickness (mm) to aortic diameter (mm) ratio and muscularis layer thickness (mm) to aortic diameter (mm) ratio in different segments of the intestine in 38 healthy cats*

Within a row, values with different superscripts are statistically different (P <0.05) (ie, mucosa:aorta: colon versus ileum or other segments)
Data are presented as median (first quartile, third quartile)
There were no significant age-related differences in GI wall thickness. The coefficient of correlation was 0.24 (P = 0.13). The coefficient of determination, R2, was 0.06, an indication that only 6% of the observed variation in total wall thickness was explained by age. Furthermore, when cats were assigned into one of two groups (1–6 years old, n = 21; 7–15 years old, n = 16), the largest observed difference was at the pylorus segment in cats aged 7–15 years old (median = 0.23 mm; first quartile = 0.19) and cats aged 1–6 years old (median = 0.21 mm; first quartile = 0.17; third quartile = 0.25), but this difference was not significant (P = 0.12).
In this study, six cats were sedated and 30 were not. The duodenum wall was thicker in sedated cats (median = 0.26; 0.22, 0.31 mm) than in cats that were awake (median = 0.24; 0.21, 0.27 mm), but this difference was not statistically significant (P = 0.25).
Twenty-one cats were fed a combination of wet and dry food, 15 cats were fed dry food only and two cats were fed wet food only. Cats fed wet food only were excluded from comparison owing to low sample size. Cats fed a combination of wet and dry food had a significantly (P <0.05) greater mucosal layer thickness in the ileum (median = 0.12 mm; 0.09, 0.15 mm) compared with those fed only dry food (median = 0.10 mm; 0.08, 0.12 mm).
Discussion
Our results indicated that the thickest intestinal layer in the feline GI tract was the mucosa, followed by muscularis, submucosa and serosa. In addition, the muscularis layer was significantly thicker than the submucosal layer in every segment, with the exception of the colon. The greater thickness of the muscularis layer compared with the serosal and submucosal layers in each segment can be explained by the primary function of the muscularis layer, which is to promote the mechanical breakdown of ingesta and the aboral movement of GI contents. 13 The significantly thicker muscularis layer of the ileum may be related to its role as a sphincter, preventing oral movement of feces and bacteria from the colon into the small intestinal tract. 13
The mucosal layer was always the thickest GI layer. Histologically, the mucosal layer contains the greatest number of sublayers, and is primarily responsible for the absorption of nutritional elements from the lumen, which may explain its greater thickness. The observed variation in mucosal thickness between each GI segment may reflect functional differences in the sublayers of the mucosa in different GI segments. For example, the greater thickness of the mucosal layer of the ileum may reflect the large amount of fluid, electrolyte, amino acid, bile salts and vitamin B12 absorption that occurs at this location compared with other locations in the GI tract. 13 In addition, there is a large number of lymphoid follicles present in the ileum, presumably providing immune defense functions at a critical junction with the colon, where there is a high content of luminal organisms. 13
Serosal and submucosal layers made relatively standardized contributions to total wall thickness in the stomach and small intestines (0.03–0.04 mm), with a lesser contribution to colon wall thickness. This result can be explained by the more standard thickness of connective tissue, blood supply and innervation amongst GI segments, 13 which varies little throughout the GI tract, despite different functions in each GI segment. This result can also be explained in the context of the extremely thin measurements and increased possibility of error.
Previous US studies have relied on aortic diameter as a consistent value associated with body size, and it has been reliably used for the calculation of ratios to be used in standardized measurements of renal length and portal vein diameter.11,12 It was our opinion that a comparison of muscularis and mucosal layer thicknesses to aortic diameter should result in a simple, easily obtained, clinically relevant value that could be used to objectively identify thickening of individual layers in response to GI diseases. The Musc:Ao ratio was greater in the ileum than in all other segments, which is not surprising considering that this segment has the greatest muscularis layer thickness of all GI segments. The Muc:Ao ratio was greater in the duodenum, gastric fundus, ileum and jejunum than in the colon, pyloric antrum or gastric body.
For the purposes of detecting muscularis layer thickening in their study, Zwingenberger et al 6 defined muscularis layer thickening as greater than half the thickness of the submucosal layer. In our study of clinically normal feline patients, all muscularis layer measurements were greater than half of the thickness of the submucosal layer, and in all segments, except the colon, the muscularis layer was as thick as, or thicker than, the submucosal layer. This finding is clinically important because the criterion reported by Zwingenberger et al 6 might result in a significant number of false-positive diagnoses.
There was no correlation between age and GI layer thickness. It is possible that with a greater sample size allowing comparison of smaller age ranges, an age-related difference might be resolved.
Cats fed a combination of wet and dry food had a small, but significantly greater, mucosal layer thickness in the ileum than cats fed dry food only. Perhaps this result can be explained by differences in the lymphoid tissue component of ileal mucosa 13 owing to exposure to different luminal contents, or by an increase in epithelial cells needed to absorb additional fluid contents at this location in the ileum. Another possible explanation is differences in carbohydrate/protein levels in these diets. Dry food diets, as a general rule, may contain more carbohydrates than canned food diets simply as part of the kibble formation process. 14 Because cats are obligate carnivores, their intestinal villi might respond differently to higher carbohydrate versus higher protein diets.
This study has some limitations. Some of the measurements made were at the limits of the spatial resolution of our transducer, specifically those of the serosal and submucosal layers, and the accuracy of these extremely small measurements can be questioned. However, measurements of the muscularis and mucosal layers were within the limits of spatial resolution, and we feel that our results provide an objective and clinically useful reference for muscularis layer and mucosal layer thickness in cats. Diet was not a controlled variable in our study. Including more cats on a wet food diet alone, or on different diet formulations, may reveal differences in layer thicknesses. Finally, it would have been ideal to confirm normalcy using full-thickness intestinal biopsies. Unfortunately, this was not performed and will be a direction of further research.
Conclusions
This study provides baseline layer thickness measurements in each segment of the GI tract in a sample of 38 healthy cats. In addition, the ratio of muscularis and mucosa thickness to aortic diameter may yield a clinically useful ratio in the diagnosis of diseases of the feline GI tract. More research is needed to assess the value of this ratio.
Footnotes
Funding
This project received partial support from the Merck-Merial Veterinary Scholars Program
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
