Abstract
Practical relevance:
Treatment of feline chronic kidney disease (CKD) tends to focus on minimising the adverse effects of reduced renal function, rather than addressing an underlying cause. Despite this, and the progressive nature of CKD, treatment can improve quality of life and enable many cats to have long survival times.
Evidence base:
Strong evidence supports the provision of renal diets, which are protein and phosphorus restricted; compliance is improved by gradual dietary transition. Additional phosphorus restriction is achieved by the use of phosphate binding agents, although it is unknown if these yield similar survival benefits to those provided by renal diets. Interventions to control hypokalaemia and hypertension in affected cats are important to prevent serious complications. Administration of benazepril to cats with proteinuric kidney disease has been shown to significantly improve their appetite but not their survival. As CKD progresses, many cats will benefit from treatment to control clinical signs of uraemic gastroenteritis and anaemia.
Therapy for CKD – what are the aims?
The ideal treatment for feline chronic kidney disease (CKD) would correct or reverse the underlying cause of CKD, identify and address factors associated with progressive CKD and minimise signs of reduced renal function. Unfortunately, the cause of feline CKD is usually unknown and treatments that slow CKD progression in other species (eg, calcitriol, benazepril) have not been proven effective in cats.1–4 The irreversible and progressive nature of CKD can be disheartening; however, treatment can improve quality of life and survival, providing fulfilling experiences for veterinarians and owners.
CKD causes retention of renally excreted wastes (eg, phosphorus) and loss of compounds (eg, potassium) that should be retained. Most therapy is aimed here, consisting of supportive and symptomatic treatments to correct hydration and address endocrine, metabolic and nutritional disturbances. Treatment is lifelong, highlighting the importance of easy medication administration to aid owner compliance (Figure 1).

Easy administration is an important consideration to aid owner compliance
Evidence-based medicine (EBM) integrates available research, clinical expertise, patient and owner preferences, and resource availability to tailor treatments in order to optimise outcome. Veterinary classification of EBM guidelines exists, and is summarised, together with the strength of evidence for currently available interventional therapeutics in feline CKD, in the box on page 30. Guidelines for CKD staging established by the International Renal Interest Society (IRIS) (www.iris-kidney.com) provide the classification system used throughout this article.

Dietary modification
Of all the CKD treatments used to date, dietary modification has the most positive long-term effect on outcome.7–9 Cats with CKD receiving renal diets instead of normal food survived significantly longer (20.8 months versus 8.7 months; 7 16 months versus 7 months 8 ). Additionally, a randomised, controlled, clinical trial (RCCT) compared feeding maintenance diets with renal diets in spontaneous CKD stages 2 and 3. 9 Cats fed the renal diet developed fewer uraemic episodes (0% versus 23%) and none died from renal disease. Thus, strong evidence exists to support the use of renal diets to prolong survival and improve the quality of life for cats with CKD.
There is no evidence supporting dietary modification in stage 1 CKD, although, in the authors’ experience, introducing a dietary change in a clinically well cat improves diet acceptance. Over 90% of cats with CKD accepted renal diets when a very gradual transition was used. 9 Attempting changes in sick, hospitalised, anxious patients can result in food aversion. Dietary modification should not be attempted until patients are well and discharged from hospital. There will always be some cats defiant of diet change. Although home-prepared renal diets are attractive to some owners, dietary assessment identified numerous nutritional inadequacies. 10 Therefore, in cats refusing renal diets, use of senior diets with phosphate binding agents (PBAs) if hyperphosphataemia is present, while not ideal, may be better than provision of maintainence diets alone.
Renal diets are restricted in protein, phosphorus and sodium and supplemented with potassium, omega-3 fatty acids, B vitamins and fat content, and are alkalinising. It is unknown which alterations are responsible for survival benefits, although studies in experimental models support phosphate restriction, 11 and essential fatty acid (EFA) supplementation as potential mechanisms. 12
Phosphate restriction
Strong evidence supports dietary phosphate restriction in animals with kidney disease. Serum phosphate is an independent predictor of disease progression in cats with CKD.11,13 In rodent models, phosphate restriction was associated with reduced tissue mineralisation and reduced glomerular hypertension. 14 Cats with induced renal disease fed phosphate-restricted diets had less severe histological renal changes than cats fed normal diets. 15
Renal diets restrict serum phosphate, and subsequently parathyroid hormone (PTH) concentrations, by limiting phosphorus-containing proteins,7,16,17 and are effective in controlling hyperphosphataemia and renal secondary hyperparathyroidism (RHPTH) and increasing survival in cats with CKD. 7 The goal of dietary therapy is to reduce phosphate concentration within 2–4 weeks of complete diet change. Target phosphate concentrations exist based solely on expert opinion (Table 1).16,18 Dietary modification alone can often control phosphate concentrations up to stage 3 CKD.17,19 In stage 4, diet alone may be insufficient. If after 4 weeks of dietary modification hyperphosphataemia (or increased PTH) persists, use of a PBA should be considered (Table 2). Regular monitoring of phosphate concentration every few months is recommended.
Recommendations for serum phosphate concentration in cats with CKD by IRIS stage

Commonly used phosphate binding agents in cats

NB All drugs must be given with food
Sodium restriction
Studies on dietary sodium intake in cats with and without CKD have produced variable results. High dietary sodium has been shown not to alter systolic blood pressure (SBP)20,21 or kidney function in healthy cats or cats with induced CKD.20,22,23 In contrast, serum creatinine concentration increased when cats changed from a low sodium to a high sodium diet. Notably, the increase in creatinine concentration was most marked in cats with higher initial creatinine concentrations. 21 However, dietary sodium restriction is not without potential risk. Cats receiving sodium-restricted diets have been shown to have renin–angiotensin– aldosterone system activation, hypokalaemia and reduced glomerular filtration rate (GFR). 20 Additionally, a case control study found that cats receiving higher sodium diets were less likely to develop CKD, although confounding factors may have affected this study. 24
In people, dietary sodium intake has been proposed to cause progressive kidney injury by mechanisms unrelated to hypertension, such as increased oxidative stress. 25
While results of the above studies indicate that hypertension is not salt sensitive in cats, the conflicting results, small study sizes and short study durations mean that the ideal dietary salt intake for cats with CKD that would minimise progressive renal damage remains unknown. There is currently no indication to alter the sodium intake of cats with CKD beyond the relatively low amounts found in renal diets.
Essential fatty acid supplementation
Feeding dogs with induced CKD diets enriched with omega-3 fatty acids decreased intraglomerular hypertension, maintained GFR and improved survival.12,26 No data is available in cats. A retrospective study reported longer survival times for cats receiving renal diets with the highest omega-3 fatty acid content, 8 and cats receiving renal diets that contain omega-3 fatty acid supplementation had fewer uraemic crises than those on maintenance diets alone. 9
Vitamins and antioxidants
Oxidative stress contributes to progression of CKD in people, 27 and has been identified in cats with CKD.28,29 A cross-over study in cats with spontaneous CKD found that vitamin E, C and beta-carotene supplementation reduced oxidative stress. 30 Studies assessing survival benefits are required.
Treatment of hyperphosphataemia and renal secondary hyperparathyroidism
Approximately 60% of cats with CKD are hyperphosphataemic. 31 The aetiopathogenesis of hyperphosphataemia and RHPTH has been described.16,17,32 Serum phosphorus concentration is a negative prognostic indicator in CKD and potentially contributes to progressive renal dysfunction.11,13,33 Methods for controlling hyperphosphataemia include rehydration, dietary phosphorus restriction and PBAs for cats with stage 2 or higher CKD. 17
In people, PTH is a major uraemic toxin, increasing intracellular calcium and resulting in neurotoxicity, immune dysfunction and exacerbation of anaemia. 34 If PTH is similarly toxic in cats, another treatment goal would be to normalise or prevent PTH increases by dietary phosphate restriction, use of PBAs and calcitriol administration. 16 In general, PTH concentrations parallel serum phosphate concentrations and the prevalence of RHPTH increases with the severity of renal disease, 31 although some cats develop RHPTH before overt azotaemia or hyperphosphataemia. 35
Phosphate binding agents
PBAs bind dietary phosphorus in the gastrointestinal tract, producing insoluble compounds that are excreted in faeces. Safety and efficacy data for some PBAs in cats are available; however, little data exist regarding their effects on survival.36–38 PBAs must be given with food and can be poorly accepted, causing inappetence that negates any potential benefits of both PBAs and renal diets. Treatment response should be assessed with regular phosphate monitoring. Obtaining fasted blood samples avoids post-prandial hyperphosphataemia. 32 Commonly used PBAs are described in Table 2.
Chitosan and calcium carbonate administration reduces hyperphosphataemia in cats.36,38 Plasma phosphate and blood urea nitrogen (BUN) concentrations decreased within 35 days in six cats with CKD receiving the supplement with a maintenance diet. 38 A RCCT in cats with induced CKD identified decreased serum phosphate and PTH concentrations and reduced urinary fractional excretion of phosphorus in animals receiving the supplement compared with maintenance diets alone. 36 No differences in BUN, creatinine, GFR or renal plasma flow were found. These studies suggest that for cats in stages 1 and 2 CKD, administration of chitosan-containing PBAs decreases phosphate concentration. Whether the survival benefit differs from that provided by dietary phosphate restriction is unknown. Calcium-containing PBAs can cause hypercalcaemia if used with calcitriol. 32
In people with renal failure, lanthanum appears efficacious with few adverse effects, 39 although tissue accumulation is reported in rats. 40 Vomiting occurs in cats at high dosages. 37 Preliminary results of lanthanum use in cats with CKD showed decreased serum creatinine and phosphate concentrations. 41
Aluminium toxicity has been demonstrated in people and dogs with renal failure.42,43 However, as aluminium hydroxide is effective, inexpensive and readily available, use has continued. Constipation occurs commonly in cats and can be addressed by conservative doses (to avoid dehydration) of lactulose (eg, 0.5–1 ml PO q12h).
Another phosphate binder widely used in humans, sevelamer, contains neither calcium nor aluminium but may bind additional vitamins and thus vitamin supplementation is required. 44

Calcitriol
Calcitriol supplementation can theoretically ameliorate excess PTH and early intervention may prevent parathyroid gland hyperplasia. 2 An uncontrolled survey reported that calcitriol administration to cats with CKD improved activity levels and appetite; 45 however, neither daily nor intermittent calcitriol administration reduced PTH concentration, 1 and a 1 year RCCT of calcitriol administration in cats with CKD failed to show any benefits. 2 Details of this last study have not been published and benefits of calcitriol have been identified in dogs 46 and people,47,48 so it remains possible that the dose, study duration or a type II study error (false-negative results) limited the ability of the study to detect a genuine effect of calcitriol. Based on current evidence, however, it is difficult to justify routine administration of calcitriol to cats. Additional RCCTs would be valuable.

If serum phosphate levels are above 1.94 mmol/l (6.0 mg/dl), calcitriol administration can cause soft tissue mineralisation. 44 Calcitriol should only be considered after dietary phosphorus restriction and PBA use (see box on page 32). In stages 1 and 2 CKD, dietary phosphate restriction may be sufficient to combat declining calcitriol levels. 17
Controlling hypertension
Hypertension occurs in 20–60% of cats with CKD,49,50 causing target organ damage (TOD)49,51,52 and potentially contributing to CKD progression. 53 Guidelines for diagnosis of hypertension are detailed elsewhere.54,55 No association between hypertension and survival has been identified;56–58 however, hypertension is associated with proteinuria, 57 and proteinuria is associated with survival in CKD, 58 which may provide justification for antihypertensive treatment in addition to the amelioration of TOD. The risk of TOD justifies screening for and treating hypertension in cats with CKD (see box above).
Amlodipine, a calcium channel blocker, is effective in reducing hypertension, proteinuria and the risk of TOD, and improving quality of life in cats.51,57,59,60 Few side effects, rapid onset and easy administration make this the first choice for hypertension control. Transdermal amlodipine is available; however, bioavailability is reduced. 61
Angiotensin-converting enzyme (ACE) inhibitors reduce both systemic and glomerular hypertension,62,63 but provide only modest antihypertensive effects,64,65 making them generally unsuitable for monotherapy. ACE inhibition may increase potassium and creatinine concentrations due to GFR reduction following efferent arteriole dilation. Increases in serum creatinine concentration of 30% above pre-treatment baselines, or inappetence associated with ACE inhibitor administration, warrant treatment cessation. 66 Benazepril (0.25–0.5 mg/kg PO q12–24h) may be better tolerated than enalapril (0.2–0.7 mg/kg q12–24h) as biliary excretion compensates for reduced renal clearance, although the potential adverse effect of reduced GFR exists for both. 67
Treatment of proteinuria
Proteinuria is a negative prognostic marker for people, dogs and cats with CKD.13,58,68–71 ACE inhibitor treatment improves survival in people with proteinuric renal disease, 68 but is not recommended in early stage CKD. 72 Treatments reducing proteinuria, specifically benazepril, have been trialled in cats with the aim of improving survival. Although benazepril reduced proteinuria, a RCCT of 61 cats with spontaneous CKD receiving benazepril or placebo failed to identify significant survival benefits, 4 a result confirmed in another study of 192 cats with spontaneous CKD. 3 In the latter study, administration of benazepril to proteinuric cats (urine protein:creatinine ratio [UPC] >1) was associated with increased appetite (and a trend towards prolonged survival, although this was not statistically significant) compared with placebo-treated cats.
Consensus recommendations to reduce renal proteinuria suggest introducing dietary protein restriction and ACE inhibitor therapy in stages 2–4 CKD when UPC >0.4. 73 ACE inhibitors cause efferent arteriolar dilation, the potential benefit of which is to reduce glomerular hypertension. The use of these agents in cats with induced CKD maintained GFR while reducing glomerular hypertension.62,63 However, in cats with severe CKD or concurrent hypovolaemia, glomerular hypertension is a mechanism for maintaining total GFR, and marked deteriorations in renal function have been associated with the use of ACE inhibitors in dogs and cats. 66
In patients with moderate to severe CKD, dose titration of ACE inhibitors should be performed cautiously, with regular monitoring, due to the risk of GFR reduction. A mild increase (10–15%) in creatinine concentration in a hydrated, appetent cat is not an indication to stop treatment but cats should be monitored for inappetence, dehydration and progressive increases in creatinine concentration. As mentioned, increases of 30% or more above pre-treatment baselines, or inappetence associated with ACE inhibitor administration, warrant treatment cessation. 66
EFA supplementation reduces renal proteinuria in dogs.12,26 If supplementation above that provided by renal diets is required, fish oil (10–200 mg/kg PO q24h) may be useful. 74
Therapy for uraemic gastroenteritis
Gastrin is excreted by the kidneys and the concentration increases with CKD progression, increasing gastric acidity and the risk of ulceration. 75 Cats in CKD stages 3–4 often demonstrate gastrointestinal signs of uraemia (eg, inappetence, nausea, vomiting, stomatitis, gastrointestinal ulceration, diarrhoea, colitis) and addressing these may improve quality of life. Commonly used antiemetic and gastroprotective agents are detailed in Table 3.
Commonly administered antiemetic and gastroprotective agents in cats
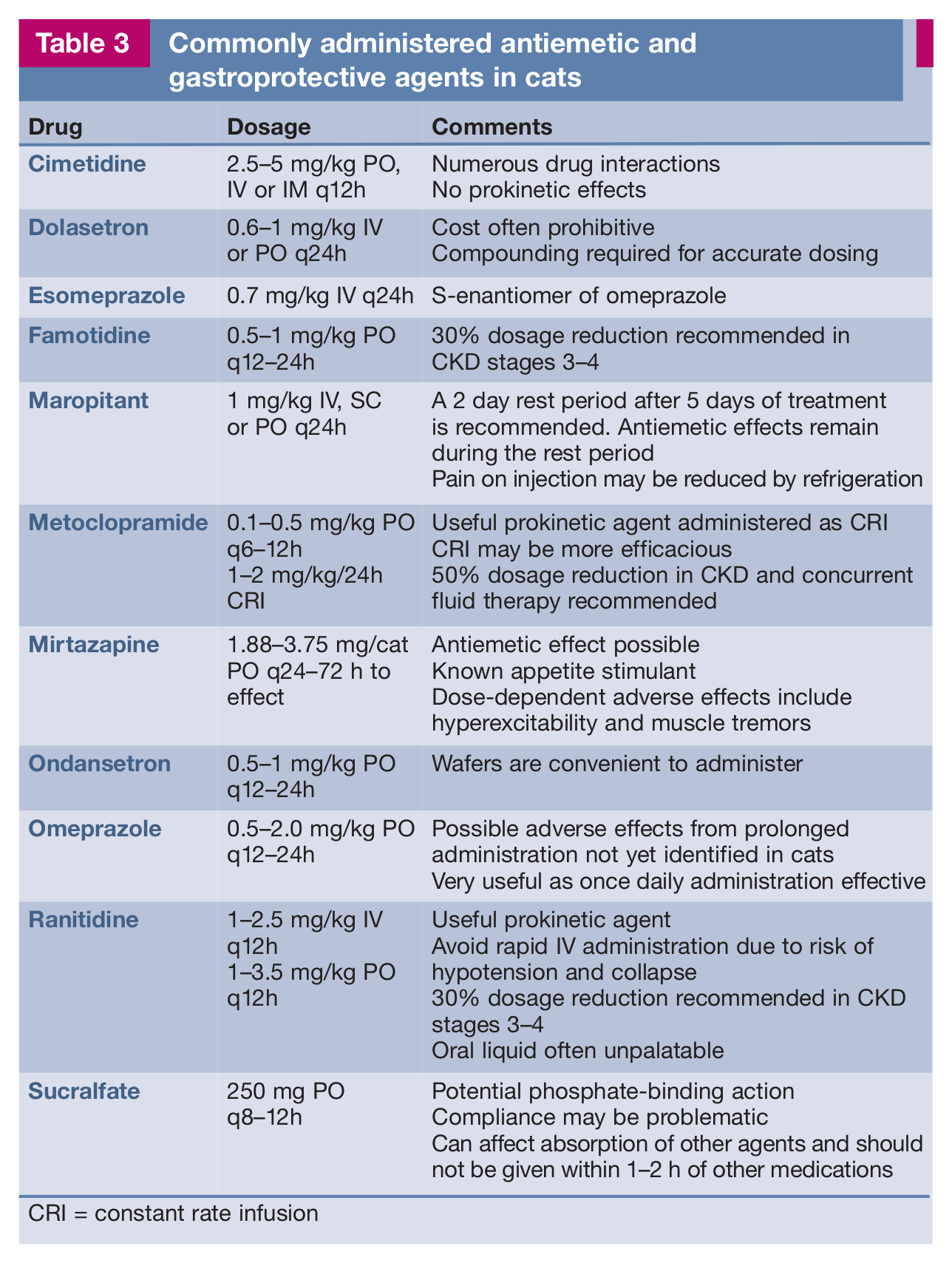
CRI = constant rate infusion
Vomiting is mediated via effects of uraemic toxins on the chemoreceptor trigger zone and gastrointestinal irritation. The effectiveness of many antiemetic agents in cats is established76–78 (and reviewed by Trepanier 2010). 79 However, few studies have evaluated these drugs in uraemic cats. 80

Maropitant inhibits neurokinin-1 receptors and is an effective, once daily antiemetic in cats with few adverse effects. The parenteral formulation is associated with pain on injection. 76 Refrigeration reduces pain in dogs, 81 and the same appears true in cats. Oral formulations are also available.
Metoclopramide is a dopaminergic antagonist, prokinetic and antiemetic agent. A short elimination half-life in other species and clinical experience suggest that constant rate infusions are substantially more effective than other routes of administration. 79 In people with renal failure, metoclopramide reduces renal plasma flow and renal failure is associated with impaired metoclopramide clearance.82,83 Dose reduction and concurrent fluid therapy should be considered in cats with CKD.
Mirtazapine has often marked appetite-stimulating properties and potential anti-emetic effects via 5-HT3 receptor antagonism. Its antiemetic efficacy in cats with CKD is unknown. Pharmacokinetic studies in cats with stage 2–4 CKD found prolonged renal clearance and doses at the lower end of the range (see Table 3) are recommended.80,84
Ondansetron and dolasetron are potent antiemetic agents, mediated via 5-HT3 receptor antagonism. 78 No studies have investigated their use in cats with CKD; however, experience with ondansetron suggests efficacy, although costs can be prohibitive.
In CKD stages 3–4, addressing gastric hyperacidity and subsequent mucosal irritation may be beneficial. H2 receptor blockers (famotidine, ranitidine, cimetidine) reduce gastric acidity. Famotidine is more potent than ranitidine, with a similar duration of action; 79 however, famotidine was no more effective than placebo in normal dogs. 85 Studies in cats are required. Uraemic gastroenteritis alters gastrointestinal motility in people, 86 and as ranitidine also has prokinetic actions, it may be the better choice. Elimination of H2 receptor blockers is reduced in people with renal failure and dose reduction should be considered in CKD.87,88 Cimetidine has a veterinary market authorisation but provides no prokinetic action and is associated with a range of drug interactions via its effects on hepatic microsomal enzymes. 89
Proton pump inhibitors (omeprazole, esomeprazole) are more potent than H2 receptor blockers and are effective once daily agents in cats. 90
Sucralfate is an aluminium compound that forms a barrier over ulcers and stimulates bicarbonate and prostaglandin E2 production, 91 which may be beneficial in stage 4 CKD.
Correcting dehydration
Dehydration occurs due to concurrent disease, inappetence or water intake that is inadequate to compensate for the polyuria that consistently accompanies CKD. Dehydration can potentiate progression of CKD – and in cats with pre-existing CKD experiencing a uraemic crisis or the newly diagnosed CKD patient that is clinically unwell, treatment of dehydration is essential. Treatment goals are to correct dehydration, restore GFR and reduce uraemia.6,44 Hydration status must be assessed regularly (Table 4) but interpreted with care. Elderly or emaciated cats have reduced skin elasticity and uraemia may cause dry mucous membranes independent of hydration. 93 Overcand congestive heart failure in patients with concurrent disease (eg, hyperthyroidism) and should be avoided (see box on page 36).
Clinical assessment of hydration status

Adapted from Chew et al 92
Water intake is encouraged by improving water access, adding water to food, using fountains and via feeding tubes. Oral intake of water avoids the sodium increase that is associated with parenteral fluids. Where oral intake is inadequate, however, alternative methods of hydration are required.
For cats with stages 3–4 CKD, subcutaneous fluid therapy may help to prevent dehydration. Once daily, alternate day or twice weekly subcutaneous fluid therapy using a balanced electrolyte solution (eg, lactated Ringer’s) appears to be helpful clinically, but has yet to be assessed with RCCTs. Dosage depends on patient size (30–100 ml per cat q24–48h to twice weekly, as required, to maintain hydration). For a step-by-step guide, owners can be directed to icatcare.org/advice-centre/cat-care/how-give-subcutaneous-fluids-your-cat.

Subcutaneous indwelling catheters (Figure 2) provide permanent ports for administration of fluids. General anaesthesia is required for placement, which could potentiate further deterioration in renal function, and catheters may increase the risk of infection. For owners who cannot otherwise manage subcutaneous fluid therapy, this may, however, be a useful option.

Subcutaneous fluid catheters provide an alternative method for fluid administration in the home
Addressing malnutrition
Malnutrition can result from inappetence secondary to uraemic gastroenteritis, dehydration, azotaemia, anaemia and concurrent disease. Dogs with CKD that are underweight have shorter survival times, 94 and malnutrition likely has similar negative effects in cats. Renal diets improve survival,7–9 so it is vital to address any causes of inappetence that could contribute to failure to implement a dietary change. Estimating resting energy requirements (70 x BWkg0.75 or 30[BWkg] + 70) determines daily calorie requirements. Dietary intake is improved by providing highly palatable, warmed food. Treatment goals are to maintain body weight and a body condition score of 5/9 or 2.5–3/5.
Mirtazapine is a useful appetite stimulant, with effects occurring within 30 minutes of administration (Table 3). Food should be offered around this time. Cyproheptadine and diazepam stimulate appetite; however, their effects are short-lived and can be unpredictable. While side effects associated with cyproheptadine (eg, sedation) are mild, oral diazepam has been associated with idiosyncratic liver failure in a small number of cats.95,96 Importantly, use of these agents does not typically result in adequate and predictable food intake and, therefore, they are not recommended. 97
Assisted enteral nutrition should be considered in cats that are inappetent or anorexic for more than 3 days. 98 Naso-oesophageal tubes allow short-term nutrition, but only limited diets can be administered. Oesophagostomy tubes allow provision of adequate nutrition and water, plus easy administration of medication (Figure 3); they can remain in situ for long periods (eg, 3–6 months). Feeding tube placement is discussed elsewhere. 97 While insertion site infection, tube migration and CKD progression following anaesthesia are potential risks, the advantages of oesophagostomy tubes tend to outweigh complications. In the authors’ experience, the ability to easily provide nutrition and medications improves quality of life.

An oesophagostomy tube allows provision of adequate nutrition and water, plus easy medication administration
Treatment for hypokalaemia
Hypokalaemia has been identified in 20–30% of cats with CKD and is more common in hypertensive cats.50,99,100 The exact cause is unknown but assumed to be a combination of reduced intake, increased urinary losses and renin–angiotensin activation. 101 Hypokalaemia can cause weakness due to myopathy (Figure 4) and potentially contributes to progressive renal injury.102,103 Potassium supplementation in hypokalaemic cats with muscle weakness often results in clinical improvement of weakness within a week. Parenteral and oral potassium administration is detailed in Table 5.

A cat with CKD demonstrating cervical ventroflexion, which is typical of hypokalaemic myopathy
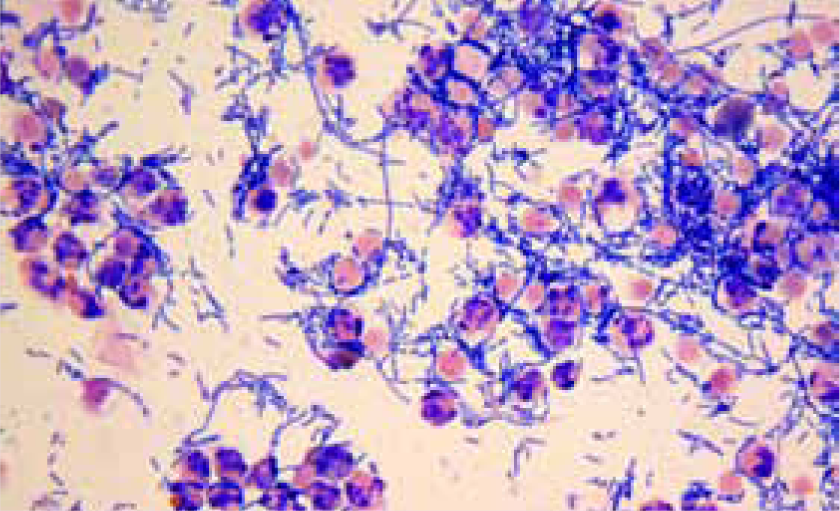
Photomicrograph of direct urine sediment findings in a cat with CKD and a UTI with Escherichia coli. x 500
Guidelines for parenteral and oral potassium supplementation in cats

Total body potassium deficits may occur before hypokalaemia; 104 however, no evidence exists to suggest that potassium administration to normokalaemic cats with CKD is beneficial. In one study, potassium gluconate administration in cats with spontaneous disease did not alter SBP, aldosterone concentration, creatinine or UPC, although studies of longer duration are needed. 105
Addressing metabolic acidosis
Metabolic acidosis affects 15% of cats in stage 3 and 52.6% of cats in stage 4 CKD. 106 Acidosis and hypokalaemia could have additive adverse effects on kidney function. 103 Whether additional alkalinisation above that provided by renal diets is required is unknown. It seems reasonable to provide additional alkalinisation in stage 3–4 CKD where blood pH is <7 22 and bicarbonate concentration <15 mmol/l in a hydrated patient. 32 Treatment options include sodium bicarbonate and potassium citrate. Unfortunately, sodium bicarbonate tends not to be palatable. Potassium citrate (Table 5) also provides additional potassium.
Monitoring by blood gas analysis should be performed every 10–14 days during stabilisation, with blood collected just prior to drug administration and pH determined within 1 h. 107 Bicarbonate concentration should be maintained between 15 and 22 mmol/l and blood pH between 7.2 and 7.4.
Treatment for anaemia
Anaemia of CKD results from insufficient renal erythropoietin (EPO) production and is often exacerbated by gastrointestinal haemorrhage, malnutrition and reduced red cell life span (reviewed by Chalhoub et al 2011). 108 It is typically normocytic, normochromic and poorly regenerative. Approximately 30–65% of cats with CKD develop anaemia, with severity proportional to disease stage.99,100,109 Whether anaemia severity affects survival is unclear;11,69,110 however, moderate to severe anaemia is likely to have a negative impact on quality of life.
All potential causes of anaemia should be addressed. Gastrointestinal haemorrhage without melaena or hypochromia occurs, 110 and should be suspected if the severity of anaemia outweighs the degree of renal dysfunction present or if urea concentration is disproportionately increased compared with serum creatinine in the absence of dehydration. Therapeutic trials with gastroprotective agents (Table 3) can be helpful.
Recombinant human erythropoietin (R-HuEPO) products, including epoetin and darbepoetin, have been used in cats with CKD, resulting in improvements in appetite and quality of life. Both products are identical to the naturally occurring hormone in people and relatively similar (83.3%) to feline erythropoietin, with darbepoetin having a prolonged half-life and, therefore, requiring less frequent administration than epoetin. 108 As R-HuEPO differs structurally from feline EPO, a major obstacle is anti-EPO antibody development; cross-reaction with the R-HuEPO agent and EPO causes pure red cell aplasia, a severe, non-regenerative anaemia that occurs in 25–30% of cats receiving R-HuEPO. 111 It is theorised that the prolonged half-life of darbepoetin compared with epoetin reduces the antigen load administered and thus the likelihood of treated cats developing antibodies. 108
There is limited published information on the efficacy and safety of R-HuEPO administration in cats with CKD and that which is available comes from uncontrolled case series.111–113 In a study of 25 cats treated with darbepoetin, most (56%) responded to treatment and responders lived significantly longer than non-responders. 112 Concurrent disease was identified more often in non-responders than in responders. 112 Notably, however, cats were only included in this study if they survived longer than 56 days after treatment was instituted. R-HuEPO may be less effective in cats with concurrent disease causing anaemia or with more severe renal disease. Further work is required to evaluate the effect of treatment with R-HuEPO on survival and the optimal time to institute treatment.
R-HuEPO agents should only be considered in cats with advanced CKD and haematocrit <22%, plus clinical signs of anaemia (eg, weakness, tachycardia, tachypnoea, pallor) without an obvious underlying cause. Additional adverse effects of R-HuEPO treatment include polycythaemia, vomiting, iron deficiency, injection site discomfort, skin reactions, fever and arthralgia.111,112 Hypertension occurred in 41–50% of cats receiving R-HuEPO agents and seizures in 16% receiving darbepoeitin.112,114,115 In human medicine, the use of epoetin has been largely replaced by darbepoetin because of its increased potency and duration of action. 116 Table 6 presents guidelines for administration of R-HuEPO agents (darbepoetin and epoetin).
Guidelines for recombinant human erythropoietin agent administration in cats

CBC = complete blood count, EPO = erythropoietin, PCV = packed cell volume, PRCA = pure red cell aplasia, R-HuEPO = recombinant human erythropoietin agent, SBP = systolic blood pressure
Oral iron supplements can be bitter, poorly absorbed and associated with gastrointestinal complications. For these reasons some authors recommend injectable iron dextran 108

Iron deficiency can occur in cats with CKD due to gastrointestinal haemorrhage and reduced absorption or intake. 108 Ideally, true iron deficiency should be differentiated from the anaemia of inflammatory disease (iron sequestered in bone marrow monocytes) because iron supplementation of the latter is ineffective and may result in iron overload. Serum iron status is difficult to assess; however, true iron deficiency should result in low serum iron, ferritin and transferrin saturation. Iron supplementation is recommended with true iron deficiency and when commencing R-HuEPO treatment (Table 6).
Anabolic steroids (eg, nandrolone cypionate, stanozolol) produce effects that are potentially beneficial in cats with CKD, including improved haematocrit, appetite and muscle mass. 117 However, results are generally mild or inapparent and stanozolol is hepatotoxic in cats. 118 The use of anabolic steroids in general is no longer recommended in cats with CKD.

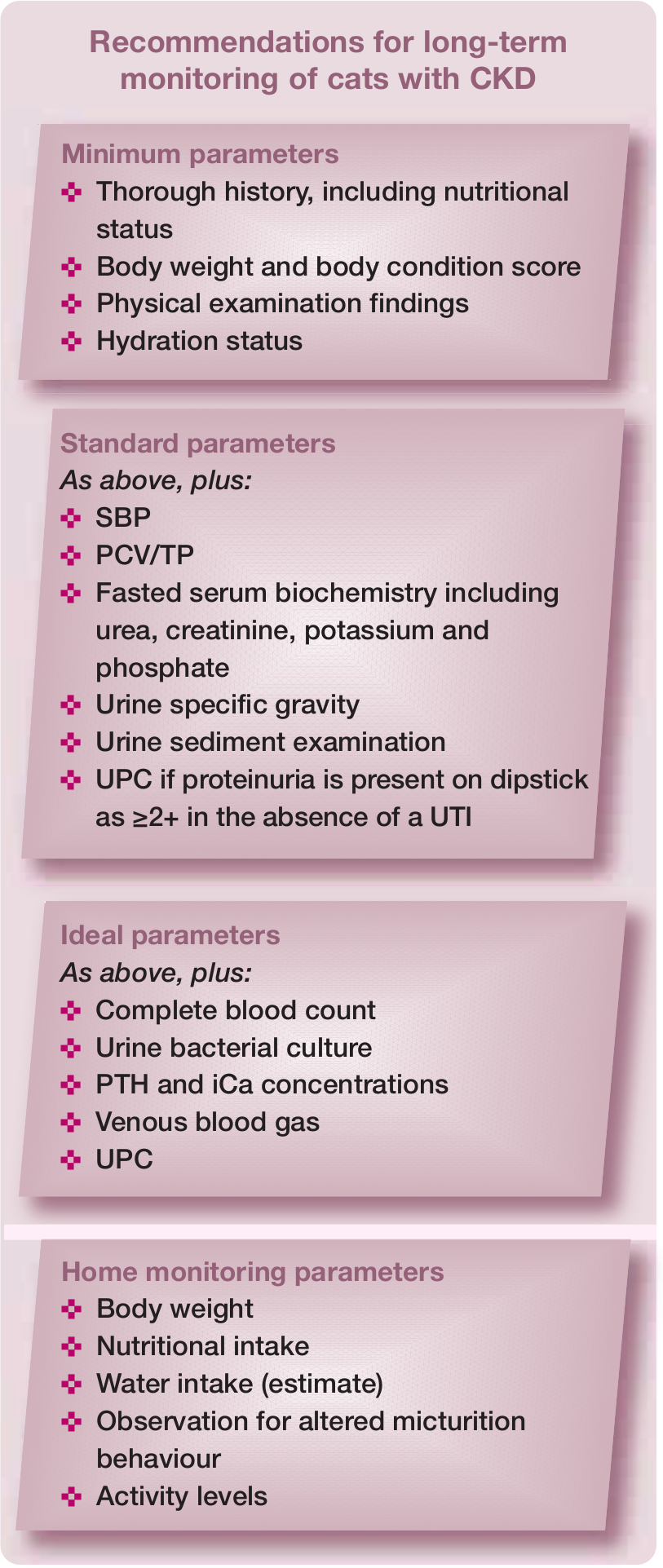
Ongoing monitoring and treatment prioritisation
CKD is a progressive condition requiring monitoring with a frequency determined by disease severity, client compliance, treatment response and financial constraints. Following diagnosis, patients should be monitored every 2–4 weeks until disease stability is established and persistent changes (eg, hypertension, proteinuria) identified. Patients in stage 1–2 could be monitored 6-monthly, and stage 3–4 every 1–3 months. Monitoring recommendations are detailed in the box on the right.
Treatment is prioritised based on the strength of evidence available (see box on page 30), together with consideration of cat and owner compliance, ease of administration, resource availability and financial constraints. Given the strong evidence supporting renal diets, ensuring successful dietary modification should be a treatment priority.
Table 7 details survival estimates for cats categorised by CKD stage. Survival times at lower stages can be long and cats receiving effective treatment often die from other diseases. 9 Currently, treatment of CKD is about management rather than cure, centred on diagnosis and staging followed by multimodal treatments to correct hydration and address endocrine, metabolic and nutritional discrepancies. With a considered approach, it is possible to improve both quality and quantity of life.
Survival estimates for cats classified by IRIS stage

IRIS = International Renal Interest Society, BP = survival estimate for hypertensive cats
Stage 2b azotaemic stage: creatinine concentration 200–250 μmol/l (2.26–2.82 mg/dl)
Footnotes
Key Points
Funding
The preparation of this article was supported by an educational grant from Boehringer Ingelheim.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
